Abstract
Despite advances in HER2-targeted therapies and CSC-directed agents, resistance remains a major barrier in breast cancer. Synthesize evidence for exercise as a precision strategy to disrupt HER2/CD44-driven resistance circuits. Preclinical and clinical data demonstrate that physical activity: (1) downregulates HER2/PI3K signaling via myokine-mediated pathways (IL-6/SPARC), (2) reduces CD44 through NK-dependent immune surveillance, and (3) synergizes with biologics to overcome cardiotoxicity and chemoresistance. Molecular subtype-specific exercise prescriptions are defined. Exercise reprograms the tumor-immune microenvironment to target therapy-resistant pathways, establishing a paradigm for exercise as adjuvant precision medicine.
Introduction
Breast cancer remains a leading cause of cancer-related morbidity and mortality worldwide, with molecular heterogeneity driving varied clinical outcomes and treatment responses. HER2 overexpression, occurring in 20% to 30% of cases, amplifies oncogenic signaling through pathways like PI3K/Akt and MAPK, accelerating tumor proliferation and metastasis while conferring resistance to conventional therapies. Similarly, CD44—a transmembrane glycoprotein and cancer stem cell (CSC) marker—promotes tumor aggressiveness by sustaining stemness, facilitating epithelial-mesenchymal transition (EMT), and enhancing chemoresistance across breast cancer subtypes. Both molecules converge on critical pathways like PI3K/Akt, creating a synergistic axis for disease progression and recurrence. 1
Physical activity has emerged as a non-pharmacological intervention that improves clinical outcomes in breast cancer patients, with meta-analyses indicating a reduction in breast cancer-specific mortality and lower recurrence risk among physically active survivors. 2 While direct mechanistic studies linking exercise to HER2 and CD44 modulation remain limited, exercise-induced systemic changes—including enhanced CD8+ T-cell cytotoxicity, 3 reduced immunosuppressive cytokines, 4 and altered metabolic profiles 5 —likely disrupt CSC maintenance and HER2-driven signaling. For instance, exercise elevates myokines like IL-6, which may indirectly suppress CD44-mediated survival pathways and PI3K/Akt activation shared with HER2.6,7 These effects collectively can remodel the tumor microenvironment, potentially attenuating the metastatic and therapy-resistant phenotypes associated with HER2 and CD44.
To date, therapeutic approaches to HER2+ breast cancer remain largely pharmacological, with exercise relegated to a supportive role for managing treatment side effects rather than targeting resistance mechanisms themselves. This represents a critical gap in our integrative approach to cancer care. Recent evidence suggests that the bidirectional communication between skeletal muscle and tumor tissue—mediated by exercise-induced myokines, immune modulation, and metabolic reprograming—may directly disrupt therapy-resistant pathways. 8 However, no comprehensive synthesis exists on how exercise precisely targets the HER2/CD44 axis that drives treatment failure in breast cancer.
This mini-review therefore establishes a novel paradigm: positioning exercise as precision medicine that can be calibrated to molecular tumor characteristics. By integrating findings from exercise oncology with molecular pathway analysis, we demonstrate how specific exercise modalities may be prescribed to disrupt HER2/CD44-driven resistance circuits—potentially enhancing treatment efficacy while reducing cardiotoxicity. This precision approach represents a paradigm shift in how we conceptualize non-pharmacological interventions in oncology, moving beyond generic ‘exercise is beneficial’ recommendations toward mechanism-based, tumor-informed exercise prescriptions.
Role of CD44 and HER2 in Breast Cancer Pathophysiology
Human epidermal growth factor receptor 2 (HER2) and cluster of differentiation 44 (CD44) are critical molecular players in breast cancer pathophysiology, each contributing to distinct aspects of tumor aggressiveness, metastasis, and therapeutic resistance. HER2, overexpressed in 20% to 30% of breast cancers, encodes a 185-kDa transmembrane receptor tyrosine kinase that lacks a known ligand but readily forms heterodimers (particularly with HER3) to activate downstream signaling cascades.9 -11 This ligand-independent dimerization triggers the PI3K/Akt and MAPK pathways, driving uncontrolled proliferation, enhanced cell survival, and suppression of apoptosis. 12 Clinically, HER2-positive tumors exhibit accelerated growth, higher recurrence rates, and increased mortality. 13 Targeted therapies like trastuzumab inhibit HER2 by blocking downstream survival signals and inducing antibody-dependent cellular cytotoxicity, though resistance often emerges through compensatory pathway activation. 14
CD44, a transmembrane glycoprotein and cancer stem cell (CSC) marker, contributes to breast cancer progression through its role in maintaining stemness, drug resistance, and metastasis. 15 Functioning as a co-receptor for hyaluronic acid (HA) and other extracellular ligands, CD44 activates PI3K/Akt, Rho GTPase, and Wnt signaling pathways upon binding, thereby regulating epithelial-mesenchymal transition (EMT), invasion, and survival. 16 Isoforms like CD44v6 are particularly implicated in aggressive phenotypes: they enhance chemoresistance to agents like doxorubicin and paclitaxel, promote tumor sphere formation, and facilitate metastasis through interactions with hepatocyte growth factor (HGF) and MET receptors. 17 In HER2-positive contexts, CD44 may synergize with HER2 signaling, as both converge on PI3K/Akt activation—a pathway critical for CSC maintenance and therapy evasion. 18 This interplay potentially amplifies tumor aggressiveness, though CD44’s influence extends across breast cancer subtypes, including hormone receptor-positive and triple-negative disease. 19
Effects of Physical Activity on CD44 Expression
Physical activity has been shown to influence the expression of various biomarkers associated with cancer, including CD44, which is a key player in cancer stem cell biology and tumor progression. Exercise can modulate the tumor microenvironment, potentially affecting the expression of CD44 and its variants. For instance, studies have indicated that physical activity may reduce the levels of inflammatory cytokines and growth factors that can upregulate CD44 expression in tumors. This modulation can lead to a decrease in the aggressive characteristics of cancer cells, including their migratory and invasive capabilities, which are often associated with high CD44 expression. 20 Furthermore, regular exercise has been linked to improved immune function, which may also play a role in regulating CD44 expression and its associated pathways in cancer cells. 21
In addition to the systemic effects of exercise, there is evidence suggesting that physical activity can directly impact the cellular mechanisms involving CD44. For example, exercise-induced mechanical stress can influence cell signaling pathways that regulate CD44 expression. This is particularly relevant in the context of cancer, where the mechanical properties of the tumor microenvironment can affect cell behavior and gene expression. Research has shown that mechanical forces can lead to the activation of signaling pathways that downregulate CD44, thereby potentially reducing the stemness and metastatic potential of cancer cells. 22 Moreover, the interaction between CD44 and other signaling molecules may also be influenced by physical activity, further affecting the invasive properties of cancer cells. 23
Effects of Physical Activity on HER2 Expression in Breast Cancer
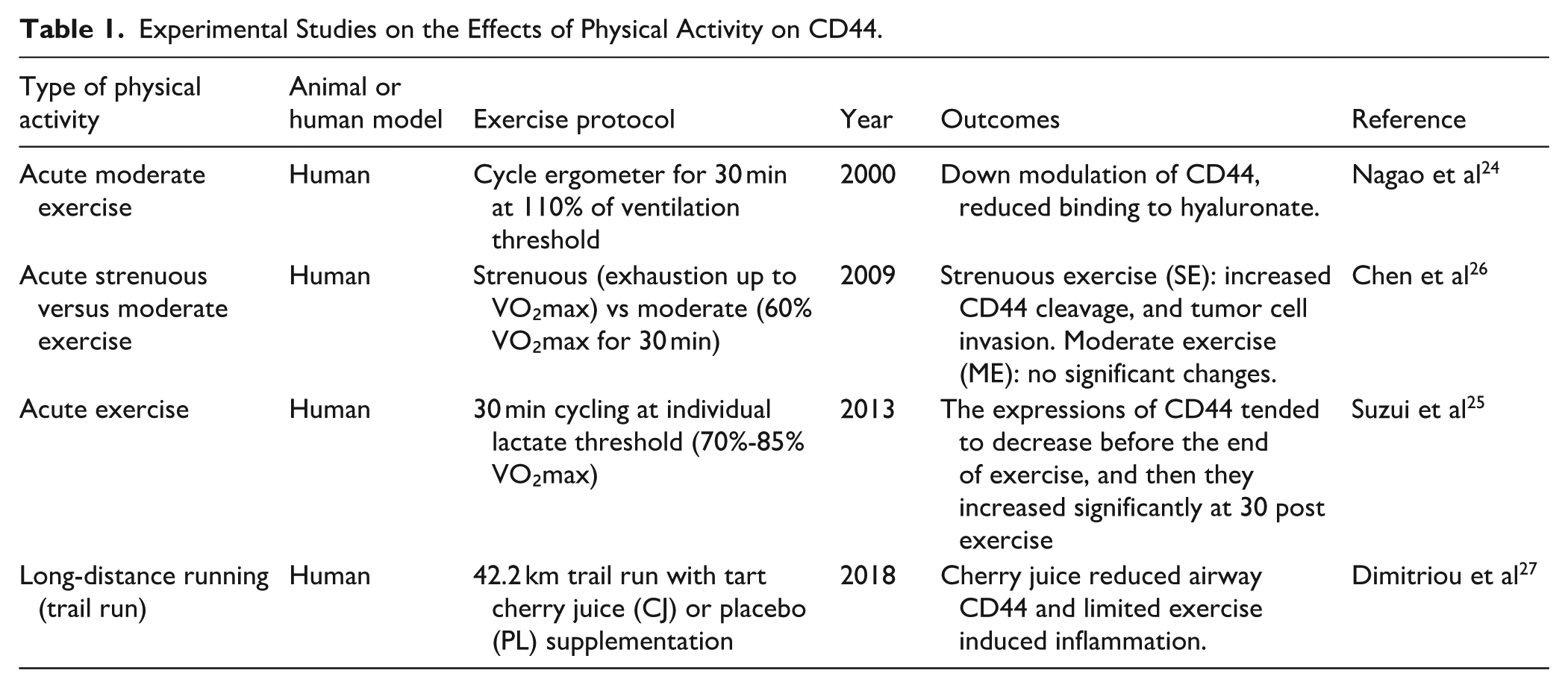
Regarding the effect of exercise on CD44, no study was available in cancer animal and human subjects. Studies that evaluated the effect of exercise on CD44 in non-cancer subjects have been presented in Table 1 which indicate that acute moderate exercise in humans (30 minutes cycling at 110% ventilation threshold) downregulated CD44 expression on NK cells and reduced their binding to hyaluronate. 24 Similarly, 30 minutes cycling at lactate threshold (70%-85% VO₂max) transiently decreased CD44 on NK cells during exercise, followed by a significant rebound 30 minutes post-exercise. 25 Strenuous exercise (to exhaustion) increased CD44 cleavage and promoted tumor cell invasion, while moderate exercise (60% VO₂max) had no such effects. 26 In a 42.2 km trail run, tart cherry juice supplementation reduced airway CD44 levels and mitigated exercise-induced inflammation compared to placebo. 27
Experimental Studies on the Effects of Physical Activity on CD44.
Findings from animal and clinical studies indicate that physical activity—whether aerobic, resistance-based, or high-intensity—can effectively downregulate HER2 markers in breast cancer. This leads to less tumor progression and improved drug effectiveness, suggesting a supportive role of exercise in cancer management strategies. Recent studies have suggested that engaging in regular physical activity may be associated with a reduced risk of developing HER2-positive breast cancer. For instance, a large nested case-control study indicated that higher levels of recreational physical activity were inversely associated with the risk of HER2-negative breast cancer, although the relationship with HER2-positive subtypes was less clear. 28 This suggests that while physical activity may not directly influence HER2 expression, it could play a role in the overall risk profile for different breast cancer subtypes.
Regarding HER2, as summarized in Table 2, available studies have evaluated the effect of exercise program on the treatment outcomes of patients with HER2-positive breast cancer and indicated a positive effect. Physical activity demonstrates distinct benefits depending on HER2 status in breast cancer. For HER2-negative tumors, recreational activity reduces cancer risk, 29 while in HER2-positive patients, supervised exercise (eg, 3-month programs combining moderate/high intensity) preserves cardiac function, mitigates trastuzumab-induced cardiotoxicity, and improves cardiorespiratory fitness (↑VO₂peak).30,31 Home-based and moderate-intensity programs also show feasibility and protective effects on cardiac parameters.32,33 In animal models, pre-treatment endurance training prevents chemotherapy-induced cardiac dysfunction. 34 Additionally, diet combined with isometric exercise aids weight/fat loss in ER+/HER2- patients. 35 Overall, tailored physical activity improves outcomes across breast cancer subtypes, emphasizing its role in risk reduction, therapy support, and quality of life. Also, a study investigated the impact of 12 weeks (36 sessions) supervised exercise program as interval cycling with intensity of personalized based on ventilatory thresholds (4 minutes @ VT1 + 1 min @ VT2, repeated 9×) on cardiotoxicity (left ventricular ejection fraction under 50% or an absolute drop of 10% from baseline) in patients undergoing treatment with trastuzumab (a HER2-targeted therapy). After 3 months, 90.5% of patients in the training group were free of cardiotoxicity, compared to 81.8% in the control group. Training also increased VO2peak and no adverse event was reported. 36 This highlights the importance of incorporating physical activity into the treatment regimen for HER2-positive breast cancer patients, as it may improve both physical and psychological well-being during therapy.
Experimental Studies on the Effects of Physical Activity on HER2 in Breast Cancer.

Implications of Physical Activity for Treatment and Patient Management
The relationship between physical activity and HER2 expression has significant implications for the management of breast cancer. Given that HER2-positive tumors are often associated with more aggressive disease and poorer outcomes, integrating physical activity into treatment plans could provide a dual benefit: potentially reducing the risk of developing HER2-positive tumors and improving the quality of life for those already diagnosed. Furthermore, evidence suggests that moderate to vigorous physical activity may enhance overall survival rates in patients with metastatic HER2-positive breast cancer. 37 This underscores the need for healthcare providers to encourage physical activity as part of a comprehensive cancer care strategy.
In conclusion, while the direct effects of physical activity on HER2 expression require further investigation, existing evidence supports the notion that exercise can play a beneficial role in the context of HER2-positive breast cancer. By potentially reducing the risk of aggressive tumor types and improving treatment outcomes, physical activity should be considered a valuable component of breast cancer management. Future research should continue to explore the biological mechanisms underlying these associations and the optimal types and intensities of exercise for patients with different breast cancer subtypes.
The relationship between physical activity and CD44 expression has also significant implications for cancer therapy and patient management. Given that high CD44 expression is often associated with poor prognosis and increased metastatic potential, strategies that incorporate physical activity could be beneficial in managing cancer progression. Exercise may serve as an adjunct therapy that not only improves overall health and quality of life but also potentially modulates the expression of key biomarkers like CD44, thereby influencing tumor behavior. 38
Conclusion
As summarized in Figure 1, the integration of physical activity into the management of breast cancer presents a promising complementary strategy for modulating critical molecular targets such as HER2 and CD44. Evidence from preclinical and clinical studies indicates that exercise can influence these biomarkers through immune-metabolic mechanisms, potentially enhancing treatment efficacy and reducing disease progression. For HER2-positive breast cancer patients, supervised moderate-intensity aerobic and resistance training programs have demonstrated benefits in preserving cardiac function, mitigating therapy-induced cardiotoxicity, and improving cardiorespiratory fitness. Similarly, physical activity has shown potential in modulating CD44 expression, particularly through transient reductions in its levels on immune cells like NK cells following moderate-intensity exercise, which may enhance immune surveillance and reduce metastatic risk. These findings underscore the importance of incorporating tailored exercise interventions into comprehensive cancer care strategies.

Proposed mechanistic pathways for exercise-induced disruption of HER2/CD44-driven therapy resistance in breast cancer. This model illustrates how structured aerobic and resistance exercise functions as a multi-system intervention. Exercise initiates systemic effects, including the release of myokines (eg, IL-6, SPARC), enhanced immune surveillance (eg, NK cell and CD8+ T-cell activation), and metabolic reprograming. These systemic changes converge on the tumor microenvironment to downregulate key oncogenic pathways: (1) suppressing HER2/PI3K/Akt signaling, (2) reducing CD44 expression and cancer stem cell (CSC) stemness, and (3) remodeling the tumor-immune landscape. Collectively, these molecular and cellular impacts disrupt the synergistic HER2/CD44 resistance axis, leading to improved clinical outcomes such as enhanced efficacy of HER2-targeted therapies (eg, trastuzumab), mitigation of treatment-related cardiotoxicity, and reduced tumor progression and metastasis.
Moving forward, while current evidence supports the beneficial role of physical activity in breast cancer outcomes, further research is needed to establish optimal exercise protocols and clarify the direct causal relationships between exercise and molecular modulation of HER2 and CD44. Randomized controlled trials with detailed biomarker analyses are essential to refine exercise prescriptions based on tumor subtype and treatment regimen. Ultimately, integrating precision exercise recommendations into standard oncological care could serve as a non-pharmacological, cost-effective approach to improve both the physiological and psychological well-being of breast cancer patients, reinforcing the growing paradigm of exercise as medicine in oncology.
Practical Implication for Breast Cancer Patients Based on HER2 and CD44
Based on current published evidence, exercise is strongly recommended for breast cancer patients across different stages of their disease, including during chemotherapy, surgery recovery, and post-treatment rehabilitation, with specific considerations for HER2-positive and CD44-associated tumor biology. For patients undergoing chemotherapy, especially those with HER2-positive breast cancer who face risks of treatment-induced cardiotoxicity, moderate-intensity aerobic exercise combined with resistance training is beneficial. A home-based program integrating aerobic and strengthening exercises during neoadjuvant chemotherapy has shown feasibility and may help maintain cardiac function while improving overall physical fitness. 32 Moderate-intensity exercise (such as brisk walking or cycling) for about 150 minutes per week, divided into sessions of 20 to 30 minutes, is generally advised. This intensity is supported by preclinical data indicating that moderate, but not low or high intensity, exercise optimally enhances antitumor immune responses, including increased infiltration and activation of CD8+ T cells that can target cancer cells expressing markers like CD44. 39
In the postoperative phase, particularly after breast cancer surgery, gradual reintroduction of physical activity is crucial. Initially, low-impact aerobic exercises such as walking, stationary cycling, or gentle yoga are recommended to improve cardiovascular health, reduce fatigue, and prevent lymphedema, especially if lymph nodes were removed. 40 Patients are advised to start with 20 minutes of aerobic exercise at least 3 times per week, progressively increasing duration and intensity as tolerated. 41 Resistance training targeting major muscle groups at moderate intensity (around 60%-80% of 1-repetition maximum) can be safely introduced after healing, helping to restore muscle strength, reduce fatigue, and improve quality of life. 42 Combining aerobic and resistance exercises has been shown to reduce cancer-related fatigue, anxiety, and depression, while improving physical function and sleep quality in breast cancer survivors.43,44
During recovery and long-term survivorship, maintaining regular moderate-intensity exercise is key to sustaining benefits. Programs that include both aerobic and resistance training performed at least 3 times per week for 8 to 12 weeks or longer are effective in improving cardiovascular fitness, muscle strength, and body composition, as well as reducing symptoms related to cancer and its treatment. 43
Overall, for clinical practice, structured, moderate-intensity aerobic and resistance exercise should be systematically integrated into the care plan for patients with HER2-positive breast cancer. Specifically, during adjuvant trastuzumab treatment, a supervised program of approximately 150 minutes per week—such as interval cycling calibrated to individual ventilatory thresholds—can directly mitigate cardiotoxicity, improve cardiorespiratory fitness, and may concurrently disrupt the HER2/CD44 resistance axis. Moving beyond a 1-size-fits-all approach, these findings advocate for the development of molecular subtype-informed exercise prescriptions as a standard component of precision oncology, empowering clinicians to use physical activity not only for supportive care but as a targeted strategy to improve treatment efficacy and patient survival. Importantly, exercise regimens should be personalized and supervised by healthcare professionals to accommodate individual treatment status, comorbidities, and risk factors such as lymphedema or cardiotoxicity.
Footnotes
Author Contributions
F.E.H and M.K.J. have contributed in collecting data, writing original manuscript and revisions.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
