Abstract
This study compared Intraneural Facilitation (INF®) therapy and standard physical therapy (PT) in preventing chemotherapy-induced peripheral neuropathy (CIPN) in women with newly diagnosed breast and gynecologic cancer. Thirty-eight women undergoing platinum and/or taxane-based chemotherapy, without prior peripheral neuropathy, were randomized into INF® therapy (n = 20) and PT (n = 18). Treatments lasted 45 minutes, twice weekly, for 6 weeks. Neuropathy severity was evaluated using the Pain Quality Assessment Scale. Assessments were at baseline, 3 weeks, 6 weeks, and 3 months post-intervention. Acceptability, burden, and satisfaction were evaluated after 6 weeks. Among 38 patients, 12 (32%) experienced CIPN, with mean pain scores remaining mild (≤3) and no pharmacotherapy required until week 6. No adverse events were reported from the interventions. The INF® therapy arm showed significant changes in numbness (F = 6.030, P = .001, partial η2 = 0.262) after week 6, while the PT arm showed significant changes in numbness (Z = −2.39, P = .017), tingling (Z = −2.84, P = .004), cramping (Z = −2.120, P = .034), surface pain (Z = −2.75, P = .006), and deep pain (Z = −1.99, P = .046) between weeks 3 and 6. Nearly 80% of patients completed chemotherapy cycles with an average relative dose intensity of 90.4% (INF® therapy: 87.73% vs PT: 73.44%). Ninety-four percent of patients were satisfied with their care, accepted the treatments, and perceived them as a low burden. The results demonstrated that INF® therapy and PT are feasible options for CIPN, improving treatment adherence, outcomes, and quality of life for women with newly diagnosed breast and gynecological cancers.
Keywords
Introduction
Chemotherapy-induced peripheral neuropathy (CIPN) remains a significant challenge, compromising chemotherapy (CT) treatment and the quality of life of patients with active cancer and cancer survivors.1 -6 It occurs from the dose-dependent and cumulative effects of certain commonly used CT agents, 4 affecting 30% to 60% of patients, up to 80% of those treated with taxanes,5,7 -9 and about 47% of women treated for breast cancer 6 years post-treatment. 10 Currently, no effective treatments are available to prevent CIPN, and the recommended standard of care of dose reduction, dose delays, and treatment discontinuation to alleviate neuropathic symptoms impacts patients’ survival and morbidity.2,4,5 Limited evidence supports pharmacologic and non-pharmacologic interventions to treat or prevent CIPN.8,11,12 Nonpharmacologic treatment recommendations include a physical therapy (PT) regimen that combines strength, balance, and aerobic exercises; however, there is a lack of standardization and limited evidence regarding the effectiveness of these exercises on CIPN symptoms.11 -13 Healthcare interventions for CIPN must effectively minimize treatment toxicity, maximize quality of life, and gain acceptance from patients to achieve the best clinical outcomes.14,15 Furthermore, prevention should remain a focus of CIPN management, given the increased survival rates among patients with breast cancer and the unpredictable development of symptoms. 4
Strategies to prevent CIPN should target the mechanism and provide neuroprotection and symptom relief without compromising treatment.6,14 Although the pathophysiology of CIPN is not entirely understood, symptoms of CIPN are predominantly sensory due to the neuronal cell damage in the dorsal root ganglion. Still, motor and autonomic effects can occur with higher doses. 16 Exercises like those used in PT can induce molecular and cellular changes in dorsal root ganglion sensory neurons, leading to nerve regeneration in as quickly as 3 or 7 days. 17 Peripheral nerve revascularization also has a role in nerve regeneration by recruiting macrophages for phagocytosis of axonal and myelin debris and allowing oxygen and nutrients to the damaged nerves to promote nerve regrowth. 18 A manual treatment called Intraneural Facilitation (INF®) therapy aims to increase blood flow to the neural connective tissue, thereby improving microvascular circulation in the neurovasculature and reversing intrafascicular ischemia. 19 A pilot study indicated that improvements in ultrasound measurements after INF® therapy could resolve neuropathy in patients with CIPN. 20 The authors posit that INF® therapy and standard PT may prevent CIPN symptoms during CT treatment, thus improving treatment adherence, clinical outcomes, and the quality of life of patients with cancer and survivors. Therefore, we conducted a randomized clinical trial aimed at evaluating INF® therapy and a standardized PT program as a preventative treatment for CIPN in patients with newly diagnosed breast or gynecologic cancer.
Methods
Design and Setting
This study was a 2-arm randomized controlled trial (Figure 1) comparing 2 treatment modalities. Patients were screened for eligibility through patient demographics and clinical information, including Eastern Cooperative Oncology Group (ECOG) scoring, tumor characteristics, previous treatment history, current medication use, and clinical staging per the eighth edition of the American Joint Committee on Cancer (AJCC 8th). Pathologists confirmed all histopathological specimens. Consented patients were randomly assigned to receive either INF® therapy (Arm 1) or a standardized PT program (Arm 2), with the first treatment occurring within 3 weeks of starting CT. A trained physical therapist (physical therapist #1) provided 45-minute treatments for both groups twice weekly for 6 weeks. A second physical therapist (physical therapist #2) assessed patients at baseline, week 3, week 6-, and 3 months post-intervention. Both physical therapists were experienced professionals with extensive training in INF® therapy, including more than 240 hours of supervised practice and advanced education in anatomy, physiology, and therapy protocols. All assessments and treatments were performed under the same conditions and in the same treatment rooms.

Consort flowchart.
Participants
A convenience sample was recruited through study advertisement posters, brochures, and clinical referrals. Eligible patients included females aged 18 years or older with newly diagnosed or previously untreated breast cancer stages I to III (AJCC eighth) with any biomarker status (ER, PR, and HER2), ECOG score of 0 or 1 and planned to receive a minimum of 4 cycles of treatment with neoadjuvant or adjuvant platinum-based (Carboplatin and Cisplatin) and/or taxane (Docetaxel and Paclitaxel) CT. Eligibility criteria were amended to include gynecologic cancers receiving treatment with the same CT agents in the curative setting, including ovarian, uterine, and cervical cancer, after the trial commenced to increase enrollment.
Patients were excluded if they had previous exposure to any of the implicated CT agents (platinum-based compounds, vinca alkaloids, and taxanes), metastatic breast or gynecologic cancers, history of significant comorbidities including medical or psychiatric illnesses that would make the patient ineligible for the study based on the investigator’s judgment, any physical or neurological disability that would preclude active participation in physical activity, prior malignancy within the past 5 years, or pre-existing peripheral neuropathy and use of pharmacologic intervention for pre-existing peripheral neuropathy. Loma Linda University’s Institutional Review Board (IRB# 5170197) approved all methods and procedures before recruitment and implementation, and all subjects gave informed consent.
INF® Therapy and Standard PT Program Interventions
INF® therapy is a technique that uses manual holds and stretches to increase epineural blood flow. 19 The first is the facilitation hold, which puts the patient’s contralateral joint in a comfortable maximal loose-pack position. 19 This stretched position is sustained throughout the entire treatment session. The second hold is believed to bias the increased epineural blood flow past the perineurium into the endoneurial capillaries, thus increasing the blood flow to the affected neural structures. 19 A third stretch, involving gentle pressure on an artery, a rib, or skin, brings circulation through the inflamed capillaries. 19 Each hold lasts approximately 2 to 5 minutes, with an average of 9 to 12 targeted areas. A hold or position starts and stops when the therapist sees improvement in muscle tone, skin color, and swelling.
The standard PT program consisted of walking, muscle strengthening, and stretching exercises known to improve CIPN symptoms.11,12 Physical therapist #1 supervised walking gait activities for 15 minutes twice weekly while monitoring breathing, pulse, and blood pressure. Lower extremity strengthening included standing knee bends, toe rises, and standing hip extension, abduction, and adduction with or without ankle weights. Stretching exercises comprised of calf and hamstring stretches. Balance exercises included single leg standing and challenging patients, standing with 1 foot in front of the other and standing with feet next to each other with eyes open and closed. Progression with these exercises was as the patient tolerated.
Outcome Measures
The primary outcome was to compare the degree of peripheral neuropathy within and between Arm 1 receiving INF® therapy and Arm 2 receiving standard PT (or usual care) based on scores from the Pain Quality Assessment Scale (PQAS). Secondary outcomes were to compare: (1) the rate of dose reductions, delays, and premature treatment discontinuations due to CIPN, (2) the CT completion rates and relative dose intensities, (3) the pharmaceutical use in patients with CIPN, and (4) the acceptability, burden, and satisfaction with the assigned treatment modality.
Pain Quality Assessment Scale
The PQAS is a 20-item questionnaire that assesses the severity and quality of neuropathic pain. The PQAS evaluates 2 global domains (pain intensity and unpleasantness), 2 spatial domains (deep and surface), and 3 quality domains, comprised of paroxysmal (the average scores of shooting, sharp, electric, hot, and radiating), surface (the average scores of itchy, cold, numb, sensitive, and tingling), and deep (the average scores of aching, heavy, dull, cramping and throbbing) subscales for neuropathic pain. 21 Patients rate the severity of their neuropathic pain on a Likert scale ranging from 0 = “no pain” or “no painful sensation” to 10 = “worst imaginable pain sensation” for each of the 20 questions. 21 Pain scores ranging from 1 to 3 were considered mild, 4 to 6 were moderate, and 7 to 10 were severe. Lower pain scores indicate a better quality of life. 21 Pain was assessed at baseline, weeks 3-, 6-, and 3 months post-intervention.
Chemotherapy Treatment Adherence
The Cancer Center clinical trials unit managed CT treatment regimens, doses, and pharmacotherapy use for CIPN. Multiple data points were used to compare CT adherence for each treatment intervention. Data was collected on the number of participants with CT dose reductions, delays, or treatment discontinuation, reasons for dose changes, number of treatment cycles planned and received, cumulative and planned CT dosing, and use of neuropathy and pain medications during clinic visits at baseline, weeks 3-, 6-, and 3-months post-intervention. The number of dose reductions included patients receiving a smaller CT dose at any time during their treatment and missed doses from patients where CT treatment was held due to CIPN. A dose delay was defined as when a patient received the held dose. Patients were deemed to have completed their CT treatment if they finished the intended number of treatment cycles, regardless of whether they received the full planned dose of their CT regimen. Relative dose intensity (RDI) was calculated by dividing the delivered total dose by the planned total dose. 22 RDI is an accepted tool for evaluating the treatment efficacy of CT administration in the curative setting. 22 An RDI of 85% or higher is widely recognized to improve patient outcomes and survival. 22
Satisfaction, Acceptability, and Burden
Objective measures such as withdrawal rates, adverse events, and self-reported measures from an investigator-developed 9-item survey determined acceptance, satisfaction, and burden. Acceptability reflects the patient’s perception of the effectiveness of the intervention and the likelihood they would continue treatment based on its effectiveness. 15 Satisfaction was defined as the patient’s perception of the care with the assigned treatment modality they received. Burden reflects the patient’s perception of the treatment in terms of time, effort, and difficulty that may impact a patient’s well-being and overall quality of life. 15 Patients rated each item on a Likert scale of 1 to 5, with 1 = “Strongly Disagree” and 5 = “Strongly Agree.” Patients completed the surveys following week 6’s treatment (Supplemental Material 1).
Randomization and Blinding
Subjects were randomized in a 1:1 ratio to one of the treatment arms by the Cancer Center clinical trials unit. The randomization process consisted of selecting 1 of 40 thoroughly shuffled sealed envelopes. Each envelope held a card concealed from the participants; 20 were marked with INF® treatment, and 20 with standardized PT treatment. Subjects drew an envelope from the shuffled pile to determine their treatment assignment. Given the nature of the interventions, it was impossible to blind patients, physicians, or the treating physical therapist to the group assignment. However, the physical therapist #2 who performed the assessments and the statistical analyst responsible for data analysis were blinded.
Sample Size and Statistical Methods
Mean and standard deviation were computed for quantitative variables (demographic and clinical characteristics, RDI, and patient satisfaction survey data) and frequency (percentage) for categorical variables (demographic and clinical characteristics, clinical data including CT dose reductions, dose delays, or treatment discontinuations, CT regimens, completion of CT treatment cycles, and pharmaceutical use). The normality of quantitative variables was assessed using the Shapiro-Wilk test and box plots. The Mann-Whitney U test was used for non-normal and ordinal data. The independent chi-square test or Fisher’s Exact test was used for categorical variables at baseline. Linear mixed effects models (repeated measures) were used to examine the effect of the between-group factor (INF® therapy vs standard PT) and within-group factor (time: baseline, 3 weeks, 6 weeks, and 3 months) on the dependent variables (pain quality descriptors and subscales from the PQAS: sharp, hot, dull, cold, sensitive, tender, itchy, shooting, numbness, electrical, tingling, cramping, radiating, throbbing, aching, heavy, intense, unpleasant, intense-deep, intense-surface, paroxysmal subscale, surface subscale, and deep subscale). 23 A Bonferroni correction was used to adjust for multiple post-hoc comparisons. Additional pairwise comparison tests using the Wilcoxon Signed-Rank test compared repeated within-subject measures due to non-normal data distribution. 24 Non-significant changes were interpreted as preventative. A power calculation was performed using G*Power (Heinrich-Heine Universität, Düsseldorf, Germany) with a similar method previously reported. 13 A minimum sample size of n = 36 was required to provide 80% power at the 5% significant level to capture a small effect size of 0.20 or higher. The data was analyzed using SPSS Statistics Software version 29.0 (SPSS, Inc, Chicago, IL, USA). All analyses were performed at an alpha level of .05.
Results
Sample Characteristics
Recruitment occurred from July 2017 to July 2022. One hundred four patients were initially screened. Forty-four patients were eligible and randomly assigned to 1 of the 2 treatment arms, an INF® therapy group (n = 23) or a standardized PT program (n = 21) group. Thirty-eight female patients were included in the final analysis: 18 in the standard PT arm and 20 in the INF® therapy arm (Figure 1).
Each arm’s study population’s characteristics are displayed in Table 1. The majority of patients were Non-Hispanic White/Caucasian (n = 17, 45%), followed by Hispanic (n = 10, 26%), Asian (n = 8, 21%), and Black (n = 3, 7.9%). Most patients had a baseline BMI under 30 (n = 26, 68%), an ECOG score of 0 (n = 23, 61%), breast cancer with hormone receptor (HR) status HR+/HER2+ and HR+/HER2- (n = 30, 79%), and received CT in the neoadjuvant setting (n = 26, 68%). The gynecologic patient in the INF® therapy arm received CT in the perioperative setting. The 2 arms had no significant differences in baseline demographic and medical characteristics, confirming effective randomization.
Patient Demographic and Medical Characteristics.

Abbreviations: ECOG, Eastern Cooperative Oncology Group; HR, hormone receptor; Taxol, Paclitaxel; Carbo, Carboplatin; Taxotere, Docetaxel; INF® therapy, intraneural facilitation; CT, chemotherapy.
Mann-Whitney U-test was used to compare continuous variables; Chi-Square tests were used to compare categorical variables. Fischer’s exact test was used when groups were less than 5.
Chemotherapy Regimens included: AC-Taxol: Adriamycin + Cyclophosphamide × 4 followed by weekly Taxol (Paclitaxel) 80 mg/m2 × 12 cycles; TCHP: Taxotere (Docetaxel) 75 mg/m2 + Carboplatin AUC 6 + Herceptin (Trastuzumab) + Perjeta (Pertuzumab) every 3 weeks × 6 cycles; TC: Taxotere 75 mg/2 (Docetaxel) + Cyclophosphamide every 3 weeks × 4 to 6 cycles; Taxol + Herceptin: Taxol 80 mg/m2 weekly × 12 + Herceptin (Trastuzumab); Taxol + Carboplatin: Taxol (Paclitaxel) 80 mg/m2 weekly × 12 + Carboplatin AUC 1.5 to 2 × 12 weeks OR Taxol (Paclitaxel) 80mg/m2 weekly × 12 + Carboplatin AUC 5 every 3 weeks × 4 OR Taxol (Paclitaxel) 175 mg/m2 weekly × 8 + Carboplatin AUC 5 every 3 weeks × 8 (Gyn cohort); AC-Taxol + Carboplatin: AC × 4 followed by weekly Taxol (Paclitaxel) 80 mg/m2 × 12 cycles with Carboplatin AUC 5 every 3 weeks; Taxotere + Carboplatin: Taxotere (Docetaxel) 75 mg/m2 + Carboplatin AUC 6 q21 days × 6 cycles.
The chemotherapy regimens have been grouped into 4 groups based on the use of taxanes: taxol, taxol and carboplatin, taxotere, taxotere and carboplatin.
Data are presented as frequency (%) for categorical variables and mean ± standard deviation for continuous variables.
The INF® therapy arm has 19 hormone receptor statuses represented given the 1 patient with gynecologic malignancy.
The INF® therapy arm has 1 gynecologic patient that received CT in the neoadjuvant setting.
Pain Quality Assessment Scale
Pain Quality Descriptors
The results of the Pain Quality Assessment Scale questionnaires are presented in Supplemental Material 2. Mean pain quality scores were classified as mild (less than 3) at each time point in both treatment arms throughout the study period. A two-way repeated measures ANOVA was conducted to evaluate the impact of time on pain quality descriptor scores. A significant change in numbness was observed in the INF® therapy arm (F = 6.030, p = .001, partial η2 = 0.262). Post-hoc pairwise comparisons with a Bonferroni adjustment indicated a significant difference between the numbness pain quality scores at baseline and 3 months (P = .028), as well as between 3 weeks and 3 months (P = .032). In contrast, no significant change was noted in the standard PT arm (F = 1.117, P = .341, partial η2 = 0.062) over time (Supplemental Material 2 and Figure 2). Additionally, no significant differences were found between the 2 groups (P = .794) for numbness (Supplemental Material 2). No other significant differences were identified within or between the 2 groups over time for any other pain quality descriptor measures (Supplemental Material 2). Additionally, the Wilcoxon Sign-Rank Test was run for the pairwise comparison and indicated that pain quality descriptor scores were significantly higher in week 6 compared to week 3 for numbness (Z = −2.39, P = .017), tingling (Z = −2.84, P = .004), and cramping (Z = −2.120, P = .034) in the standard PT arm. Numbness pain quality scores were also significantly higher at 3 months post-intervention than week 6 (Z = −2.19, P = .029) in the INF® therapy arm. No other significant differences were found within the group pain quality descriptor scores (Supplemental Material 2).

Repeated measures ANOVA graph of numbness pain quality scores over time.
Global Domains: Intensity and Unpleasantness
In the global domains, both arms showed an initial decrease in pain intensity and unpleasantness from baseline to week 3, then an increase between week 3 and week 6, and a decrease from week 6 to 3 months post-intervention (Supplemental Material 2 and Figure 3A and B). Two-way repeated measures ANOVA was performed to evaluate the effect of time on global domains (intense and unpleasantness). No significant changes were found for intense pain in the standard PT arm (F = 0.767, P = .518, partial η2 = 0.043) or the INF® therapy arm (F = 0.176, P = .826, partial η2 = 0.010) over time. No significant changes were found for unpleasant pain in the standard PT arm (F = 0.589, P = .625, partial η2 = 0.034) or the INF® therapy arm (F = 0.903, P = .446, partial η2 = 0.050) over time. No significant differences were found between the 2 groups for intense (P = .255) or unpleasant (P = .506) pain scores over time (Supplemental Material 2 and Figure 3A and B). The Wilcoxon Sign-Rank Test was run for the pairwise comparison and no significant differences were found within groups at each time interval for intense or unpleasant pain (Supplement Material 2).

Repeated measures ANOVA graphs of global (A and B) and spatial domain (C and D) pain scores over time. Global domains: intensity (A), unpleasantness (B), and spatial domains, intense-deep (C), and intense-surface (D).
Spatial Domains: Intense-Deep and Intense-Surface
Two-way repeated measures ANOVA was performed to evaluate the effect of time on spatial domains (intense-deep and intense-surface). No significant changes were found for intense-deep pain in the standard PT arm (F = 1.211, P = .308, partial η2 = 0.067) or the INF® therapy arm (F = 0.858, P = .469, partial η2 = 0.048) over time. No significant changes were found for intense-surface pain in the standard PT arm (F = 0.258, P = .855, partial η2 = 0.015) or the INF® therapy arm (F = 0.831, P = .483, partial η2 = 0.047) over time. No significant differences were found between the 2 groups for intense-deep (P = .443) or intense-surface (P = .195) pain scores over time (Supplemental Material 2 and Figure 3C and D). The Wilcoxon Sign-Rank Test was run for the pairwise comparison and no significant differences were found within groups at each time interval for intense-deep or intense-surface pain (Supplemental Material 2).
Quality Domains: Paroxysmal, Surface, and Deep
Average baseline measures for the paroxysmal, surface, and deep neuropathic pain quality domains were higher in the standard PT arm than the INF® therapy arm and remained higher overall during the 6-week intervention (Supplemental Material 2 and Figure 4). Paroxysmal pain scores in the INF® therapy arm were stable, gradually decreasing until the intervention was stopped at week 6. In contrast, the standard PT arm initially declined, peaked at week 6, and then reduced (Figure 4A). No significant changes were found for paroxysmal pain in the standard PT arm (F = 0.645, P = .542, partial η2 = 0.037) or the INF® therapy arm (F = 0.239, P = .723, partial η2 = 0.014) over time. No significant differences were found between groups for paroxysmal pain (P = .173) over time (Supplemental Material 2 and Figure 4A). The Wilcoxon Sign-Rank Test was run for the pairwise comparison and no significant differences were found within groups at each time interval for paroxysmal pain (Supplemental Material 2).

Repeated measures ANOVA graphs of quality domains: paroxysmal (A), surface (B) and deep (C) pain scores over time.
Surface pain scores were stable for both interventions until week 3, then the standard PT arm showed a sharp increase in pain, peaking at week 6 when the intervention was stopped, while the INF® therapy arm showed a gradual increase through 3 months post-intervention (Figure 4B). No significant changes were found for surface pain in the standard PT arm (F = 1.155, P = .324, partial η2 = 0.064) or the INF® therapy arm (F = 2.490, P = .091, partial η2 = 0.135) over time. No significant differences were found between groups for surface pain (P = .404) over time (Supplemental Material 2 and Figure 4B). The Wilcoxon Sign-Rank Test indicated that surface pain (Z = −2.75, P = .006) was significantly higher in week 6 than week 3 within the standard PT arm (Supplemental Material 2). No other significant differences were found within groups at each time interval for surface pain (Supplemental Material 2).
Deep pain scores show stability in the INF® therapy arm until the intervention was stopped at week 6, while the standard PT arm fluctuated between treatment periods (Figure 4C). No significant changes were found for deep pain in the standard PT arm (F = 1.133, P = .335, partial η2 = 0.062) or the INF® therapy arm (F = 1.137, P = .605, partial η2 = 0.027) over time. No significant differences were found between groups for deep pain (P = .244) over time (Supplemental Material 2 and Figure 4C). The Wilcoxon Sign-Rank Test showed that deep pain (Z = −1.99, P = .046) was significantly higher in week 6 than week 3 in the standard PT arm (Supplemental Material 2). No other significant differences were found within groups at each time interval for deep pain (Supplemental Material 2).
Rate of Dose Reductions, Delays, and Treatment Discontinuation due to CIPN
Table 2 summarizes the characteristics and clinical data of the patients who developed CIPN. The 12 (32%) patients who developed CIPN were diagnosed with breast cancer, primarily with HR+/ HER2 negative tumors, and received neoadjuvant CT. While there were no treatment delays, 11 out of 38 patients (28.9%) had dose reductions or discontinued CT treatment due to CIPN (Table 2). Fewer treatment changes were observed in the INF® therapy arm, with no modifications occurring until after the intervention stopped at week 6, compared to treatment changes noted as early as week 3 in the standard PT arm (Table 2). A Fisher’s exact test showed no significant association between treatment groups and the number of treatment changes by time period at week 3 (P = 1.0), week 6 (P = .182), or 3 months (P = 1.0; Table 2). Two patients (10%) in the INF® therapy arm and 3 (16.6%) in the standard PT arm discontinued CT treatment prematurely. No serious adverse events were reported with the assigned interventions, and no patients withdrew from the study due to dissatisfaction with the intervention received. One patient in the INF® therapy arm chose to withdraw at the end of the 6-week intervention to continue INF® therapy due to worsening CIPN. Other reasons for dose adjustments and discontinuation were all related to CT side effects, including patient preference (n = 1), tinnitus (n = 1), fatigue and weakness (n = 2), transaminitis (n = 2), hearing loss (n = 1), decreased creatinine clearance (n = 1), poor treatment tolerance (n = 1), nail infection (n = 1), and port placement (n = 1). Ten out of 11 patients (91%) who required CT dose reduction or discontinuation due to CIPN received a taxol-based CT regimen.
Characteristics and Clinical Data of Patients that Developed CIPN.

Abbreviations: ECOG, Eastern Cooperative Oncology Group; HR, hormone receptor; Taxol, Paclitaxel; Carbo, Carboplatin; Taxotere, Docetaxel; CIPN, chemotherapy-induced peripheral neuropathy; INF® therapy, intraneural facilitation; CT, chemotherapy.
Mann-Whitney U-test used to compare continuous variables; Chi-Square for Independence and Fischer’s exact tests were used to compare categorical variables. Kruskal-Wallis H test was used to compare the treatment changes across treatment groups.
Data are presented as n (%) for categorical variables and mean ± standard deviation for continuous variables.
CT treatment changes represent the number of patients that had dose reductions or discontinuations due to CIPN. No patients had dose delays.
CT Completion Rates and Relative Dose Intensities
CT completion rates and relative dose intensities are summarized in Table 3. A total of 30 patients (78.9%) completed their planned CT treatment cycles, receiving an average RDI of 90.4% (SD 14.37), with 76% (n = 29) meeting the recommended threshold of 85% or higher (Table 3). Of the 8 patients who did not complete their treatment cycles, 3 were in the INF® therapy arm, citing reasons such as patient preference (n = 1), CIPN and worsening tinnitus (n = 1), and CIPN only (n = 1). In the standard PT arm, 5 did not complete their planned CT treatment cycles due to transaminitis (n = 1), poor treatment tolerance (n = 1), fatigue and CIPN (n = 1), and CIPN only (n = 2). The mean RDI in the INF® therapy arm was higher overall (INF 92.76% vs PT 87.95%) and across the taxane-based CT regimens, except the taxotere and carboplatin combined regimen (Table 3 and Figure 5). Among patients with CIPN, 7 out of 12 (58%) completed their planned treatment cycles with dose reductions (n = 6) or support of neuropathy medication (n = 1; Table 3). Patients with CIPN also received a numerically higher % RDI in the INF® therapy arm (87.73%) compared to the standard PT arm (73.44%; Table 3 and Figure 6). Figure 6 illustrates that the mean RDI in the INF® therapy arm is more consistent, indicating greater reliability than the standard PT arm, which shows more variability. Furthermore, the median RDI of the INF® therapy arm is higher than that of the standard PT arm and exceeds the 85% RDI threshold. The skew to the left shows that more patients in the INF® therapy arm (n = 4, 67%) received an RDI of 85% or higher than those in the standard PT arm (n = 3, 50%). The RDI decreased by 14.5% in the standard PT arm and 5.0% in the INF® therapy arm due to CIPN. No significant associations were found between treatment type and completion rates (Fisher exact test, P = .438), mean RDI scores between groups (Mann-Whitney U, U = 172.50, P = .815), or mean RDI scores between groups for those that experienced CIPN (Mann-Whitney U, U = 7.0, P = .078; Table 3 and Figure 6).
CT Completion Rates and Relative Dose Intensities.

Abbreviations: INF® therapy, intraneural facilitation; RDI, relative dose intensity; CT, chemotherapy; CIPN, chemotherapy-induced peripheral neuropathy.
Mann-Whitney U-test used to compare continuous variables; Chi-Square tests were used to compare categorical variables. Fischer’s exact test was used when groups were less than 5. ANOVA tests were used to compare %RDI grouped by taxanes.
Data are presented as frequency (%) for categorical variables and mean ± standard deviation for continuous variables.
The chemotherapy regimens have been grouped into 4 groups based on the use of taxanes: taxol, taxol and carboplatin, taxotere, taxotere and carboplatin.
Relative Dose Intensity was calculated as Planned/Given Chemotherapy Dose.
% RDI calculation includes all CT regimens, CT regimens with multiple agents were averaged.
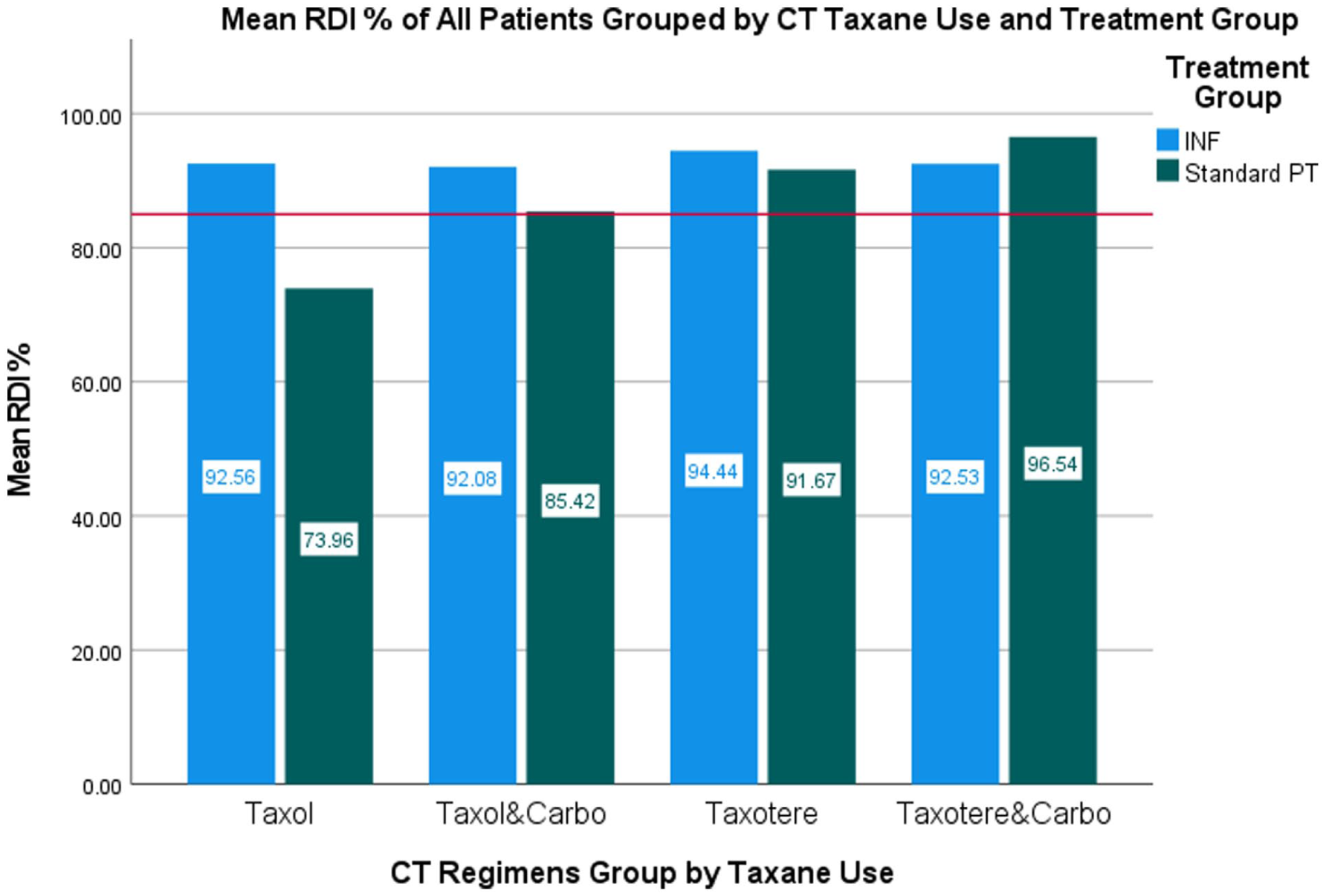
Mean percentage RDI of all patients grouped by taxane use and treatment group.

Mean percentage RDI of patients with CIPN by treatment group.
Pharmaceutical Medication Use
In this study, no patients required opioids, but 6 out of 12 patients (50%) received neuropathy medication for CIPN (Table 4). Gabapentin was the most prescribed, with 1 patient in the standard PT arm also taking Duloxetine, Pregabalin, Venlafaxine, and Nortriptyline. No patients in either treatment arm received neuropathy medications until week 6, when the interventions were stopped. After week 6, a higher percentage of patients in the standard PT arm (n = 4, 67%) were taking neuropathy medications to continue CT treatment with dose-related changes compared to the INF® Therapy arm (n = 1, 17%; Table 4). Data is unavailable after week 6 for the patient who withdrew from the INF® therapy arm to continue INF® therapy when the intervention ended. A Fisher’s exact test did not indicate significant associations between treatment groups and neuropathy medication use (P = .248) or treatment groups and patients taking neuropathy medications at week 6 (P = .387), end of CT treatment (P = .303), or 3 months (P = .887; Table 4).
Pharmacologic Intervention for CIPN.

Abbreviations: CIPN, chemotherapy-induced peripheral neuropathy; INF® therapy, intraneural facilitation.
Chi-squared test or Fisher’s exact tests use to compare categorical variables.
Data are presented as n (%) unless otherwise noted.
One patient withdrew from the INF® therapy arm when the intervention ended at week 6 to continue INF® therapy; data after week 6 for this patient is unavailable.
Treatment Satisfaction, Acceptability, and Burden
Figure 7 shows that over 94% of patients in both intervention groups were satisfied with their treatments, rating them a 4 or 5 out of 5. Patients found the department easy to work with, parking convenient, and testing manageable, and they reported that treatments were appropriately timed and worth the effort. While average scores for patient satisfaction, acceptability, and burden were similar between the 2 groups, more patients reported that INF® therapy effectively reduced nerve discomfort, did not cause unpleasant symptoms, and treatment length and timing between treatments were appropriate compared to standard PT (Table 5). A Mann-Whitney U test indicated that the INF® therapy arm did not perceive progress testing as a significant burden compared to those in the standard PT arm (Mann-Whitney U, U = 113.0, P = .024). No other significant differences were found in patient-reported outcomes between the 2 interventions (Table 5).

Percentage of satisfaction survey scores rated agree or strongly agree.
Patient-Reported Outcome Survey Data of Treatment Satisfaction, Acceptability, and Burden.

Abbreviations: INF® therapy: intraneural facilitation.
Calculated with the use of the two-tailed Mann-Whitney Test.
P-value was significant at the .05 level.
Data are presented as mean ± standard deviation.
Discussion
Interpretation
Cancer treatment drugs, like platinum-based, vinca alkaloids, and taxanes, can damage peripheral sensory and motor neurons, resulting in disabling symptoms such as burning and shooting pain, numbness, and impaired fine motor skills.6,8 Currently, there are no effective methods to prevent CIPN, and the standard of care, which includes dose reduction, dose delays, and treatment discontinuation for alleviating its symptoms, can negatively affect a patient’s survival and overall well-being.2,4,5 This study presents a novel nonpharmacological alternative, INF® therapy, for preventing and managing CIPN. Key findings indicate that both INF® therapy and standard PT are effective treatments for CIPN in patients with early-stage breast and gynecologic cancers undergoing CT. CIPN is a significant burden, with a prevalence rate of 68% within 1 month of CT and 61.5% among patients with breast cancer treated with paclitaxel.6,9 This study reported a nearly 50% reduction in the prevalence of CIPN, with 32% of patients developing it while receiving CT treatment with taxanes.
Consistent with systematic reviews and meta-analyses, the present study demonstrates that combining muscle strengthening and balance exercises improves neuropathic pain, physical function, and quality of life.11,12,25,26 However, there are conflicting reports on the effectiveness of exercise in preventing CIPN symptoms.11,12,26 In our study, the standard PT group showed symptoms typical of CIPN, with increased numbness, tingling, and cramping between weeks 3 and 6 and variable paroxysmal, surface, and deep pain fluctuating over time and decreasing after week 6. Since CIPN symptoms usually appear within the first 2 months of treatment, worsen with each cycle, may stabilize, and then improve once treatment ends,25,27 it’s plausible that the standard PT arm may be more susceptible to the increasing intensity of the CT agents and the cumulative effects of the CT doses. These findings support earlier research that, although exercise may not prevent CIPN symptoms like numbness and tingling, it can lessen their severity and help maintain a patient’s quality of life.13,28
In comparison, the INF® therapy arm demonstrated greater consistency in preventing neuropathy pain and symptoms, requiring fewer doses and delaying the use of neuropathy medication until after the intervention ended at week 6. This finding is supported by the stable trends observed in paroxysmal, surface, and deep pain associated with INF® therapy. In contrast, patients receiving standard PT began using neuropathy medication in week 3. These observations align with the onset of CIPN symptoms and the necessary treatment adjustments in the standard PT arm during the study period. Consequently, INF® therapy may be more effective than standard PT at preventing symptoms and pain during CT. Given that impaired vascularization is implicated in toxic and diabetic peripheral neuropathies, 18 INF® therapy might prevent symptoms by enhancing peripheral nerve vascularization in the intraneural vascular system, supplying oxygen to peripheral nerves damaged by CT. This revascularization could also account for the slight rise in intense-surface pain scores from baseline to week 3, as increased blood flow moves toward the neural connective tissue.
The mild pain, lack of opioid use, and limited reliance on neuropathy medications in this study support findings from other studies that therapeutic exercises can effectively manage CIPN-related pain. 11 Although both groups increased their medication use after the interventions ended at week 6 to the end of CT treatment, pain intensity and unpleasantness remained mild and even decreased despite the onset of symptoms post-intervention. The rise in medication use likely resulted from ongoing CT treatment after the interventions ended, underscoring the importance of continued therapy to mitigate the toxicity of CT agents. Without treatment, patients may develop persistent or worsening symptoms post-CT treatment, known as the “coasting phenomenon.”3,5,27 A systematic review reported that 11 to 80% of patients with early-stage breast cancer experienced persistent neuropathic symptoms for 1 to 3 years after CT treatment. 29 In this study, activity levels were not tracked after week 6, so the role of these interventions in maintaining these effects warrants further investigation. Current guidelines recommend that exercise programs for CIPN last at least 10 weeks. 11 However, regimens involving exercises 3 times weekly for up to 4 weeks post-CT treatment are associated with improved CIPN symptoms and higher CT completion rates.13,30 Interestingly, nerve fibers appear almost normal in a supportive microenvironment 3 months after Wallerian degeneration, 18 implying that a program lasting at least 12 weeks after CT may be beneficial in preventing CIPN symptoms, including the coasting phenomenon.
Completing the planned CT regimen improves patient outcomes, including increased treatment efficacy and survival rates. 22 In this study, about 80% of patients completed their planned CT treatment cycles, achieving an average of 90.48% RDI, with 76% reaching the 85% RDI threshold. The high overall RDI suggests that INF® therapy and standard PT support treatment tolerance and compliance with a prescribed CT regimen. This aligns with findings from Bland et al, 13 which indicated that exercise can improve tolerance of taxane treatment despite worsening symptoms, allowing patients to achieve an RDI of 85% or above. However, RDI can be influenced by dose reductions, delays, and discontinuations. 31 In this study, there were no dose delays, and only 28.9% required dose reductions or discontinuation of CT treatment. This reflects an 8.4% improvement from Speck et al’s 32 retrospective study of 488 women with breast cancer who received docetaxel or paclitaxel, where more than a third (n = 56, 37.3%) of patients experienced dose delays, dose reductions, or treatment discontinuations due to CIPN. The lower prevalence rates, absence of dose delays, fewer dose reductions, and fewer treatment discontinuations contributed to high CT treatment adherence and RDI.
For patients who developed CIPN in the standard PT arm, the average RDI was comparable to Bhatnagar et al’s 33 retrospective study, which reported a median RDI of 73.4% in newly diagnosed patients with breast cancer treated with taxane-based neoadjuvant or adjuvant CT without any interventions. In contrast, the INF® therapy arm had a higher average RDI of 87.73% compared to those who received standard PT or no intervention. The difference in average RDIs between the 2 study interventions approached significance. More patients in the INF® therapy arm also achieved an RDI of 85% or higher, regardless of their CT treatment regimen or increasing symptoms. These findings indicate that INF® therapy enhances both treatment adherence and effectiveness during CT. This improvement is likely attributed to its consistent management of dose-limiting toxicities related to CT, evidenced by fewer dose reductions and a delayed onset of treatment adjustments. Therefore, this study indicates that a 6-week, twice-weekly regimen of INF® therapy or standard PT can reduce prevalence, improve treatment adherence, and prevent CIPN symptom onset, pain, and reliance on pharmaceuticals. Still, longer treatment durations or a home exercise program may offer additional benefits, resulting in sustained relief from CIPN and improved patient outcomes such as higher CT completion rates and RDI.
Research shows that patients undergoing CT treatment often struggle to adhere to exercise programs due to factors such as activity tolerance, cancer stage, co-existing health conditions, and severity of CIPN.12,34 Low-intensity exercises can effectively manage CIPN, particularly for those unwilling or unable to participate in more rigorous exercise programs. 30 This study included low-intensity movements suitable for diverse patient populations and abilities. No adverse events related to the interventions were reported, and patients in both groups expressed satisfaction with their care and viewed it as a low-burden treatment modality. The interventions were well-tolerated and accepted by patients, suggesting they would adhere to the prescribed treatments. Similar positive experiences were reported in Kleckner et al’s 28 secondary analysis of all cancer types, where 94% of the exercise group would recommend an exercise program of walking and strength training while receiving CT. While not statistically significant, more patients in the INF® therapy arm reported decreased nerve discomfort, that the treatment did not cause unpleasant symptoms, and that the treatment length and timing were appropriate compared to standard PT. These findings further reinforce the positive observations of this study regarding INF® therapy, suggesting it is an acceptable treatment option for CIPN.
Limitations and Generalizability
This study has several limitations. First, we compared INF® therapy with an active treatment control, as a sham group was not feasible due to ethical concerns and the novel nature of the therapy. Consequently, we cannot exclude the possibility of a placebo effect influencing symptom improvement. Despite this, our study is one of the few prospective analyses on the prevalence and progression of CIPN symptoms, treatment adherence, and medication use. It establishes a standardized PT protocol and introduces an alternative nonpharmacological treatment to prevent CIPN. Second, the intervention period lasted 6 weeks, while the CT treatment protocols varied from 6 weeks to 3 months. Although assessing long-term effects poses challenges, our findings provide valuable insights for future practice and research. Third, we did not consider baseline activity levels or control for physical activity outside the intervention, which may have affected results. Finally, although a larger sample size could yield more pronounced differences between treatments, our study included a diverse population, offering valuable insights and replicable methods for other cancer diagnoses and demographics.
Conclusions
INF® therapy and standard PT could be used as feasible nonpharmacological treatment options to prevent and manage CIPN. In this study, there was a 50% reduction in the prevalence of CIPN compared to prior studies. Both treatment modalities were well-tolerated and positively impacted CT adherence by controlling pain and alleviating CIPN symptoms that often result in cessation or reduction in CT. Although preliminary, the need for pharmaceutical intervention, CT dose reductions, and early cessation were less prevalent and more consistent in the INF® therapy arm than in the standard PT arm, resulting in better compliance and a higher average RDI. While these effects are modest, they are likely clinically significant as the findings indicate that active management of dose-limiting toxicities of CT treatment improved completion rates which in turn help optimize the quality of life and overall survival of patients with cancer diagnosis. Given these benefits, these interventions are recommended to be considered part of a multidisciplinary approach at the start of CT. However, larger studies with longer treatment duration should be performed to confirm these findings.
Supplemental Material
sj-docx-1-ict-10.1177_15347354251413222 – Supplemental material for A Novel Approach of Intraneural Facilitation Versus Standard Physical Therapy for the Prevention of Chemotherapy-Induced Peripheral Neuropathy: A Randomized Controlled Trial
Supplemental material, sj-docx-1-ict-10.1177_15347354251413222 for A Novel Approach of Intraneural Facilitation Versus Standard Physical Therapy for the Prevention of Chemotherapy-Induced Peripheral Neuropathy: A Randomized Controlled Trial by Jamie Hankins, Mark Bussell, Lida Gharibvand, Won Jin Jeon, Dani Ran Castillo, Ellen D’Errico and Gayathri Nagaraj in Integrative Cancer Therapies
Supplemental Material
sj-xlsx-2-ict-10.1177_15347354251413222 – Supplemental material for A Novel Approach of Intraneural Facilitation Versus Standard Physical Therapy for the Prevention of Chemotherapy-Induced Peripheral Neuropathy: A Randomized Controlled Trial
Supplemental material, sj-xlsx-2-ict-10.1177_15347354251413222 for A Novel Approach of Intraneural Facilitation Versus Standard Physical Therapy for the Prevention of Chemotherapy-Induced Peripheral Neuropathy: A Randomized Controlled Trial by Jamie Hankins, Mark Bussell, Lida Gharibvand, Won Jin Jeon, Dani Ran Castillo, Ellen D’Errico and Gayathri Nagaraj in Integrative Cancer Therapies
Footnotes
Acknowledgements
We express our sincere gratitude to everyone who contributed to the completion of this research manuscript. Our heartfelt thanks go to Dr. Salem Dehom for his invaluable insights, expertise, and support. We also wish to acknowledge the dedication and hard work of our research teams at the Cancer Center—Annette Boggs and Lorena Garcia—and at the Neuropathic Therapy Center—Ronald Coleman and Karla Pieters. Their collaborative efforts have been essential to the success of this project. We extend special thanks to Loma Linda University for providing the necessary resources and facilities that facilitated our research.
Ethical Considerations
This study was performed in accordance with the principles of the Declaration of Helsinki. The Institutional Review Board of Loma Linda University (IRB # 5170197) granted approval.
Consent to Participate
Written informed consent was obtained from all individual participants included in the study.
Consent for Publication
Written informed consent was obtained from all individual participants included in the study.
Author Contributions
GN, ED, and MB contributed to the study’s conception, design, conduct, and interpretation of the data. JH, WJ, and DC prepared the tables and data for analysis. JH and LG performed the statistical analysis and contributed to the data interpretation. JH drafted the initial version of the manuscript, while GN, MB, LG, ED, DC, and WJ provided feedback on all revisions. All authors have read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Grants for Research and School Partnerships (GRASP) grant provided by the Loma Linda University Research Administration (Award # 2160303-01).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mark Bussell, DPT, OCS developed the technique known as Intraneural Facilitation (INF®). As the creator of INF® therapy, he has a vested interest in its application, research, and dissemination. The remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
