Abstract
Purpose:
Hepatocellular carcinoma (HCC) poses a significant global health burden with limited therapeutic options. Traditional Chinese medicine (TCM), particularly Yangyin Fuzheng Jiedu Prescription (YFJP), has shown promise in improving patient outcomes, but its mechanisms are poorly understood. This study aimed to elucidate the key components and mechanisms of YFJP in treating HCC using an integrative approach combining network pharmacology, molecular docking, and experimental validation.
Patients and Methods:
We analyzed data from 1021 HCC patients (481 treated with YFJP and 540 with Western medicine alone) using propensity score matching to minimize bias. Network pharmacology identified key components and targets of YFJP, with a focus on Jiedu Prescription (JDP). Molecular docking and dynamics simulations validated the binding affinity between core components and targets. GO and KEGG analyses elucidated biological functions and pathways. In vivo experiments using a tumor-bearing mouse model further validated the mechanisms.
Results:
YFJP significantly improved overall survival (P < .0001) and increased CD4+T and CD8+T cell counts (P < .05) in HCC patients compared to the control group. Network pharmacology analysis identified JDP as the core component of YFJP, with quercetin, luteolin, and apigenin as the key active compounds. GO and KEGG pathway analyses revealed that JDP modulates HCC through the regulation of cell death, immune response, and the JAK-STAT signaling pathway. In vivo experiments demonstrated that JDP increases the proportion of CD8+T cells in the tumor microenvironment and inhibits apoptosis by downregulating the IL-6/STAT3 pathway. Molecular docking and dynamics simulations further confirmed the strong binding affinity of JDP’s key compounds to STAT3, supporting their role in modulating this pathway.
Conclusion:
YFJP, particularly its core component JDP, enhances anti-tumor immunity and improves survival in HCC patients by modulating the IL-6/STAT3 pathway. These findings highlight YFJP as a promising adjuvant therapy for HCC, offering a multi-target approach to enhance anti-tumor immunity.
Keywords
Introduction
Primary liver cancer, predominantly hepatocellular carcinoma (HCC), is the sixth most common cancer and the third leading cause of cancer-related deaths worldwide. 1 Although surgical resection is effective for early-stage HCC, most patients present at advanced stages, necessitating systemic therapies like molecularly targeted drugs and immune checkpoint inhibitors (ICIs). 2 Despite the promise of ICIs, their efficacy is limited, with an overall response rate of less than 30%,3,4 and they are often associated with severe side effects. 5 These limitations highlight the urgent need for alternative or adjunctive therapies that can improve patient outcomes with minimal toxicity.
Traditional Chinese medicine (TCM), with its multi-component and multi-target properties, has shown promise in enhancing immune function and reducing tumor recurrence in cancer treatment.6,7 Our previous studies confirmed that TCM adjuvant therapy was an independent protective factor for the 5-year overall survival (OS) of HCC patients (hazard ratio [HR] = 0.46, 95% confidence interval [CI] 0.40-0.52, P < .0001), which could improve patients’ OS and progression-free survival (PFS). 8 Moreover, the safety of YFJP has been preliminarily evaluated in an H22 tumor-bearing mice, showing no significant adverse effects on immune homeostasis. 9 However, the specific mechanisms and targets remain unclear. In this study, we employed an integrative approach combining network pharmacology, molecular docking, and in vivo experiments to elucidate the key components and mechanisms of YFJP in treating HCC, with a focus on its core component, Jiedu Prescription (JDP).
From the perspective of TCM, the pathogenesis of cancer is characterized by the deficiency of positive attributes and accumulation of detrimental attributes. Hedysarum Multijugum Maxim (HMM; Chinese name: Huangqi) is used for tonifying Qi, accentuating Yang, and for hepatoprotection.10,11 Atractylodes Macrocephala Koidz (AMK; Chinese name: Baizhu) strengthens the spleen and relieves dampness, which benefits cancer patients with Qi deficiency. 12 Hedyotis diffusae Herba (HDH; Chinese name: Baihuasheshecao) clears heat, detoxifies and dissipates nodules, and removes swelling and water 13 ; Sophorae flavescentis Radix (SFR; Chinese name: Kushen) can clear heat, relieve moisture, and kill insects; it contains oxymatrine and other alkaloids and displays antibacterial, anti-inflammatory, and anti-tumor activities, which are suitable for cancer patients with internal accumulation of toxic heat. 14 Yangyin Fuzheng Jiedu prescription (YFJP), an empirical TCM formula, has demonstrated clinical efficacy in HCC treatment. Animal experiments have shown that this prescription can effectively inhibit tumor growth and increase the proportion of CD8+T cells in blood, spleen, and tumor tissues. Decreased expression of programed cell death protein 1 (PD-1), T cell immunoglobulin and ITIM domain (TIGIT), and T cell immunoglobulin and mucin domain 3 (TIM-3) in CD8+T lymphocytes reduces the apoptosis of tumor-infiltrating T cells. 9 However, the core formula of YFJP and mechanisms of action remain unclear.
Network pharmacology can elucidate the interactions between compounds and targets and the potential mechanisms of multi-component, multi-target TCM at the molecular level by constructing a compound-target-disease network.15,16 In this study, we evaluated the efficacy of YFJP in HCC patients. Network pharmacology analysis revealed that the effect of YFJP was mainly due to JDP Chinese medicine. The key components and core targets of JDP in the treatment of HCC were explored. The potential signaling pathways were obtained by KEGG and GO enrichment analysis. The regulatory effect and function of JDP on the number of CD8+T cells in HCC immune microenvironment were observed through in vivo experiments.
Materials and Methods
Patients
This retrospective cohort study involved a total of 1509 HCC patients were consecutively recruited from Beijing Ditan Hospital between June 2009 and December 2019. This study was approved by the Ethics Committee of the Beijing Ditan Hospital. The patient inclusion criteria were: (1) diagnosis of HCC, (2) age 18 to 75 years, and (3) availability of complete clinical data. The exclusion criteria were: (1) cholangiocarcinoma (n = 122), (2) metastatic liver cancer (n = 84), (3) other types of cancer (n = 33), (4) loss to follow-up (n = 120), and (5) incomplete clinical data (n = 129). Ultimately, 1021 patients were included in this study.
Important clinical data included age, sex, family history, personal life history (history of smoking and alcohol consumption), etiology of HCC, hepatitis B virus (HBV) indicators that included HBV DNA, hepatitis B e antigen, tumor-related indicators (tumor number, tumor size, portal vein tumor thrombus [PVTT], and extrahepatic spread), and laboratory parameters that included white blood cells (WBC), neutrophil-lymphocyte ratio (NLR), platelet (PLT) count, alanine aminotransferase (ALT), total bilirubin (TBIL), albumin (ALB), γ-glutamyl transferase (γ-GGT), creatinine, prothrombin time activity (PTA), International Normalized Ratio (INR), C-reactive protein (CRP), alpha fetoprotein (AFP), and T lymphocytes.
Follow-Up and Endpoint
Computed tomography or magnetic resonance imaging scan, ultrasonography, or serum AFP tests were performed every 6 months. The definition of progression conformed to the Modified RECIST (mRECIST) criteria. 17 Progression was defined as development of vascular metastasis or extrahepatic diffusion. The period from admission to death or the last follow-up on September 30, 2023, was designated as the survival time.
YFJP Preparation and Treatment
YFJP is composed of 8 TCMs: Glehniae Radix (GR; Chinese name: Beishashen, 15 g), Ophiopogon japonicus (OJ; Chinese name: Maidong, 15 g), Hedysarum Multijugum Maxim (HMM; Chinese name: Huangqi, 20 g), Atractylodes Macrocephala Koidz (AMK; Chinese name: Baizhu, 9 g), Radix Bupleuri (RB; Chinese name: Chaihu, 9 g), Sophorae flavescentis Radix (SFR; Chinese name: Kushen, 9 g), Hedyotis diffusae Herba (HDH; Chinese name: Baihuasheshecao, 20 g),and Radix Cynanchi paniculati (RCP; Chinese name: Xuchangqing, 12 g). The drug effects were divided into JDP (SFR 9 g, RCP 12 g, HDH 20 g), Fuzheng prescription (FZP; HMM 20 g, AMK 9 g), and Yangyin prescription (YYP; GR 15 g, OJ 15 g). The prescriptions were provided by the Pharmacy of Traditional Chinese Medicine at Beijing Ditan Hospital (Beijing, China). Patients in the YFJP group received comprehensive treatment with Western medicine supplemented with YFJP for ≥3 months. Patients in the control group received only Western medicine based comprehensive treatment. To reduce the influence of confounding factors, we performed propensity score matching at a 1:1 ratio to divide the participants into the YFJP and control groups.
Network Pharmacology Analysis
Collection of Components and Targets
The traditional Chinese medicine systems pharmacology database and analysis platform (TCMSP) 18 was used to query and screen the compounds of YFJP. Screening criteria were DL ≥ 0.18 and OB ≥ 30%.19,20 The BATMAN-TCM database 21 was used to search for OJ compounds. Screening criteria were score cut-off ≥ 20 and P ≥ .05. After obtained the YFJP active components, PubChem 22 or Open Babel software (version 2.4.1) were used for conversion to the SMILES format to determine the molecular structure of each compound. SwissTargetPrediction 23 was used to predict the potential targets of the compounds.
Screening of Targets for HCC
HCC-associated targets were screened using DisGeNET, 24 TTD, 25 DrugBank, 26 OncoDB.HCC, 27 and Liverome. 28 The intersecting targets of these 5 databases were HCC targets.
Network Construction
We established a YFJP compound-target network and YFJP-HCC overlapping target-compound network to obtain TCMs with compounds that have strong interactions with the targets in the network, which are considered to play a major role in YFJP. We then mapped a network of JDP compounds and the overlapping targets of JDP and HCC. In order to explore the interaction among target proteins, the overlapping targets of JDP and HCC were uploaded to the STRING database (https://string-db.org/), and “species” was set as “human” to construct the protein-protein interaction (PPI) network to screen the core targets for JDP efficacy. Networks were constructed using Cytoscape 3.7.2.
Enrichment Analysis
DAVID platform (https://david.ncifcrf.gov/) was used for gene ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) analyses of the overlapping targets of JDP and HCC to explore the cell functions and signaling pathways that JDP may affect during the treatment of HCC.
Molecular Docking
The binding activity of the key active ingredients and core targets of JDP in the treatment of HCC were analyzed by molecular docking. Compounds and targets with the best binding energies were selected. Four steps were used in the molecular docking procedure. First, the protein structures of the core targets were downloaded from the PDB database. 29 Pymol was used to remove the protein crystal water and original ligand. The protein structure was imported into AutoDockTool (version 1.5.6) for hydrogenation, and charge calculation and assignment. The atom type was specified and saves in the “pdbqt” format. Second, the 3-dimensional structure of the key compound ligands from the PubChem database were saved in the sdf format and imported into ChemBio3D Ultra 14.0 for energy minimization. The Minimum RMS Gradient was set to 0.001 and small molecules were saved in the mol2 format. The optimized small molecules were imported into AutodockTools-1.5.6 for hydrogenation, charge calculation, charge distribution, and to set the rotable key. The data were saved in the “pdbqt” format. Third, the processed core proteins and key ligands were imported into the AutoDockTool software to determine the active site. Finally, the docking score was calculated and the docking results were visualized using PyMOL software.
Molecular Dynamics Simulation
To ensure the stability of the obtained compound-target combination, MDS of the ligand-receptor conformation with the best molecular docking binding energy were performed using GROMACS 2023 (www.gromacs.org). The protein with the lowest energy calculated by the docking process was used as the starting geometry for the MDS process. The charmm36-jul2022 force field was used to obtain the topology file and parameters of TET from the PRODRG server. These complexes were then inserted into an 0.85 × 0.85 × 0.85 nm periodic cubic box with an extended simple point charge (SPC/E) model of water used for dissolution. An adequate amount of sodium ions was added to the neutralization system. Three-dimensional periodic boundary conditions are applied to the system. To release unfavorable contacts in all MDSs, energy minimization was performed using the steepest-descent method followed by 100 ps equilibration in the NPT and NVT ensembles. A velocity-rescale thermostat was used to obtain a constant temperature of 310 K. A Parrinello-Rahman barostat was used to achieve a constant pressure of 1 bar. The LINCS algorithm was used to limit all bonds. The particle mesh Ewald method and a 12 Å cut-off distance were used to assess the long-range interactions. The Verlet cut-off scheme was also used for neighbor search, and the neighbor lists were updated every 10 steps. The leapfrog algorithm was ultimately used to perform 100 ns MDSs. The binding free energy between the ligand and protein was calculated using the molecular mechanics/Poisson–Boltzmann surface area (MM/PBSA) method.
Experimental Verification
Establishment of H22 Tumor-Bearing Mice Model and Drug Treatment
The herbal components of JDP, including SFR 9 g, RCP 12 g, and HDH 20 g, were prepared according to the standardized decoction protocol previously established by our group. 30 Each herb was soaked in water (450 mL) for 20 minutes and decocted for 40 minutes. The liquids were then filtered into a beaker and boiled for 30 minutes with 300 mL of water. The 2 extracts were combined and filtered. Each decoction was then evaporated to concentrate under reduced pressure and stored at −20°C or 4°C for subsequent use.
Eighteen specific pathogen-free male BALB/c mice (6-8 weeks old, 18 ± 2 g) were purchased from Beijing Sibeifu Biotechnology Co. Ltd. (Beijing, China). Mouse H22 cells were purchased from the China Center for Type Culture Collection (CCTCC number: 3142C0001000000110). Animals were provided free access to water and food under standard conditions. After 1 week of adaptive feeding, the mice were randomly divided into 6 blank control group and 12 mice for modeling, the cell concentration was adjusted to about 2×l06/ml, and the cell survival rate was ≥95%. The cells were collected, resuspended, and injected into the right underarm of each mouse. Before administration on the second day, tumor-bearing mice were randomly divided into two groups (n = 6 in each group): model group (normal saline) and JDP group (6.2 g/kg body weight), 30 while blank control group was given normal saline. All mice were given 0.2 mL once daily for 14 days. Twenty-four hour after the last intragastric administration, the cervical vertebrae were removed and the spleen and tumor tissues were taken.
Flow Cytometry Analysis
The antibodies used in this study included CD3-BV786 (BD-740854; clone: 500A2), CD4-BUV395 (BD-565974; clone: GK1.5), CD8-BV510 (Biolegend-100752; clone: 53-6.7), Annexin V-PE (Biolegend-640907), IL-6R-BV605 (BD-554462; clone: D7715A7), and STAT3 (pY705)-AF488 (BD-557814; clone: 4/P-STAT3). Phenotypic staining and phosphorylated antibody staining were performed. All antibodies were diluted at the recommended ratio of 1:50 as per the instructions, and incubated at 4°C in the dark for 30 minutes. After staining, the samples were centrifuged at 300g for 5 minutes and then resuspended in 500 μL PBS for detection. In this study, LSR Fortessa cell analyzer (BD Biosciences) was used, and data analysis was performed using FlowJo software.
Immunohistochemical Staining
Paraffin-embedded sections of tumor tissues were dewaxed with xylene and immersed in an ethanol gradient before being rehydrated with water. The tumor tissue was repaired using ethylenediaminetetraacetic acid (EDTA) antigen retrieval solution (pH 9.0; Servicebio) in a microwave. After cooling at 25°C, the slides were placed in PBS (pH 7.4) and shaken on a decolorizing shaker 3 times for 5 minutes each time. The slides were immersed in 3% hydrogen peroxide solution for 25 minutes at room temperature before washing 3 times for 5 minutes each time on a decolorizing shaker. The slides were incubated with 3% bovine serum albumin at 25°C for 30 minutes before incubating with primary antibody, including mouse antibody against interleukin-6 (IL-6; G111117, Servicebio), signal transducer and activator of transcription 3 (STAT3; GB111776, Servicebio), tumor necrosis factor-alpha (TNF-α; GB11188, Servicebio) and vascular endothelial growth factor (VEGF; GB15165, Servicebio). This was followed by incubation with horseradish peroxidase-conjugated goat anti-rabbit secondary antibody for 50 minutes. 3,3-Diaminobenzidine chromogenic fluid was used for color development. Images were captured under identical exposure settings using fluorescence microscopy. Quantitative analysis was performed using ImageJ software by applying a consistent threshold across all images to calculate the percentage of positive staining area.
Statistical Analyses
Statistical analyses were performed using SPSS 25.0, GraphPad Prism 8.0, and R version 4.0.5. Continuous variables that fit the normal distribution were presented as means ± standard deviations and compared using the t-test. Non-normally distributed continuous variables were presented as medians (Q1 and Q3) and compared using the Wilcoxon rank-sum test. Categorical variables were compared using Pearson’s chi-square test. To account for multiple statistical comparisons, P-values were adjusted using the Bonferroni correction method where applicable. A Bonferroni-corrected P-value < .05 was considered statistically significant.
Results
Baseline Characteristics of HCC Patients
Table 1 summarizes the baseline characteristics of 1021 HCC patients included in this study. Of these, 481 had received YFJP treatment for more than 3 months and 540 had only received comprehensive Western medicine treatment without TCM treatment. Before propensity score matching (PSM), the YFJP group had a higher proportion of patients with AFP < 400 ng/mL, single tumor, and tumor size < 5 cm compared to the corresponding proportions in the control group. In addition, YFJP group patients had lower WBC, NLR, ALT, TBIL, γ-GGT, and INR, and higher ALB, PTA, CRP, and T CD8+T, and CD4+T cell counts compared to those observed in the control group. Compared to those in the control group, the YFJP group had a higher proportion of BCLC A patients and a lower proportion of BCLC C and D patients. More patients in the YFJP group underwent minimally invasive treatment or surgical resection. Other clinical characteristics were not significantly different between the 2 groups. To reduce the influence of baseline heterogeneity between the 2 groups, 1:1 PSM was performed using variables including WBC, NLR, γ-GGT, INR, PTA, CRP, AFP, liver function indicators (ALB, ALT, TBIL), tumor characteristics (BCLC stage, tumor size and number), immune-related parameters (CD8+T, and CD4+T cell counts), and treatment type. Eventually, 307 patients were included in each group, and there was no significant differences between the 2 groups (P > .05; Table 1).
Baseline Characteristics of HCC Patients Before and After Propensity Score Matching.

Abbreviations: YFJP, Yangyin Fuzheng Jiedu Prescription; SD, standard deviation; HBV, hepatitis B virus; HCV, hepatitis C virus; WBC, white blood cells; NLR, neutrophil-lymphocyte ratio; ALT, alanine aminotransferase; TBIL, total bilirubin; ALB, albumin; γ-GGT, γ-glutamyl transferase; PTA, prothrombin time activity; INR, international normalized ratio; CRP, C reactive protein; AFP, alpha-fetoprotein.
Clinical Effect of YFJP
We evaluated the clinical efficacy of YFJP through survival analysis and changes in T lymphocyte count after 1 year of treatment. Kaplan-Meier analysis revealed significantly higher overall survival (OS) and progression-free survival (PFS) in YFJP-treated patients compared to controls (P < .0001). We observed changes in CD8+T and CD4+T cell counts after 1 year of treatment. There were no significant differences between the 2 groups at baseline, and after 1 year of treatment, CD8+T and CD4+T cell counts were significantly higher in the YFJP group than in the control group (Figure 1E and F).

The changes of overall survival (OS), progression-free survival (PFS), and peripheral blood T lymphocytes after treatment. Comparison of OS and PFS of Yangyin Fuzheng Jiedu prescription (YFJP) and the control group before (A and B) and after (C and D) propensity score matching (PSM). After PSM, the changes of CD8+T cell (E) and CD4+T cell counts (F) at 1-year follow-up in the control and YFJP groups.
YFJP Compound-Target Network and Analysis
Using TCMSP database screening (OB ≥ 30%, DL ≥ 0.18), we identified 98 active compounds from YFJP. They included 8 GR, 20 HMM, 7 AMK, 17 RB, 45 SFR, 7 HDH, and 6 RCP. Because OJ could not be retrieved from the TCMSP database, 22 active compounds were obtained from the BATMAN-TCM database for a final total of 120 active YFJP compounds. The targets of these compounds were further screened. Target information was not retrieved for 3 compounds; 1046 potential targets were obtained after removing duplicate values.
As shown in Supplemental Figure S1, a network of 8 herbs, 117 active components, and 1046 potential targets of YFJP was constructed. The green triangles in the network represent the 8 herbs of YFJP, the different colored circles around the triangles represent the active components of each herb. The 7 yellow circles refer to the shared components of herbs. The red squares represent the 1046 potential targets of YFJP. The network topology analysis revealed that the higher the degree of nodes, the stronger the function. Supplemental Table S1 summarizes the top 10 compounds ranked by degree and their corresponding herbs. A certain herb appears more frequently, and we believe that this herb may play a major role in YFJP.
YFJP-Compound-Target-HCC Network and Analysis
After deleting reduplicated targets, a total of 1287 HCC-associated targets were obtained from DisGeNET, TTD, Drugbank, OncoDB.HCC, and liverome databases. Based on 1287 targets in HCC and 1046 targets in YFJP, 238 overlapping targets between HCC and YFJP were obtained, which were considered potential targets for YFJP treatment of HCC (Figure 2A). As shown in Figure 2B, a network of 8 herbs, 117 components, and 238 overlapping targets was constructed. Topological analysis revealed that compounds with the top 10° had strong therapeutic effects (Supplemental Table S2). Similarly, we believe that a certain herb appears more frequently and may play a major role in YFJP treatment of HCC.
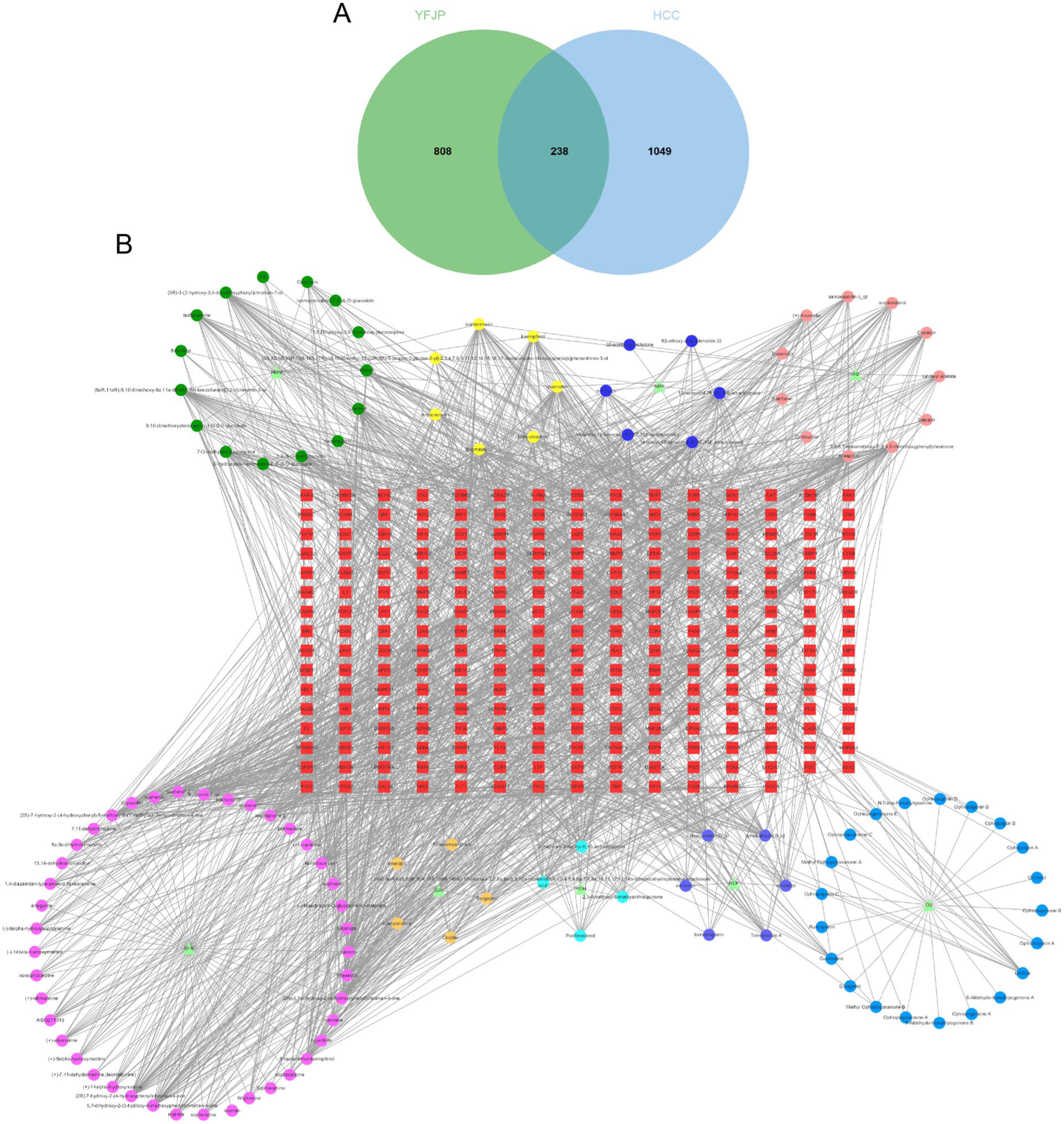
(A)Target mapping for YFJP and HCC. (B) Compound-target network of YFJP on HCC. The triangle denotes the herb. The circle denotes the herb active compound. Seven yellow circles denote the shared components in 2 or more herbs of YFJP. The square represents the overlapping targets of YFJP on HCC.
JDP-Compound-target-HCC Network and Analysis
As shown in Supplemental Tables S1 and S2, JDP herbs (SFR, RCP, and HDH) in YFJP may play an important role in the treatment of HCC. Accordingly, a network pharmacological analysis of JDP was performed to explore the pharmacological effects of JDP in the treatment of HCC. DL ≥ 0.18 was the criterion to screen the active compounds of JDP. After removing duplicate values, 130 compounds and 873 potential targets were obtained. Combined with the aforementioned 1287 HCC-related targets, 205 overlapping targets for JDP and HCC were identified (Figure 3A), and a network of 3 herbs, 130 components, and 205 overlapping targets was established (Figure 3B). As shown in Table 2, we performed topological analysis of the network and selected the top 4 compounds with the highest degree of activity as the key active compounds for JDP treatment of HCC: quercetin, luteolin, (2R)-5,7-dihydroxy-2-(4-hydroxyphenyl) chroman-4-one, and apigenin.

Network pharmacological analysis predicts key components and potential targets of JDP therapy for HCC. (A) Overlapping targets of JDP and HCC. (B) Compound-target network of JDP on HCC. The triangle denotes the herb, the circle denotes the herb active compound, and the yellow circle denotes the shared components in 2 herbs of JDP. The red square denotes overlapping targets of JDP on HCC. (C) Construction of the PPI network to identify potential targets of JDP. (D) The top 15 target networks are shown.
Top 4 Compounds Information in the JDP and HCC Network.

PPI Network and Analysis
A PPI network was constructed by submitting 205 overlapping targets of JDP- and HCC-associated genes to the STRING database. As shown in Figure 3C, the PPI network contained 205 nodes and 3020 edges. In addition, topological analysis was performed. The higher the degree of the node, the darker is the color. We selected the top 15 targets with the highest degrees as core targets, as shown in Figure 3D: GAPDH, CTNNB1, EGFR, VEGFA, TNF, HRAS, ESR1, IL6, HSP90AA1, CASP3, JUN, MAPK3, ERBB2, STAT3, and SRC.
GO Enrichment and KEGG Pathway Analyses
To explore the biological mechanism of JDP treatment of HCC, we used the DAVID platform to conduct GO biological process and KEGG pathway enrichment analyses on 205 overlapping targets of JDP and HCC. The GO results were mainly related to the regulation of cell death and immune system processes, phosphate-containing compound metabolic processes, JAK-STAT, and other related biological processes. As shown in Figure 4A, we screened 10 biological process (BP), cellular component (CC), and molecular function (MF) terms based on the number of targets. In addition, 163 KEGG pathway terms were obtained. As shown in Figure 4B, 20 pathways in the KEGG database were selected based on the-log p-values. Green to red indicate -lg p-values ranging from small to large. The size of the circle indicates the number of gene proteins in the pathway. In the KEGG pathways, most of the genes were related to hepatitis B, HCC, and hepatitis C diseases, and mainly related to the phosphoinositide 3-kinase (PI3K)-AKT, TNF, and JAK-STAT signaling pathways.

(A) GO analysis of 205 overlap targets for JDP treatment of HCC. (B) KEGG analysis of 205 overlap targets for JDP treatment of HCC.
Molecular Docking and Molecular Dynamics Verification
A combined GO, KEGG, and PPI network analysis revealed 4 core hub target proteins: IL-6, STAT3, VEGF, and TNF. These were used for molecular docking analysis with the 4 key compounds obtained by the JDP-compound-target-HCC network. As shown in Figure 5A, the 4 compounds and 4 proteins all had strong binding affinity, and the affinity of the 4 compounds quercetin, luteolin, apigenin, and (2R)-5,7-dihydroxy-2-(4-hydroxyphenyl) chroman-4-one with STAT3 is −8.2, −8.4, −8.4, and −8.2 kcal/mol, respectively, indicating that these 4 compounds have effective binding capacity with STAT3 protein. Finally, we visualized the docking results (Figure 5B-E).

The core components and targets were validated using molecular docking and molecular dynamics simulations. (A) Heatmap of binding affinity between compounds and targets (kcal/mol); Docking of STAT3 with quercetin (B), luteolin (C), apigenin (D) and (2R)-5,7-dihydroxy-2-(4hydroxyphenyl) chroman-4-one (E); (F-H) Molecular dynamics simulation verified the strongest binding complex STAT3 with luteolin and apigenin. (F) RMSD diagram during molecular dynamics simulations. (G) RMS fluctuation in molecular dynamics simulations. (H) Radius of gyration during molecular dynamics simulations.
We further verified the 2 strongest binding pairs of luteolin-STAT3 and apigenin-STAT3 by molecular dynamics simulation. As shown in Figure 5F, both complexes reached equilibrium after approximately 60 ns. The RMSD value of the apigenin-STAT3 complex stabilized around 0.25 ± 0.015 nm, indicating better conformational stability compared to the luteolin complex. In Figure 5G, the luteolin-STAT3 complex exhibited a higher RMSF value, suggesting increased flexibility of the binding interface. Moreover, the Rg of the luteolin-STAT3 complex remained slightly lower and more stable throughout the simulation, indicating that the complex maintained a more compact structure (Figure 5H).
JDP Increases the Proportion and Functionality of CD8+T Cells in H22-Bearing Mice
To investigate whether JDP has immunomodulatory effects on HCC, we established a mouse model of H22 bearing tumor. The results showed that the proportion of CD8+T cells in spleen of mice in the model group was significantly lower than that in normal control group (P < .05). In addition, the expression ratio of CD8+T cells in spleen and tumor tissues of mice in the JDP treatment group was significantly increased (P < .01; Figure 6A-C). We further compared the apoptosis level of CD8+T cells in tumor tissues of mice in the model group and the JDP treatment group, and found that the apoptosis level of CD8+T cells in tumor tissues of mice in the treatment group was significantly lower than that in the model group (P < .01; Figure 6D and E). Furthermore, tumor photographs were taken on the day of tissue collection. As shown in Supplemental Figure S2, tumor size in the JDP treatment group was markedly reduced compared to the model group, providing macroscopic evidence of the anti-tumor efficacy of JDP.

JDP can increase the proportion of CD8+T cells in spleen and tumor microenvironment of H22 tumor-bearing mice, and reduce the apoptosis of CD8+T cells in tumor microenvironment. (A-C) Proportion map and statistical analysis of CD8+T cell expression in spleen and tumor-infiltrating lymphocytes (TIL) of H22 tumor-bearing mice. (D and E) Annexin V expression level on CD8+T cells in tumor tissues of H22-bearing mice and statistical analysis. N = 6 in each group, *P < .05, **P < .01.
JDP Reduces the Expression of IL-6/STAT3 Signaling Pathway Proteins in CD8+T Cells
In order to further explore the effect of JDP on IL-6/STAT3 signaling pathway in HCC immune microenvironment, flow cytometry was used to detect the expression levels of IL-6R and p-STAT3 in tumor tissues of H22 tumor-bearing mouse model group and JDP treatment group (Figure 7A-D). The results showed that the expression ratio of IL-6R and p-STAT3 on CD8+T cells in JDP treatment group were significantly decreased (P < .05). Additionally, immunohistochemistry staining of tumor sections revealed significantly decreased expression of IL-6 (P < .001) and STAT3 (P < .01) in the JDP-treated group (Figure 7E-G), suggesting that JDP downregulates the IL-6/STAT3 signaling pathway in the HCC microenvironment.

JDP decreased the expression of IL-6/STAT3 signaling pathway protein in CD8+T cells of H22 tumor-bearing mice. (A-D) Flow cytometry was used to detect the expression ratio of IL-6R and p-STAT3 on CD8+T cells in TILs of tumor-bearing mice. (E-G) The expression of IL-6 and STAT3 in TILs of tumor-bearing mice was detected by immunohistochemical method (200 ×). N = 6 in each group, *P < .05, **P < .01, ***P < .001.
To validate these findings in another HCC model, we further employed an orthotopic liver cancer model established by intrahepatic injection of Hepa1-6 cells. Mice were treated with the same dosage of JDP, and tumor tissues were analyzed by immunofluorescence. Consistently, the expression of IL-6⁺, STAT3⁺, and IL-6⁺/STAT3⁺ double-positive cells was significantly reduced in the JDP-treated group compared to controls (Figure S3A-D). These results confirm that JDP effectively inhibits the IL-6/STAT3 signaling axis in multiple HCC models, further supporting its role in modulating T cell exhaustion and improving the immune microenvironment.
Discussion
The clinical treatment of patients with malignant tumors is determined through multidisciplinary comprehensive analysis, combined with tumor characteristics and specific conditions of patients. The onset of HCC is occult and the prognosis is poor. In the immune microenvironment of HCC, there are many inhibitory factors, especially the decrease of T cells and the exhaustion of function. 31 Therefore, increasing the number of T lymphocytes and reducing apoptosis are particularly important for improving the immunosuppressive microenvironment of HCC. In recent years, there have been more and more researches on TCM assisted treatment of cancer. The adjuvant treatment of HCC can reduce the toxic side effects, enhance tumor inhibition and prolong the survival time of patients.6,32
In this study, we demonstrate that YFJP improves overall survival (OS) and progression-free survival (PFS) in HCC patients, accompanied by increased CD8+T and CD4+T cell counts. And the network pharmacological analysis showed that the 3 kinds of Chinese herbs may be the core Chinese medicines of YFJP in treatment of HCC. In order to further explore the mechanism of JDP, the active ingredients and targets of JDP involved in HCC therapy were screened by network pharmacological analysis, molecular docking, molecular dynamics simulation, enrichment analysis, and other methods. The potential molecular mechanism of H22 tumor bearing mouse model was verified.
JDP includes SCR, HDH and RCP. Matrine is one of the main active components of matrine. Studies have shown that matrine can play an anti-HCC role through various signaling pathways. For example, Qian et al found that matrine inhibited proliferation, invasion, migration and induce apoptosis of HCC cells by inhibiting EGFR/ERK/MMP-2 and PTEN/AKT signaling pathways. 33 The polysaccharide, flavonoids and other components of Hedyotis alba have remarkable anti-tumor effects. 34 For example, Ma et al found that the polysaccharide of Hedyotis diffusa enhanced the antitumor activity of cytokine-induced killer cells. 35 Some studies have shown that certain components of Hedyotis diffusa can restore the function of tumor-infiltrating T cells and increase levels of CD8+T cells. 36 This suggests that the herb may promote the infiltration and function of CD8+T cells by improving the tumor microenvironment, so as to enhance the immune surveillance and killing of liver cancer. RCP extract reportedly has a good inhibitory effect on the proliferation of cultured HepG-2 HCC cells in in vitro and also inhibits the growth of ascites carcinoma in mice transplanted with H22 HCC cells. 37
Network pharmacology identified quercetin, luteolin, and apigenin as the key active compounds of JDP, with STAT3 as a central target. The core target of JDP treatment of HCC was screened by constructing PPI network. Previous studies have shown that quercetin can inhibit the G0/G1 phase of SMMC-7721 HCC cells, inhibit the activation of AKT, and activate Caspase-9 to promote cell apoptosis. 38 This suggests that quercetin may enhance the sensitivity of tumor cells to the immune system by regulating cell cycle and apoptosis-related pathways. Luteolin can induce autophagy and promote apoptosis of hepatocellular carcinoma cells by inhibiting the expression of pyruvate kinase M2 subtype (PKM2) and NF-Κb. 39 IL-6 is a pleiotropic cytokine whose expression level is significantly elevated in the tumor microenvironment and promotes tumor development by activating the JAK/STAT3 signaling pathway.40,41 It is worth noting that the pathway enrichment analysis in this study showed that JDP has a therapeutic effect on HCC through the JAK-STAT signaling pathway. GO bioenrichment analysis also showed that JDP was involved in the regulation of T cell immunity.
To support this hypothesis, we conducted molecular docking and MD simulations. Luteolin and apigenin exhibited the strongest binding affinity to STAT3, and their complexes stabilized after 60 to 80 ns of simulation, demonstrating good structural stability, and compactness. Moreover, studies have shown that IL-6/STAT3 signaling pathway plays an important role in the differentiation and function of T cells. 42 Therefore, we speculated that JDP might improve the expression of CD8+T cells in the microenvironment of HCC by regulating IL-6/STAT3 signaling pathway. Finally, we established an H22 tumor-bearing mouse model to verify that JDP can increase the proportion of CD8+T cells in HCC immune microenvironment, while alleviating the apoptosis of CD8+T cells. Our findings suggest that JDP exerts its anti-HCC effects by downregulating the IL-6/STAT3 pathway, a key regulator of T cell exhaustion in the tumor microenvironment. This mechanism is supported by the decreased expression of IL-6R and p-STAT3 in CD8+T cells following JDP treatment.
However, this study has some limitations. First, the clinical retrospective cohort studies were not randomized, controlled, or double-blind. PSM was applied to reduce this bias. Second, the molecular docking and MD simulations are predictive tools and lack experimental validation through techniques such as surface plasmon resonance (SPR). Third, although the findings were verified in tumor tissues of mice, a more definitive conclusion requires further verification in cell experiments.
Conclusion
In conclusion, our study demonstrates that JDP, the core component of YFJP, enhances anti-tumor immunity and inhibits HCC progression by modulating the IL-6/STAT3 pathway. Through an integrative approach combining network pharmacology, molecular docking, and in vivo experiments, we identified quercetin, luteolin, and apigenin as the key active compounds of JDP, with STAT3 as a central target. These findings provide a scientific basis for the use of YFJP as an adjuvant therapy in HCC treatment, offering a multi-component, multi-target approach to address the limitations of current therapies. Future studies should focus on the clinical translation of these findings, particularly in combination with immune checkpoint inhibitors, to improve outcomes for HCC patients.
Supplemental Material
sj-docx-1-ict-10.1177_15347354251380219 – Supplemental material for Mechanism of Jiedu Prescription in Hepatocellular Carcinoma: Integrating Network Pharmacology and Experimental Validation
Supplemental material, sj-docx-1-ict-10.1177_15347354251380219 for Mechanism of Jiedu Prescription in Hepatocellular Carcinoma: Integrating Network Pharmacology and Experimental Validation by Yuan Wu, Xiaoli Liu, Yuqing Xie, Lihua Yu, Huiwen Yan, Qing Pu, Xue Cai, Yuling Liang, Yaxian Kong and Zhiyun Yang in Integrative Cancer Therapies
Footnotes
Abbreviations
TCM, Traditional Chinese medicine; YFJP, Yangyin Fuzheng Jiedu Prescription; JDP, Jiedu prescription; SFR, Sophorae flavescentis Radix (Chinese name: Kushen); RCP, Radix Cynanchi paniculate (Chinese name: Xuchangqing); HDH, Hedyotis diffusae Herba (Chinese name: Baihuasheshecao); PSM, propensity score matching; OS, overall survival; PFS, progression-free survival; MDS, molecular dynamics simulation; GO, gene ontology; KEGG, Kyoto Encyclopedia of Genes and Genomes; PPI, protein–protein interaction; RMSD, root mean square deviation; RMSF, root mean square fluctuation.
Ethical Considerations
This study follows the Declaration of Helsinki and has been approved by the ethical committee of Beijing Ditan Hospital [NO. JDLKZ (2019) D (009) -01]. This was a retrospective study. The ethics committee approved it to be used in this study while ensuring patient privacy, not interfering with the patient’s treatment methods, and not bringing physiological risks to patients. And animal experiments were approved by the Animal Ethics Committee of the Animal Care and Use Committee of Beijing University of Chinese Medicine and in strict accordance with the Guidelines for the Care and Use of Experimental Animals published by the US National Institutes of Health (license no. BUCM-4-2021062301-2067).
Author Contributions
Zhiyun Yang and Yaxian Kong designed the research. Yuan Wu and Xiaoli Liu wrote the manuscript, collected and analyzed the data. Huiwen Yan, Lihua Yu, Qing Pu, and Xue Cai were responsible for the patients’ inclusion and follow-up. Yuqing Xie and Yuling Liang conducted animal experiment and assisted with statistical analysis. All authors contributed to the article and approved the submitted version.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Science Foundation of China (No. 82104781 and 82274479); Laboratory for Clinical Medicine, Capital Medical University; Beijing Research Ward Excellence Program, BRWEP (NO. BRWEP2024W10217010); National Key Research and Development Program (2024YFC3044700); High-level Public Health Technical Personnel Construction Project (Subject leaders-02-16); Key Medical Professional Development Program (No. ZYLX202127); Beijing Hospitals Authority Youth Program (QML20231801); Research and Development of Yangyin Fuzheng Jiedu Prescription for Liver Cancer Medical Institutions (DTTP-202301); and Beijing Municipal Administration of Hospitals’ Ascent Plan (DFL20241802).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
