Abstract
Chimeric antigen receptor (CAR)-T cell-based immunotherapy has emerged as a path-breaking strategy for certain hematological malignancies. Assessment of the response to CAR-T therapy using quantitative imaging techniques such as positron emission tomography/computed tomography (PET/CT) has been broadly investigated. However, the definitive role of PET/CT in CAR-T therapy remains to be established. [18F]FDG PET/CT has demonstrated high sensitivity and specificity for differentiating patients with a partial and complete response after CAR-T therapy in lymphoma. The early therapeutic response and immune-related adverse effects such as cytokine release syndrome and immune effector cell–associated neurotoxicity syndrome can also be detected on [18F]FDG PET images. In otherwise asymptomatic lymphoma patients with partial response following CAR-T therapy, the only positive findings could be abnormal PET/CT results. In multiple myeloma, a negative [18F]FDG PET/CT after receiving B-cell maturation antigen-directed CAR-T therapy has been associated with a favorable prognosis. In leukemia, [18F]FDG PET/CT can detect extramedullary metastases and treatment responses after therapy. Hence, PET/CT is a valuable imaging tool for patients undergoing CAR-T therapy for pretreatment evaluation, monitoring treatment response, assessing safety, and guiding therapeutic strategies. Developing guidelines with standardized cutoff values for various PET parameters and tumor cell–specific tracers may improve the efficacy and safety of CAR-T therapy.
Introduction
Immunotherapy has profoundly improved the clinical management of cancer and opened new therapeutic possibilities in the past decade. One such modality is chimeric antigen receptor (CAR)-T cell therapy. Chimeric antigen receptor-T cells are engineered by introducing a CAR fusion protein into autologous T cells using retrovirus or lentivirus-based recombinant DNA technology. 1 These synthetically designed receptors guide T-cells to identify and kill malignant cells expressing the targeted antigen. 2 Unlike the typical response of T-cells, activation of CAR-T cells and potent antitumor responses occur when the CAR binds to antigens in tumor cells in a manner independent of the major histocompatibility complex. 3 Chimeric antigen receptor-T cell therapy has been studied in hematologic malignancies such as lymphoma, multiple myeloma, and leukemia with curative potential.4-10 The United States Food and Drug Administration (FDA) approved CAR-T therapy against CD-19 (CAR T19) for aggressive B-cell lymphoma4,9 and multiple myeloma. 11 Noninvasive imaging via positron emission tomography/computed tomography (PET/CT) is a well-established method to assess the response to treatment in patients with hematological malignancies. However, information on PET/CT imaging to monitor the response following CAR-T therapy is currently limited. 12
Chimeric antigen receptor-T therapy can cause adverse effects such as lymphodepletion, prolonged cytopenia, hematopoietic reconstitution, infections, and immune-mediated toxicity. 13 Immune-mediated toxicity occurs in the form of cytokine release syndrome (CRS), immune effector cell-associated neurotoxicity syndrome (ICANS), and macrophage activation syndrome. 13 These side effects can restrict the use of CAR-T therapies. Therefore, the use of PET/CT imaging to predict or diagnose these complications early could improve the results of CAR-T immunotherapy. Developing imaging-guided therapeutic strategies typically involves evaluating possible responses (hyperprogression and pseudoprogression) and immune-mediated adverse events, often through metabolic imaging with 18F-Fluorodeoxyglucose ([18F]FDG) or other radiotracers. In addition to briefly describing the biology and generations of CAR-T cells, this literature review reexamines studies that have assessed the efficacy and safety of CAR-T cell therapy using [18F]FDG and other radiotracer-based PET/CT imaging in patients with lymphoma, multiple myeloma, and leukemia.
Chimeric Antigen Receptor-T Cell Biology and History
In the 1980s, groundbreaking research demonstrated the potential of lymphocytes in cancer treatment when allogeneic hematopoietic grafts eradicated leukemic cells in mice.14,15 Over the following 3 decades, significant progress was made in human studies. 3 An important milestone occurred in August 2017 when the FDA approved the first CAR-T cell therapy, “Tisagenlecleucel-T,” for young patients with B-cell acute lymphocytic leukemia (B-ALL), achieving a complete remission rate of 60% and an 81% overall response rate. 5 Three generations of CAR-T cells have evolved since their initial development 13 (Table 1).
Summary of CAR-T Cell Generations.

Abbreviation: CAR-T, Chimeric antigen receptor-T cell.
The third generation of CAR-T cells combines CD28 and 4-1BB costimulatory molecules, enhancing their clinical efficacy against cancer.1,16,17 CD28-based cells expand through B7 signaling, while 4-1BB-based cells expand through TNF-associated factors signaling, activating NF-κB to boost T-cell activity.18,19 These CAR-T cells consist of 4 components: an antigen-binding domain, a hinge region, a transmembrane domain, and intracellular signaling molecules. 20 They proliferate, target specific cancer antigens, and prevent tumor infiltration through innate and adaptive immunity.21,22
Despite these advances, the widespread clinical use of CAR-T therapy has been limited by 2 main factors: poor transfer to tumor sites limiting clinical response and the development of life-threatening adverse events, including CRS, as a result of massive T cell proliferation, and immune activation.28,29 Moreover, the persistent exposure of CAR-T cells to cancer antigens has been shown to cause their differentiation into a dysfunctional state, inhibiting their effector functions and ultimately leading to CAR-T cell exhaustion. These cells become dysfunctional in the exhausted state, contributing to the failure to clear cancer cells and, thus, resulting in late-stage disease relapse. 30 Furthermore, CAR-T cells can also become senescent, leading to cell-cycle arrest while cells are still viable and metabolically active. The senescence of immune cells is related to age factors and is often present in patients administered with chemotherapy.31-33
The use of functional PET is imperative for the noninvasive monitoring of the response to CAR-T therapy by imaging the homing pattern, expansion, and activation markers.34,35 For example, CAR-T cells are usually monitored by administering a reporter gene substrate, which creates a signal detectable by PET/CT. Many immune markers have already been developed; others are in the investigational phase. In addition to localizing the anatomy of the lesion with the help of CT, these radiotracers provide a valuable toolbox for initial diagnosis and monitoring of clinical response and tumor immune-microenvironment.36,37 Multiple PET reporter genes have been identified, including HSV-tk, hdCKDM, hTK2DM, hNET, and others. 38 Among these, hdCKDM and hTK2DM are pyrimidine-specific PET reporter genes used for imaging with [18F]FEAU, [18F]FMAU, and [124I]FIAU, which are all substrates of HSV-tk.39,40 These genes are of human origin and, hence, they are nonimmunogenic. In addition, these tracers have not been found to impact T-cell function in in-vitro assays. However, the sensitivity of reporters in CAR-T cells is difficult to predict due to differences in uptake and expression with variable transduction of T-cells.41,42
Time Frame of [18F]FDG PET/CT Imaging in CAR-T Therapy
In CAR-T therapy, patients typically undergo 4 key evaluations with [18F]FDG PET/CT.12,43 First, 2 scans are performed before CAR-T cell infusion: one at the time of decision to guide treatment planning and another at the time of transfusion just prior to CAR-T infusion. 44 Subsequently, 2 more scans are used at 1 month and 3 months after infusion to assess therapy response, with very high sensitivity and specificity. 44 Although multiple scans are generally well tolerated by patients, not all medical centers conduct both pre-therapy scans.45,46 This has led to uncertainty regarding the importance of early response assessments after CAR-T infusions. The optimal timing for imaging during CAR-T therapy remains a topic of discussion. 47
Patterns of Response to CAR-T Therapy
In addition to typical response patterns associated with complete or partial regression, stable and progressive disease, CAR-T therapy can cause atypical response patterns on [18F]FDG PET/CT. 48 Atypical responses in cancer immunotherapy can be classified into 4 categories: pseudoprogression, hyperprogression, durable response, and dissociated response. 49 Pseudoprogression can occur in certain patients before a favorable clinical outcome 50 ; it is more like a flare produced by excessive immunological activation. 48 Delayed immunologic response may also cause pseudoprogression. 51 The highest increase in tumor burden associated with pseudoprogression has been reported to be 20% to 163%. 52 Immune cells may be recruited into the tumor microenvironment during early therapy, temporarily increasing tumor size and metabolic activity, falsely giving the impression of increased tumor burden. 48 This frequently involves tumor size growth but not a decline in clinical condition. Histopathological examination and follow-up imaging can confirm pseudoprogression by tumor regression and good clinical outcomes. 53 However, true disease progression, or hyperprogression, generally worsens the patient's clinical condition. Although criteria such as Immune Response Evaluation Criteria in Solid Tumors, Immunotherapy Response Assessment in Neuro-Oncology, or Lymphoma Response to Immunomodulatory Therapy Criteria can be used to evaluate pseudoprogression, there is currently no established method to evaluate hyperprogression. There have been several proposed assessment criteria for hyperprogression; however, future international collaborative efforts will be necessary for establishing a consensus. 54 The dissociated response implies regression of some tumor cells and progression of others, similar to the mixed response seen after chemotherapy. 49 The survival outcome has been found to be higher in the dissociated response compared to patients with true progression of the disease. 55
Role of [18F]FDG PET in CAR-T Therapy for Lymphoma
[18F]FDG PET/CT is well established for the staging and evaluation of chemotherapy treatment response in patients with non-Hodgkin lymphoma (NHL). 56 After chemotherapy, residual [18F]FDG uptake predicts long-term survival.57,58 The different metabolic parameters measured with [18F]FDG PET/CT are maximum standardized uptake value (SUVmax), the total metabolic tumor volume (MTV, obtained from the sum of the metabolic volumes of all nodal and extranodal lesions), and whole-body total lesion glycolysis (TLG, the product of the metabolic volume and the mean standardized uptake value (SUVmean) for all lesions) (TLG = ∑MTV × SUVmean). 59 In patients with NHL receiving chemotherapy, quantifying the metabolic tumor burden before infusion as MTV or TLG has demonstrated a considerable prognostic value.56,60
In a retrospective study involving 72 patients who underwent CAR-T cell therapy for aggressive B-cell lymphoma, Sesques et al assessed how effectively [18F]FDG PET/CT before leukapheresis, before CAR T-cell infusion, and one month after infusion could predict patients’ prognosis. 59 Metabolic parameters such as MTV, TLG, SUVmax, and tumor kinetics were assessed. The changes in MTV and TLG before therapy, as measured by ΔMTVpre−CAR and ΔTLGpre−CAR (where, Δpre−CAR = (value at preinfusion − value preleukapheresis)/value at preleukapheresis × 100), were predictive of progression-free survival, and SUVmax at one month after CAR-T infusion was predictive of overall survival. In simple terms, changes in MTV and TLG before CAR-T cell infusion were found to be significant in predicting progression-free survival. 59 Specifically, lower increases in these metrics were associated with longer PFS. Similarly, SUVmax one month after infusion was associated with overall survival. A higher SUVmax at this time point indicated a poorer prognosis. 59
In another retrospective clinical study of large B-cell lymphoma by Ruff et al, the sensitivity and specificity of [18F]FDG PET/CT to detect the lesion were 99% and 100% at the time of the first response assessment (median time was 86 days). 12 Forty-two percent showed a complete response and another 14% showed a partial response. In patients with partial response, pretreatment SUVmax initially decreased with later increases and progression of the disease. Furthermore, although most of the patients with partial response and progressive disease were asymptomatic, routine surveillance with [18F]FDG PET/CT could detect those asymptomatic patients with progressive disease. 12
In a retrospective study by Reinert et al, patients who achieved partial response had significantly higher baseline MTV and TLG than those who obtained a complete morphological and metabolic response. 61 Following CD19 CAR-T therapy, MTV and TLG were not detectable in the cohort that achieved a complete response, while MTV and TLG decreased significantly in the group that achieved partial response. 61 Another retrospective study by Hong et al showed that early post-treatment average standardized uptake value (SUVavg) and MTV were independent risk factors associated with overall survival and progression-free survival, respectively. 62 Patients with a baseline SUVavg <4.36 had a higher overall survival rate at 1 year (100.0 vs 44.9%, P = .019) than those with SUVavg >4.36. Furthermore, patients with reduced early post-therapy SUVavg (<2.60), MTV (<0.55 cm3), and TLG (<1.54) values had 1-year progression-free survival rates higher than those of the comparison groups (Figure 1). It was also discovered that patients with larger baseline tumor loads had considerably higher cytokine levels and incidence of CRS. 62

Typical [18F]FDG PET/CT radiographic changes after CAR-T therapy in a patient with diffuse large B cell lymphoma. (C1) Lymphoma cells in the abdominal aorta, the right iliac vessels, and the mediastinum showed high metabolic activity before CAR-T therapy; (C2) One month after CAR-T cell infusion, the metabolic activity of the abdominal aorta and the right iliac vessels decreased significantly. (Image reprinted without changes from Hong et al. 62 under the Creative Commons Attribution 4.0 International License (CC BY). https://creativecommons.org/licenses/by/4.0/).
Derlin et al conducted a retrospective clinical study to determine whether an early metabolic response was necessary for remission. 63 Interestingly, a significant decrease in both spleen and lymph node signals was observed between baseline and follow-up PET at one month in patients with unfavorable prognoses (Figure 2). 63 Moreover, higher SUVmax at baseline was associated with neurotoxicity, while MTV and TLG were not. None of the baseline PET metabolic parameters were associated with the development of CRS and other side effects. 63

Metabolism of lymphoid organs and results of CAR-T cell treatment. [18F]FDG metabolically active disease at baseline (PET 1), partial remission in PET 2, and progressive disease in PET 3 (unfavorable outcome) are all visible on the PET/CT (top row). Significant reduction in early spleen activity in terms of absolute quantification (SUVmean) and in relation to liver metabolism (lower row). (Image reprinted without changes from Derlin et al. 63 under the Creative Commons Attribution 4.0 International License (CC BY). https://creativecommons.org/licenses/by/4.0/).
Breen et al retrospectively studied NHL to investigate the prognostic significance of different metabolic and volumetric PET parameters a month after CAR-T and determine patient outcomes. They reported SUVmax ≥ 10 at one month of follow-up after CAR-T infusion in patients with partial response and SUVmax <10 on stable disease. 64 Total MTV > 55, TLG > 250, SUVmax >10, or >2 total lesions had an increased risk of progressive disease. Similarly, total MTV >180, presence of bone or parenchymal disease, SUVmax > 10, single lesion TLG > 245, or >2 total lesions were associated with an increased risk of death 64 (Figure 3).
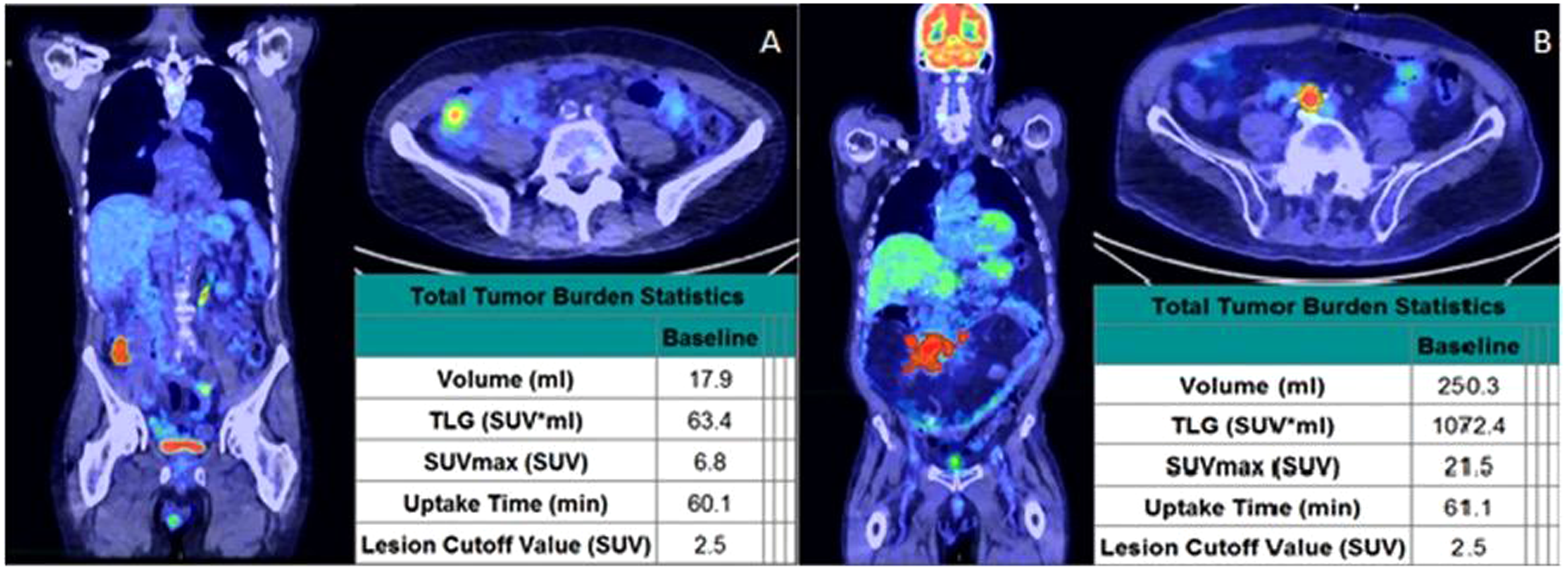
Post-CAR-T PET imaging and analysis at one month. Two patients were found to have a partial response one month after receiving CAR-T therapy. In contrast to the patient on the right (B, SUVmax > 10) who later progressed and ultimately died, the patient on the left (A, SUVmax < 10) eventually achieved complete response. (Image reprinted without changes from Breen et al. 64 under the Creative Commons Attribution 4.0 International License (CC BY). https://creativecommons.org/licenses/by/4.0/).
Furthermore, retrospective analysis of diffuse large B-cell lymphoma (DLBCL) by Cohen et al showed that SUVmax >17.1 at the time of treatment decision was associated with shorter overall survival. 65 Besides, patients with a Deauville score >3 at 1-month post-CAR-T PET/CT had significantly shorter overall survival than patients with a Deauville score ≤ 3. While the baseline SUVmax obtained during the treatment decision or transfusion predicted overall survival, the SUVmax obtained in the first month did not predict overall survival. 65 Notably, baseline PET metabolic parameters did not predict side effects. 65 Table 2 describes the major findings of PET/CT studies for evaluating CAR-T therapy in lymphoma.
Summary of Studies Related to the Use of PET/CT for the Evaluation of CAR-T Therapy in Lymphoma.

Abbreviations: NA, not applicable; CAR-T, Chimeric antigen receptor-T cell; PET/CT, positron emission tomography/computed tomography; CRS, Cytokine release syndrome; NHL, non-Hodgkin lymphoma; MTV, metabolic tumor volume; TLG, total lesion glycolysis.
Immune-Related Toxicities Due to CAR-T Therapy
Chimeric antigen receptor-T cell therapy can cause serious side effects, such as CRS, ICANS, or hemophagocytic lymphohistiocytosis/macrophage activation syndrome.67,68 PET imaging may play a useful role in evaluating CRS and ICANS due to the CAR-T cell therapy. 67 Cytokine release syndrome occurs within 10 days of CAR-T cell transfusion and symptoms usually last up to 10 days. The frequency of occurrence ranges from 42% to 100%, and severe cases happen in up to 46% of patients.68,69 It is the most common complication of CAR-T therapy that can have a mild to life-threatening presentation. 70 In multicenter studies exploring CAR-T therapy for relapsed/refractory aggressive lymphoma, there was a clear association between the severity of CRS and the extent of tumor burden.4,71,72 Recent studies by Hong et al, 62 Dean et al, 73 and Wang et al 74 also demonstrated that a high tumor burden, reflected by high MTV, TLG, and SUVmean measured during screening [18F]FDG PET/CT as a significant risk factor for the development of CRS. However, in contrast to these findings, none of the baseline PET parameters were associated with developing CRS in a study by Derlin et al 63 and Zhou et al. 75 Notably, PET/CT was not performed in any patient during CRS symptoms.
Neurological issues linked to CAR-T therapy, such as ICANS, can manifest with various symptoms, including confusion and coma.9,10 Immune effector cell-associated neurotoxicity syndrome usually show up 1 to 3 weeks after the CAR-T cell transfusion and are severe in up to 50% of cases. It is important to note that CRS and ICANS often occur at the same time and are associated, but can sometimes occur on their own.68,69 Standard imaging techniques such as CT and MRI often appear normal in cases of ICANS. 76 However, brain [18F]FDG PET/CT scans have proven useful in identifying metabolic changes in the brain associated with this condition. These scans have revealed areas of reduced brain metabolism, even in cases where traditional imaging methods did not show abnormalities. Rubin et al demonstrated these findings by studying the neurological effects of CAR-T therapy up to 2 months post-infusion. 77 They examined 6 patients with neurotoxicity and abnormal electroencephalogram results using brain [18F]FDG PET/CT. In 5 cases, areas of reduced brain metabolism on [18F]FDG PET/CT matched electroencephalogram abnormalities. In 3 cases, elevated blood flow velocities aligned with [18F]FDG PET hypometabolism. Importantly, these findings did not have a structural basis and remained unexplained. Paccagnella et al reported a patient with DLBCL with hypometabolism on brain [18F]FDG PET/CT but no abnormalities on MRI. 78 Vernier et al documented a similar case where [18F]FDG PET/CT done on the 14th day of CAR-T transfusion revealed hypometabolism in various regions of the brain while MRI was normal. 79 This hypometabolism appeared to be essential in evaluating CAR-T neurotoxicity.
Application of Artificial Intelligence in CAR-T Therapy for Lymphoma
The advent of immunotherapy has marked a novel era in the field of cancer therapy, highlighting the importance of precision medicine and the evaluation of early response. 54 However, assessing treatment outcomes in immunotherapy poses a challenge due to the rapid development of immunotherapeutic agents, such as various generations of CAR-T cells, and the emergence of atypical responses such as pseudoprogression and hyperprogression. To address these issues, experts have conducted research on innovative approaches for evaluating responses, such as the use of artificial intelligence techniques such as radiomics and machine learning. Keeping up to date with current advances in CAR-T therapy and novel techniques to assess treatment responses may be useful for treating physicians to deliver efficient patient care.44,54,80
Tong et al applied deep learning–based image analysis techniques using pretreatment [18F]FDG PET, diagnostic CT, and low-dose CT, in addition to rule-based reasoning, to predict the response to treatment with CAR-T therapy in B-cell lymphomas. 81 The study utilized transfer learning by initially using a preexisting neural network model, which was subsequently retrained for this particular objective to predict treatment responses at the lesion level, separately for [18F]FDG PET, diagnostic CT, and low-dose CT images. 81 They analyzed 770 lymph node lesions from pretreatment images of 39 adult patients and found that this approach has a high precision in predicting lesion-level treatment responses (accuracy = 0.82), as well as patient-level responses (accuracy = 0.81), outperforming traditional prognostic methods such as the international prognostic index (accuracy = 0.54). 81 This method could be clinically useful for decision making before the initiation of CAR-T therapy.
In the context of CAR-T therapy, there is limited application of artificial intelligence techniques in the evaluation and management of lymphoma, multiple myeloma, and leukemia. However, attempts could be made to replicate studies conducted outside of the CAR-T therapy setting for patients who undergo CAR-T therapy. Milgrom et al evaluated the characteristics of refractory Hodgkin lymphoma in a retrospective study and suggested that the use of a PET radiomic model could enhance initial risk assessment and treatment strategies for patients with this condition. 82 Frood et al demonstrated the utility of pretreatment [18F]FDG PET/CT–derived machine learning models derived from pretreatment [18F]FDG PET/CT for prediction of outcome (2-year EFS) in patients with classical Hodgkin lymphoma who underwent chemotherapy, radiation therapy, or were treated as an advanced disease. 83 Sollini et al developed a methodological framework for applying radiomics to Hodgkin's lymphoma. 84 Their approach involved (1) the introduction of a novel method for selecting characteristics, (2) the examination of the similarity of radiomic characteristics within lesions from the same patient, and (3) distinguishing between patients with relapsing refractory (R / R) Hodgkin lymphoma and those without it. The results indicated that the radiomic features within Hodgkin lymphoma lesions exhibited variability between individual patients, suggesting that the random selection of the lesions should be avoided in radiomics analyses. 84 Additionally, the classifier performed most effectively when considering all lesions to predict the status of R / R versus non-R/R status in patients. 84 Such efforts are currently lacking in the setting of CAR-T therapy, but they would be highly useful regardless. Radiomics could also be applied to differentiate pseudoprogression from hyperprogression in patients undergoing CAR-T therapy, similar to how Barabino et al 85 did for non-small cell lung cancer and Basler et al 86 did for metastatic melanoma treated with immune checkpoint inhibitors.
Role of [18F]FDG PET in CAR-T Therapy for Multiple Myeloma
Recently, the FDA approved the use of B-cell maturation antigen (BCMA) directed CAR-T therapy (idecabtagene vicleucel) in patients with multiple myeloma. 87 B-cell maturation antigen is a tumor necrosis factor receptor family protein expressed only in multiple myeloma (mature B and plasma) cells and allows their prolonged survival.88,89 Although unprecedented results were achieved, the use of BCMA CAR-T was limited due to poor trafficking to tumor sites and CRS as a side effect.28,90-92 To better visualize its trafficking at multiple myeloma sites, assess expansion, and diagnose toxicity, sodium iodide symporter (NIS) has been incorporated into the CAR-T construct as a novel approach.66,93 After incorporating NIS into CAR-T cells, they can be visualized with gamma- or positron-emitting radioisotopes such as technetium-99m (99mTc) pertechnetate, iodide-123 (123I), iodide-124 (124I), iodide-131 (131I), and 18F-tetrafluoroborate ([18F]TFB43). 94 In particular, malignant plasma cells in multiple myeloma are often distributed in the bone marrow or bones while NIS accumulates physiologically in the extramedullary areas; this reduces the possibility of erroneous results.93,95,96 Moreover, studies have shown that NIS in CAR-T cell constructs does not affect CAR-T cell function and efficacy.66,93 Furthermore, nonimmunogenic NIS is suitable for longitudinal imaging studies in multiple myeloma. 97
A single institutional analysis by Wong et al performed in 54 patients with multiple myeloma receiving BCMA-CAR-T therapy recognized serum-free light chains (iFLC) and minimal residual disease testing by next-generation sequencing (MRD-NGS) as novel predictors of progression-free survival following this new therapy. 98 The study analyzed the overall response rate of each patient through iFLC and MRD-NGS. PET/CT evaluation was performed after infusion and between 3 and 6 months. 98 This addition of PET/CT to MRD-NGS allowed for further differentiation of the patients; MRD-NGS and PET/CT negativity were strongly associated with a more favorable progression-free survival. 98
Role of [18F]FDG PET in CAR-T Therapy for Leukemia
Recently, anti-CD19 CAR-T cells have shown potential in treating B-ALL.99,100 However, extramedullary involvement in B-ALL remains a therapeutic challenge due to limitations related to poor response and neurotoxicity.101,102 Further studies are underway to develop a third-generation CAR-T cell vector with the Toll/interleukin-1 receptor domain of Toll-like receptor 2 to target CD19 with the generated 1928zT2T cells.103,104 In a case series, Weng et al found that all 3 patients with relapsed or refractory B-ALL with extramedullary involvement achieved complete remission after 1928zT2T-cells infusion. 103 In this study, PET/CT along with bone marrow smear, B-ultrasonography, histology, flow cytometry, quantitative polymerase chain reaction, enzyme-linked immunoassay, and Luminex assay were used to evaluate clinical responses after therapy. The PET/CT was initially used to diagnose areas of relapse in extramedullary sites (kidney, breast, pleura, peritoneum, pelvic fascia, pancreas, and multiple lymph nodes), which showed a highly intense metabolic mass in the affected region. 103 From 2 months to a year after 1928zT2T cell therapy, PET/CT showed hypometabolic lesions without abnormal signals in all 3 patients. 103 These PET/CT findings were consistent with histological and clinical analyses. Therefore, PET/CT can monitor leukemia relapse and treatment response following CAR-T therapy 103 (Figure 4).

A small dose of 1928zT2T cell infusion eradicated leukemia and induced a complete response in a patient with ALL with extramedullary involvement. (A) Represents the therapeutic process of therapy. (B) PET/CT images—The left breast had an abnormally intensive, highly metabolic mass at the beginning of the study. On day 30, after cell infusion, a repeat PET/CT showed that the lesion had entered a hypometabolic state and no aberrant signal had been seen since. (C) Photograph of the proven biopsy of extramedullary involvement at baseline. (D) Before the cells were infused, B-mode ultrasonography revealed an inhomogeneous hypoechoic mass that was approximately 2.8 × 1.6 cm in diameter. On day 14, the mass had shrunk to 2.3 × 1.1 cm. On day 46 and thereafter, the unusual hypoechoic mass disappeared. (Image reprinted without changes from Weng et al. 103 under the Creative Commons Attribution 4.0 International License (CC BY). https://creativecommons.org/licenses/by/4.0/).
Holland et al reported similar results from more than 180 patients with refractory/relapsed B-ALL. Of these, 38 (21.1%) patients were found to have isolated extramedullary disease (EMD) of the non-central nervous system (CNS) (n = 5) or combined medullary/non-CNS EMD (n = 33), as detected by [18F]FDG PET/CT. 105 Following infusion of CAR-T cells, a subset of 18 patients underwent both preinfusion and post-infusion [18F]FDG PET/CT to monitor response. 105 Among them, 13 patients (72.2%) showed minimal residual medullary disease, that is, negative complete remission with a complete (n = 7) or partial (n = 6) non-CNS EMD response. 105 The study concluded that serial [18F]FDG PET/CT scans were required to identify and monitor non-CNS EMD and patients for remission or disease recurrence after CAR-T therapy. 105
Conclusion
The potential of CAR-T immunotherapy in the treatment of cancer is significant, offering the possibility of complete remission in hematologic malignancies. Noninvasive imaging techniques, such as PET/CT, are beneficial for tracking treatment responses and side effects in patients with hematological malignancies, including lymphoma, multiple myeloma, and leukemia. However, the broad clinical application of PET/CT in CAR-T therapy is currently hindered by the inadequate sensitivity and specificity of the available radiotracers. Improving the efficacy and safety of CAR-T therapy may be possible by developing tumor-specific tracers and establishing standardized threshold values for PET parameters. Consequently, more research into specific radiotracers and new technologies is essential to enhance their sensitivity and specificity to establish PET/CT as the preferred diagnostic method in CAR-T cell therapy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
