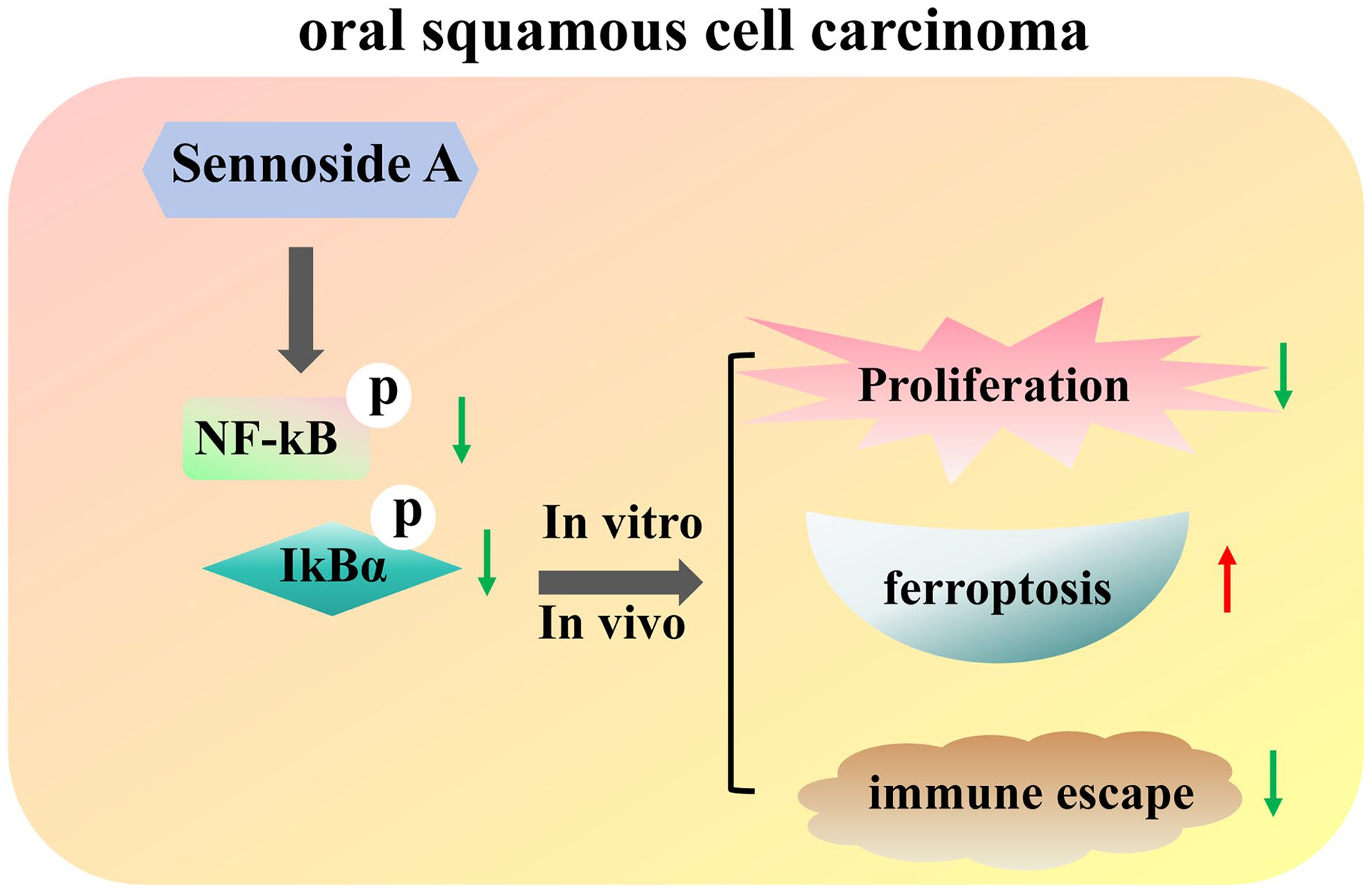
Abstract
Background:
Oral squamous cell carcinoma (OSCC) is an invasive cancer with a high rate of metastasis and recurrence. Anthraquinone natural active element sennoside A (SA) has demonstrated a repressive effect on several malignancies. Its impact on OSCC is still unknown, though.
Methods:
The toxicity effect of SA on OSCC cells was examined by CCK8, to screen the appropriate concentrations for following assays. The effect of SA on proliferation, ferroptosis and immune evasion of OSCC was assessed by CCK-8, DCHF-DA staining, biochemical detection, ELISA, and western blotting. The mechanism of SA on OSCC was determined by western blotting and immunofluorescence. Besides, in vivo effect of SA was investigated on tumor-bearing mice using HE staining, immunohistochemistry, and western blotting.
Results:
SA reduced SCC7 and CAL27 cell viability, with a IC50 values of 94.38 and 77.41 μM, respectively. SA downregulated the expressions of GPX4 and xCT expression and the SOD level, but elevated the levels of ROS, MDA, and Fe2+ in SCC7 and CAL27 cells. SA decreased the PD-L1 expression, whereas increased the cytotoxicity of CD8+ T cells and the concentrations of IFN-γ, IL-2, and TNF-α of SCC7 and CAL27 cells. Mechanically, SA reduced the phosphorylation of NF-κB and IκBα in SCC7 and CAL27 cells. Also, RANKL treatment reversed the outcomes of indicators involved in proliferation, ferroptosis and immune evasion in SCC7 cells. In vivo, SA reduced tumor weight and size, the expression levels of GPX4 and PD-L1 and the phosphorylation of NF-κB and IκBα, but enhanced the IFN-γ level in mice xenografted with SCC7 cells.
Conclusion:
SA suppressed proliferation and immune evasion, but induced ferroptosis through the inactivation of NF-κB pathway in OSCC.
Graphical abstract
Background
Oral squamous cell carcinoma (OSCC) is a usual subtype of head and neck cancer, which affects the oral cavity and oropharynx, thereby critically disrupting the patient’s feeding, speech, and others.1,2 OSCC is the seventh most common cancer globally accounting for 4.5% of all malignancies and contributes to 916 827 deaths every year. 3 Except for surgery, as its main form of treatment, other approaches, such as chemotherapy, radiotherapy, immune checkpoint inhibitors, molecularly targeted drugs as well as their collaborations, have been immensely improved the outcomes of OSCC. 4 However, due to its asymptomatic characteristics at early stages and subtle mucosal changes, the overall 5-year OSCC survival rate is still about 60%. 5 Moreover, aggression, recurrence and metastasis further aggravate its poor prognosis. 2 Multitudinous molecular mechanisms, such as apoptosis, angiogenesis, metastasis, ferroptosis, and immune escape, have been identified.6-8 Thus, agents targeted these molecular mechanisms, should be persistently developed to bring well clinical outcomes of OSCC.
It is well recognized that cancers are influenced by the immune checkpoint-based programed cell death 1 receptor (PD-1) and its ligand, programed cell death ligand 1 (PD-L1). The immunological checkpoint protein PD-1 is expressed on the surface of immune cells, including cytotoxic T lymphocytes. 9 PD-L1 gene is widely expressed in tumor cells. 10 The PD-1/PD-L1 pathway functions as an auto-tissue defense mechanism to preserve peripheral tolerance under typical physiological circumstances. 11 However, immunosuppression results from T lymphocyte malfunction brought on by the tumor cells’ PD-L1 ligand binding to PD-1. When tumors develop immunological tolerance, immune escape is encouraged, enabling tumor cells to multiply and spread by evading immune detection and destruction.12,13
The natural substance sennoside A (SA), an anthraquinone, which comes from Rheum officinale Baill 14 and Rheum palmatum L., is well-known for its laxative qualities as well as its pharmacological benefits such hypoglycemic, anti-obesity, and anti-inflammatory actions.15-18 Moreover, its anti-tumor property has been demonstrated in diverse tumors. For instance, SA suppresses the non-small cell lung cancer cell tumor immune microenvironment and pernicious phenotypes. 19 SA inactivates the PI3K/AKT/mTOR axis, which causes prostate cancer to undergo autophagic death. 20 SA represses growth, mobility and invasion of hepatocellular carcinoma cells. 21 However, its effect on the progression of OSCC remains unclear.
The goal of this study was to investigate how SA affected the development of OSCC both in model of cells and mice. In addition, the mechanism of SA on OSCC involved in NF-κB pathway was determined by the application of RANKL. RANKL is a type II transmembrane protein that is the ligand of the NF-κB receptor activator (RANK), thereby using as an NF-κB activator. 22 This work is the first functional investigation of SA on OSCC that we are aware of. The findings will offer a more thorough comprehension of how SA inhibits the advancement of OSCC.
Methods
Cell Culture
Human oral keratinocytes (HOK; aoruicell, Shanghai, China, Cat# ORC1095, RRID: CVCL_B404), CAL27 cells (SUNNCELL, Wuhan, China, Cat# SNL-179, RRID: CVCL_1107), and SCC7 cells (Cat# SNL-571, RRID: CVCL_V412) were used in this study. The identity and mycoplasma-free status of all cell lines were confirmed by the short tandem repeat (STR) profiling, and the STR profiles of CAL27 and SCC7 cells could be found in website (https://www.ubigene.us/product/WT-Cell-Line/CAL-27.html; https://www.ubigene.us/product/WT-Cell-Line/SCC7.html). All 3 cell lines were cultivated in RPMI-1640 media (Zhong Qiao Xin Zhou Biotechnology, Shanghai, China, Cat# ZQ-230), containing 10% fetal bovine serum (FBS, Zhong Qiao Xin Zhou Biotechnology, Cat# ZQ500-A) at 37°C with 5% CO2. Besides, RPMI-1640 medium was used to cultivate human CD8+ T cells from the American Type Culture Collection (ATCC, Manassas, VA, Cat# PCS-800-017) at 37°C with 5% CO2.
Cell Counting Kit-8 (CCK-8) Assays
HOK cells with a density of 1 × 105 cells/well were seeded in 96-well plates and cultured at 37°C with 5% CO2 overnight. Cells were then washed with phosphate buffer saline (PBS, Solarbio, Beijing, China, Cat# P1020) and incubated with SA with concentrations of 0, 6.25, 12.5, 25, 50, 100, 200, and 400 μM for 48 hours based on the previous study. 20 Subsequently, cells in each well were incubated with 10 μl of CCK-8 reagents (YEASEN, Shanghai, China, Cat# 40203ES76) for 3 hours at 37°C after cells were washed with PBS. Next, the absorbance was recorded at 450 nm by a microplate reader (Thermo Fisher Scientific, Waltham, MA, USA), to calculate the cell viability of HOK cells. SA (Cat# HY-N0365) was bought from MedChemExpress (Monmouth Junction, NJ, USA), dissolved in DMSO (YEASEN, Cat# 60313ES60), and then diluted in PBS (YEASEN, Cat# 41403ES76) for use.
To measure the IC50 for SA in HOK cells, HOK cells with a density of 1 × 105 cells/well were seeded in 96-well plates and cultured at 37°C with 5% CO2 overnight. Then, HOK cells were incubated with 0, 100, 200, 400, 600, 800, 1000, and 1200 μM for 48 h, and washed with PBS. Subsequently, the cell viability of HOK cells was examined by CCK-8 assays as the above description, to calculate the IC50 for SA in HOK cells.
To examine the effect of SA on the cell viability of OSCC, SCC7, and CAL27 cells with a density of 1 × 105 cells/well were seeded in 96-well plates and cultured at 37°C with 5% CO2 overnight. Then, cells were washed with PBS. According to the results from the cell viability of HOK cells, as well as the previous report, 20 0, 6.25, 12.5, 25, 50, 100, and 200 μM of SA were selected to incubate with SCC7 and CAL27 cells for 48 hours. Subsequently, the cell viability of SCC7 and CAL27 cells was examined by CCK-8 assays as the above description.
To assess the effect of SA on immune escape, SCC7 and CAL27 cells were seeded into 24-well plates with a density of 2.5 × 105 cells per well, and cultured at 37°C with 5% CO2 overnight. Then, cells were treated with 25, 50, and 100 µM of SA for 48 h. At the same time, CD8+ T cells were activated as the following description. CD8+ T cells with a density of 1.5 × 106 cells per well were seeded into 6-well plates, and cultured at 37°C with 5% CO2 overnight. Then, CD8+ T cells were treated with Dynabeads™ Human T-Activator CD3/CD28 (Gibco, Rockville, MD, USA, Cat# 11161D) at a bead-to-cell ratio of 1:1. Subsequently, CD8+ T cells were incubated with 100 IU/mL IL-2 (Beyotime, Shanghai, China, Cat# P5115) at 37°C with 5% CO2 for 2 weeks. Next, SCC7 and CAL27 cells treated with SA were co-incubated with activated CD8+ T cells in a 12-well plate at a ratio of 1:10. After 24-hour incubation at 37°C with 5% CO2, the cell viability of CD8+ T cells was examined by CCK-8 assays as the above description.
To verify the direct role of the NF-κB pathway in SA-mediated proliferation in OSCC, SCC7 cells were seeded into 24-well plates with a density of 2.5 × 105 cells per well and cultured at 37°C with 5% CO2 overnight. Subsequently, SCC7 cells were treated with 100 ng/ml of RANKL for 60 minutes, and then incubated with 100 μM of SA for 48 hours. The incubation concentration and time were based on the previous study. 23 The cell viability of SCC7 cells was examined by CCK-8 assays as the above description. RANKL was bought from Sigma-Aldrich (Cat# R0525, St. Louis, MO, USA) and dissolved in PBS for use.
To verify the direct role of the NF-κB pathway in SA-mediated immune escape in OSCC, SCC7 cells were seeded into 24-well plates with a density of 2.5 × 105 cells per well and cultured at 37°C with 5% CO2 overnight. Subsequently, SCC7 cells were treated with RANKL and SA as above description. Meanwhile, CD8+ T cells were activated as the above description. Next, the treated SCC7 cells and activated CD8+ T cells were co-cultured in a 12-well plate at a ratio of 1:10. After 24-hour incubation at 37°C with 5% CO2, the cell viability of CD8+ T cells was examined by CCK-8 assays as the above description.
DCHF-DA Staining
To assess the effect of SA on ferroptosis, SCC7 and CAL27 cells were seeded into 24-well plates with a density of 2.5 × 105 cells per well, and cultured at 37°C with 5% CO2 overnight. Then, cells were treated with 25, 50, and 100 µM of SA for 48 hours. Subsequently, SCC7 and CAL27 cells were washed with PBS and incubated with 5-(and-6)-chloromethyl-2-,7-dichlorofluorescin diacetate (DCHF-DA, MedChemExpress, Cat# HY-D0940) at 37°C for 30 minutes without light. Next, SCC7 and CAL27 cells were collected and analyzed on a FACScan flow cytometry (BD Biosciences, NJ, USA), to determine the relative fluorescence intensities.
To verify the direct role of the NF-κB pathway in SA-mediated ferroptosis in OSCC, SCC7 cells were seeded into 24-well plates with a density of 2.5 × 105 cells per well and cultured at 37°C with 5% CO2 overnight. Subsequently, SCC7 cells were treated with 100 ng/ml of RANKL for 60 minutes, and then incubated with 100 μM of SA for 48 hours. The relative fluorescence intensities were obtained based on the DCHF-DA staining as the above description.
Biochemical Measurement
To assess the effect of SA on ferroptosis, SCC7 and CAL27 cells were seeded into 24-well plates with a density of 2.5 × 105 cells per well, and cultured at 37°C with 5% CO2 overnight. Then, cells were treated with 25, 50, and 100 µM of SA for 48 hours. Subsequently, SCC7 and CAL27 cells were washed with PBS and collected by the centrifugation with 3000g for 5 minutes. Subsequently, the levels of malondialdehyde (MDA) and superoxide dismutase (SOD) were measured by using the commercial kits from Sangon Biotech (Shanghai, China) with the product number of D799761 and D799593. In detail, the collected SCC7 and CAL27 cells with a number of 5 × 106 were resuspended into 1 mL extracting solution (from the kits), and destroyed by ultrasound (power 200 W, ultrasound for 3 seconds, interval 10 seconds, repeated 30 times). After centrifugation at 8000g for 10 minutes at 4°C, the supernatant was removed and placed on ice until measured. The levels of MDA and SOD in the supernatant were calculated after the absorbance was measured at 532 nm (MDA) and 560 nm (SOD) using a microplate reader (Thermo Fisher Scientific).
Detection of the Iron Level
To assess the effect of SA on ferroptosis, SCC7 and CAL27 cells were seeded into 24-well plates with a density of 2.5 × 105 cells per well, and cultured at 37°C with 5% CO2 overnight. Then, cells were treated with 25, 50, and 100 µM of SA for 48 hours. Subsequently, SCC7 and CAL27 cells were washed with PBS and collected by the centrifugation with 3000g for 5 minutes. Next, SCC7 and CAL27 cells with a number of 5 × 106 were resuspended into 1 mL PBS, and destroyed by ultrasound (power 200 W, ultrasound for 3 seconds, interval 10 seconds, repeated 30 times). After centrifugation at 8000g for 10 minutes at 4°C, the supernatant was removed and placed on ice until measured. The level of Fe2+ in the supernatant was measured by using the commercial kits from Abcam (Cambridge, UK) with the product number of ab83366. The absorbance was measured at 593 nm using a microplate reader (Thermo Fisher Scientific).
To verify the direct role of the NF-κB pathway in SA-mediated ferroptosis in OSCC, SCC7 cells were seeded into 24-well plates with a density of 2.5 × 105 cells per well and cultured at 37°C with 5% CO2 overnight. Subsequently, SCC7 cells were treated with 100 ng/ml of RANKL for 60 minutes, and then incubated with 100 μM of SA for 48 hours. Subsequently, SCC7 cells were collected for ultrasound, and the supernatant was collected. The relative level of Fe2+ was measured as the above description.
Enzyme-Linked Immunosorbent Assay (ELISA)
To assess the effect of SA on immune escape, SCC7 and CAL27 cells were seeded into 24-well plates with a density of 2.5 × 105 cells per well, and cultured at 37°C with 5% CO2 overnight. Then, cells were treated with 25, 50, and 100 µM of SA for 48 hours. At the same time, CD8+ T cells were activated as the above description. Next, SCC7 and CAL27 cells treated with SA were co-incubated with activated CD8+ T cells in a 12-well plate at a ratio of 1:10. After 24-hour incubation at 37°C with 5% CO2, the supernatant of cells was collected by the centrifugation at 500g for 5 minutes at 4°C. The concentrations of interferon-γ (IFN-γ), interleukin-2 (IL-2), and tumor necrosis factor-α (TNF-α) in the supernatant were measured by using the commercial kits from Beyotime with the product number of PI511, PI580, and PT518. The absorbance was measured at 450 nm using a microplate reader (Thermo Fisher Scientific).
To determine the effect of SA on the release of IFN-γ prior to adding CD8+ T cells in NOK cells, NOK cells were seeded into 24-well plates with a density of 2.5 × 105 cells per well, and cultured at 37°C with 5% CO2 overnight. Then, NOK cells were treated with 100 µM of SA for 48 hours. At the same time, CD8+ T cells were activated as the above description. Next, SCC7 and CAL27 cells treated with SA were co-incubated with activated CD8+ T cells in a 12-well plate at a ratio of 1:10. After 24-hour incubation at 37°C with 5% CO2, the supernatant of cells was collected by the centrifugation at 500g for 5 minutes at 4°C. The concentration of IFN-γ was measured as the above description.
To verify the direct role of the NF-κB pathway in SA-mediated immune escape in OSCC, SCC7 cells were seeded into 24-well plates with a density of 2.5 × 105 cells per well and cultured at 37°C with 5% CO2 overnight. Subsequently, SCC7 cells were treated with RANKL and SA as above description. Meanwhile, CD8+ T cells were activated as the above description. Next, the treated SCC7 cells and activated CD8+ T cells were co-cultured in a 12-well plate at a ratio of 1:10. After 24-hour incubation at 37°C with 5% CO2, the concentration of IFN-γ was measured as the above description.
Immunofluorescence (IF)
To verify the direct role of the NF-κB pathway in SA-mediated progresses of OSCC, SCC7 cells were seeded into 24-well plates with a density of 2.5 × 105 cells per well and cultured at 37°C with 5% CO2 overnight. Subsequently, SCC7 cells were treated with RANKL and SA as above description. Then, SCC7 cells were collected and seeded on the glassy plates, and grown in a cell incubator at 37°C with 5% CO2 overnight. Next, SCC7 cells were fixed using 4% paraformaldehyde (YEASEN, Cat# 60536ES60) at room temperature for 15 minutes. After SCC7 cells were treated with blocking solution (Solarbio, Cat# SW3015) and 0.2% Triton X-100 (Solarbio, Cat# T8200), they were incubated with a rabbit polyclonal antibody against NF-κB p65 (1:100 dilution, Abcam, Cat# ab16502, RRID: AB_443394) at 4°C overnight. The next day, SCC7 cells were incubated with the secondary antibody (1:1000 dilution, Abcam, Cat# ab150077, RRID: AB_2630356) at room temperature for 1 hour. Finally, SCC7 cells were stained with mounting media (Solarbio, Cat# S2110) and imaged using a fluorescent microscope (Olympus, Tokyo, Japan, Cat# IX71).
Animal Experiment
Subjects in this study were BALB/c naked mice (RRID: MGI:2161072) with 4-week-old from Junke (Nanjing, China), which were kept in a SPF environment with a controlled temperature and 12-hour light-dark cycle. Mice were accommodated for 1 week before the experiment. Then, the left flank of mice was subcutaneously injected with 2 × 105 of SCC7 cells, and monitored for consecutive 4 weeks. The mice were assigned at random to 2 groups: the control group and the 10 mg/kg SA group, with each group consisting of 6 individuals. The doses of injected cells and SA were following the earlier reports.24,25 Mice in 10 mg/kg SA group were intraperitoneally injected with 10 mg/kg SA for 10 days daily. Mice in sham group received intraperitoneal injections of an equivalent volume of PBS for 10 days daily. Tumor volume was quantified by the following formula: volume = 0.5 × length × width2. At the end of the 4-week period, the mice were euthanized using excessive inhalation of isoflurane (RWD, Guangdong, China, Cat# R510-22). Subsequently, the tumor tissues were excised, their weights were recorded, and they were preserved for subsequent analyses. The conduct of all animal studies was approved by the Institutional Animal Care and Use Committee of our medical facility.
Hematoxylin and Eosin (H&E) Staining
The tumor tissues were fixed in 4% paraformaldehyde solution overnight, and then underwent the dehydration through a series of ethanol concentrations. Subsequently, the samples were embedded in paraffin (Solarbio, Cat# YA0011), and then cut into slices with a thickness of 5 μm. These slices were stained with H&E (Solarbio, Cat# G1120) and examined using an Olympus light microscope along with the Image-Pro Plus 6.0 image analysis software (Media Cybernetics, USA, RRID: SCR_009729).
Immunohistochemistry
Paraffin-embedded slices were treated for antigen retrieval using a sodium citrate buffer solution (pH 6.0, Beyotime, Cat# P0081) at 94°C for 15 minutes. Slices were blocked with 1% bovine serum albumin (BSA, Beyotime, Cat# ST2249) for 60 minutes. Then, sections were stained with a rabbit polyclonal antibody against GPX4 (1:500 dilution, Abcam, Cat# ab231174, RRID: AB_3073732), a mouse monoclonal antibody against PD-L1 (1:2500 dilution, Proteintech, Wuhan, China, Cat# 66248-1-Ig, RRID: AB_2756526), and a rabbit polyclonal antibody against IFN-γ (1:100 dilution, Invitrogen, Carlsbad, CA, USA, Cat# PA5-95560, RRID: AB_2807362) at 4°C overnight. Subsequently, slices were incubated with secondary antibodies (Goat Anti-Rabbit IgG H&L (HRP; 1:1000 dilution, Abcam, Cat# ab6721, RRID: AB_955447) or Goat Anti-Mouse IgG H&L (HRP; 1:2000 dilution, Cat# ab6789, Abcam, RRID: AB_955439) for 30 minutes at 37°C. Finally, slices were counterstained with hematoxylin (Solarbio, Cat# G1080) and images under an Olympus light microscope.
Western Blotting
To assess the effect of SA on ferroptosis and immune escape, SCC7 and CAL27 cells were seeded into 24-well plates with a density of 2.5 × 105 cells per well, and cultured at 37°C with 5% CO2 overnight. Then, cells were treated with 25, 50, and 100 µM of SA for 48 hours, and collected for the examination of the expression levels of GPX4, xCT, and PD-L1.
To determine the mechanism of SA in the progresses of OSCC, SCC7, and CAL27 cells were seeded into 24-well plates with a density of 2.5 × 105 cells per well, and cultured at 37°C with 5% CO2 overnight. Then, cells were treated with 25, 50, and 100 µM of SA for 48 h, and collected for the examination of the expression levels of NF-κB, phosphorylated NF-κB (p-NF-κB), IkBα, and p-IkBα.ours
To verify the direct role of the NF-κB pathway in SA-mediated ferroptosis and immune escape in OSCC, SCC7 cells were seeded into 24-well plates with a density of 2.5 × 105 cells per well and cultured at 37°C with 5% CO2 overnight. Subsequently, SCC7 cells were treated with RANKL and SA as above description. Then, cells were collected for the examination of the expression levels of GPX4, xCT, and PD-L1.
To determine the effect and mechanism of SA with the same concentrations as used in SCC7 and CAL27 cell lines on the expression of proteins involved in ferroptosis in NOK cells at the same SA concentrations as used in SCC7 and CAL27 cell lines, NOK cells were seeded into 24-well plates with a density of 2.5 × 105 cells per well and cultured at 37°C with 5% CO2 overnight. Subsequently, NOK cells were treated with 100 µM of SA for 48 hours, and collected for the examination of the expression levels of GPX4, xCT, NF-κB, p-NF-κB, IkBα, and p-IkBα.
To evaluate the effect of SA on the progresses of OSCC in vivo, tumor tissues were collected for examination of the expression levels of NF-κB, p-NF-κB, IkBα, and p-IkBα. The relative protein expression levels were examined by western blotting. The procedures of western blotting were as following.
Tumor cells and tissues were lysed with RIPA buffer (YEASEN, Cat# 20101ES60) to extract the total protein. The concentrations of protein were quantified using the BCA Protein Assay Kit (YEASEN, Cat# 20201ES76). About 20 μg of the protein samples were subjected to electrophoresis on 10% SDS-PAGE gels and then transferred to PVDF membranes (YEASEN, Cat# 36126ES03). The membranes were blocked with BSA Blocking Buffer for 60 minutes at room temperature, and then stained with a rabbit polyclonal antibody against GPX4 (1:1000 dilution, Abcam, Cat# ab231174, RRID: AB_3073732), a rabbit monoclonal antibody against xCT (1:1000 dilution, Abcam, Cat# ab307601, RRID: AB_3094570), a rabbit polyclonal antibody against PD-L1 (1:1000 dilution, Thermo Fisher Scientific, Cat# PA5-20343, RRID: AB_11153819), a rabbit polyclonal antibody against NF-κB (1:1000 dilution, Abcam, Cat# ab16502, RRID: AB_443394), a rabbit monoclonal antibody against p-NF-κB (1:1000 dilution, Abcam, Cat# ab76302, RRID: AB_1524028), a rabbit polyclonal antibody against IkBα (1:5000 dilution, Abcam, Cat# ab97783, RRID: AB_10680152), a rabbit monoclonal antibody against p-IkBα (1:1000 dilution, Abcam, Cat# ab92700, RRID: AB_10562951) and a rabbit polyclonal antibody against GAPDH (1:1000 dilution, Abcam, Cat# ab9485, RRID: AB_307275) at 4°C overnight. Subsequently, the membranes were incubated with the secondary antibody Goat Anti-Rabbit IgG H&L (HRP; 1:10 000 dilution, Abcam, Cat# ab6721, RRID: AB_955447) for 60 minutes at room temperature. Bands were visualized using the BeyoECL Plus kit (Beyotime, Cat# P0018S), and the band intensity was quantified with Image-ProPlus software.
Statistical Analysis
Data were presented as the mean ± standard deviation and were processed using SPSS 20.0 software (IBM, Armonk, New York, USA, RRID:SCR_002865). To identify statistical disparities, we employed Student’s t-test for comparisons between 2 groups or one-way ANOVA for comparisons involving more than 2 groups, complemented by the post hoc Bonferroni test. A P-value of less than .05 was considered to indicate statistical significance.
Results
SA Inhibited the OSCC Proliferation
To survey the action of SA (Figure 1A) on the progression of OSCC, the toxic effect of SA was first examined, to screen the SA concentration. Only SA at the dose of 400 μM markedly lessened the HOK cell viability as compared with the control (0 μM SA) with a IC50 of 718.37 μM based on CCK-8 assays (Figures 1B and S1A). Thus, the other 6 concentrations of SA were further used to detect the toxic effect of SA on OSCC lines, including CAL27 and SCC7 cells. In contrast to the control group, the application of SA at concentrations of 25, 50, 100, and 200 μM notably decreased the viability of SCC7 cells, with an IC50 value of 77.41 μM. Similarly, SA at 50, 100, and 200 μM markedly reduced the viability of CAL27 cells, with an IC50 value of 94.38 μM (Figure 1C). Consequently, SA concentrations of 25, 50, and 100 μM were selected for further investigation. SA attenuated the proliferation of OSCC.

SA restrained the proliferation of OSCC. (A) The molecular structure of SA. (B) The cell viability was detected by CCK-8 assays after HOK cells were treated with 0, 6.25, 12.5, 25, 50, 100, 200 and 400 μM of SA for 48 hours. (C) SCC7 and CAL27 cells were treated with 0, 6.25, 12.5, 25, 50, 100, and 200 μM of SA for 48 hours. The cell viability was detected by CCK-8 assays.
SA Elicited Ferroptosis of OSCC
Ferroptosis is strongly involved in the progression of OSCC, and has been elucidated to be a candidate for the treatment of OSCC.7,26-28 Moreover, SA has been demonstrated to regulate the development of Alzheimer’s disease. 18 However, there are currently no relevant studies on whether SA regulates the processes of OSCC through ferroptosis. There was no statistical difference in the expression of GPX4 and xCT after the treatment of 100 μM SA (Figure S1B and C). However, results in Figure 2A revealed that the expressions of GPX4 and xCT were notably shrunk by all the 3 concentrations of SA in SCC7 cells, while by only 2 higher concentrations of SA in CAL27 cells, with a concentration-dependent fashion. However, the relative levels of ROS and MDA were prominently elevated in SCC7 cells treated with 25, 50, and 100 μM of SA, whereas in CAL27 cells treated with 50 and 100 μM of SA (Figure 2B and C). Besides, all 3 concentrations of SA observably slashed the SOD level, while markedly fostered the relative Fe2+ level in SCC7 cells (Figure 2D and E). Similarly, the prominent changes in levels of SOD and Fe2+ level were observed in CAL27 cells following the treatment of 50 and 100 μM of SA (Figure 2D and E). Therefore, SA evoked ferroptosis of OSCC.

SA evoked the ferroptosis of OSCC. (A) The relative protein expression of GPX4 and xCT was examined by western blot after SCC7 and CAL27 cells were treated with 25, 50 and 100 μM SA for 48 hours. Data were expressed after being normalized with GAPDH. (B) The relative lipid ROS level was examined after SCC7 and CAL27 cells were incubated with 25, 50 and 100 μM SA for 48 hours. Scale bar = 100 µm. (C) Examination of the MDA level in SCC7 and CAL27 cells incubated with 25, 50 and 100 μM SA for 48 hours. (D) Measurement of the SOD level in SCC7 and CAL27 cells incubated with 25, 50 and 100 μM SA for 48 hours. (E) The relative Fe2+ level was detected after SCC7 and CAL27 cells were treated with 25, 50 and 100 μM SA for 48 hours.
SA Suppressed the Immune Escape of OSCC
The PD-L1 expression was prominently abridged in SCC7 cells treated with all the 3 concentrations of SA, whereas in CAL27 cells with 50 and 100 μM of SA (Figure 3A). Besides, all the 3 concentrations of SA boosted the cell viability of CD8+ T cells after co-culture with SCC7 cells, and enhanced the contents of IFN-γ, IL-2, and TNF-α (Figure 3B and C). Just 50 and 100 µM of prominently fostered the cell viability of CD8+ T cells after co-culture with CAL27 cells, and increased the contents of IFN-γ, IL-2, and TNF-α (Figure 3B and C). However, no statistical difference was found in the concentration of IFN-γ after NOK cells treated with 100 μM of SA were co-cultured with CD8+ T cells (Fig S1D). Overall, SA repressed immune escape of OSCC.

SA reduced immune escape of OSCC. (A) The expression of PD-L1 was examined by western blot. Data were expressed after being normalized with GAPDH. (B) CD8+ T cell viability was detected by CCK-8 assays. (C) Measurement of IFN-γ, IL-2 and TNF-α by ELISA.
SA Restrained the NF-κB Pathway in OSCC
Mechanistically, our study revealed that treatment with SA at the 3 concentrations prominently declined the ratios of phosphorylated NF-κB to total NF-κB and phosphorylated IkBα to total IkBα in SCC7 cells (Figure 4A and B). Just 50 and 100 μM of SA significantly diminished the relative protein levels, whereas the 25 μM concentration of SA did not exert a statistically significant effect on the expression of these proteins in CAL27 cells compared to the control group with 0 μM of SA (Figure 4A and B). Moreover, the diminishment in the NF-κB levels due to the treatment of 100 μM SA were markedly rescued with the administration of RANKL (Figure 4C). However, there was no statistical difference in the expression levels of p-NF-κB/NF-κB and p-IkBα/IkBα after the treatment of 100 μM SA (Figure S1B and C). Together, SA dampened the level of NF-κB pathway in OSCC.

Effect of SA on the expression of NF-κB pathway in OSCC. (A and B) The relative protein levels of p-NF-κB, NF-κB, p-IkBα, and IkBα were detected by western blot after SCC7 and CAL27 cells were incubated with 25, 50 and 100 μM SA for 48 hours. Data were expressed after being normalized with GAPDH. *P < .05 and **P < .01 versus 0 μM SA. (C) SCC7 cells were incubated with 100 μM SA for 48 hours, or with 100 μM SA for 48 hours and 100 ng/ml RANKL for 60 minutes. The relative protein levels of NF-κB were detected by immunofluorescence.
SA Attenuated Proliferation and Immune Escape, But Enhanced Ferroptosis Through Repressing NF-κB Pathway in OSCC
Treatment of SA observably reduced the cell viability of SCC7 cells, which was markedly rescued with the RANKL management (Figure 5A). Besides, the decrease in the expression of GPX4 and xCT, and the enhancement in the relative level of ROS and Fe2+ in SCC7 cells following the treatment of SA, were significantly reversed with the administration of RANKL (Figure 5B-D). In addition, 100 µM of SA prominently elevated the cell viability of CD8+ T cells after co-culture with SCC7 cells with an increased release of IFN-γ, which were markedly counteracted with the incubation of RANKL (Figure 5E and F). Taken together, SA inhibited cell viability and immune evasion, and evoked ferroptosis by repressing NF-κB pathway in OSCC.

SA repressed cell viability and immune escape, but induced ferroptosis by inactivating NF-κB pathway in OSCC. (A) SCC7 cell viability was determined by CCK-8 assays. (B) The relative protein expression of GPX4, xCT and PD-L1 were detected by western blot. Data were expressed after being normalized with GAPDH. (C) The relative lipid ROS level was examined after SCC7 cells were tread with DCFH-DA. Scale bar = 100 µm. (D) Examination of the relative Fe2+ level. (E) CD8+ T cell viability was detected by CCK-8 assays. (F) Measurement of IFN-γ by ELISA.
SA Impeded the OSCC Growth in Mice
The administration of SA notably reduced both the tumor volume and mass, while it did not affect the body weight of the mice (Figure 6A-C). Treatment of SA prominently reduced the expression of GPX4 and PD-L1, whereas fostered the IFN-γ expression (Figure 6D and E). Additionally, tumor-bearing mice exhibited loosely arranged cells with an irregular pattern, which were overtly ameliorated with the application of SA (Figure 6E). Moreover, SA injection also decreased the level of NF-κB signaling in tumor tissues (Figure 6F). Thus, SA restrained OSCC cell growth involving in the NF-κB pathway in mice. Taken, together, SA curbed the proliferation and immune evasion of OSCC while enhancing ferroptosis by deactivating the NF-κB signaling pathway.

SA regulated OSCC growth, ferroptosis and immune escape involving in the NF-κB pathway in vivo. Mice were subcutaneously inoculated with a total of 2 × 105 of SCC7 cells into the right flank of nude mice, and then intraperitoneally received with 10 mg/kg SA and the same dose of PBS every day for 10 days. (A) Representative images of neoplasms from nude mice (Left). Monitor of tumor volume every 1 week for successive 4 weeks. Tumor volume was quantified by the following formula: volume = 0.5 × length × width2 (Right). (B) Supervision of mice body weight every 1 week for successive 4 weeks. (C) Measurement of tumor weight after 4 weeks. (D and E) Pathological examination by HE staining, and the expression levels of GPX4, PD-L1, and IFN-γ in tumor tissues were detected by immunohistochemistry. (F) The relative protein expression of p-NF-κB/NF-κB and p-IkBα/IkBα was examined by western blotting. Data were expressed after being normalized with GAPDH.
Discussion
In our research, we discovered that SA inhibited both the proliferation and immune evasion of OSCC and stimulated ferroptosis in both cellular and animal models. At the molecular level, SA reduced the activity of the NF-κB pathway in OSCC. The activation of the NF-κB pathway counteracted SA’s effects on cell proliferation, ferroptosis, and immune evasion. In animal studies, SA was found to impede the growth of OSCC. In summary, SA curtailed the proliferation and immune evasion of OSCC while boosting ferroptosis by inhibiting the NF-κB pathway.
Growth is a key feature of cancerous tumors. 29 Consequently, substances that counteract this growth could potentially serve as therapeutic agents for cancer treatment. Plentiful studies have identified SA as potential therapeutic drug by functioning on proliferation in various cancers. Xia et al 19 report that SA dampens non-small cell lung cancer cell viability in cells and their growth in mice. Similar results are also exhibited by Qiao et al 20 in prostate cancer. SA also has been revealed to suppresses proliferation of hepatocellular carcinoma cells. 21 In accordance with these results, this study found that SA diminished cell viability of OSCC in vitro, and reduced tumor size and weight in xenografted mice, suggesting that SA inhibited OSCC growth.
Extensive researches have reported that the process of ferroptosis is closely related to the advancement and growth of OSCC, in which targeting ferroptosis is demonstrated to be a potential treatment for OSCC.7,26-28 Moreover, ferroptosis-related genes are identified as prognostic factors in OSCC.27,30,31 Besides, natural active ingredients, such as resveratrol, 32 quercetin, 33 and baicalin, 34 that induce ferroptosis, have exhibited anti-tumor effect on OSCC. Similarly, SA increased relative Fe2+ level, the level of ROS and MDA, but diminished the SOD level as well as the relative protein expression of GPX4 and xCT in the 2 cells. Besides, SA decreased the GPX4 level in tumor mice. SA has been demonstrated to strongly bind with EZH2, a hub ferroptosis-related gene in diabetic kidney disease. 35 Besides, the modulatory function of SA on ferroptosis has been exhibited in Alzheimer’s disease. 18 Overall, these outcomes suggested SA evoked ferroptosis in OSCC.
Studies have concentrated on the change in treatment paradigm from conventional cytotoxic chemotherapy to immunotherapy, which has been brought about by the quick development of tumor immune therapy in various tumors, including OSCC.36,37 PD1 and PD-L1, checkpoints for tumor immune escape, are crucial for immunotherapy. 38 Blocking PD-1/PD-L1, which can enable cytotoxic T cells to carry out their fatal effects on a number of malignancies, including OSCC, is a crucial strategy for tumor immunotherapy.39,40 This study discovered that SA diminished the PD-L1 level. Besides, the development of pro-inflammatory factors in the tumor microenvironment can reduce PD-L1 levels, blocking immune escape. 41 In this study, SA boosted the cytotoxicity of CD8+ T cells against OSCC, and the levels of IFN-γ, IL-2, and TNF-α. In accordance with our findings, Kearney et al’s 42 group reveal that CD8+ T cells and NK cells emit pro-inflammatory chemicals such TNF-α, IL-2, and IFN-γ that impact tumor and immune cells. One important mechanism of immune escape is the tumor cells’ reduced vulnerability to TNF. 43 Moreover, SA has been revealed to suppress tumor immune evasion of non-small cell lung cancer, as evidenced by the increased CD8+ T cell viability and the concentrations of TNF-α, IL-2, and IFN-γ, and the decreased expression of PD-L1. 19 In summary, SA restrained the immune evasion capabilities of OSCC.
Mechanically, SA reduced the expression of proteins in the NF-κB pathway, a finding that was validated through the use of RANKL. After IkB protein is separated and broken down by IkB kinase (IKK), NF-κB is activated. Then, the NF-κB signaling pathway triggers the transcription and expression of inflammatory mediators. 44 Hence, NF-κB signaling is reported to be involved in immune escape of OSCC.40,45,46 Additionally, NF-κB pathway is complicated with the progression of OSCC.47,48 Moreover, SA inhibits the TRAF6/NF-κB signaling pathway, thereby reducing the aggressive characteristics and altering the tumor immune context of non-small cell lung cancer cells. 19 SA improves Alzheimer’s disease by modulating inflammation, ferroptosis and cognitive impairment via TRAF6/NF-κB pathway. 18 Gene expression profiling of SA’s impact on hepatocellular carcinoma cell metastasis showed that NF-κB signaling pathway is the dominate pathways that are strongly inhibited by SA. 21 In line with these findings, SA also deactivated the NF-κB pathway in OSCC. Consequently, RANKL counteracted the beneficial effects of SA on proliferation, ferroptosis, and immune evasion in OSCC. Collectively, these outcomes demonstrate that SA inhibits proliferation and immune evasion while promoting ferroptosis through the NF-κB pathway.
Conclusion
In conclusion, this study expounded that SA repressed growth and immune evasion, and evoked ferroptosis in OSCC through inhibiting NF-κB pathway. Nevertheless, future research should examine SA’s impact on additional aspects of tumor progression, including cell migration and metastasis. Moreover, it is essential to explore the direct influence of the NF-κB pathway in vivo settings. Besides, multiple doses of SA should be applied in animal experiments to consolidate the results. In summary, our findings have pinpointed a promising candidate for the therapeutic intervention of OSCC.
Supplemental Material
sj-tif-1-ict-10.1177_15347354251375464 – Supplemental material for Sennoside A Modulates the Ferroptosis and Immune Evasion of Oral Squamous Cell Carcinoma Cells Through Inhibiting the NF-κB Pathway
Supplemental material, sj-tif-1-ict-10.1177_15347354251375464 for Sennoside A Modulates the Ferroptosis and Immune Evasion of Oral Squamous Cell Carcinoma Cells Through Inhibiting the NF-κB Pathway by Jiaying Huo, Jianhua Qi and Qixuan Ren in Integrative Cancer Therapies
Footnotes
Acknowledgements
Not applicable.
Ethical Considerations
The research was authorized by the Board and Ethics Committee of Hebei Medical University.
Consent for Publication
Not applicable.
Author Contributions
JY H: Conceptualization, Methodology, Software, Data curation. JY H and JH Q: Writing-Original draft preparation, Visualization, Investigation, Supervision. JY H and QX R: Validation, Writing-Reviewing and Editing. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
