Abstract
Introduction. African American breast cancer survivors engage in less physical activity compared with their Caucasian counterparts. There is a need for exercise intervention research that focuses on improving the overall health and long-term survivorship of African American breast cancer survivors, especially because they often have worse outcomes than Caucasian survivors. Study objectives were to determine whether African American participants increase physical activity and explore whether exercise had a positive impact on fitness and health. Methods. African American breast cancer survivors, stage 0 to IIIA, within 2 years of completing primary cancer treatments were recruited for a 16-week home-based aerobic and resistance training exercise pilot study. Outcome measures assessed at baseline and postintervention included physical activity questionnaires and accelerometry, cardiopulmonary function (VO2peak) with gas exchange, muscle strength, Selective Functional Movement Assessment, and dual energy X-ray absorptiometry scans for body composition analysis. Assessments for fatigue and quality of life (QOL) were also completed at baseline and postintervention. Motivational interviewing was utilized to determine goals and explore exercise facilitators/barriers. Participants completed weekly exercise logs and received weekly phone calls. Wilcoxon signed rank tests were used to detect significant changes in physical activity and also changes in fitness/health parameters, fatigue, and QOL. Spearman correlation coefficients were used to examine relationships between physical activity and health measures. Results. A total of 17 women enrolled; 13 completed the intervention (76%). Mean age of the participants was 51 years. There was a significant increase in total minutes of weekly physical activity postintervention (M = 271 minutes, SD = 151; P = .001). Significant improvements were found in cardiopulmonary fitness as measured by VO2peak with a mean increase of 2.03 mL/kg/min (P = .01). Several strength measures significantly increased and also functional movement (P = .005). Positive correlations existed between physical activity and several physical measures, with significant relationships between functional movement and some strength measures (eg, left arm extension: rs = 0.61, P = .002). Total QOL and fatigue scores improved, but neither was significant. Conclusions. The intervention led to increased physical activity. As a result of increased levels of physical activity, improvements on several fitness/health parameters occurred.
Introduction
There are approximately 2.9 million breast cancer survivors in the United States and while Caucasian women have the highest incidence of breast cancer, African American women are more likely to be diagnosed with advanced disease and have higher mortality rates.1,2 These disparities are felt to be related to myriad factors, including differences in screening, treatments, genetics, and lifestyle behaviors, such as physical inactivity. 1 The unique health care needs of breast cancer survivors are a result of the cancer experience itself and adverse effects from cancer treatments. Breast cancer survivors may experience long-term and late effects from surgery, chemotherapy, hormonal therapy, and/or radiation therapy. Potential long-term physical effects include fatigue, postsurgical pain, and lymphedema, while psychosocial challenges may include anxiety and depression. 3 Certain chemotherapeutic agents and radiation therapy can be cardiotoxic leading to late effects, such as cardiomyopathy. 3 A late effect of both chemotherapy and hormonal therapy is osteoporosis, which increases a woman’s risk for bone fractures. 3 In addition to some of these long-term and late effects, these women are at increased risk for recurrence, secondary malignancies, comorbidities such as weight gain and diabetes, as well as premature death.4,5 African American breast cancer survivors have a higher incidence of comorbidities (ie, hypertension, cardiovascular disease, diabetes, and obesity) which can adversely affect quality of life and overall survival. 6
Exercise training among cancer survivors appears to be safe, is well tolerated, protects against weight gain, has a positive effect on cardiorespiratory fitness, fatigue, anxiety, depression, and overall quality of life.7 -11 There is also evidence that physical activity reduces all-cause mortality in breast cancer survivors and recent observational data reveals that moderate physical activity may reduce breast cancer–specific mortality.5,12,13
The majority of breast cancer survivors who have completed treatment are not meeting physical activity recommendations for cancer survivors endorsed by the American Cancer Society and the American College of Sports Medicine of at least 30 minutes of moderate-intensity physical activity on 5 or more days per week.14 -16 In fact, Blanchard et al 14 reported that only 37.1% of breast cancer survivors were meeting this physical activity recommendation. Compared with Caucasian breast cancer survivors, African American survivors typically engage in even less physical activity following diagnosis and treatment.17 -19 Furthermore, they are more likely than their Caucasian counterparts to experience overweight and sarcopenic obesity (weight gain with increased fat mass and loss of muscle) following diagnosis and treatment. 20 Weight changes following cancer treatments in African American breast cancer survivors can cause significant psychological distress. 21 Halbert et al 21 found that African American women who gain weight after treatment often have difficulty managing weight, which women attribute partly to reduced physical activity. Smith et al 19 found that African American breast cancer survivors who met recommended physical activity levels reported significantly better quality of life than those not meeting physical activity recommendations.
While research on the beneficial role of exercise in breast cancer survivor’s physical and psychological outcomes continues to grow, further research is needed on how best to deliver exercise programs to women who may have significant barriers toward regular engagement in exercise. Most exercise intervention studies conducted among breast cancer survivors have included primarily Caucasian women. Because African American breast cancer survivors often have worse outcomes than their Caucasian counterparts, there is a great need for interventions (eg, exercise programs) that focus on improving their overall health and long-term survivorship.
The purpose of this pilot study was to examine if physical activity levels increased as a result of a 16-week home-based motivational exercise study (ie, brisk walking and resistance training) and to determine whether changes in physical activity had positive effects on fitness/health parameters, including body composition, cardiorespiratory fitness, muscle strength, selective functional body movement, fatigue, and quality of life. A secondary goal was to assess relationships between physical activity and fitness/health parameters.
Methods
Participants and Setting
Participants were recruited through study brochures, e-mail, and referral by oncology clinicians at a National Cancer Institute–designated comprehensive cancer center and an affiliated community cancer center. Institutional review board approval was obtained through the primary cancer center. Potential participants were also recruited through the Cancer Registry of the primary institution after initiation of passive physician consent. Potential participants on the cancer registry received study introduction letters along with a return postage paid opt-out card to be mailed back if they did not wish to be contacted. If opt-out cards were not received after 1 month, women were called to discuss the study and determine initial eligibility if interested. Women were eligible if they meet the following criteria: ≥18 years old, histological evidence of stage 0 to IIIA invasive breast cancer, within 24 months of completion of all primary breast cancer treatments (eg, surgery, radiation, and chemotherapy with the exception of women on adjuvant Herceptin therapy), currently sedentary (ie, exercising less than twice per week for 30 minutes), and received medical clearance from oncologist or primary care physician to participate. Exclusion criteria included the following: history of another cancer (exception for nonmelanoma skin cancers), comorbidities that would preclude safe participation in exercise at home (eg, symptomatic cardiovascular or pulmonary disease), plans for major surgery (including breast reconstruction) during the intervention time frame, and psychiatric illness that would make it difficult to participate. Women were also asked to avoid any changes in their dietary patterns during the study that would be intended for weight loss. Once medical clearance was obtained, participants were contacted to discuss informed consent and to schedule a convenient time to come into the Integrative Exercise Oncology Laboratory (IEOL) for testing at the primary institution. On arrival to the IEOL, informed consent was obtained followed by baseline assessments. Participants received two $10 gas cards for travel to and from the IEOL and were provided free parking. For their efforts in participating, women received a $25.00 check on completion of the study.
Design and Exercise Intervention
A prospective single-arm pretest/posttest design was used for this 16-week progressive aerobic (ie, walking) and resistance training pilot study. After completing baseline assessments, participants were instructed on the intervention and received training on the resistance exercises from a certified personal trainer, which included return demonstrations for proper and safe use of resistance bands. Each participant received 3 Therabands, each a different strength (ie, low, medium, and heavy) to perform the resistance exercises and a pedometer to help encourage walking activity, even though steps per day was not included as a measure in this study. The rationale for providing pedometers is that they have been thought to serve as a motivator for walking.
Home-Based Combined Aerobic and Resistance Exercise Plan
The home-based exercise intervention began with low-intensity walking and resistance (ie, strength) training, which progressed to follow the previously tested and successful Diabetes Prevention Program exercise training model of 150 min/wk of low-to-moderate intensity exercise. 22 The combined aerobic and resistance training protocol followed guidelines set forth by the American College of Sports Medicine. 16 The progressive standardized aerobic exercise plan involved walking, which has been identified as the preferred form of exercise by breast cancer survivors, including African American women.18,23 Resistance training included upper, lower, and abdominal core strengthening exercises. Upper body strength exercises included lateral and front raises, wall or modified floor push-ups, chest press, bent row, arm curls, and tricep stretches. Lower body strength exercises included chair squats, chair leg raises, hamstring curls, and calf raises. The four core strengthening exercises were the bridge, crunches, reverse crunches, and obliques. Most upper and lower body exercises were done with a resistance band. Participants were also instructed to perform stretching exercises of major muscle groups after the walking component of their exercise program. Participants were given a heart rate monitor, accelerometer (ie, physical activity monitor), and an exercise training workbook that included weekly exercise logs, the exercise training plan, illustrations of African American breast cancer survivors engaging in resistance exercises and stretches, as well as motivational quotes. Participants were instructed on how to monitor their personal exercise training intensity (ie, heart rate training zones for aerobic training) with a heart rate monitor watch. To stimulate a training effect following recommendations from the American College of Sports Medicine, from week 1 to completion of the study, walking increased from 15 to 30 minutes and intensity from 40% to 65% of heart rate reserve. Therabands were used for the resistance training. Participants were instructed to perform 12 to 15 repetitions per exercise beginning with the light- or medium-strength band and progressing to the heaviest strength band as ability permitted. The training volume increased from 1 set per exercise on week 1 to 3 sets by week 16. See Figure 1 for a description of the exercise plan and percentage heart rate reserve. Also, participants received information on using an Actical accelerometer for capturing objective physical activity data on weeks 1, 8, and 16. Stamped, addressed padded envelopes were provided for mailing the devices after weeks 1 and 8 and participants were instructed to bring the Actical with them on their last visit, along with the heart rate monitor. Resistance bands and pedometers were theirs to keep. Finally, they received encouragement to obtain the support of a friend or family member to join them in their walks when possible.

Recommended exercise plan for progressive walking and resistance training.
Initial and Weekly Telephone Motivational Interviewing Sessions
At the conclusion of the initial visit the nurse researcher conducted a motivational interviewing (MI) session in a quiet room in the IEOL to determine exercise goals, facilitators, and potential barriers. She conducted all weekly telephone sessions with participants as well. The nurse researcher had prior professional training in MI counseling techniques. MI techniques help elicit participant’s own motivations for engaging and adhering to a regular physical exercise practice. 24 Initial counseling sessions were based on the 4 guiding principles of MI, one of which emphasizes empowering the individual. 24 Supporting self-efficacy or self-confidence in one’s ability to achieve behavior change (ie, exercise) is a major factor in MI. The initial session began with opening guiding questions, such as the following (a) “What are your top reasons for wanting to exercise?,” (b) “What do you think you are able to do at this time?,” and (c) “What else have you accomplished that helps you know you can do this?” Various MI strategies were also used as needed in the weekly sessions to help address physical activity barriers and promote exercise self-efficacy.
During weekly sessions, the nurse researcher assessed progress with the exercise plan by reviewing the weekly exercise log and any health changes. Also, information was provided on modifications to the exercise plan to stimulate a training effect. To augment the intervention, culturally-appropriate tailored newsletters were mailed monthly over the four month intervention and each focused on a specific exercise-related topic (eg, goal setting, exercise benefits, exercise barriers, and social support).
Random samples of ten initial MI sessions were audio-recorded for the purpose of determining fidelity on the use of MI components in interviews. These audio-recordings were reviewed and coded by a certified Motivational Interviewing National Trainer who then provided feedback about use of appropriate techniques. Subsequent interviews were modified as needed based on feedback.
Data Collection
Participants completed baseline measures to collect information on sociodemographics and health history, including medical and treatment-related information.
Physical Fitness/Function Assessments
Physical fitness/function testing followed the same protocol at baseline and postintervention. After arriving to the IEOL, preassessment guidelines (eg, no eating 2 hours before testing, no exercise 12 hours before testing, and no alcohol 48 hours before testing) were reviewed to ensure all participants followed testing guidelines. Following completion of all questionnaires, physical fitness assessments were initiated. Waist and hip measurements were taken with a flexible fabric tape measure to determine waist-to-hip ratio. Weight and height were measured using a balance beam physician scale equipped with a height rod (Health-o-meter 402KL, Rye, NY). Body composition was then evaluated using DEXA (dual-energy X-ray absorptiometry) W series (Hologic, Bedford, MA). Functionality was then assessed through the Selective Functional Movement Assessment (SFMA). The SFMA is the clinical adaptation of the well-studied Functional Movement Screen for patients presenting with pain and/or dysfunction at time of evaluation.25,26 The SFMA provides a systematic way of testing and scoring 7 fundamental movement patterns: forward flexion, backward extension, trunk rotation, cervical mobility, shoulder mobility, single leg stance, and the deep squat. 27 SFMA scoring is based on 4 different categories (ie, Functional/Nonpainful, Functional/Painful, Dysfunctional/Nonpainful, and Dysfunctional/Painful) for each of the movement patterns. To evaluate functional ability, categories were dichotomized to functional (painful and nonpainful) or dysfunctional (painful and nonpainful). Following a 5- to 10-minute rest period in a seated position, resting heart rate, blood pressure, oxygen saturation, and a 12-lead electrocardiogram were performed. The electrocardiogram was performed using a GE Case Stress Test System. These assessments were followed by a cardiorespiratory function exercise test. Cardiorespiratory function (VO2peak) was assessed using an electric braked cycle ergometer (Lode BV, Groningen, Netherlands) and a metabolic chart for collection of respiratory gases for determining peak oxygen consumption via VO2peak (Parvo Medics TrueMax2400 Metabolic System, Parvo Medics, Salt Lake City, UT) with 12-lead electrocardiogram monitoring following standardized procedures. This protocol has been previously demonstrated to be appropriate for measuring VO2peak in patients with cancer.28,29 Muscular strength was assessed using the Biodex System 3 Isokinetic Dynamometer (Biodex Medical Systems, Shirley, NY). Two multijoint exercises, leg extension for assessment of quadriceps strength and low row for the assessment of latissumus dorsi and rhomboid major and minor, were used for assessment of lower and upper body strength, respectively. Strength was assessed by having participants perform 3 consecutive trials at a concentric speed of 90°/s, following manufactures’ recommended procedures. Peak torque from each trial was recorded for upper and lower body muscular strength.
Physical Activity Measurements
Actical accelerometers were used to objectively measure physical activity (Mini Mitter, Respironics, Bend, OR). Actical data were collected minute-by-minute. Participants were instructed to wear the Actical on a waistband positioned just above the right hip for seven days during waking hours on weeks 1, 8, and 16. They were also instructed to keep a log of when the device was put on and taken off. Acticals were tested for proper operation through the software program prior to distributing to participants. Data were downloaded at each time point into the Actical software program. All downloaded data were then processed using a customized program that generated data specific to our outcome measures (ie, mean counts/min/d and mean minutes of moderate–vigorous physical activity/day for valid days worn). A valid day was defined as ≥8 hours of wear time as suggested by Martin-Matillas et al. 30 Data that recorded ≥60 minutes of consecutive zeros were considered non–wear time and therefore were removed from analysis. At least 4 days, including 1 weekend day, was chosen as the minimum number of days for a valid week of physical activity observations, which has been previously suggested.31,32 The average number of days across the 3 assessment periods was 6.7. Cut-points for moderate-to-vigorous intensity activity were based on those established by Colley and Tremblay 33 and were programmed into the customized software program.
The short version of the International Physical Activity Questionnaire (IPAQ) was used to collect self-report physical activity data. The IPAQ assesses physical activity over the past 7 days in adults. The IPAQ has 7 items and assesses the following physical activity domains: house and yard work, leisure or recreational activities, sports, transportation-related activities, and time spent in sedentary behaviors. Separate scores are calculated for walking, moderate and vigorous physical activity. Total scores were then calculated based on the IPAQ scoring protocol (http://www.ipaq.ki.se). For analysis purposes, total physical activity was calculated in minutes per week, as well as MET (metabolic equivalents)-minutes per week. The IPAQ has undergone extensive reliability and validity testing across diverse cultures. 34 Reported correlation coefficients for reliability were .80 and criterion validity revealed a median ρ of .30, which is comparable to other physical activity self-report measures. 34
Quality-of-Life Measures
The Functional Assessment of Cancer Therapy–Breast (FACT-B + 4; version 4) was used to collect quality-of-life data. The FACT-B has been validated for assessment of quality of life in breast cancer patients of various racial/ethnic backgrounds.35,36 This instrument has 42 items and consists of the FACT–General (FACT-G) and a breast cancer–specific subscale. The FACT-G includes the following 4 domains: Physical Well-being (7 items), Social/Family Well-being (7 items), Emotional Well-being (6 items), and Functional Well-being (7 items). The breast cancer subscale has 15 items, 4 of which are specific to lymphedema. A 5-point Likert-type scale is used and ranges from 0 = not at all to 4 = very much with higher scores indicating higher quality of life. Scores were calculated based on the FACT scoring guidelines (http://www.facit.org) with total scores ranging from 0 to 144. This self-administered instrument is brief and written at fourth grade reading level. The α coefficient, indicative of internal consistency, of the FACT-B is high at .92, with subscale α coefficients ranging from .77 to .88 in African American breast cancer survivors. 35
Data on fatigue were collected using the Functional Assessment of Chronic Illness Therapy–Fatigue (FACIT-Fatigue, version 4). The FACIT-Fatigue is a brief 13-item instrument using a 5-point Likert-type scale with response options ranging from 0 = not at all to 4 = very much. Scores range from 0-52 with higher numbers indicating less fatigue. Scores were calculated based on FACIT scoring guidelines available at http://www.facit.org. This instrument is written at the fourth grade reading level and takes less than 5 minutes to complete. The instrument has high internal consistency among cancer patients, with coefficients ranging from .93 to .95 and good test stability over time (r = 0.89). 37
Postintervention Visit
Participants returned to the IEOL for their postintervention assessment within 1 week of completing the 16-week intervention. On arrival, the postintervention questionnaires (ie, IPAQ, FACT-B + 4, FACIT-Fatigue) were reviewed for completeness prior to physical fitness testing. To account for circadian variations, attempts were made to schedule participants for final assessments at a similar time (within a window of 2 hours maximum) of baseline testing. Once testing was done, women were provided with a comparative review of their pre- and postintervention testing. They then completed a program evaluation and received a thank you letter and $25.00 check for their time and effort as study participants.
Statistical Analysis
Descriptive statistics were calculated for all variables, including counts and percentages for categorical variables and means (standard deviations) for continuous variables. Changes in physical activity levels, measured by change in score on the IPAQ and on Actical data from baseline to 4 months, were calculated and paired Wilcoxon signed rank tests were used to test if changes were significantly different from 0. Spearman correlations were used to examine relationships between IPAQ and Actical physical activity data at baseline and week 16, as well as between subjective physical activity data collected from exercise logs and Actical data from weeks 1, 8, and 16. Wilcoxon sign tests were also used for determining mean changes in health/fitness parameters, as well as for mean changes on FACT-B + 4 and FACIT-Fatigue scores. Spearman correlation coefficients were used to examine relationships between changes in physical activity levels and changes in health/fitness parameters. An α-level of .05 was used to determine statistical significance. All analyses were conducted using SAS Version 9.2 (SAS Institute, Cary, NC).
Results
Participant Characteristics
Among 17 women who enrolled in the study, 13 completed the intervention and postintervention assessment and none reported any adverse events as a result of the exercise program. Four women withdrew because of various reasons (ie, breast cancer recurrence, unexpected surgery, new job with increased time demands, and no reliable phone service). Sociodemographic as well as health and clinical characteristics are reported in Table 1. Women ranged in age from 30 to 69 years (mean 51.6 years). Approximately half were married (54%). All women reported having at least some college education, 54% worked full-time and 31% had a total annual household income less than $40 000. None were current smokers and the majority (69%) did not consume alcoholic beverages. Among women receiving chemotherapy (61.5%), 6 received a regimen which included Doxorubicin and 6 received Taxane. Four women also received Herceptin therapy.
Demographic and Clinical Characteristics (N = 13).
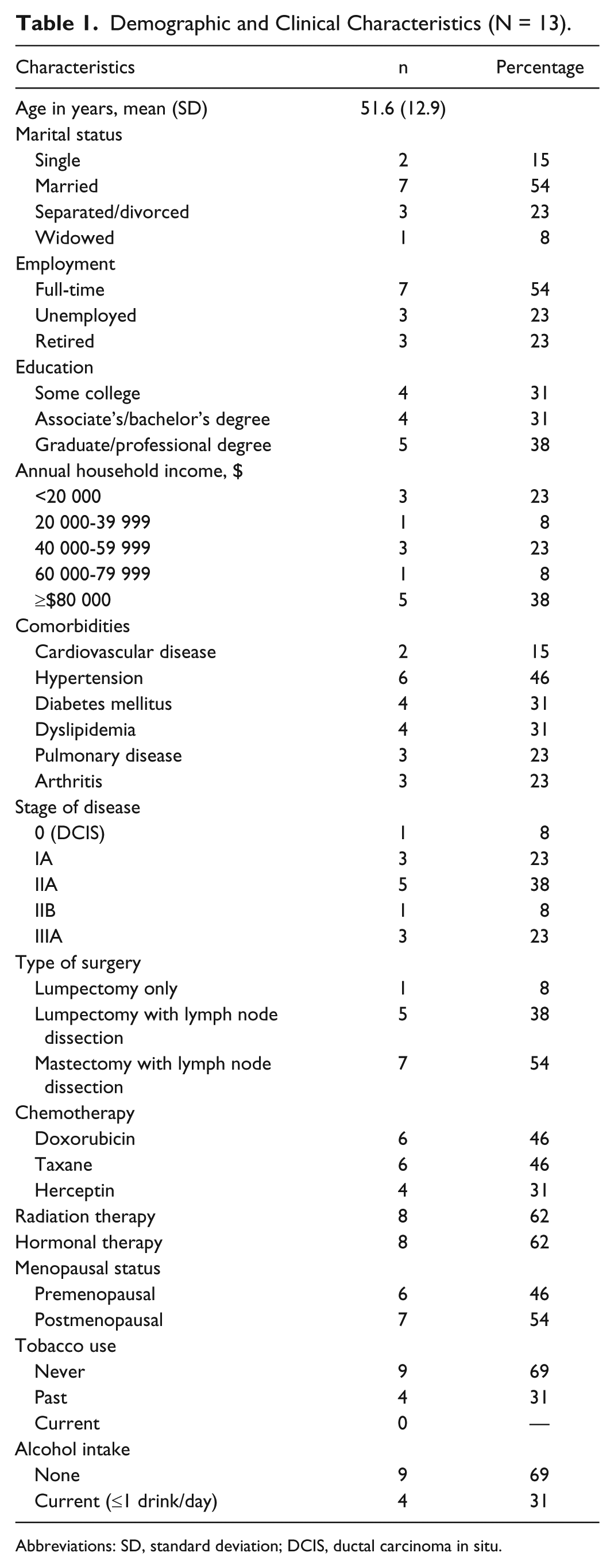
Abbreviations: SD, standard deviation; DCIS, ductal carcinoma in situ.
Changes in Physical Activity, Fitness/Health Parameters, and Quality of Life
Table 2 shows the mean scores for physical activity and fitness/health variables. IPAQ data revealed a significant increase in total minutes of weekly physical activity postintervention (M ± SD = 252 ± 142; P = .001). Actical data showed that both mean activity counts and moderate-to-vigorous activity increased significantly postintervention (P = .005 and P = .02, respectively). IPAQ total activity was positively correlated with Actical counts across the 3 time points (rs = 0.57, P < .001) with the highest correlation at week 8 (rs = 0.75, P = .02) and the lowest at week 1 (rs = 0.22, P = .48).
Physical Activity and Physical Fitness Measures at Baseline and 4 Months Post–Exercise Intervention (N = 13).

Abbreviations: SD, standard deviation; IPAQ, International Physical Activity Questionnaire (self-report); MET, metabolic equivalents; Actical, physical activity monitor (n = 11 because of missing Actical data); DEXA, dual energy X-ray absorptiometry; SFMA, Selective Functional Movement Assessment.
Significant mean differences, P ≤ .05.
Body weight and body mass index remained stable. No significant changes were found in percentage of body fat or in bone mineral density (ie, T score) on DEXA scans. Significant improvements were found in cardiorespiratory fitness as measured by VO2peak with a mean increase of 2.03 mL/kg/min (P = .01). All upper body and lower body strength measures increased, some significantly such as left arm flexion (P = 0.03) and extension (P < .001) and left and right leg extension (P < .001 and P = .005, respectively). Functional movement changed with SFMA scores revealing significant improvements in functional movement patterns (P = .005) and a decrease in dysfunctional movement patterns (P = .005). Although FACIT-Fatigue scores increased, they were not significant (P = .31). Total FACT-B scores increased but were nonsignificant; however, scores significantly improved on the FACT-B Trial Outcome Index (P = .05). Table 3 shows the specific subscale changes on the FACT-B Trial Outcome Index with significant improvements revealed on the FACT-B Breast Cancer Subscale (BCS; P = .03). Percentages to responses on the 9-item BCS subscale are reported in Figure 2.
Fatigue and Quality-of-Life Scores at Baseline and 16 Weeks Postintervention (N = 13).

Abbreviations: SD, standard deviation; FACIT, Functional Assessment of Chronic Illness Therapy; FACT, Functional Assessment of Cancer Therapy; B, breast; G, general; TOI, Trial Outcome Index; BCS, Breast Cancer Subscale.

Percentages for Functional Assessment of Cancer Therapy–Breast (FACT-B) Breast Cancer Subscale items pre- and postintervention.
Relationships Between Physical Activity and Fitness/Health Outcomes
Positive correlations existed for both physical activity measures (ie, IPAQ and Actical) with several fitness/health variables (see Table 4). Significant moderately strong positive relationships existed between total physical activity (minutes per week) and specific strength measures (eg, left and right arm extension: rs = 0.61, P < .01 and rs = 0.56, P < .01, respectively) and functional movement (rs = .40, P = .05). A moderately strong positive relationship was also found between IPAQ data and FACT-B BCS (rs = 0.54, P < .01). A moderate positive relationship occurred between the IPAQ and VO2peak, but it was nonsignificant. Interestingly, there was a strong positive correlation (rs = 0.84) between counts per minute (Actical) and VO2peak (P < .001) indicating that higher physical activity was related to better cardiopulmonary function. Strong relationships were found between counts/minute and lower extremity strength. In contrast, IPAQ scores showed moderate correlations with lower extremity strength. Strong significant associations existed between both functional (rs = 0.60) and dysfunctional movement (rs = −0.61) and counts per minute (both P = .002). A moderate positive correlation was found between counts per minute and the total FACT-B (rs = 0.40), which was nonsignificant. However, there was a strong significant relationship between fatigue scores and counts per minute suggesting that higher physical activity levels were associated with less fatigue.
Correlations for IPAQ and Actical Physical Activity Data and Health and Fitness Measures (N = 13).
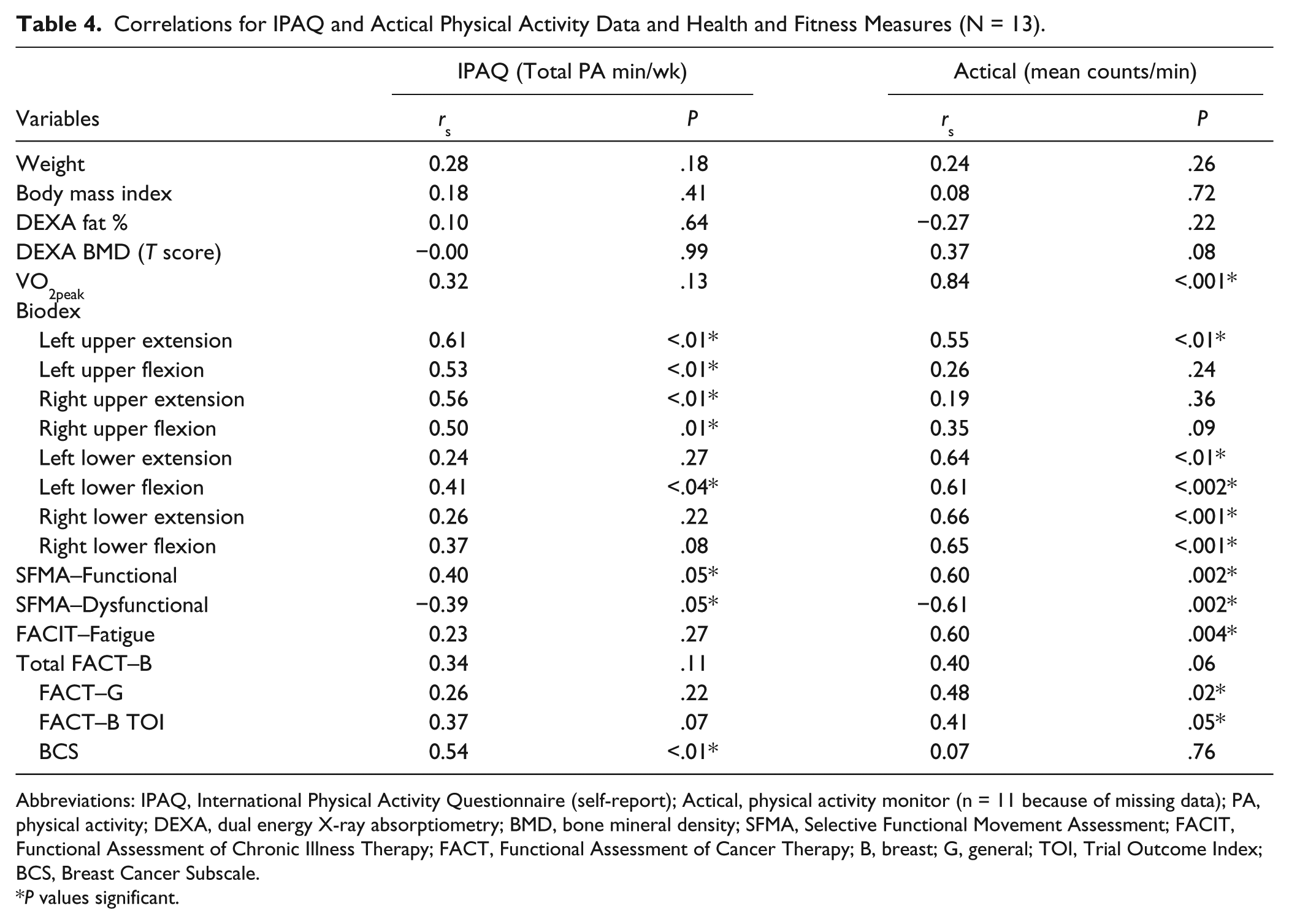
Abbreviations: IPAQ, International Physical Activity Questionnaire (self-report); Actical, physical activity monitor (n = 11 because of missing data); PA, physical activity; DEXA, dual energy X-ray absorptiometry; BMD, bone mineral density; SFMA, Selective Functional Movement Assessment; FACIT, Functional Assessment of Chronic Illness Therapy; FACT, Functional Assessment of Cancer Therapy; B, breast; G, general; TOI, Trial Outcome Index; BCS, Breast Cancer Subscale.
P values significant.
Discussion
This study evaluated whether a home-based motivational progressive walking and resistance training exercise program could lead to increased physical activity and improve fitness/health among a sedentary group of African American breast cancer survivors. Findings revealed that women significantly increased their physical activity levels as reflected in both IPAQ and Actical data. Most women achieved higher physical activity levels than those recommended for breast cancer survivors (ie, at least 150 minutes per week of moderate-intensity activity based on the IPAQ). This increase in weekly physical activity is consistent with other home-based exercise studies for breast cancer survivors.38,39 Pinto et al 39 found an increase in total minutes of weekly exercise after 12 weeks in the intervention group (baseline M = 83.5; postintervention M = 202.4) with a significant difference between intervention and control groups (P = .001). In the walking study by Matthews et al, 38 total weekly walking time increased significantly at 12 weeks (P = .01). Both of these studies were conducted primarily among Caucasian women. Findings from an 8-week home-based walking study, which included weekly group educational sessions, among African American breast cancer survivors revealed that women significantly increased mean steps per day as measured by pedometer from baseline (M = 4791) to postintervention (M = 8297, P = .001). 40 These studies were found to be safe and effective at positively influencing women’s physical activity behavior.
Although no changes occurred in body mass index or body composition, they at least remained stable through the 16-week intervention, a finding similar to that of another study where DEXA scans were used to measure percent body fat. 38 The fact that weight was maintained is viewed positively since many breast cancer survivors tend to gain weight after diagnosis, which is associated with an increased mortality risk.41,42 Considering that our intervention was focused on exercise without any dietary component it was not anticipated that a change in body composition would occur. For a significant decrease in body fat to occur, it is likely that an exercise program with a dietary component and of longer duration than 4 months would be necessary.
Increases in physical activity appeared to result in cardiorespiratory function improvement. The VO2peak increased 2.03 mL/kg/min postintervention, which is similar to findings from a recent review by Jones et al, 43 who found a mean VO2peak increase of 2.3 mL/kg/min among adult cancer survivors participating in various exercise studies. While the clinical relevance of these improvements among cancer survivors is not fully known, VO2peak has been found to be a strong predictor of mortality. 44 Improvements in both upper and lower body strength were also found in the current study, which is consistent with other exercise studies in breast cancer survivors that included resistance training.45 -47 Although direct comparisons are not possible because of different muscle strength assessment modes (eg, bench and leg press, grip strength) and study duration, it is notable that despite our relatively short (ie, 16 weeks) exercise program, compared with those that ranged from 12 to 24 months, significant strength increases occurred. With regard to functional assessment, no reports on the use of the SFMA in cancer populations were found. However, it appeared to be a useful test for participants in our study. For the majority of women who initially presented with movement dysfunction, most of which was upper body dysfunction, a significant benefit was achieved as assessed through the SFMA. This improvement may have been in part due to the resistance training and recommended stretches.
Results from the current study did not show significant improvements in fatigue, which is contrary to other reports from home-based exercise studies where significant positive effects were found in breast cancer survivors.48,49 While Pinto et al 48 assessed fatigue with the same scale used in our study, Musanti 49 used the Piper Fatigue Scale. Of note is that mean baseline and postintervention fatigue scores were higher in our study compared with those reported by Pinto et al, 48 which was conducted in a sample of Caucasian women. Fatigue scores in the current study increased slightly indicating less fatigue, but the sample size may have been too small to achieve significance. With regard to quality of life, significant improvements were reported on the FACT-B BCS, but not on the other subscales even though mean changes increased postintervention on all subscales. Our baseline and postintervention BCS mean scores were similar to those reported from primarily Caucasian breast cancer survivors in a home-based diet and exercise study. 50 Ottenbacher et al 50 found a mean BCS score at baseline of 25.2 and 26.3 at 1 year compared to mean scores from the present study (baseline M = 25.1 and postintervention M = 28.3). We also found a significant positive correlation between the BCS scores and physical activity levels on the IPAQ, which indicate that higher levels of physical activity are associated with less breast cancer concerns. Ottenbacher et al 50 did not find a correlation, but FACT-B scores have been correlated positively with physical activity in other studies. 9
Limitations and Strengths
The major limitations of the study are the small sample size and lack of a control group. However, the purpose of this pilot study was to determine whether the delivery of a home-based combined aerobic and resistance training exercise program could lead to increased physical activity and subsequent improvements in health and fitness prior to developing a larger randomized exercise intervention trial. A third limitation was that recruitment was based on self-selection, which may have led to sample bias. Additionally, the sample was not representative of the general population of African American breast cancer survivors because of the fact that all study participants had some level of college education. Efforts to strengthen recruitment of women with a broader range of education should be considered in future exercise studies of African American breast cancer survivors.
Strengths included the use of objective assessments for measuring most fitness/health outcomes. Objective measurements for testing cardiorespiratory function and body composition (DEXA scans) are considered gold standards in oncology settings. Muscle strength testing has typically been conducted in the oncology population with less sophisticated assessment tools than the Biodex system, which may provide higher precision. Additionally, physical activity data was collected through an accelerometer for the purpose of comparing subjective and objective data to determine whether correlations existed.
Conclusions
The 16-week motivational exercise pilot study provides preliminary evidence that a home-based progressive brisk walking and resistance training exercise program can increase physical activity and lead to improvements in both physical and psychological outcomes. Now that the pilot study is complete and our findings indicate the intervention was successful in increasing levels of physical activity, our next step is to develop a randomized clinical trial that will test the intervention in a larger sample. If similar results are found, we may have the ability to extend home-based motivational exercise programs to a larger number of African American breast cancer survivors. Many breast cancer survivors are unable to attend exercise programs offered through cancer centers, wellness centers or gyms due to a myriad of barriers such as cost, distance, family and/or job obligations, and so forth. A home-based exercise program with telephone counseling support would also be a cost-effective strategy to implement in health care facilities aiming to address health disparities among African American breast cancer survivors.
Footnotes
Acknowledgements
We would also like to acknowledge and thank Dr Derek Hales from the Gillings School of Global Public Health at the University of North Carolina at Chapel Hill for his assistance with Actical data assessment. A special thanks to the women who participated and contributed to this study.
Author’s Note
Sadly, one of our authors, Keith D. Amos, MD, is now deceased.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Health-e-NC, an initiative of the University Cancer Research Fund at the University of North Carolina at Chapel Hill. The first author was supported through a Cancer Health Disparities postdoctoral training grant (5T32CA128582-03) at the time the study was conducted. Dr Hales is supported through the Nutrition Obesity Research Center (grant number NIH DK56350).
