Abstract
Objective:
To evaluate the clinical efficacy and safety of Shuanghuang Yangxue Decoction in treating lung cancer patients with cancer-related anemia (CRA) accompanied by serum iron deficiency.
Method:
One hundred lung cancer patients with CRA and serum iron deficiency were included in this prospective randomized controlled study and randomly divided into control and treatment groups. The treatment group received Shuanghuang Yangxue Decoction plus ferrous succinate, while the control group received ferrous succinate alone. Related indicators were observed and recorded before and after treatment. Statistical analysis was performed using SPSS 26.0.
Results:
The effective correction rate of anemia in the treatment group was significantly higher than that in the control group (64 vs 34%, P < .01). Through stratified analysis of patients with moderate anemia, the effective correction rate in the treatment group was significantly higher than that in the control group (75 vs 45%, P < 0.01). After treatment, the treatment group showed statistically significant differences (P < .05) in peripheral blood routine parameters (hemoglobin, erythrocyte count, and hematocrit), anemia indicators (serum iron, ferritin, transferrin saturation, and erythropoietin [EPO]), immune function markers (cytotoxic T lymphocytes [Tc] and natural killer [NK] lymphocytes), and inflammatory factors (interleukin-1β [IL-1β], IL-6, tumor necrosis factor-α [TNF-α], and interferon-γ [IFN-γ]) compared to baseline values. Between-group comparisons showed that mean hemoglobin in the treatment group increased from 106.60 to 119.86 g/L, which was not significantly different from the control group increase from 110.00 to 116.18 g/L (P > .05). However, mean serum iron in the treatment group increased from 7.03 to 12.33 μmol/L, which was significantly higher than the control group increase from 7.33 to 9.45 μmol/L (P < .01). Throughout the study, no adverse reaction events occurred in either group.
Conclusion:
Shuanghuang Yangxue Decoction demonstrates superior efficacy compared to ferrous succinate alone in treating lung cancer CRA with serum iron deficiency. It effectively corrects anemia, increases serum iron levels, reduces inflammation, and is both safe and effective.

Keywords
Introduction
Cancer-related anemia (CRA) is one of the most common comorbid conditions in patients with malignant tumors. 1 It is frequently observed in individuals with advanced cancer, those with tumor recurrence or metastasis, and those undergoing anti-cancer treatment, 2 where its incidence is relatively high and its disease burden has attracted widespread attention worldwide. A prospective investigation of CRA in Europe found that approximately 67% of malignant tumor patients suffer from anemia. This condition is particularly prevalent in lung cancer, with the incidence of lung cancer CRA reaching approximately 77%. 3 However, only a few patients receive anemia treatment. 4 According to an epidemiological survey in China, the incidence in lung cancer reaches as high as 50.69%, whereas 92.84% of CRA patients have not received any treatment for their anemia. 5 Despite being a common complication of lung cancer, CRA has not received adequate attention, 6 which seriously affects patient prognosis. The severity of anemia correlates directly with poorer prognosis.7,8 Research data reveal that cancer patients with CRA have a 65% higher overall risk of death compared to those without CRA. 1
Iron deficiency is a common complication of malignant tumors and an important cause of anemia. In CRA, iron deficiency can be categorized as absolute or functional, 4 both of which are characterized by serum iron deficiency. Furthermore, absolute iron deficiency is manifested by reduced iron stores and is closely associated with malnutrition, digestive system surgery, and chronic blood loss. In contrast, functional iron deficiency is characterized by normal or elevated iron stores and decreased erythropoiesis, which is related to chronic inflammation 9 and is the most prevalent type of iron deficiency in cancer patients. 10 When iron metabolism and homeostasis are disrupted in patients with malignant tumors, the iron supply to red blood cells becomes inadequate, leading to anemia. 11 Moreover, when the body experiences iron deficiency, hemoglobin synthesis in red blood cells is reduced, leading to smaller red blood cells and decreased oxygen-carrying capacity, resulting in iron deficiency anemia. 12 In clinical practice, iron deficiency is usually assessed using 2 indicators: ferritin and transferrin saturation. Ferritin levels reflect the body’s ability to store iron, and the lower the ferritin level, the higher the possibility of iron deficiency anemia. Therefore, ferritin is currently considered to be closely related to iron deficiency in the body. However, under certain specific conditions, such as inflammation or malnutrition, serum ferritin may exhibit pseudo-elevation,13,14 which affects accurate diagnosis of iron deficiency and subsequent treatment selection. Serum iron is an important component in hemoglobin synthesis. Studies have shown that iron deficiency is related to serum iron deficiency levels and increased total iron-binding capacity (TIBC). 15 Additionally, decreased serum iron levels indicate a reduction in iron bound to transferrin in circulation. Therefore, although serum iron cannot independently evaluate the body’s iron storage and metabolism capacity, it reflects the concentration of iron in the body and iron levels during transportation, which is considered a good predictor for diagnosing iron deficiency. Thus, exploring the role of serum iron levels in CRA is significant.
Currently, treatment of CRA mainly includes blood transfusion, erythropoiesis-stimulating agents (ESAs), and iron supplementation, but the effects are not satisfactory and side effects may occur to varying degrees, making them suboptimal as first-line treatments. Traditional Chinese Medicine (TCM) has unique advantages in treating CRA, as it can not only correct anemia symptoms but also enhance anti-cancer efficacy and improve patients’ quality of life. Meta-analysis suggests that TCM is significantly superior to Western medicine alone in improving hemoglobin levels in patients with CRA. Renshen Yangrong Decoction originates from the classical medical text Treatise on the Three Causes of Diseases. The formula harmonizes qi, blood, and spirit, possessing functions of tonifying the spleen and kidneys, replenishing qi, generating blood, and calming the mind. Shuanghuang Yangxue Decoction is derived from Renshen Yangrong Tang, with modifications based on the dosage used in the original formula and the experience of renowned TCM physician Pu Fuzhou. 16 In this modification, Polygonati Rhizoma (Huangjing) is added to the original formula to nourish the kidneys and replenish essence, and the doses of Astragali Radix (Huangqi) and Paeoniae Alba Radix (Baishao) are increased to enhance its qi-tonifying and blood-nourishing effects. Positive therapeutic effects were obtained in clinical application of Shuanghuang Yangxue Decoction for improving lung cancer CRA. Therefore, it provides strong evidence for precise treatment of lung cancer CRA.
This study is a prospective randomized controlled trial to systematically evaluate the clinical efficacy of Shuanghuang Yangxue Decoction combined with ferrous succinate in treating lung cancer CRA patients with serum iron deficiency. The aim was to confirm the efficacy and safety of Shuanghuang Yangxue Decoction in treating lung cancer CRA patients with serum iron deficiency, reveal the synergistic mechanism of integrated Chinese and Western medicine, and provide a scientific basis for clinical application of Shuanghuang Yangxue Decoction.
Materials and Methods
Case Recruitment
Lung cancer CRA patients with serum iron deficiency who met the inclusion and exclusion criteria were recruited from inpatients and outpatients at Shanghai Fourth People’s Hospital affiliated with Tongji University and Yueyang Hospital of Integrated Chinese and Western Medicine affiliated with Shanghai University of Traditional Chinese Medicine.
Diagnostic Criteria
Western Medicine Diagnostic Criteria
(1) Patients had pathologically and/or cytologically confirmed lung cancer and met diagnostic criteria for anemia and serum iron deficiency.
(2) The anemia standards refer to the World Health Organization’s anemia grading standards in the Clinical Practice Guidelines for Cancer-related Anemia (2022 edition) by the Chinese Society of Clinical Oncology (CSCO). 17
① Grade 0 (normal): Hemoglobin (Hb) levels should not be lower than 130 g/L for adult men, 120 g/L for non-pregnant adult women, and 110 g/L for pregnant adult women;
② Grade 1 (mild): 110 g/L to normal value;
③ Grade 2 (moderate): 80 to 109 g/L;
④ Grade 3 (severe): <80 g/L.
(3) Diagnostic criteria for serum iron deficiency: According to the Criteria for Diagnosis and Efficacy of Blood Diseases, serum iron <8.95 μmol/L is the standard for serum iron deficiency. 18
TCM Diagnostic Criteria
According to the tenth edition of TCM diagnostics published by China Traditional Chinese Medicine Publishing Company (the textbook for the 13th Five-Year Plan) 19 and TCM syndrome diagnosis criteria of the Guiding Principles for Clinical Research of New Chinese Medicine, 20 the diagnostic criteria for qi and blood deficiency syndrome were formulated:
Primary symptoms: pale or sallow complexion, dizziness and blurred vision, palpitation, shortness of breath, fatigue.
Secondary symptoms: spontaneous sweating, reluctance to speak, insomnia, numbness of hands and feet, delayed menstruation, scanty and pale menstrual flow, or even amenorrhea.
Tongue and pulse: pale or slightly swollen tongue, weak or deficient pulse.
Two senior physicians at or above the attending level made separate dialectical analyses and unified the dialectical results. If there was any disagreement, another physician with a senior professional title made dialectical confirmation. When 1 primary symptom and 2 secondary symptoms were present, combined with tongue and pulse examination, a diagnosis of qi and blood deficiency syndrome could be made.
Inclusion Criteria
(1) Patients with lung cancer confirmed by pathology or cytology and meeting the above diagnostic criteria for mild to moderate anemia (male: 80-130 g/L, female: 80-120 g/L);
(2) Participants meeting the above criteria for serum iron deficiency (serum iron < 8.95 μmol/L);
(3) Patients who had not received radiotherapy or chemotherapy previously, or who had completed radiotherapy or chemotherapy for more than 1 month;
(4) Patients whose TCM syndrome classification corresponded to the qi and blood deficiency type;
(5) Patients who had not been treated with iron, EPO, blood transfusion, qi-nourishing and blood-nourishing prescriptions, or proprietary Chinese medicine 1 month before admission;
(6) Karnofsky Performance Status (KPS) score ≥ 60;
(7) Age ≥ 18 years and ≤80 years, with no gender restrictions;
Exclusion Criteria I
(1) Patients with acute hemorrhage or hemolysis;
(2) Individuals who had participated or were participating in other drug-related clinical trials within the last month;
(3) Patients allergic to the investigational drug;
(4) Those with severe dysfunction of the heart, liver, kidneys, or other vital organs;
(5) Pregnant and lactating women;
(6) Those expected to receive anti-tumor therapy such as radiotherapy, chemotherapy, and targeted therapy during the treatment period that would affect efficacy evaluation;
(7) Patients using other proprietary Chinese medicines with qi-nourishing and blood-nourishing effects during the treatment period.
Exclusion Criteria II
(1) Patients with poor compliance who were unable to continue cooperating with the treatment plan of this study;
(2) Individuals who did not adhere to prescribed medication during treatment, those whose clinical efficacy could not be evaluated, or those with incomplete information that might impact safety assessment of therapeutic effect.
Dropout Criteria
(1) Patients who experienced serious adverse reactions and were unable to tolerate the treatment;
(2) Patients who voluntarily withdrew from the study;
(3) Participants who discontinued naturally or were lost to follow-up.
Criteria for Treatment Termination
(1) Patients who voluntarily revoked their original consent or refused further treatment (reasons should be recorded in the clinical observation sheet);
(2) Individuals who experienced disease progression or deterioration leading to death;
(3) Patients who could not comply with the protocol in the project plan;
(4) When management decisions were made by the project leader.
Research Design
Research Methods
This study was designed, conducted, and reported strictly in accordance with the CONSORT statement. The study was a prospective, randomized, controlled clinical trial.
Ethical Review and Clinical Trial Registration
This study was approved by the Ethics Committee (Numbers: 2023-077 and 2025026-001) and completed registration with the China Clinical Trial Center (Number: ChiCTR2300077699).
Sample Size Calculation
By referring to relevant literature, 21 it was estimated that the effective rate of the treatment group was 87.5% and that of the control group was 62.5%. The sample size calculation formula was adopted:
Cases in the treatment and control groups were allocated in a 1:1 ratio. A bilateral test was conducted, with standards set at α = .05 and β = .20. Therefore, according to the above formula, the minimum sample size per group was 44 cases. Accounting for a 20% dropout rate, the minimum sample size of each group was adjusted to 50 cases, totaling 100 cases for both groups.
Treatment Plan
Grouping Method
This study strictly adhered to the CONSORT statement, and a completely randomized design method was applied for group assignment. Participants were randomly assigned to either the treatment or control group in a 1:1 ratio, with 50 individuals in each group. The randomization sequence was generated using a computer-generated random number list by an independent statistician. To ensure allocation concealment, the group assignments were placed in sequentially numbered, opaque, sealed envelopes. These envelopes were prepared and kept by an research assistant who had no contact with potential participants. Upon enrollment of an eligible participant, the assigned research staff opened the next sequentially numbered envelope to reveal the group assignment. Written informed consent was obtained from all participants prior to their enrollment in the study.
Treatment Protocol
(1) Control group: basic treatment
(2) Treatment group: basic treatment + traditional Chinese medicine treatment
Basic treatment: According to the Clinical Practice Guidelines for Cancer-related Anemia (2022 edition) of the Chinese Society of Clinical Oncology (CSCO), oral iron supplementation was given: ferrous succinate (specification: 0.2 g/tablet, 24 tablets/box), manufacturer: Nanjing Jinling Pharmaceutical Co., Ltd., 1 tablet per time, once daily, for 28 days.
Chinese medicine treatment: Shuanghuang Yangxue Decoction is an empirical formula developed in our department, derived by modifying Renshen Yangrong Decoction with the addition of Polygonati Rhizoma (Huangjing). Traditional Chinese medicine treatment was administered using Shuanghuang Yangxue Decoction. The prescription was prepared by Shanghai Tongjitang Pharmaceutical Co., Ltd., under the supervision of our hospital’s preparation department. Quality control was performed using high-performance liquid chromatography-mass spectrometry fingerprinting.
Decoction method: The traditional Chinese medicine was placed in a container and soaked in cold water for 30 minutes. For the first decoction, the soaked traditional Chinese medicine was poured into a clay pot, 400 mL of water (including the water used for soaking the herbs) was added to the pot, brought to a boil over high heat, then reduced to low heat and continued to simmer for 30 minutes. The liquid was poured out and the residue filtered. For the second decoction, the medicinal residue was poured into a clay pot, 300 mL of water was added to the pot, brought to a boil over high heat, then reduced to low heat and continued to simmer for 20 minutes. The medicinal liquid was poured out. The medicinal liquids from the 2 decoctions were mixed evenly. Doses were taken warm half an hour after breakfast and dinner, 200 mL each time, for 28 days. (All decoction pieces of Shuanghuang Yangxue Decoction in this study were purchased from regular suppliers that met national Good Manufacturing Practice for drugs and underwent strict quality inspections. The preparation of Shuanghuang Yangxue Decoction conformed to national drug standards and provincial processing norms.)
The composition of Shuanghuang Yangxue Decoction is as follows:
Polygonati Rhizoma (Huangjing) 30 g, Astragali Radix (Huangqi) 30 g, Angelicae Sinensis Radix (Danggui) 9 g, Cinnamomi Cortex (Rougui) 6 g, Glycyrrhizae Radix Et Rhizoma Praeparata Cum Melle (Zhigancao) 9 g, Citri Reticulatae Pericarpium (Chenpi) 9 g, Atractylodis Macrocephalae Rhizoma (Chaobaizhu) 9 g, Ginseng Radix Et Rhizoma (Renshen) 6 g, Paeoniae Alba Radix (Chaobaishao) 18 g, Rehmanniae Radix Praeparata (Shudihuang) 12 g, Schisandrae Chinensis Fructus (Wuweizi) 9 g, Poria (Baifuling) 12 g, Polygalae Radix (Yuanzhi) 9 g, Zingiberis Rhizoma Recens (Shengjiang) 3 g, Jujubae Fructus (Dazao) 6 g.
Quality Control
(1) Patients were fully informed of the significance and process of this experiment to improve patient compliance, and instructions on how to fill in the record form were provided.
(2) Uniform training was provided for researchers participating in the trial, focusing on informed consent methods and evaluation criteria.
(3) At least 2 people were assigned to evaluate various indicators each time.
Observation Indexes and Efficacy Evaluation
Clinical Efficacy Observation Index
(1) Primary outcome indicators: the effective rate of anemia correction
(2) Secondary outcome indicators: peripheral blood routine, anemia indicators, peripheral blood immune indicators, inflammatory factors, nutritional status, TCM syndrome score, Functional Assessment of Cancer Therapy-Anemia (FACT-An) score scale.
(3) Observation period: Drug administration was followed up once weekly, 1 day before the experiment and once after the end of the experiment at 28 days. Moreover, relevant data were recorded.
(4) Safety indicators: patients’ basic vital signs (body temperature, heart rate, blood pressure, respiration), blood routine, liver and kidney function, and 12-lead electrocardiogram were evaluated. Follow-up assessments were conducted 1 day before the experiment and 28 days after the experiment, with relevant data recorded.
Evaluation Criteria of Therapeutic Effect
(1) The effective rate of anemia correction: refer to the Criteria for Diagnosis and Efficacy of Blood Diseases and related studies.18,22
Markedly effective: Clinical symptoms have significantly improved, and Hb level after treatment has increased by more than 20 g/L or Hb has returned to normal value.
Effective: Clinical symptoms have lessened, and Hb increase after treatment is ≤20 g/L but greater than 10 g/L.
Ineffective: Clinical symptoms did not improve notably compared to before, with Hb rise ≤10 g/L, no change in Hb levels, or even a decline.
Total effective rate = markedly effective + effective
(2) TCM syndrome score: Patients’ primary symptoms and secondary symptoms were scored according to the Guiding Principles for Clinical Research of New Chinese Medicine (trial) 20 symptom scoring standard. Primary symptoms included pale or yellowish complexion, dizziness, palpitations, shortness of breath, and fatigue. Secondary symptoms included spontaneous sweating, reluctance to speak, insomnia, numbness of hands and feet, delayed menstruation, pale menstruation, and even amenorrhea. Based on the degree of severity—normal, mild, moderate, and severe—primary symptoms were scored 0, 2, 4, and 6 points, respectively, while secondary symptoms were scored 0, 1, 2, and 3 points accordingly. Efficacy was evaluated according to syndrome scores of patients before and after treatment, as follows:
Markedly effective: symptoms and signs after medication were significantly improved compared with those before medication, and syndrome score was reduced by ≥70%;
Effective: symptoms and signs after medication showed improvement compared to before medication, with syndrome score reduction of ≥30%;
Ineffective: Post-medication symptoms and signs exhibited no notable improvement or even worsened compared to the pre-medication state, with reduction in syndrome score of less than 30%.
Note: The Nimodipine method formula was applied to calculate: [(pre-treatment score − post-treatment score)/pre-treatment score] × 100%.
Total effective rate = markedly effective + effective
(3) FACT-An score scale 23 : The Functional Assessment of Cancer Therapy-Anemia scale (FACT-An) is a core scale developed by Northwestern University to evaluate quality of life in patients with cancer-related anemia. The FACT-An scale, which incorporates the FACT-G as its core component, consists of 27 items distributed across 4 domains: 7 items in the physical well-being domain, 7 items in the social/family well-being domain, 6 items in the emotional well-being domain, and 7 items in the functional well-being domain. GP1-GP7, GE1, and GE3-GE6 are reverse-scored items. Based on the FACT-G scale, the FACT-An scale added another 20 anemia-specific module scales (negative), including 13 items related to fatigue and 7 items related to non-fatigue symptoms. The FACT-An scale employs a five-level scoring system: “Not at all,” “A little bit,” “Somewhat,” “Quite a bit,” and “Very much.” Positive items are scored directly as 0, 1, 2, 3, 4 points, and reverse items are scored as 4, 3, 2, 1, 0 points. The score of each domain item is added to the total score of the domain, and the total score of each domain is summed to the total score of the scale. The higher the score, the better the quality of life.
Statistical Analysis
Statistical experts conducted statistical analysis of the study results. All clinical data collected were entered into a database for comprehensive data entry. Data analysis followed the principle of intention-to-treat (ITT), and patients who failed to complete the study were regarded as having no change from baseline. All data in this experiment were analyzed using SPSS 26.0 software. The chi-square test was used for categorical data, and the t-test was applied for continuous data. Additionally, non-normally distributed continuous data and ordinal data were analyzed using the rank-sum test. In this study, a P-value of less than .05 was considered statistically significant.
Results
Demographic Characteristics
A total of 100 patients with lung cancer CRA accompanied by serum iron deficiency were included in this study (see Figure 1). In the treatment group, 50 patients consisted of 35 males and 15 females, with a mean age of 66.46 ± 9.22 years. In the control group, 50 patients consisted of 34 males and 16 females, with a mean age of 68.54 ± 7.38 years. There was no significant difference in gender, age, pathological type, stage, surgery and chemotherapy (in the past) between the 2 groups (P > .05), indicating that the 2 groups were comparable, as shown in Table 1.

Flow diagram.
Demographic Characteristics.

Effective Rate of Anemia Correction
Clinical effect was evaluated by referring to the effective rate of anemia correction in the Criteria for Diagnosis and Efficacy of Blood Diseases. 18 After treatment, the total effective rate in the treatment group was 64%, compared to 34% in the control group. The difference was statistically significant (P < 0.01), suggesting that the treatment group demonstrated superior clinical effect compared to the control group. According to the World Health Organization’s anemia grading standard in the Clinical Practice Guidelines for Cancer-Related anemia (2022 edition) of the Chinese Society of Clinical Oncology (CSCO), anemia patients were stratified and analyzed. After treatment, the total effective rate of patients with moderate anemia in the treatment group was 75%, while in the control group it was 45%. After statistical analysis, the difference was found to be statistically significant (P < .05), demonstrating that clinical efficacy for patients with moderate anemia in the treatment group was superior to that in the control group, as shown in Table 2.
The Effective Rate of Anemia Correction.

Compared with the control group P < .05. **Compared with the control group P < .01.
Comparison of Peripheral Blood Routine
After treatment, significant differences were found in hemoglobin, red blood cell count, and hematocrit levels in the treatment group (P < .01), while differences in C-reactive protein (CRP) and albumin levels were not statistically significant (P > .05). In the control group, only hemoglobin level showed a statistically significant difference (P < .01), but no significant differences were observed in red blood cell count, hematocrit, CRP, or albumin levels (P > .05). When comparing the treatment group and control group after treatment, no statistically significant differences were found in hemoglobin, red blood cell count, hematocrit, CRP, or albumin levels (P > .05), as shown in Table 3.
Peripheral Blood Parameters Before and After Treatment (x ± s).

Compared with before treatment P < .05. **Compared with before treatment P < .01. #Compared with control group P < .05. ##Compared with control group P < .01.
Comparison of Anemia Indexes
Following treatment, the treatment group showed statistically significant differences in serum iron, ferritin, transferrin saturation, and EPO levels (P < .01), while the difference in TIBC was not statistically significant (P > .05). In the control group, there were statistically significant differences in serum iron and transferrin saturation (P < .01), but no significant differences were observed in ferritin, total iron-binding capacity, and EPO (P > .05). Interestingly, there was significant heterogeneity in the ferritin results. The effect value of ferritin before and after treatment in the treatment group was −3.731, and that in the control group was −1.03. The larger the absolute value of the effect value, the stronger the effect and the more obvious the phenomenon. Although heterogeneity was relatively large here, it still showed differences between the 2 groups before and after treatment. Moreover, comparisons between the treatment and control groups after treatment revealed statistically significant differences in serum iron and transferrin saturation (P < .01), while there were no statistically significant differences in ferritin, TIBC, and EPO levels (P > .05), as shown in Table 4.
Anemia Indexes Before and After Treatment (x ± s).
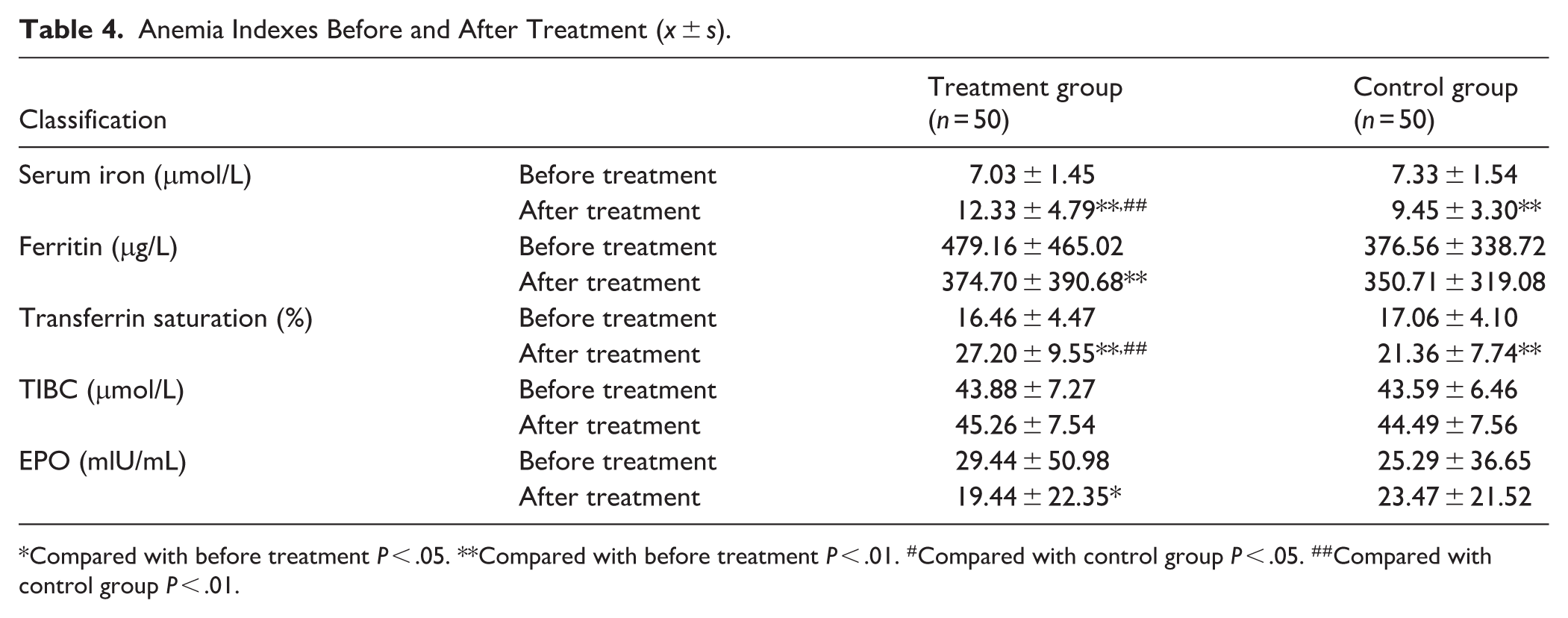
Compared with before treatment P < .05. **Compared with before treatment P < .01. #Compared with control group P < .05. ##Compared with control group P < .01.
Comparison of Immune Function
Post-treatment, the treatment group exhibited statistically significant differences in Tc lymphocytes and NK lymphocytes (P < .05), while differences in T lymphocytes, helper T (Th) lymphocytes, CD4+/CD8+ cells, and B lymphocytes were not statistically significant (P > .05). In the control group, no statistically significant differences were observed in T lymphocytes, Th lymphocytes, Tc lymphocytes, CD4+/CD8+ cells, B lymphocytes, and NK lymphocytes (P > .05). After treatment, comparisons between the treatment and control groups showed no statistically significant differences in T lymphocytes, Th lymphocytes, Tc lymphocytes, CD4+/CD8+ cells, B lymphocytes, and NK cells (P > .05), as shown in Table 5.
Immune Function Parameters Before and After Treatment (x ± s).

Compared with before treatment P < .05. **Compared with before treatment P < .01. #Compared with control group P < .05. ##Compared with control group P < .01.
Comparison of Inflammatory Factors
Following treatment, the treatment group indicated statistically significant differences in IL-1β, IL-6, TNF-α, and IFN-γ (P < .05). In the control group, a statistically significant difference was detected only in IFN-γ (P < .05), while IL-1β, IL-6, and TNF-α did not display significant differences (P > .05). Comparisons between the treatment and control groups post-treatment revealed no statistically significant differences in IL-1β, IL-6, TNF-α, and IFN-γ (P > .05), as shown in Table 6.
Inflammatory Factors Parameters Before and After Treatment (x ± s).
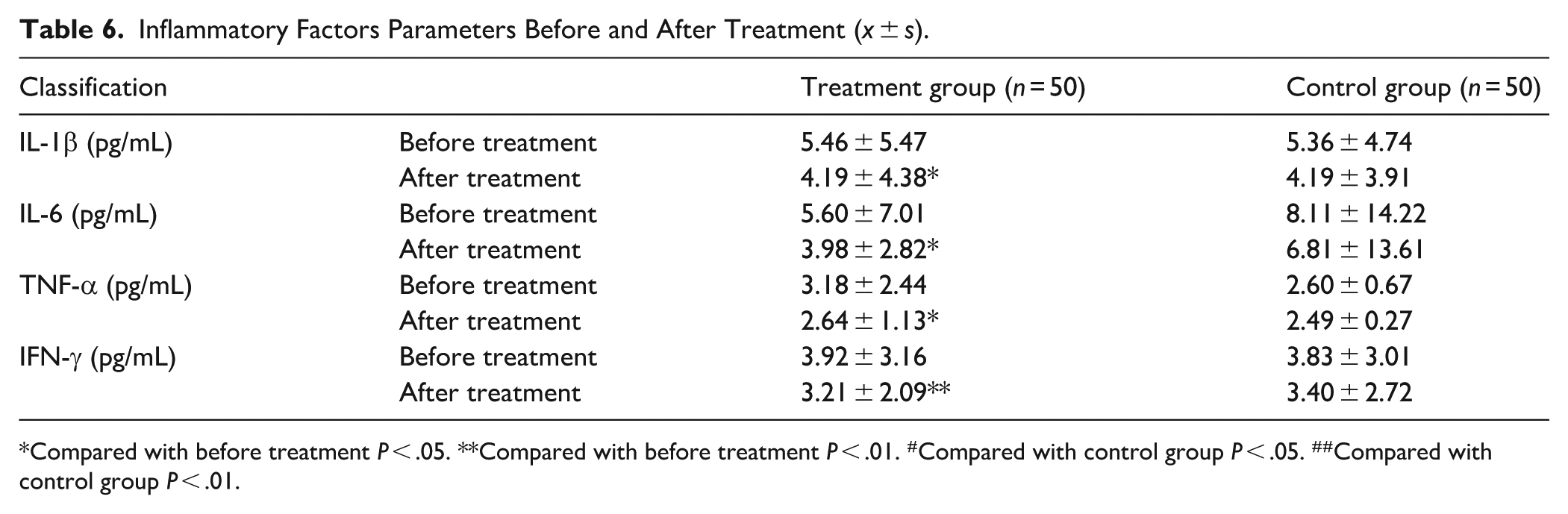
Compared with before treatment P < .05. **Compared with before treatment P < .01. #Compared with control group P < .05. ##Compared with control group P < .01.
Comparison of TCM Syndrome Scores
(1) Comparison of TCM syndrome efficacy: After treatment, the total effective rate of the treatment group was 62% and that of the control group was 34%. Furthermore, the difference was statistically significant through statistical analysis (P < .05), indicating that the efficacy of TCM syndromes in the treatment group was superior to that in the control group, as shown in Table 7.
The Curative Effects on TCM Syndromes.

Compared with control group P < .05. ##Compared with control group P < .01.
(2) Overall comparison of TCM syndrome total scores and individual symptoms: After treatment, there were statistically significant differences in the total score of TCM syndrome and individual symptoms such as pale or sallow complexion, dizziness, palpitation, shortness of breath, fatigue, spontaneous sweating, reluctance to speak, insomnia, and numbness of hands and feet in the treatment group (P < .01). In contrast, the control group did not show statistically significant differences in total TCM syndrome scores or individual symptoms (P > .05). Additionally, comparisons between the treatment and control groups revealed statistically significant differences in total TCM syndrome scores and symptoms, including pale or sallow complexion, spontaneous sweating, reluctance to speak, and numbness of hands and feet (P < .01), as shown in Table 8.
TCM Syndrome Scores and Individual Symptoms Before and After Treatment (x ± s).

Compared with before treatment P < .05. **Compared with before treatment P < .01. #Compared with control group P < .05. ##Compared with control group P < .01.
Comparison of FACT-An Scale Scores
After treatment, the total score of the FACT-An scale and subscale domains, including physical well-being, social/family well-being, emotional well-being, functional well-being, and anemia subscale in the treatment group showed significant differences (P < .01). However, there was no significant difference in the total FACT-An scale score and 5 subscale domains in the control group (P > .05). In addition, statistically significant differences were noted between the treatment group and the control group in FACT-An total score, physical well-being, social/family well-being, emotional well-being, and anemia subscale after treatment (P < .05), as shown in Table 9.
FACT-An Scale Scores Before and After Treatment (x ± s).

Compared with before treatment P < .05. **Compared with before treatment P < .01. #Compared with control group P < .05. ##Compared with control group P < .01.
Safety Evaluation
Throughout the study, there were no abnormalities in vital signs, blood routine, liver and kidney function, or electrocardiogram in both groups, and no adverse events were observed, indicating that Shuanghuang Yangxue Decoction has high oral safety.
Discussion
CRA is a common complication of malignant tumors and is usually induced by various factors, such as tumor invasion of bone marrow, resulting in impaired hematopoietic microenvironment and hematopoietic dysfunction of bone marrow, thus inhibiting red blood cell production and leading to anemia.24,25 Currently, it is widely believed that the formation mechanism of CRA is closely related to inflammation, malnutrition, kidney dysfunction, iron metabolism disorders, and bone marrow hematopoietic abnormalities. 17 The main symptoms are fatigue, pallor, dizziness, and abdominal distension, and even some manifestations of tumor-related fatigue, which gradually develop into chronic diseases, 26 seriously affecting patients’ quality of life and the effectiveness of anti-cancer therapy.
The immune function of CRA patients is compromised because the body secretes reactive oxygen species (ROS) which inhibit the immune response mediated by CD8+ T cells, resulting in decreased immune function in CRA patients. The underlying mechanism is that erythroid differentiation of CRA patients is blocked, resulting in production of CD45+ erythroid progenitor cells (CD45+ EPCs). Furthermore, CD45+ EPCs are closely related to myeloid-derived suppressor cells (MDSCs), which are induced to differentiate into MDSC-like cells that possess immunosuppressive functions stronger than those of T cells and MDSCs, thereby further inhibiting the body’s immune function. 27 Additionally, CRA can cause various related symptoms, 28 which significantly affects the efficacy of anti-tumor therapy and patients’ quality of life. Therefore, anemia has also become a major factor in determining poor prognosis in cancer patients. 11
In traditional Chinese medicine, lung cancer CRA is classified under categories such as “blood depletion” and “consumptive disease,” with a predominant characteristic of qi and blood deficiency. The disease is primarily located in the lung, spleen, and kidney, and is closely related to the liver. In lung cancer patients, prolonged illness leads to deficiency in the organs, or depletion of vital energy due to chemotherapy and radiotherapy, resulting in impaired spleen and stomach function, which disrupts the production and transformation of qi and blood. This deficiency, combined with common pathological factors in lung cancer such as qi stagnation, blood stasis, phlegm retention, dampness accumulation, and heat toxins, further impairs the liver’s function of storing blood. As a result, blood storage is reduced, and chronic illness depletes the kidneys, leading to insufficient kidney essence and inability to generate blood. The liver’s blood deficiency, coupled with essence depletion, under the combined influence of “cancer” and “toxins,” forms a key pathogenesis characterized by predominant “deficiency,” with elements of “toxicity” and “stasis.” 29 Traditional Chinese medicine has unique advantages in treating CRA, as it can not only correct anemia symptoms but also improve anti-tumor efficacy and patients’ quality of life. Currently, treatment of CRA mainly involves invigorating the spleen and kidneys, tonifying qi, and nourishing blood. Shuanghuang Yangxue Decoction is composed of Polygonati Rhizoma (Polygonatum sibiricum Redouté) 30 g, Astragali Radix (Astragalus membranaceus (Fisch.) Bunge) 30 g, Angelicae Sinensis Radix (Angelica sinensis (Oliv.) Diels) 9 g, Cinnamomi Cortex (Cinnamomum cassia (L.) D. Don) 6 g, Glycyrrhizae Radix Et Rhizoma Praeparata Cum Melle (Glycyrrhiza spp.) 9 g, Citri Reticulatae Pericarpium (Citrus reticulata Blanco) 9 g, Atractylodis Macrocephalae Rhizoma (Atractylodes macrocephala Koidz.) 9 g, Ginseng Radix Et Rhizoma (Panax ginseng C. A. Mey.) 6 g, Paeoniae Alba Radix (Paeonia lactiflora Pall.) 18 g, Rehmanniae Radix Praeparata (Rehmannia glutinosa (Gaertn.) Libosch. ex Fisch. & C. A. Mey.) 12 g, Schisandrae Chinensis Fructus (Schisandra chinensis (Turcz.) Baill.) 9 g, Poria (Poria cocos (Fr.) Wolf) 12 g, Polygalae Radix (Polygala tenuifolia Willd) 9 g, Zingiberis Rhizoma Recens (Zingiber officinale Roscoe) 3 g, and Jujubae Fructus (Ziziphus jujuba Mill.) 6 g. The formula regulates the liver, spleen, and kidneys, achieving the effects of invigorating the spleen and kidneys, replenishing qi and blood. This recipe emphasizes a balance of acid and sweetness. Acid is absorbed into Yin, while sweetness supplements Yang. Meanwhile, the combination of acidic and sweet helps generate Yin, with the production of Yin fluids continuously supported by the movement of Yang qi. 30 The combined use of acid and sweetness can not only replenish the essential substances needed for blood production in cases of blood deficiency but also promote the transformation of kidney essence into Yin blood through the action of acidic herbs that nourish the liver and kidneys. 31 In this prescription, Polygonati Rhizoma (Huangjing) and Astragali Radix (Huangqi) serve as the principal herbs. Polygonati Rhizoma tonifies the kidneys by replenishing essence, focusing on consolidating and strengthening vitality. Essence and blood share a common origin: the liver stores blood, and kidney essence relies on liver blood for supplementation, while liver blood depends on kidney essence for nourishment. The 2 complement each other, resulting in abundant qi and blood. Astragali Radix excels at replenishing and tonifying qi and blood. When combined with Polygonati Rhizoma, the 2 herbs not only strengthen the kidneys and replenish essence but also enhance their ability to boost qi and nourish blood. Angelicae Sinensis Radix (Danggui), Rehmanniae Radix Praeparata (Shudihuang), Paeoniae Alba Radix (Baishao), Schisandrae Chinensis Fructus (Wuweizi), Ginseng Radix Et Rhizoma (Renshen), Atractylodis Macrocephalae Rhizoma (Baizhu), Poria (Baifuling), and Citri Reticulatae Pericarpium (Chenpi) serve as subordinate herbs. The combination of Angelicae Sinensis Radix and Rehmanniae Radix Praeparata nourishes Yin and blood, directly replenishing deficient Yin-blood. Paeoniae Alba Radix softens the liver and nourishes blood, while Schisandrae Chinensis Fructus replenishes qi, generates fluids, nourishes the kidneys, and calms the heart. Together, they work to astringe and stabilize the pulse, harmonizing Yin and securing the nutritive qi. Ginseng Radix Et Rhizoma greatly replenishes vital qi, while Atractylodis Macrocephalae Rhizoma strengthens the spleen and boosts qi. Both herbs primarily enter the spleen meridian, tonifying qi and invigorating the spleen. Combined with the principal herbs, they enhance the overall function of tonifying qi and strengthening the spleen. The spleen and stomach are considered the foundation of the acquired constitution. The middle burner (spleen and stomach) receives and processes food and water, extracting the essence from grains and fluids, which then enters the vessels to form blood. Citri Reticulatae Pericarpium regulates qi and strengthens the spleen, while Poria drains dampness through its bland properties. When used together, these herbs support the middle burner’s function, promoting blood production from the essence of food and drink. Angelicae Sinensis Radix, Rehmanniae Radix Praeparata, Paeoniae Alba Radix, and Schisandrae Chinensis Fructus nourish Yin and blood. Ginseng Radix Et Rhizoma, Atractylodis Macrocephalae Rhizoma, Poria, and Citri Reticulatae Pericarpium replenish qi. These herbs work synergistically with the principal herbs to enhance the effect of replenishing qi. By replenishing qi in cases of blood deficiency, the principle of “nourishing yang to promote the growth of Yin” is applied. Cinnamomi Cortex (Rougui) and Polygalae Radix (Yuanzhi) serve as assistant herbs in the formula. Cinnamomi Cortex replenishes fire and supports Yang, guiding the fire back to its source, while Polygalae Radix calms the mind and enhances cognitive function. Zingiberis Rhizoma Recens (Shengjiang) and Jujubae Fructus (Dazao) harmonize the nutritive and defensive qi, and Glycyrrhizae Radix Et Rhizoma Praeparata Cum Melle (Zhigancao) tonifies the middle qi and harmonizes the actions of all herbs in the formula, making them the envoy herbs. In the formula, Ginseng Radix Et Rhizoma, Astragali Radix, and Schisandrae Chinensis Fructus supplement the lung, while Paeoniae Alba Radix and Angelicae Sinensis Radix nourish the liver. Citri Reticulatae Pericarpium, Glycyrrhizae Radix Et Rhizoma Praeparata Cum Melle, Poria, and Atractylodis Macrocephalae Rhizoma strengthen the spleen. Rehmanniae Radix Praeparata and Polygonati Rhizoma tonify the kidney, while Polygalae Radix helps to connect the heart and kidney. Cinnamomi Cortex guides the herbs into the nutritive qi to generate blood. With the 5 organs nourishing and supporting each other, the formula effectively replenishes qi and nourishes blood.
The results of this study found that after treatment, Shuanghuang Yangxue Decoction can remarkably correct anemia in patients compared with iron treatment alone, and the therapeutic effect is significant, which is consistent with previous studies.22,32 Through stratified analysis of anemia patients, the results found that the efficacy of traditional Chinese medicine on moderate anemia patients was more significant.
Serum iron is an essential trace element for the human body and a primary raw material for hemoglobin synthesis. It typically maintains dynamic balance within the body through the ferroptosis program and hepcidin regulation pathways 33 and also exerts its function by binding to transferrin.27,34 A decrease in serum iron level corresponds to a reduction in iron bound to transferrin in circulation. The primary reasons for lower circulating iron levels include retention of iron in macrophages of the reticuloendothelial system35,36 and reduced absorption of iron by the intestinal mucosa. Therefore, although serum iron cannot independently evaluate the storage and metabolism of iron in the body, it does reflect the concentration of iron in the body and iron levels during transport. The results of this study showed that serum iron levels in both groups increased significantly after treatment, and the therapeutic effect of the treatment group was markedly better than that of the control group, indicating that Shuanghuang Yangxue Decoction can significantly enhance iron concentration and iron levels during transport. Serum iron reflects the iron concentration in the body and iron levels during transport, while ferritin reflects the body’s iron storage. In this study, it was found that Shuanghuang Yangxue Decoction not only failed to improve ferritin levels but decreased them, which might be due to the fact that the body was either in or approaching an inflammatory state or malnutrition, leading to a false elevation of ferritin levels. In reality, the ferritin levels were likely lower, and the post-treatment ferritin levels should have been higher compared to pre-treatment, meaning that the current ferritin levels represent an elevated state following treatment. Furthermore, under inflammatory conditions, inflammatory factors promote ferritin synthesis by liver cells, resulting in increased ferritin levels, which indirectly explains the pseudo-increase in ferritin in this study. Therefore, it is suggested that Shuanghuang Yangxue Decoction can increase the content of serum iron and ferritin (when not in a state of inflammation and malnutrition), indicating that the decoction can significantly improve iron storage in the body. Researchers have measured the elemental content of 40 traditional Chinese medicines using atomic absorption spectroscopy and found that many herbs, such as Angelicae Sinensis Radix, Rehmanniae Radix Praeparata, Schisandrae Chinensis Fructus, Astragali Radix, Polygonati Rhizoma, Chuanxiong Rhizoma (Chuanxiong), Atractylodis Macrocephalae Rhizoma, Poria, Glycyrrhizae Radix, and other Chinese herbs have relatively high iron content. Schisandrae Chinensis Fructus had the highest iron content at 664 μg/g, followed by Angelicae Sinensis Radix with 500 μg/g, Rehmanniae Radix Praeparata with 438 μg/g, and both Polygonati Rhizoma and Astragali Radix around 300 μg/g. Therefore, these herbs are commonly used in clinical practice for treating anemia. The treatment of anemia through these herbs is consistent with the rationale for modern medicine using iron supplements to treat anemia and blood loss. This concept is exemplified in Wang Qingren’s use of Astragali Radix in large doses within Shaofu Zhuyu Decoction to treat gynecological blood disorders. 37 These Chinese medicines with high iron content are also important components of Shuanghuang Yangxue Decoction in this study. Through treatment of lung CRA with serum iron deficiency, the study found that Shuanghuang Yangxue Decoction significantly increased serum iron levels. This effect is closely related to the high iron content of the individual herbs in the formula. Total iron-binding capacity refers to the total amount of iron that can bind to transferrin in serum, reflecting the body’s transferrin levels. Transferrin saturation is the ratio of serum iron to TIBC, expressed as a percentage, indicating the proportion of transferrin that is bound to iron. This study found that Shuanghuang Yangxue Decoction can slightly raise TIBC levels, though the effect was not obvious. However, the decoction significantly improved transferrin saturation compared to the control group, indicating that Shuanghuang Yangxue Decoction can effectively enhance iron metabolism in the body.
Inflammatory factors are involved in the development of CRA, primarily by promoting the secretion of pro-inflammatory cytokines, such as IL-1, IL-6, IFN, TNF, which can not only inhibit erythropoiesis but also promote apoptosis of red blood cells, resulting in inflammatory anemia or chronic anemia.33,34 At the same time, the release of inflammatory cytokines is related to the generation of reactive oxygen species, which further inhibit erythropoiesis and interfere with nutritional status, leading to reduction in essential materials for red blood cell production, such as iron, vitamin B12, and folic acid, thereby worsening anemia. 38 Besides, ROS can suppress CD8+ T-cell-mediated immune responses, which further contributes to the development of anemia. 27 In addition, inflammatory cytokines can stimulate the liver to synthesize hepcidin, 39 which can bind to iron transport-related proteins on the surface of duodenal cells, weakening their ability to export iron. This regulates iron transport through the intestinal mucosa, inhibits iron absorption, and restricts the release of intracellular iron, leading to iron metabolism disorders and eventually resulting in anemia.35,36,40 Therefore, it is of great significance to actively explore changes in inflammatory cytokines in this study. By comparing levels of inflammatory factors in the 2 groups, it was found that levels of inflammatory factors in the treatment group were significantly reduced compared to before treatment. This indicates that Shuanghuang Yangxue Decoction can effectively reduce inflammatory cytokine levels and inflammatory responses in patients with lung CRA. Thus, it can be inferred that the decoction may improve anemia in these patients by reducing inflammation. In addition, CRP is considered one of the most valuable indicators for evaluating the inflammatory state of cancer patients, 41 with elevated levels reflecting the body’s inflammatory status. Iron deficiency in lung CRA is mainly manifested by decreased serum iron levels, which is associated with reduced iron storage and metabolism disorders. The reduction in iron storage is closely related to malnutrition, gastrointestinal surgeries, and chronic blood loss, while iron metabolism disorders are largely linked to chronic inflammation within the body. 9 This study focused on lung cancer CRA with serum iron deficiency as the main research subject, exploring the relationship between inflammation response, represented by CRP, and lung cancer CRA with serum iron deficiency.
The results showed that CRP levels decreased in both groups after treatment, but there was no significant difference in therapeutic effect. CRP is an acute-phase protein produced during inflammatory states and serves as a nonspecific marker of inflammation. Previous studies have shown that in lung cancer patients, CRP levels positively correlate with tumor stage and negatively correlate with serum iron levels. When the body is in an inflammatory state, CRP stimulates ferritin synthesis, which binds to intracellular iron, thereby reducing serum iron levels and increasing the risk of anemia and iron deficiency. 42 However, in this study, despite a remarkable reduction in CRP levels, no difference in therapeutic efficacy was observed. This could be because some patients were either in or about to enter an inflammatory state, which may have led to a paradoxical rise in CRP levels even after treatment, despite the general trend of reduction.
Tumor immunity is mainly based on T lymphocytes, and both cellular and humoral immunity participate in the anti-tumor process. 43 Patients with CRA have compromised immune function. In this study, it was found that Tc lymphocytes and NK cells in the treatment group were significantly higher after treatment than before treatment, and the difference was statistically significant. Tc lymphocytes are cytotoxic T cells that primarily secrete various cytokines to participate in immune functions, while NK cells are natural killer cells. Studies have found that NK cells can not only directly kill tumor cells but also express various cytokines and chemokines to recruit other immune cells and promote adaptive immune responses of T cells and B cells. 44 This indicated that Shuanghuang Yangxue Decoction could improve patients’ immune function by increasing levels of Tc lymphocytes and NK cells. Moreover, through evaluation of TCM syndromes and quality of life scales in patients with lung CRA and serum iron deficiency, the results showed that Shuanghuang Yangxue Decoction can significantly improve pale or sallow complexion, dizziness, palpitations, shortness of breath, fatigue, spontaneous sweating, reluctance to speak, insomnia, and numbness of hands and feet. Furthermore, there were also significant improvements in quality of life domains, including physical well-being, social/family well-being, emotional well-being, functional well-being, and anemia-specific conditions. It can be seen that compared to treatment with iron supplements alone, combination with Shuanghuang Yangxue Decoction can not only improve functional well-being of patients with lung cancer CRA but also improve physical and emotional well-being, leading to improved quality of life.
Conclusion
The therapeutic effect of Shuanghuang Yangxue Decoction in treating lung cancer CRA with serum iron deficiency is significantly superior to that of ferrous succinate iron supplements alone. It can markedly correct anemia in patients, increase serum iron levels, significantly improve peripheral blood parameters, anemia indicators, and TCM syndromes, and reduce inflammation. Additionally, it is proven to be safe and effective.
Limitations
Due to limitations in research conditions and funding, the study has some limitations. Firstly, the treatment duration in this clinical study was restricted to 4 weeks (28 days). The increase in hemoglobin value was modest, and the overall clinical effective rate was relatively low. This may be attributed to the short study duration, which did not allow for observation of changes as red blood cells returned to normal levels. Secondly, this study was conducted solely in China, which limited the generalizability of the population, led to selection bias, and lacked blinding, increasing the risk of bias in subjective results. Finally, the sample size of this study was relatively small, and some patients withdrew from the study, resulting in certain missing data. In future studies, multicenter clinical trials will be carried out in subsequent research, the treatment duration or observation period will be extended, and follow up the survival data of the patients, with multiple time points monitored to track hemoglobin change trends, and blinding will be used to enhance the design and reduce bias, enabling a more objective evaluation of the clinical efficacy of Shuanghuang Yangxue Decoction in treating lung cancer CRA.
Footnotes
Acknowledgements
The authors would like to thank all the researchers in our working group.
Abbreviations
CD45+ EPCs: CD45 erythroid progenitor cells
CRA: Cancer-related anemia
CRP: C-reactive protein
CSCO: Chinese Society of Clinical Oncology
ESA: Erythropoiesis stimulating agents
FACT-An: Functional Assessment of Cancer Therapy-Anemia scale
HB: Hemoglobin
ITT: Intention-to-treat
MDSC: Myeloid-derived suppressor cells
ROS: Reactive oxygen species
TIBC: total iron-binding capacity
Ethics Approval
This study was approved by the Ethics Committee (Number: 2023-077 and 2025026-001), and was registered with the China Clinical Trial Center (Number: ChiCTR2300077699).
Consent to Participate
Written informed consent was obtained from all individual participants included in the study.
Author Contributions
SJ obtained funding for the study. SJ and XL designed the trial. LQY, YWX, LY, and LD wrote the first draft of the manuscript. All authors were responsible for recruiting the patients, MH, WQ, YJL, LH, and YLQ participated in the statistics, LX, QWC, XPP, and QC participated in the survey questionnaire, LQY, YWX, LY, and LD participated in the revision of the subsequent draft. SJ and XL review the manuscript. All the authors approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Special Scientific Research Project of Shanghai Fourth People’s Hospital (sykyqd10201).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that the study was conducted in the absence of any commercial or financial relationships that could be construed as potential conflict of interests.
Data Availability Statement
All data used or analyzed during this study are available from the corresponding author on reasonable request.

