Abstract
Background: The majority of pediatric oncology patients report use of complementary and alternative medicine. Some naturopathic doctors (NDs) provide supportive pediatric oncology care; however, little information exists to formally describe this clinical practice. A survey was conducted with members of the Oncology Association of Naturopathic Physicians (OncANP.org) to describe recommendations across four therapeutic domains: natural health products (NHPs), nutrition, physical medicine, and mental/emotional support. Results: We had 99 respondents with a wide variance of clinical experience and aptitude to treat children with cancer. Of the majority (52.5%) of respondents who choose not to treat these children, the three primary reasons for this are lack of public demand (45.1%), institutional or clinic restrictions (21.6%), and personal reasons/comfort (19.6%). The 10 most frequently considered NHPs by all NDs are fish-derived omega-3 fatty acid (83.3%), vitamin D (83.3%), probiotics (82.1%), melatonin (73.8%), vitamin C (72.6%), homeopathic Arnica (69.0%), turmeric/curcumin (67.9%), glutamine (66.7%), Astragalus membranaceus (64.3%), and Coriolus versicolor/PSK (polysaccharide K) extracts (61.9%). The top 5 nutritional recommendations are anti-inflammatory diets (77.9%), dairy restriction (66.2%), Mediterranean diet (66.2%), gluten restriction (61.8%), and ketogenic diet (57.4%). The top 5 physical modality interventions are exercise (94.1%), acupuncture (77.9%), acupressure (72.1%), craniosacral therapy (69.1%), and yoga (69.1%). The top 5 mental/emotional interventions are meditation (79.4%), art therapy (77.9%), mindfulness-based stress reduction (70.6%), music therapy (70.6%), and visualization therapy (67.6%). Conclusion: The results of our clinical practice survey highlight naturopathic interventions across four domains with a strong rationale for further inquiry in the care of children with cancer.
Keywords
Introduction
A recent survey of pediatric specialty outpatient clinics in Canada found that 61% of pediatric oncology patients use some form of complementary and alternative medicine (CAM). 1 CAM use decisions by patients and their families are often self-directed and independent of the conventional oncology team. Furthermore, only 77% of CAM users informed their physician of this use and only 57% consulted a pharmacist. 1 This may, in part, be due to the lack of mechanisms at the institutional level for conventional care providers to assist their patients in making informed decisions when it comes to CAM therapies and practitioners. 2 While CAM use remains high, the literature indicates that significant gaps exist in research.3,4 The high prevalence of use, potential for harm, and lack of evidence and resources warrants further research in order to meet the evidentiary demand for CAM efficacy and safety.
This is the first survey to our knowledge that examines current practice in the naturopathic medical community regarding pediatric oncology. Naturopathic oncology, provided by licensed naturopathic doctors (NDs), aims to improve quality of life, manage side effects, facilitate recovery, prevent recurrence, and educate the patient regarding adopting a healthy lifestyle. 5 Naturopathic oncology practice is not to be provided as an alternative form of medicine but ideally in conjunction and inclusive of conventional oncology care in a manner consistent with integrative oncology. 6
This survey gathered information from NDs who see patients with cancer regarding: (1) practitioner demographics (education, clinical practice, etc), (2) natural health product (NHP) use (recommendations, dosing, reasons for use, and contraindications), (3) nutrition counselling, (4) physical medicine interventions, and (5) mental-emotional treatments.
The primary objectives of this survey are the following: (1) to identify and enumerate the most common therapeutic recommendations (among the 4 principal domains); (2) to identify the principal reasons for use of the primary NHPs recommended; (3) to identify contraindications reported by the respondents regarding NHPs recommended; and (4) to identify characteristics of NDs focused in oncology with regard to their care of pediatric patients with cancer. A secondary objective of the survey was to identify similarities and differences between oncology-focused NDs who do and do not treat children with regard to their pediatric cancer care recommendations.
The survey structure was primarily based on a previously published project from the same institution (Ottawa Integrative Cancer Center) that investigated naturopathic thoracic cancer care, which influenced treatment decisions for a subsequent clinical trial (Thoracic Peri-Operative Integrative Surgical Care Evaluation [TPOISE]). 7 Similarly, to the TPOISE project, this survey will act as a starting point for intervention information collection in order to decide on treatments that will be offered in a clinical setting. Question formulation and organization, intervention choices, data presentation, and general architecture of the Integrative Pediatric Oncology Program (IPOP) survey was influenced by the TPOISE survey. 7 IPOP will provide and develop a safe and evidence-informed hospital-based IPOP, influenced by survey results.
Methods
Survey Content and Development
This survey was developed using the SurveyMonkey platform (https://www.surveymonkey.com/) and consisted of 2 main parts. Part 1 included questions pertaining to the respondent’s education, clinical practice, and whether or not they treated pediatric cancer cases. If they indicated that they did treat pediatric patients, they were queried about their clinical experience and pediatric case exposure. If the ND indicated that they did not treat pediatric patients, they were additionaly asked to provide additional information through open-ended questions as to why they do not treat pediatric cases and the resources that would help them if they decided to pursue pediatric oncology care. Part 2 collected information regarding integrative therapies that respondents might recommend for pediatric cancer patients. Participants, including both those who do and do not treat children, were queried across 4 main domains of treatment including NHPs (56 options), Nutrition Counselling (12 options), Physical Interventions (12 options), and Mental-Emotional Interventions (14 options). A complete list of all 56 NHPs that were presented to respondents to choose from is available as a supplementary file (Supplementary File 1, available online). Open-text fields were available to enter interventions not listed. Respondents were asked to provide additional information on the NHP interventions selected regarding route of administration, dosing, reasons for use, and contraindications. In addition to the response options in the Nutrition Counselling domain, respondents were asked to provide general information regarding foods they encouraged and discouraged their patients to consume. No additional information was collected beyond selected interventions for the Physical Interventions and Mental-Emotional domains. It is important to note that respondents could skip sections/questions.
Prior to survey dissemination, 5 pilot testers with expertise in naturopathic oncology were recruited to take the survey and provide feedback. After refining, the survey was sent out to the study sample to a total of 447 recipients in May 2017. To encourage survey completion, the survey was advertised through social media (Twitter and Facebook). Seven rounds of reminder emails were sent to nonresponders and partial responders to encourage completion. An incentive prize draw was also promoted after completion of the study. The survey received ethics approval from the Canadian College of Naturopathic Medicine Research Ethics Board (CCNMREB017).
Respondent Characteristics and Selection Method
The survey was sent out to members of the Oncology Association of Naturopathic Physicians (OncANP), a professional organization composed of licensed NDs, naturopathic medical students, and allied health care providers primarily across North America. A membership list was acquired from the OncANP, and survey invitations were sent to those meeting the following inclusion criteria: having an “Active,” “On Hold,” or “Pending” subscription status and holding a membership classification of “FABNO membership” or “Associate ND Membership.” Members with an “Expired” subscription status, a “Student Membership,” or “Allied Membership” classification were excluded. Of the 583 members on the OncANP mailing list, invitations were sent to 446 NDs. One ND who showed interest as a result of the social media campaign, but was not part of the OncANP membership, was subsequently sent an invitation. As such, 447 invitations were initially sent out. Altogether, there were 450 NDs, including 3 of the pilot testers, who were invited to complete the survey.
Data Analysis
Survey results were exported into Microsoft Excel 2016 (Microsoft Office, Redmond, WA) for analysis. Results are presented using descriptive statistics (counts and percentages). Open-ended questions were manually coded into general response categories by a single author (AP) using inductive coding with sampling and iterative re-coding. In the NHP domain, interventions that were selected by >50% of pediatric cancer treaters were considered as primary recommendations. A more detailed analysis of these NHP recommendations, including reason for use and contraindications, was conducted for these primary recommendations. For all other domains, recommendations selected by >50% of all respondents were also considered as primary recommendations. Differences between recommendations or considerations for interventions between pediatric treating NDs and non-pediatric treating NDs were compared using either the χ2 test 8 or Fisher’s exact test. 9
Results
Survey Dissemination and Respondent Characteristics
A total of 99 responses were collected (96 through invitation and 3 from pilot testers), reflecting a 22.1% response rate. Of these, 67 (67.7%) were complete responses and 32 (32.3%) were partial responses. A total of 62.6% of respondents practice in the United States, 36.4% practice in Canada, and 1.0% practice in Greece. The top educational institution at which the NDs were trained was the Canadian College of Naturopathic Medicine (34.3%). Over one third of the respondents had between 11 and 19 years of clinical experience (37.4%). A total of 47.5% of respondents self-identified as treating pediatric oncology cases, whereas 52.5% stated that they do not treat pediatric oncology cases. Detailed respondent characteristics are summarized in Table 1.
Respondent Characteristics.

The National College of Natural Health has since changed to the National University of Naturopathic Medicine, and so “other” responses that included the new name were pooled all together under “National University of Naturopathic Medicine.”
Additional Information Collected From NDs Who Do Not Treat Pediatric Cases (N = 51)
The most frequent reason reported by respondents to explain why they do not treat pediatric cancer cases was an absence of public demand (ie, patients and families/caregivers not seeking the direct care of an ND; 45.1%). Overall, 59.7% of non-treater respondents reported a need for evidence-related resources (dosing, safety, clinical data/research), and 35.8% required educational resources (training, conferences, mentorship). Further details from the survey respondents as to reasons for not treating pediatric cancer cases and the resources that would be needed to better support a change in this practice are outlined in Tables 2 and 3, respectively.
Reason for Not Treating Pediatric Cancer Cases a (N = 51).
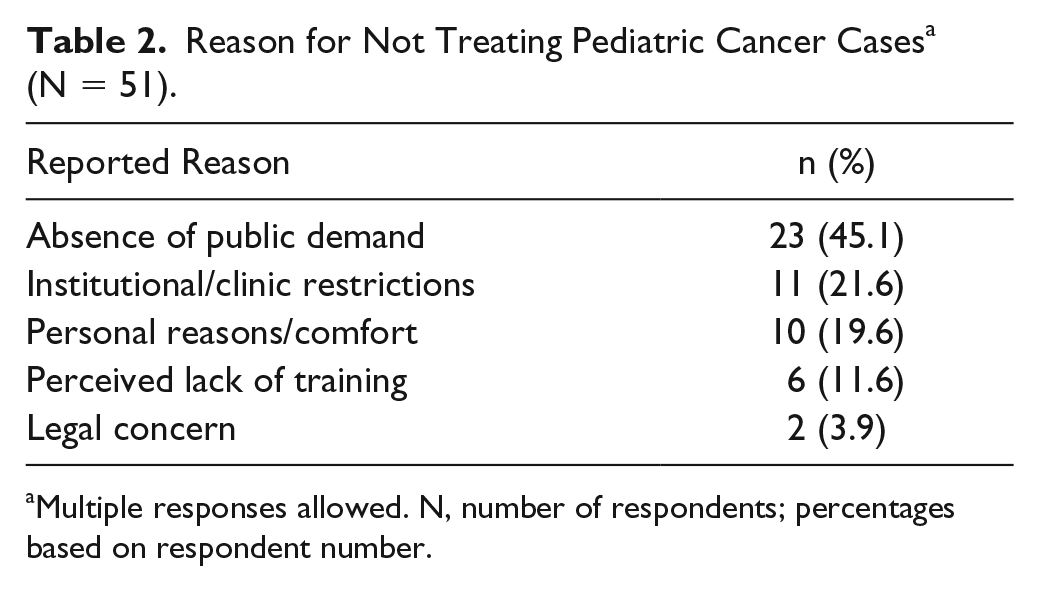
Multiple responses allowed. N, number of respondents; percentages based on respondent number.
Resources Required and Requested to Begin Treating Pediatric Cases a (N = 67).

Multiple responses allowed. N, number of respondents; percentages based on respondent number.
Natural Health Product Domains
Primary Recommendations
Eighty-four respondents provided complete information within the NHP domain (recommendations, reasons for use, reported contraindication, dosing, and administration). Eighteen NHPs were identified as primary recommendations (selected by >50% of those who do treat pediatric cancer cases): vitamin D (88.4%), fish-derived omega-3 (86.0%), probiotics (86.0%), vitamin C (79.1%), melatonin (76.7%), turmeric (Curcuma longa, curcumin extract; 74.4%), glutamine (72.1%), homeopathic Arnica (67.4%), Astragalus membranaceus (62.8%), magnesium (citrate, bisglycinate, etc; 62.8%), Coriolus versicolor (60.5%), zinc (58.1%), Boswellia serrata (frankincense; 53.5%), coenzyme Q10 (53.5%), ginger (Zingiber officinale; 53.5%), green tea (Camellia sinensis, epigallocatechin-3-gallate [EGCG]; 53.5%), reishi (Ganoderma lucidum; 53.5%), and vitamin B complex (53.5%; Table 4).
Survey Recommended Natural Health Products.

Abbreviations: NDs, naturopathic doctors; PSK, polysaccharide K; EGCG, epigallocatechin-3-gallate.
Reasons for Use of Primary NHP Recommendations
Based on responses by both those who do treat and do not treat pediatric cancer cases, the reasons for use are presented in Table 5 for each of the 18 primary NHP recommendations. Respondents were allowed multiple responses that were in open-text format. The total number of respondents inputting for each NHP varied, with participants being able to skip the section if they chose. Notably, 4 reasons for use were significantly different between treaters and non-treaters. Both melatonin and green tea were recommended for their antineoplastic effects significantly more by those who do not treat pediatric cases compared with those who do (P = .04 and P = .02, respectively). Vitamin C was recommended significantly more for “other” uses than provided in the survey by non-treaters compared with treaters (P = .02). Homeopathic Arnica was recommended for general side effect management significantly more by non-treaters compared with treaters (P = .003).
Primary NHP Uses/Contraindications/Administration a .

Abbreviations: NHP, natural health products; N, number of respondents (% based on this value); supp, support; QOL, quality of life; Tx, treatment; Sx, side effects; injectable, includes both subcutaneous and intramuscular administration; PSK, polysaccharide K; EGCG, epigallocatechin-3-gallate.
Data presented refers to response rates from all ND’s (both treaters and non-treaters). Percentages based on number of respondents. Note that multiple responses from respondents were allowed. Response was selected significantly more by non-treaters compared with treaters (P = .05).
Overall, 6 NHPs were primarily recommended for augmentation of the immune system (vitamin D, vitamin C, Astragalus membranaceus, Coriolus versicolor, Ganoderma lucidum, and zinc). Three NHPs were recommended primarily for anti-inflammatory effects (fish-derived omega-3, turmeric/curcumin, and Boswellia serrata). Two are primarily used for organ and tissue support (CoQ10 and homeopathic Arnica), which encompass endpoints such as postoperative recovery and cardiovascular support. Only one NHP was primarily recommended for antineoplastic activity (green tea/Camellia sinensis). All the remaining NHPs were primarily recommended for side effect–related endpoints; probiotics for gastrointestinal support (including diarrhea, constipation, and painful bowel movements), melatonin (insomnia and sleep quality), ginger (nausea/vomiting), magnesium for gastrointestinal support (including constipation and bowel movement regulation), and glutamine for general side effect management.
Contraindications of Use of Primary NHP Recommendations
All primary NHP recommendations, except homeopathic Arnica, had identified contraindications recorded by respondents. Contraindications identified for the primary NHP recommendations are presented in Table 5. No reported contraindications were significantly different between the 2 groups; however, radiation therapy almost achieved significance for treaters reporting it more often than non-treaters as a contraindication for green tea (P = .05). NHPs with specific cancer-type contraindications identified include melatonin (hematological cancers), Astragalus membranaceus (hematological cancers), Coriolus versicolor (hematological cancers), Ganoderma lucidum (hematological cancers), and Boswellia serrata (brain tumors). NHPs with specific chemotherapy agent interactions include fish-derived omega-3 (platinum-based chemotherapy), turmeric/curcumin (cyclophosphamide), Coriolus versicolor (cyclophosphamide), CoQ10 (anthracyclines), vitamin C (Velcade and methotrexate), vitamin D (tamoxifen), and vitamin B complex (5-fluorouracil chemotherapy). Turmeric/curcumin, glutamine, green tea, vitamin C, CoQ10, and Boswellia serrata were also flagged for unspecified antineoplastic treatments.
NHPs reported to have perioperative/bleeding risk include fish-derived omega-3, turmeric/curcumin, Ganoderma lucidum, ginger, CoQ10, and Boswellia serrata. Recommendations that participants felt required further general investigation, without a specific contraindication reported, included fish-derived omega-3, Ganoderma lucidum, melatonin, green tea, Coriolus versicolor, vitamin C, vitamin D, zinc, Astragalus membranaceus, vitamin B complex, and Boswellia serrata.
It is of note to interpret these findings with caution, as while contraindications were reported by respondents, this does not necessarily reflect the current literature. A comprehensive review of NHP contraindications would be warranted and allow for a clear representation of true risks according to the best available data.
Dosing Approaches and Administration of Primary NHP Recommendations
Participants were asked to both identify dosing rules/approaches, which they commonly use, and also to provide typical dose ranges for each of the NHPs they chose in the previous section. Dosing approaches ranged widely between responders, primarily being based on different weight-adjusted rules. The route of administration (Table 5) for individual NHPs also varied between responders. The broad range of dosing could be as result of different NHP formulations (solid extract, dry herb, patent products, etc) and NDs accessing different sources of evidence to base recommendations on. The variance in dosing approaches and ranges is consistent with the need identified by non-treaters as an area that requires better elucidation.
Nutrition Counselling Interventions: Primary Recommendations
Sixty-eight of the respondents selected nutrition recommendations that they would generally use in practice from a list provided. Primary nutrition recommendations (selected by >50% of all respondents) included the following: anti-inflammatory focused diet (77.9%), dairy restriction (66.2%), Mediterranean diet (66.2%), gluten restriction (61.8%), ketogenic diet (57.4%), and low glycemic diet (52.9%). No significant differences were found for any nutrition recommendation between treaters and non-treaters (Table 6).
Nutrition Domain a .

Abbreviation: NDs, naturopathic doctors.
Multiple responses allowed, and percentages based on respondent numbers.
Physical Medicine Interventions: Primary Recommendations
Sixty-eight respondents selected physical medicine interventions that they would generally recommend in practice from a list provided. Primary physical medicine recommendations (selected by >50% of all respondents) included the following: exercise (94.1%), acupuncture (77.9%), acupressure (72.1%), craniosacral therapy (69.1%), yoga (69.1%), hydrotherapy (67.6%), and massage therapy (66.2%). No significant differences were found for any physical medicine recommendation between treaters and non-treaters (Table 7).
Physical Domain a .

Abbreviation: NDs, naturopathic doctors.
Multiple responses allowed, and percentages based on respondent numbers.
Mental-Emotional Interventions: Primary Recommendations
Sixty-eight respondents selected mental-emotional interventions that they would generally recommend from a list provided. Primary mental-emotional recommendations (selected by >50% of all respondents) included the following: meditation (79.4%), art therapy (77.9%), mindfulness-based stress reduction (70.6%), music therapy (70.6%), visualization (67.6%), cognitive-behavioral therapy (61.8%), progressive muscle relaxation (57.4%), diaphragmatic breathing (55.9%), psychotherapy (54.4%), and reiki (52.9%). No significant differences were found for any mental-emotional physical medicine recommendation between treaters and non-treaters (Table 8).
Mental and Emotional Domain a .
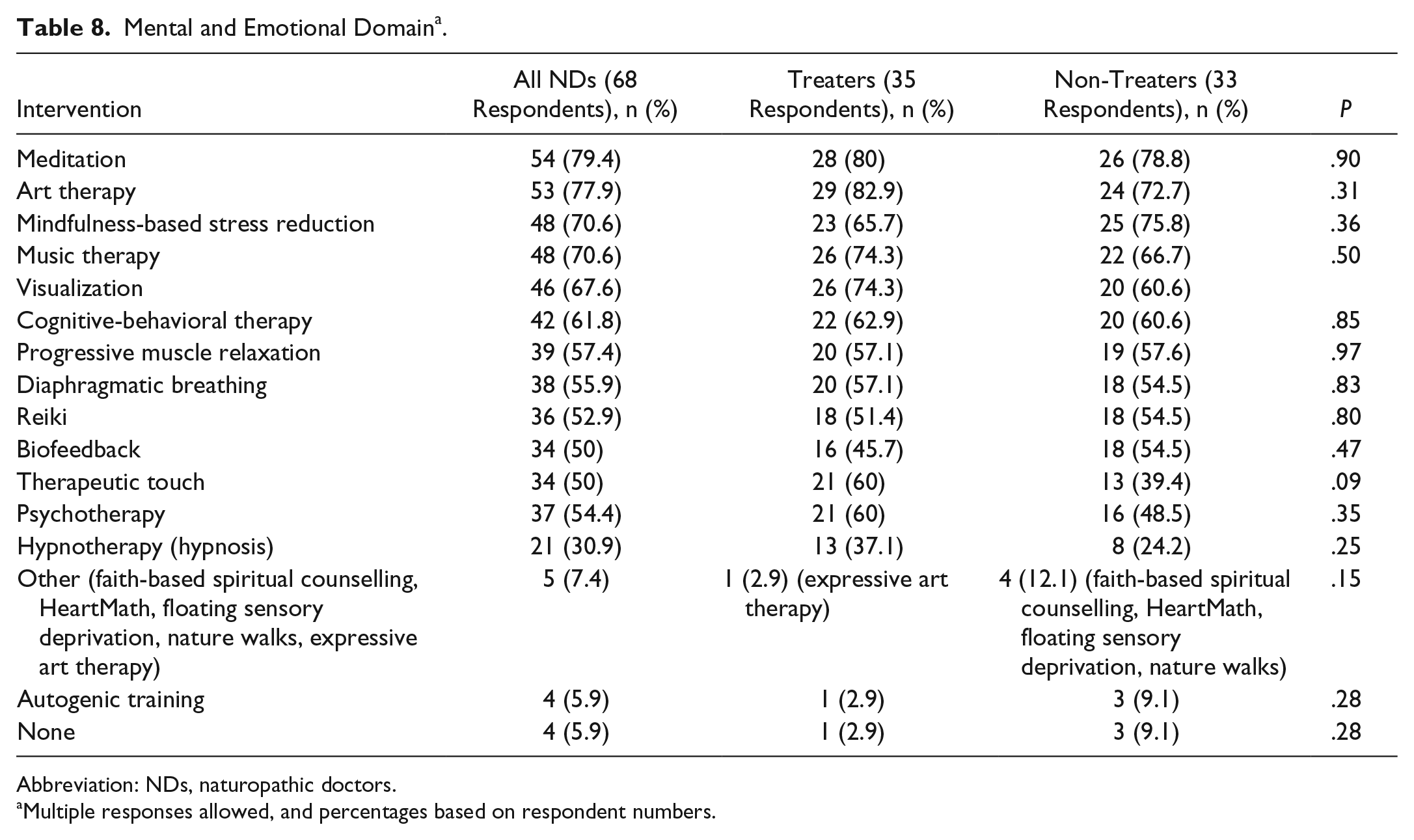
Abbreviation: NDs, naturopathic doctors.
Multiple responses allowed, and percentages based on respondent numbers.
Discussion
This report provides important information to better understand common recommendations made by NDs and their clinical applications with regard to pediatric cancer care. The survey highlights naturopathic interventions spanning 4 interventional domains, collecting pertinent information regarding what is currently being recommended, what are the principal reasons for the recommendations, and what safety concerns are perceived to exist. The findings describe and characterize current demographics of NDs who offer cancer care for patients, including their educational background, place of practice, years of clinical experience, degree of involvement in pediatric cancer care, and resources requested for support if treating children with cancer. Furthermore, the results describe which interventions, along with their reasons for use, are likely to be recommended in clinical practices with regard to the 4 domains investigated. Given the broad scope of practice of NDs, these findings help us to better understand common trends of clinical focus currently occurring in naturopathic practice as they apply to pediatric integrative cancer care.
Results from this survey indicate that the primary reason for use of NHPs in pediatric oncology care is side effect/symptom management and supportive care, rather than direct treatment of the cancer. This finding contrasts certain aspects of the literature, which has found that patients and consumers of CAM therapies may not explicitly know NHPs are intended for supportive management and not direct antineoplastic effects.10,11 The most common reason for use of all 18 primary NHPs (except green tea/EGCG/Camellia sinensis) identified were side effect and symptom management related, both general and specific, and overall general supportive care. For example, while melatonin had 19 responses out of 78 relating to antineoplastic effects, it had 59 responses pertaining to supportive symptom management, including helping with insomnia, immune support, and conventional treatment support. It appears that NDs are currently using NHPs primarily to help support patients through conventional treatment and manage associated side effects/symptoms, with an emphasis on immune support. Furthermore, while there was variance regarding NHP recommendations between respondents, it appears that a small number of NHPs were selected by the majority of all respondents (>80%). These majority selected NHPs included omega-3 (fish-derived; 83.3%), vitamin D (83.3%), and probiotics (82.1%). These 3 NHPs can be considered as generally accepted interventions in the naturopathic profession, which are highly likely to be encountered in clinical practice by both patients and other health care providers. Perceived contraindications for NHPs do exist and were identified by respondents which would influence interventions in a clinical setting. With regard to the 3 other interventional domains (nutrition, physical medicine, and mental-emotional), the highest selected choice by all respondents included the anti-inflammatory diet, exercise, and meditation, respectively.
Broadly speaking, with regard to the practitioner characteristics in North America (members of the OncANP), half of the NDs surveyed with a focus in cancer care are open to providing pediatric oncology care. Our results show that the primary reason (45.1% of non-treaters) that NDs are not treating pediatric patients with cancer is an absence of public demand. Future research is required to understand the disconnect between the reported high use of CAM by families 1 and patients accessing CAM providers (such as NDs). It is encouraging that there is clear support for the real-world applicability of the program that this survey will help create. The majority of non-treaters (59.7%) report that they require more information about clinical standards (dosing, safety, and clinical data/research), which we plan to expand on and elucidate through the future IPOP.
Comparing the findings of the current survey to our previous work in the context of perioperative care of thoracic cancers provides interesting insight into naturopathic oncology practice. Fish-derived omega-3, vitamin D, probiotics, melatonin, vitamin C, and homeopathic Arnica were found in the top 10 most frequently selected NHPs by NDs for both pediatric cancers and thoracic cancers. 7 For pediatric settings, turmeric/curcumin, glutamine, Astragalus membranaceus, and Coriolus versicolor were also in the top 10, whereas modified citrus pectin, zinc, whey protein, and a multivitamin/mineral were most common in the thoracic cancer setting. 7 Recommendations in the remaining domains (physical medicine, mental/emotional, and nutrition interventions) were quite similar in the 2 surveys, with some approaches being recommended more commonly in one context compared with the other. Overall, it seems that there is not a substantial difference between recommendations for an adult population with thoracic cancer and a pediatric cancer population. Findings from this survey will be utilized, as were the responses of the TPOISE survey, in order to guide an evidence-driven literature search to form a clinical trial within a hospital setting.
Limitations to this work include a relatively small sample size and response rate compared with the number of practicing NDs who treat cancer. Thus, the responses may not effectively represent the recommendations and thoughts of the entire professional body accurately. In order to organize the data collected, open-ended responses were interpreted and categorized into more common terms, which could have led to misinterpretation of what responders actually intended to describe. It is important to note that we only collected what NDs would consider recommending and not how these translate into actual clinical recommendations in practice. Moreover, we created the survey to assess pediatric oncology in general, and it is likely that recommendations may differ based on the primary cancer type, age, health status, and other comorbidities.
Strengths of this work include that we were able to collect information across multiple treatment domains. We reached out to an organization, the OncANP, that represents NDs with the greatest focus on cancer care and thus most likely representative of some of the best naturopathic treatment considerations available for review. In addition, there was an almost even split between those who do and do not treat pediatric cancer cases, allowing us to obtain and compare information from both practitioner populations.
While formal guidelines pertaining to CAM use are limited, survey research from around the globe shows a high prevalence of use in the pediatric oncology population. A 2009 systematic review of 20 studies incorporating 2871 participants found the prevalence of CAM use among pediatric cancer patients ranged from 6% to 91%. 12 The high prevalence of CAM use by pediatric cancer patients and their families, with limited research and clinical guidance of its clinical use, supports the creation of programs, such as IPOP, which will assess safety and efficacy. Results from this survey provide a starting point on which to explore evidence relating to frequently considered and recommended therapies in CAM. A scoping review for the primary recommendations is currently underway by our team.
Conclusion
According to our survey, approximately half of NDs who treat cancer include pediatric cases within their practice. Those who do not treat pediatric cancer patients state that they primarily require more clinical information before they would treat this population. While recommendations varied between responders, there appears to be a few main interventions that are consistently most recommended among the majority of respondents, including both treaters and non-treaters. There is a need to evaluate the safety and efficacy of the recommendations identified in this survey. Relevant evidence syntheses in this area of pediatric integrative oncology will help develop a safe and evidence-informed hospital-based IPOP.
Supplemental Material
Supplementary_File_1_NHP_list – Supplemental material for Naturopathic Oncology Care for Pediatric Cancers: A Practice Survey
Supplemental material, Supplementary_File_1_NHP_list for Naturopathic Oncology Care for Pediatric Cancers: A Practice Survey by Athanasios Psihogios, Jullie K. Ennis and Dugald Seely in Integrative Cancer Therapies
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support grant from the Children’s Hospital of Eastern Ontario (CHEO) Foundation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
