Abstract
Background:
Colorectal cancer is one of the most common and fatal cancers worldwide. Despite therapeutic advancements, patients with stage II and III colon cancer often experience recurrences and metastases, particularly to the liver, leading to suboptimal disease-free survival (DFS) rates and decreased long-term survival. Qu-Shi-Jie-Du decoction (QSJDD), a traditional Chinese herbal formula, may prevent cancer recurrence and spread by boosting immunity, reducing inflammation, and inhibiting tumour growth. Preliminary studies have demonstrated that QSJDD reduces liver metastasis in patients with colon cancer. However, robust clinical evidence is required to confirm its efficacy and safety.
Objective:
This study aims to evaluate the safety and efficacy of QSJDD in preventing colon cancer recurrence and liver metastasis, thereby offering a potential adjunctive therapy to improve patient outcomes.
Methods:
This multicentre, double-blind, randomised, placebo-controlled trial involves 336 high-risk stage II or III colon cancer patients from 10 Chinese hospitals. Post-surgery and chemotherapy, 168 patients will receive either QSJDD compound granules or a placebo for 6 months, with a 3-year follow-up and evaluations every 6 months. The primary endpoint is to measure the 3-year DFS rate, while secondary endpoints include 1- and 2-year DFS rates, overall survival, and changes in the Traditional Chinese Medicine Dampness Syndrome Scale. Safety and adverse events will be tracked, and blood and gut microbiomes will be analysed to assess QSJDD’s impact on delaying colon cancer metastasis.
Discussion:
This trial will determine the efficacy and safety of QSJDD and provide evidence regarding its role in the adjuvant treatment of colon cancer.
Keywords
Introduction
Colorectal cancer (CRC) is one of the most common malignancies worldwide, with over 1.9 million new cases and approximately 900,000 deaths reported in 2022, accounting for nearly 10% of all cancer-related incidences and fatalities. 1 In China, more than 60% of patients with primary colon cancer are diagnosed at stage II or III. 2 For these patients, curative surgery followed by adjuvant chemotherapy remains the standard treatment approach. According to the National Comprehensive Cancer Network (NCCN) guidelines, 3 to 6 months of adjuvant chemotherapy is recommended for patients with high-risk stage II or stage III colon cancer, typically involving FOLFOX or CAPOX regimens.3,4 Recent follow-up data from the International Duration Evaluation of Adjuvant Therapy (IDEA) trial support a 3-month course of CAPOX as an effective adjuvant treatment for patients with high-risk stage II and most stage III colon cancer.5,6 However, despite standard adjuvant chemotherapy, up to 40% of high-risk stage III patients (T4 or N2 disease) experience recurrence or metastasis within 3 years after surgery. 7 Among these, 15 to 25% develop liver metastases.8,9 Once metastasis occurs, the 5-year survival rate drops sharply to around 10%, 10 and more than 40% of patients ultimately die from colorectal cancer liver metastases (CRLM). 11 The anti-recurrence and anti-metastasis phase following adjuvant chemotherapy represents a critical period in colon cancer management. However, effective maintenance strategies during this phase are currently limited. According to NCCN guidelines, patients typically enter a surveillance and follow-up phase after completing standard adjuvant chemotherapy, with no clearly recommended treatments proven to prevent CRLM. This underscores the urgent need for novel interventions to prevent recurrence and improve long-term outcomes.
Traditional Chinese Medicine (TCM) has a long history of clinical application in China and is widely used as a complementary strategy in cancer care. In colorectal cancer, TCM is often continued after the completion of adjuvant chemotherapy to support recovery, improve immune function, and reduce the risk of recurrence and metastasis. A multicentre prospective study by Xu et al 12 showed a significantly lower 3-year rate of recurrence and metastasis in patients with high TCM exposure (17.7%) compared to low exposure (27.9%, P = .01). Yang et al 13 reported that integrated TCM and Western treatment reduced the 5-year recurrence and metastasis rate to 21.3%, versus 38.2% with Western therapy alone (P < .001). Tang et al 14 found that ≥18 months of oral herbal therapy significantly prolonged disease-free survival (P = .0012). Shi et al 15 observed improved 6-year DFS with TCM, especially in stage III patients (43.3 vs 22.0%). The study recommends 2 years of TCM for stage II and continuous treatment for stage III over a 6-year follow-up. In a randomized, double-blind trial, Jia et al 16 showed that adding the TCM formula PRM1201 to chemotherapy raised the 3-year DFS rate (77.1 vs 68.6%, P = .024) and reduced recurrence and metastasis risk by 37% (HR = .63). These findings suggest that TCM could play a beneficial role in the post-chemotherapy maintenance phase, particularly in preventing recurrence and metastasis.
Through clinical practice, we observed that Dampness Syndrome is the most prevalent TCM syndrome in colorectal cancer,17,18 for which Professor Zhang developed QSJDD. The herbal ingredients used in QSJDD are listed in Table 1. This decoction boosts the spleen Qi, removes dampness, and has antitumour properties, aiding colon cancer prognosis post-surgery. In a preliminary single-arm trial conducted at our centre, 45 patients with high-risk stage III colon cancer received QSJDD following standard adjuvant chemotherapy. After 2 years of follow-up, the DFS rate was 82.2%. Although this appeared higher than the 70 to 75% reported in the IDEA trial, 7 the comparison should be interpreted with caution, as the pilot study was limited by small sample size, lack of statistical adjustment for baseline differences, and absence of independent validation. These exploratory findings therefore cannot provide definitive evidence, but they suggest a possible benefit of QSJDD and underscore the need for a rigorously designed multicentre, double-blind, placebo-controlled trial to formally evaluate its efficacy in patients with high-risk stage II or III colon cancer.
Standard Formulation of Qu-Shi-Jie-Du Decoction (QSJDD).

Methods and Design
Study Design
This multicentre, double-blind, randomised controlled trial is scheduled to be conducted between July 2022 and June 2026 at the Guangdong Provincial Hospital of Chinese Medicine and 9 other major hospitals. The trial was registered with ClinicalTrials.gov (ChiCTR2300078971) and follows the SPIRIT (Standard Protocol Items: Recommendations for Interventional Trials) 19 and CONSORT (Consolidated Standards of Reporting Trials) CHM 20 guidelines for methodology and quality control. The trial flowchart is depicted in Figure 1.

Trial flowchart.
Inclusion Criteria
Patients eligible for the study must meet the following criteria:
a. Diagnosis of colon cancer by tissue or cytopathology.
b. Radical surgery with pathological staging of stage III or high-risk stage II (based on NCCN clinical practice guidelines for colorectal cancer [version 1. 2022]). 21
c. Completed adjuvant chemotherapy with 4 to 6 cycles of CAPEOX, 6 to 12 cycles of FOLFOX, or fluorouracil monotherapy, with no additional chemotherapy planned.
d. Final chemotherapy treatment within 3 months prior to enrolment.
e. Eastern Cooperative Oncology Group (ECOG) score of ≤2 points.
f. Individuals aged 18 to 75 years.
g. Normal hepatic and renal function. Normal renal function is defined as levels of serum creatinine ≤ 1.5 mg/dL (133 μmol/L) or creatinine clearance ≥ 60 mL/minutes. Normal liver function is defined as serum total bilirubin levels ≤1.5 times the upper limit of normal (ULN), and serum glutamic oxaloacetic transaminase (AST) and glutamic pyruvic transaminase (ALT) levels ≤2.5 times ULN.
h. Signed informed consent, willingness to follow the treatment regimen, ability to adhere to prescription, and good compliance.
Exclusion Criteria
Participants meeting any of the following will be excluded:
a. Inability to complete the baseline assessment form.
b. Complete or incomplete bowel obstruction.
c. Other serious illnesses, including uncontrolled infection, severe electrolyte disturbances, and/or bleeding tendencies.
d. Pregnant or lactating women.
e. Comorbid depression, mania, obsessive-compulsive disorder, or schizophrenia.
f. History of autoimmune disease, haematological disease, organ transplantation (including bone marrow auto transplantation and peripheral stem cell transplantation), or long-term use of hormones or immunosuppressive agents.
g. Other uncontrolled tumours.
h. Received or are scheduled to receive preoperative neoadjuvant radiotherapy.
i. Severe brain or psychiatric disorders that affect the ability to self-report.
j. Individuals without legal capacity, medical, or ethical concerns that may impact the study.
Elimination Criteria
Participants meeting any of the following criteria will be excluded from the analysis:
a. Withdrawal of informed consent by the participant.
b. Other explicit request to exclude data from the analysis.
c. Erroneous enrolment of a participant.
Withdrawal Criteria
The investigator may discontinue a participant from the study under the following circumstances while continuing to record follow-up data:
a. Severe adverse reactions or disease progression during treatment.
b. Protocol violations including discontinuing the study medication for over 2 months, using traditional Chinese medicines outside the study for more than 2 months, and receiving anti-tumour therapies (such as chemotherapy, biological agents, radiotherapy, targeted therapy, or immunotherapy) for 2 months or longer.
Participants
This study will be conducted at ten medical institutions in China. Guangdong Provincial Hospital of Chinese Medicine is leading the research and the others include: Sun Yat-sen University Cancer Center, The Sixth Affiliated Hospital of Sun Yat-sen University, The First Affiliated Hospital of Sun Yat-sen University, Guangdong Provincial People’s Hospital, Zhujiang Hospital of Southern Medical University, Dongguan People’s Hospital, The First People’s Hospital of Foshan, Mei Zhou Hospital of Traditional Chinese Medicine, and Guangdong Provincial Agricultural Reclamation Central Hospital. All centres are Grade A general or tumour specialty hospitals who adhere to the NCCN guidelines for colon cancer. Inpatient and outpatient participants will voluntarily complete an informed consent form.
Interventions
Intervention Group
Patients in the intervention group will receive QSJDD granules daily for 6 months or until tumour recurrence or metastasis are detected, or intolerable side effects occur. Treatment starts within 3 months of completion of standard adjuvant chemotherapy with continuous compliance monitoring. QSJDD is administered once daily with warm water.
Control Group
Patients in the control group will receive a placebo (QSJDD mimetic agent) consisting of 5% QSJDD granules and other ingredients such as colour and taste correctors and malt dextrin. The placebo granules will match the test drugs in appearance, dosage form, weight, colour, and odour. The duration of the placebo treatment will be identical to that of the intervention group.
Procedure
Figure 1 displays the study flowchart and Table 2 details the measurement methods and data collection timelines. Treatment starts within 3 days of randomization and lasts 6 months. Patients will be assessed every 3 months during treatment, and then every 6 months after surgery, until death or study conclusion at 3 years. Patient and treatment details, along with demographic information, such as age, sex, and living conditions, will be obtained from medical records and baseline questionnaires. Medical data will be obtained from the patients’ medical records.
Measurement Methods and Data Collection Timeline.

•: mi; ◎: nmi.Abbreviations: CT, computed tomography; MRI, magnetic resonance imaging; CTC, circulating tumour cells.
CTC and CIC Isolations by Microfluidic Chips
A total of 5 mL peripheral blood (PB) samples from cancer patients were collected in EDTA-containing tubes, stored at room temperature, and processed within 4 hours of collection. PB samples were diluted with same volume of phosphate buffered solution (PBS), and the mixture was processed through the cascaded filter deterministic lateral displacement microfluidic chip (CFD-Chip).22,23 The obtained cell suspensions consisting of CTCs, circulating cell-in-cell tumour cells (CICs) and leukocytes were centrifuged at 250 g for 10 minutes and resuspended in PBS.
Detection of CTCs and CICs Using the Immunofluorescence
The cell suspensions after CFD-Chip separation were seed onto 96-well plates. Cells were fixed for 15 minutes with 4% paraformaldehyde (Affymetrix, Santa Clara, CA) at room temperature. Fixed cells were washed 4 times using PBS, and then permeabilized by 0.1% TritonX-100 (Sigma-Aldrich, St. Louis, MO) for 10 minutes at room temperature. After blocking with 3% BSA solution for 1 hour, cells were incubated with a cocktail of antibodies containing PE anti-CD45 antibody (1:100, BD Biosciences), 488 anti-pan cytokeratin antibody (1:500, Thermo Fisher Scientific) overnight at 4°C. After washing 6 times, cells were stained with 15 μL 1 mg/mL DAPI solution (Solarbio, Cat# C0060, Beijing, China) for 10 minutes. After washing 2 times by PBS, cells were observed using a fluorescence microscope. The numbers of CTCs were manually counted which were determined by DAPI+/CD45-/CK+ immunofluorescence staining cells with integrated neoplastic cell morphology. The numbers of CICs were determined by DAPI+/CD45+/CK+ immunofluorescence staining tumour cells with mononuclear but double-layered cell membrane morphology. 24
Primary Outcome
The primary endpoint is the 3-year DFS rate. DFS is defined as the time from randomisation to the first occurrence of tumour recurrence, metastasis, second primary cancer, or death from any cause. 25 Patients will be assessed through scheduled imaging examinations (eg, CT, MRI) and tumour marker evaluations (eg, CEA) at regular follow-up intervals.
The choice of a 3-year DFS rather than a traditional 5-year endpoint is based on clinical evidence showing that most recurrences of high-risk stage II/III colorectal cancer occur within the first 3 years after curative surgery and adjuvant treatment. Several studies have demonstrated that 3-year DFS is a reliable early surrogate for long-term outcomes, including overall survival (OS), particularly in adjuvant therapy trials for colon cancer. 26 Moreover, multiple large-scale clinical trials have already adopted 3-year DFS as the primary or co-primary endpoint for evaluating treatment efficacy, 7 further supporting its validity as a robust and pragmatic outcome measure. This time frame allows for earlier evaluation of the efficacy of interventions and is increasingly accepted in clinical trials as a pragmatic and predictive endpoint.
Secondary Outcomes
Secondary endpoints include 1- and 2-year DFS rates and median DFS. Additional secondary endpoints include recurrence rates (RR) and overall survival (OS) at 1 and 2 years.
Exploratory and Mechanistic Endpoints
The intestinal microbiota, circulating tumour cells (CTCs), peripheral blood immune cells, and cytokines will be assessed at 3 time points randomisation, the third, and sixth month of treatment. The 16S rDNA sequencing of faecal samples for gut microbiota profiling was conducted by KMHD Gene Tech Co., Ltd. (Shenzhen, China), while CTCs classification and detection were performed using technologies provided by Shenzhen Zigzag Biotechnology Co., Ltd. (Shenzhen, China). These endpoints will also serve as indicators for exploring the mechanisms by which QSJDD prevents liver metastases. Additionally, the Dampness Syndrome Scale of Chinese Medicine (DSSCM) provided by the State Key Laboratory of Dampness Syndrome of Chinese Medicine under the joint establishment of the Ministry of Provincial Affairs of the People’s Republic of China will be used to evaluate TCM-related symptoms and changes (refer Figures 2 and 3).
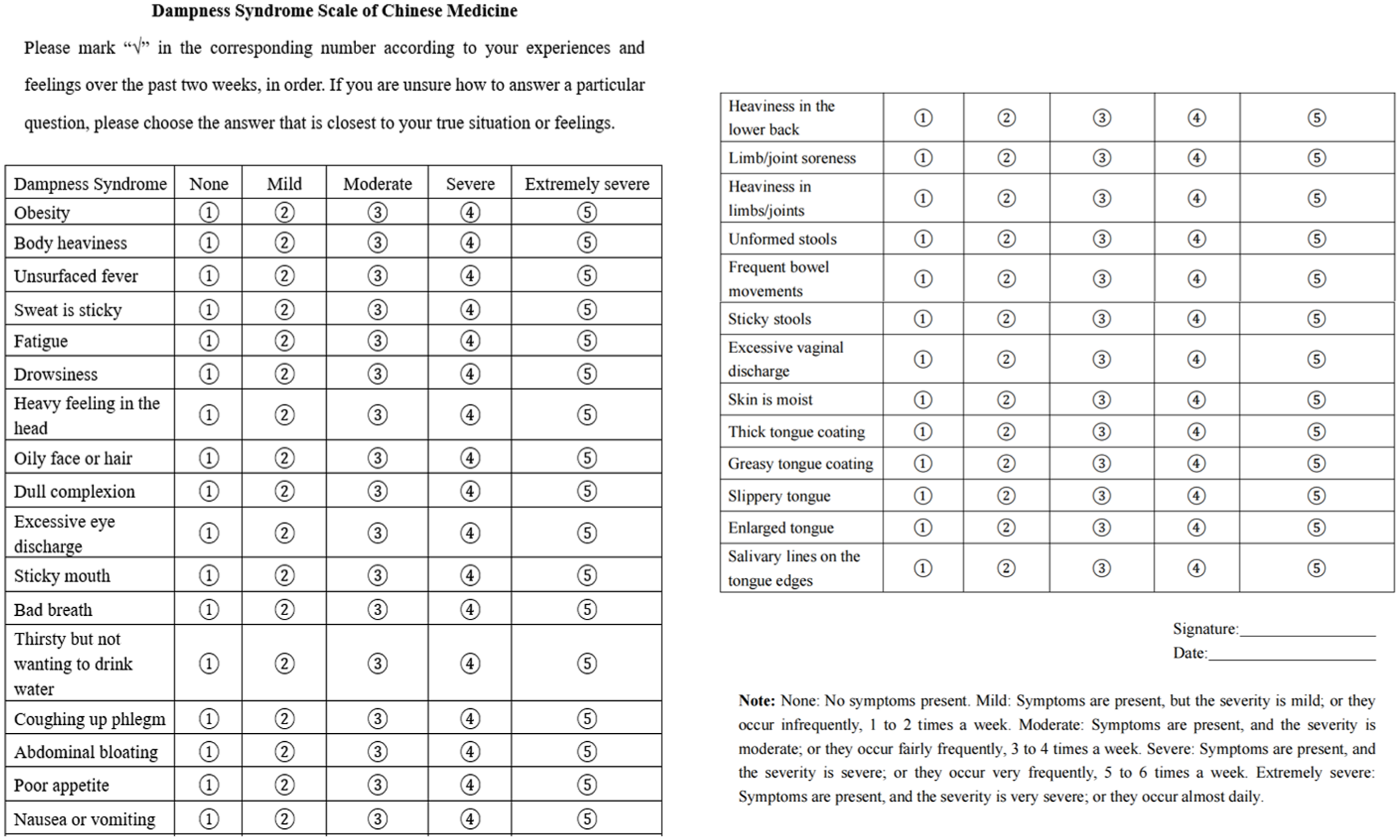
Dampness syndrome scale of Chinese medicine. 27

Dampness clinical diagnosis scale. 28
To assess the severity of dampness syndrome, we employed the DSSCM, which was developed based on TCM theory, expert consensus, and clinical investigation. The scale has undergone comprehensive psychometric validation in a large cross-sectional study involving 869 participants. 29 The reliability of the DSSCM was demonstrated by high internal consistency (Cronbach’s α coefficients of .809, .891, .816, and .780 across its 4 dimensions) and acceptable test–retest reliability (ICCs ranging from 0.674 to 0.806). Confirmatory factor analysis supported the structural validity of the scale, with RMSEA = 0.091, SRMR = 0.074, CFI = 0.766, and TLI = 0.745. Furthermore, the DSSCM showed good discriminant and criterion validity when compared against clinicians’ diagnoses. These findings support the DSSCM as a scientifically sound and clinically relevant instrument for evaluating dampness syndrome as an exploratory endpoint in our study.
The enrolment of 60 patients in the pre-trial phase is expected to be completed within 12 months. During this phase, CTC counts will be assessed as an exploratory biomarker to monitor disease progression. While CTCs levels have been associated with PFS and OS in colorectal cancer,30 -32 their role as a surrogate endpoint for DFS requires further validation. As such, DFS will remain the primary clinical outcome in this trial, and CTCs will serve as a secondary, exploratory biomarker.
Safety Outcomes
Adverse events and postoperative complications will be evaluated according to the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0, established by the National Cancer Institute (NCI). 33 Safety assessments include: complete blood count, urinalysis, biochemical tests, electrocardiogram, general physical examination, and documentation of other adverse events. In addition, potential adverse events specifically associated with Traditional Chinese Medicine (TCM) will be recorded in detail, including rash, gastrointestinal reactions, skin allergy, and liver or kidney function abnormalities. All observed or suspected AEs will be documented in case report form (CRF) with information on onset time, severity, management, and outcome, to ensure a comprehensive and transparent evaluation of safety.
Randomization and Blinding
This study adopted a double-blind, centrally randomized design. A centre-stratified, permuted block randomization sequence (block size = 4) was generated using SAS 9.4 software (SAS Institute Inc., Cary, USA) by the Center for Clinical Research (CCR), Guangdong Provincial Hospital of Chinese Medicine. Eligible participants were randomly assigned in a 1:1 ratio to the QSJDD or placebo group via the Interactive Web Response System developed by CCR. The randomization list and blinding codes were kept strictly confidential and accessible only to CCR staff. QSJDD and matching placebo granules were manufactured and supplied by Guangdong YiFang Pharmaceutical Co., Ltd., China. Details of the preparation process, including measures to ensure indistinguishable appearance, taste, and packaging, are provided in Supplemental Material 1. Investigators, participants, and medical staff remained blinded to treatment assignments throughout the trial. Unblinding was permitted only under emergency conditions where knowledge of the assigned treatment was essential for patient management (eg, serious adverse events). In such cases, the investigator initiated the unblinding request in consultation with the sponsor or medical monitor, unless immediate disclosure was required for emergency care. All unblinding events were documented in the CRF, including the reason, timing, and personnel involved. To preserve the integrity of blinding, unblinding information was restricted to essential clinical personnel and not disclosed to outcome assessors. 34
Data Collection and Monitoring
Participants will provide baseline information before the first treatment, and the Dampness Syndrome Scale will be assessed at randomisation and during follow-up at 3 and 6 months. Data will be recorded in the CRF by trained investigators, with any revisions noted, including the reason and date. Two data managers will enter the data independently using EpiData software (version 3.1), and discrepancies will be cross-checked and resolved to ensure accuracy. The final database will be locked after data verification. To ensure compliance with Good Clinical Practice (GCP) standards, an independent Data Monitoring Committee (DMC) was established under the Clinical Research Center of Guangdong Provincial Hospital of Chinese Medicine. The DMC is responsible for conducting regular audits across all participating sites. These audits will include the review of research documentation, source data verification, and assessment of protocol adherence. All quality control procedures will be performed in accordance with institutional standard operating procedures (SOPs) and applicable regulatory requirements.
Sample Size Calculation
This study aims to assess the 3-year recurrence and metastasis rates. Recent trials, including SCOT and IDEA, have reported recurrence rates of approximately 23 to 25%.6,35 -37 However, subgroup analyses indicated that patients with T4 or N2 disease had significantly worse outcomes, with 3-year DFS as low as 60%. According to a multicentre observational study in China, 2 stage III patients comprise nearly 30% of all newly diagnosed colorectal cancer cases, and a substantial proportion of them present with high-risk features. Given that our study is expected to enrol a relatively high number of patients with such high-risk characteristics, assuming a 30% recurrence rate for the control group remains both a reasonable and conservative estimate. We hypothesize that the QSJDD group will achieve a reduction to 15% compared to 30% in the placebo group. Hence, 268 patients (134 per group) are required, with a two-sided alpha of 0.05 and 80% power. Accounting for a 20% dropout rate, 336 participants are required (168 per group).
Statistical Analysis
Data will be analysed using SPSS 17.0 and Stata 11.0 software. For normally-distributed quantitative data, the results will be presented as mean ± standard deviation. For non-normal distributions, the data will be described using the median, lower quartile (Q1), upper quartile (Q3), minimum, and maximum values. Intergroup comparisons will be conducted using the t-test. For non-normal data distributions or unequal variances, the Wilcoxon rank-sum test will be applied. Categorical data will be described using proportions and ratios, and inter-group comparisons will be performed using the χ² test or Fisher’s exact test. The Kaplan–Meier method will be used to analyse and calculate cumulative DFS and OS, with comparison using the log-rank test. A multivariate analysis will be performed using the Cox regression model. All tests will be two-sided, and the significance level (α) will be set at .05.
Subgroup analyses will be conducted to explore potential differential effects of the intervention across predefined patient categories. Planned subgroup analyses will include stratification by disease stage (II vs III), age group (<60 vs ≥60 years), sex, tumour location (left-sided vs right-sided), and baseline CTC status. These analyses will be primarily exploratory and hypothesis-generating, and interaction terms will be tested to assess heterogeneity of treatment effects.
Discussion
Postoperative recurrence and metastasis remain the leading causes of treatment failure in colon cancer, especially in high-risk stage II and III patients.38 -41 To address the challenge of reducing metastasis in colon cancer, several adjunctive therapies have been studied. Low-dose non-steroidal anti-inflammatories, particularly aspirin, may lower the risk of postoperative recurrence and mortality in colon cancer.42 -45 However, it has not yet been confirmed by a large randomized controlled trial. 46 Moreover, growing evidence has investigated the links between vitamin D47 -49 and metformin50 -53 with the risk of recurrence and mortality in colon cancer, although the results have been inconclusive. Traditional Chinese medicine has shown significant potential in reducing the metastasis and recurrence of colorectal cancer. In recent years, an increasing number of studies have demonstrated that TCM can inhibit the progression and metastasis of colorectal cancer through multiple mechanisms. First, TCM can influence the onset, progression, and prognosis of colorectal cancer by modulating gut microbiota. Research indicates that TCM can increase the abundance of beneficial bacteria such as bifidobacteria in the gut, thereby inhibiting the development and metastasis of colorectal cancer.54,55 In addition, TCM can inhibit the proliferation and metastasis of tumour cells by affecting multiple signalling pathways. For example, certain components of TCM can reverse epithelial-mesenchymal transition (EMT) by modulating the PI3K/AKT signalling pathway, thereby suppressing the migration and invasion of colorectal cancer cells.56,57 Furthermore, TCM can enhance the body’s resistance to tumours by regulating the immune system, thus reducing tumour recurrence and metastasis.58,59 In clinical practice, numerous studies have suggested that TCM has potential benefits in reducing recurrence and metastasis of colorectal cancer.12 -16 However, the study by Shi et al 15 did not evaluate the efficacy of TCM in high-risk stage II patients. Although Jia et al 16 conducted a randomized, double-blind, placebo-controlled trial using PRM1201 in combination with adjuvant chemotherapy, it did not assess the role of TCM in preventing recurrence and metastasis during the post-chemotherapy phase. Therefore, there is an urgent need for high-quality randomized controlled trials to validate the clinical value of TCM in preventing recurrence and metastasis after adjuvant chemotherapy in colon cancer patients.
Our study specifically focused on colon cancer, aiming to provide a more precise understanding of its distinct biological characteristics and therapeutic needs. Anatomically, rectal cancer differs significantly from colon cancer in terms of venous drainage. The former primarily drains into the systemic circulation through the middle rectal vein and internal iliac vein, which results in a higher incidence of lung metastasis and a relatively lower incidence of liver metastasis. 60 In contrast, colon cancer primarily drains into the portal system, making the liver the most common site of distant metastasis. 61 Therapeutically, rectal cancer presents unique challenges due to its location in the pelvis and its proximity to critical urogenital organs. As a result, treatment of rectal cancer often involves a more complex approach, typically including neoadjuvant chemoradiotherapy, surgical resection, and adjuvant chemotherapy based on 5-fluorouracil. 62 These differences underscore the importance of studying colon and rectal cancers separately in clinical research. Preclinical in vivo studies demonstrated that QSJDD was well tolerated, without significant effects on body weight or food intake, and improved hepatic steatosis and colonic inflammation without causing renal toxicity, while also alleviating 5-FU–induced intestinal injury. In addition, QSJDD improved serum lipid metabolism and inflammatory profiles by reducing TG, TC, LDL-C, TNF-α, and bile acids while elevating HDL-C (see Supplemental Material 1). These results provide preclinical evidence supporting the safety, antitumor efficacy, and immunometabolic regulatory effects of QSJDD, thereby justifying its evaluation in clinical trials. Notably, our recent mechanistic studies using a murine dampness-cancer model revealed that dampness syndrome was associated with systemic inflammation (elevated TNF-α, IL-6), dyslipidaemia, bile acid metabolism abnormalities (upregulation of FXR, CYP7A1, CYP8B1, ASBT), and impaired intestinal barrier function (decreased ZO-1, Occludin). Furthermore, tumour tissues in dampness syndrome mice exhibited enhanced proliferation (Ki67, PCNA) and activation of lipid synthesis pathways (FASN, ACC, ACLY). These findings suggest that dampness syndrome may promote tumour progression through inflammatory activation, metabolic reprogramming, and bile acid dysregulation, thus providing a biological basis that bridges TCM syndrome theory and modern oncology. In our prior single-arm study of QSJDD with over 2 years of follow-up, no cases of liver metastasis were observed among patients who experienced recurrence or distant spread. This preliminary finding suggests that QSJDD may help reduce the risk of liver metastasis in colon cancer, which is clinically meaningful given the liver is the most common site of distant spread in this subtype. Additionally, compared to rectal cancer, colon cancer presents an approximately 50% higher incidence and risk of mortality, emphasizing its public health significance. 63 A focused approach on colon cancer allows for tailored therapeutic strategies. In this context, beyond regular surveillance and standard adjuvant chemotherapy, interventions aimed at reducing liver metastasis risk, such as QSJDD, may offer a novel and valuable strategy to improve prognosis in high-risk stage II and III patients.
An important component of our study is the incorporation of circulating tumour cell (CTC) monitoring. Currently, the CellSearch™ system is the only FDA-approved method for CTC detection and is considered a standardized approach for use in metastatic breast, colorectal, and prostate cancers.64 -67 However, due to the heterogeneity of CTCs, especially the presence of mesenchymal CTCs (M-CTCs), EpCAM-based systems like CellSearch™ may miss certain subtypes. Therefore, alternative technologies such as size-based isolation (ISET) and microfluidic-based enrichment have been developed to improve detection sensitivity and subtype coverage. 68 In our study, we used a microfluidic chip combined with immunofluorescence staining to identify CTCs, which enhances both detection sensitivity and specificity. Wu et al 69 classified CTCs into 3 subgroups: E-CTCs (EpCAM), M-CTCs (vimentin), and E/M-CTCs (both). They reported that M-CTCs and circulating tumour micro emboli were more strongly linked to tumour metastasis than other CTC subgroups. Similarly, Zhong et al 70 showed that M-CTCs correlate with tumour size, T stage, TNM stage, vascular invasion, and carcinoembryonic antigen levels, and demonstrated that M-CTCs > 1 were associated with poor DFS. Although some researchers have focused on CTC subgroup analyses, CTC cell subtypes have received limited focus in previous studies. We identified a new CTC subtype of circulating cell-in-cell tumour cell (CIC) using microfluidic detection technology. CICs have a single nucleus, but the cell membrane is double-layered and has neutrophil characteristics. The number of CICs usually exceeds the number of conventionally-defined CTCs, and may be more effective for monitoring prognosis. CICs have a significant regulatory effect on chemotaxis, epithelial-mesenchymal transition, and gene expression on immune checkpoints. 24 Therefore, these CTC will be used for secondary outcome measurement and for validating its feasibility and accuracy as a predictor. Although CTCs cannot replace imaging or tumour markers for determining the exact location of recurrence, they can provide complementary prognostic information that may help optimize follow-up strategies.30 -32 Furthermore, combining CTC detection with other clinical markers may improve overall diagnostic accuracy.
This multicentre, double-blind, randomised controlled trial evaluates the effectiveness and safety of QSJDD in preventing recurrence and metastasis in high-risk stage II/III colon cancer and evaluates the effectiveness of CTCs and their subtypes as indicators of recurrence and potential therapeutic targets. We will employ a standard RCT methodology to validate the efficacy of QSJDD, and this rigorous approach provides a solid foundation for our findings. However, this study has certain limitations. The treatment duration of QSJDD was limited. Considering that prolonged use of QSJDD may lead to decreased patient adherence and an increased follow-up loss rate, a 6-month treatment period was preferred, which is considered an effective duration for traditional Chinese medicine treatments. 71 As for CICs, we recognize that they are a novel exploratory subtype of CTCs. Although research on their prognostic value is still limited, we included CICs to assess their potential utility in complementing conventional CTC analysis. Further investigation is warranted to validate their clinical relevance. In addition, while DSSCM has demonstrated solid psychometric properties, its clinical relevance in colorectal cancer remains unexplored. Despite these limitations, our study assesses the benefits of TCM interventions, provides a TCM-based therapeutic strategy, and lays the groundwork for future large-scale studies.
Supplemental Material
sj-docx-1-ict-10.1177_15347354251396002 – Supplemental material for Qu-Shi-Jie-Du Decoction for Prevention of Recurrence and Metastasis in High-Risk Stage II/III Colon Cancer: Study Protocol for a Double-Blind, Randomized, Placebo-Controlled Trial
Supplemental material, sj-docx-1-ict-10.1177_15347354251396002 for Qu-Shi-Jie-Du Decoction for Prevention of Recurrence and Metastasis in High-Risk Stage II/III Colon Cancer: Study Protocol for a Double-Blind, Randomized, Placebo-Controlled Trial by Ya-dong Chen, Zi-yan He, Wen-jie Zhao, Yi-han He, Yi-hong Liu, Yan-juan Zhu, Ya-ya Yu, Zhen-zhen Xiao, Xue-song Chang, Zong-bin Liu and Hai-bo Zhang in Integrative Cancer Therapies
Footnotes
Acknowledgements
We thank the Methodology Group of Guangdong Provincial Hospital of Chinese Medicine for providing the central randomisation method. In addition, we thank Shenzhen Zigzag Biotechnology Co., Ltd. for the CTCs sorting and detection technology used in this study, and KMHD Gene Tech Co., Ltd. (Shenzhen, China) for performing the 16S rDNA sequencing of faecal samples for gut microbiota profiling. We extend our gratitude to all the healthcare professionals who provided support throughout the course of this study. We would like to thank Editage (![]() ) for English language editing.
) for English language editing.
List of Abbreviations
5-FU: fluorouracil
ALT: serum glutamic pyruvic transaminase
Anti-CRLM: anti-colorectal cancer liver metastasis
CAPE-OX: capecitabine and oxaliplatin
CICs: circulating cell-in-cell tumour cells
CONSORT: Consolidated Standards of Reporting Trials
CRC: colorectal cancer
CRLM: colorectal cancer liver metastases
CRF: case report form
CTCAE: Common Terminology Criteria for Adverse Events
CTCs: circulating tumour cells
DFS: disease-free survival
DMC: data monitoring committee
ECOG: Eastern Cooperative Oncology Group
EpCAM: epithelial cell adhesion molecule
FOLFOX: leucovorin (folinic acid), fluorouracil, and oxaliplatin
mFOLFOX6: modified FOLFOX 6 regimens
MOSAIC: Multicenter International Study of Oxaliplatin/ 5-Fluorouracil/ Leucovorin in the Adjuvant Treatment of Colon Cancer Study
NCCN: National Comprehensive Cancer Network
NCI: National Cancer Institute
NSABP: National Surgical Adjuvant Breast and Bowel Project
OS: overall survival
QSJDD: Qu-Shi-Jie-Du decoction
RCT: randomized, controlled trial
RR: relapse rates
SPIRIT: Standard Protocol Items: Recommendations for Interventional Trials
TAM: Tumour-associated macrophage
TCM: traditional Chinese medicine
ULN: upper limit of normal
Ethical Considerations
This study follows the Declaration of Helsinki and adheres to the SPIRIT guidelines. It is registered with the Chinese Clinical Trial Registry (ChiCTR2300078971) and has received ethical approval from the Guangdong Provincial Hospital of Traditional Chinese Medicine’s ethics committee (BF2021-204).
Consent to Participate
Eligible participants from inpatient and outpatient departments will voluntarily complete an informed consent form.
Author Contributions
YC and HZ conceived the study. YC, WZ, YH, YL, YZ, YY, ZX, XC, FH and HZ participated in the study design and protocol development. YC, ZH, and WZ wrote the manuscript. ZL provided testing techniques. HZ led the development of the proposal and protocol and applied for funding. All the authors have read and approved the final manuscript.
Funding
This study is funded by State Key Laboratory of Dampness Syndrome of Chinese Medicine, The Second Affiliated Hospital of Guangzhou University of Chinese Medicine (grant number: SZ2021ZZ40, SZ2023ZZ14), Science and Technology Planning Project of Guangdong Province (No. 2023B1212060063), Science and Technology Planning Project of Guangzhou City (No. 2025A03J4133), Guangzhou Municipal Science and Technology Bureau, China (grant number: 2024A03J0709, 2025A03J3156), The Traditional Chinese Medicine Bureau of Guangdong Province (grant number: 20254059), The Project of Guangdong Provincial Hospital of Chinese Medicine (grant number: YN2023MS32), and The Project of State Key Laboratory of Traditional Chinese Medicine Syndrome (grant number: SKLKY2024B0002Z).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
No datasets were generated or analysed during the current study.
Supplemental Material
Supplemental material for this article is available online.
Trial Status
The last updated protocol was version 3.0. Study execution period: Between 1 July 2021 and 30 June 2026; recruitment period: 24 August 2023 to 30 June 2026. At the time of manuscript submission, patient recruitment was ongoing, with a total of 336 patients expected to be recruited for both the intervention and control groups.
Trial Registration
Chictr.org.cn Identifier: ChiCTR2300078971. Registered on 22nd December 2023.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
