Abstract
Context:
Hepatocellular carcinoma (HCC) is a malignant tumor with high global incidence and mortality rates. Jianpi Huayu Formula (JPHY) has demonstrated therapeutic potential in HCC, but its underlying mechanisms remain unclear.
Objective:
This study aimed to investigate the anti-HCC effects of JPHY and its molecular mechanisms, focusing on its regulatory roles in vasculogenic mimicry (VM) formation and epithelial-mesenchymal transition (EMT) progression.
Methods:
Sk-Hep-1 cells were treated with 5%, 10%, or 20% JPHY-medicated serum (JPHY-MS). Cell proliferation, migration, VM, and EMT were assessed using CCK-8, wound healing, tube formation, and Western blot assays. Molecular docking was employed to analyze the binding between core components of JPHY and CXCR4. A stable CXCR4-overexpressing cell line was established, and the regulatory effect of CXCR4 on the Smad2/3 pathway was observed following intervention with the Smad2/3-specific inhibitor SB431542. Finally, a retrospective study was conducted using JPHY-MS intervention. In vivo experiments: Mice bearing HCCLM3 xenografts were divided into control, model, low-dose (6.24 g/kg), and high-dose (24.96 g/kg) JPHY groups. Tumor size and body weight were measured every 3 days. The effects of drug intervention on the CXCR4-Smad2/3 pathway, EMT, and VM were examined via immunohistochemistry (IHC), CD31/PAS double staining, and Western blot analysis.
Results:
JPHY may impede HCC progression and metastasis through CXCR4 downregulation-mediated suppression of Smad2/3 signaling pathway activation, concomitantly modulating VM formation and EMT processes.
Conclusion:
JPHY suppresses HCC progression by targeting the CXCR4-Smad2/3 pathway, highlighting its potential as a novel HCC therapeutic.
Keywords
Introduction
Primary Hepatocellular Carcinoma (HCC) is one of the most common and fatal malignant tumors worldwide, with a mortality rate ranking among the top 3 for all cancers. 1 The progression of this disease is closely associated with angiogenesis, which provides essential support for tumor growth and metastasis.2,3 Anti-angiogenesis therapy has thus become an important approach for treating HCC. However, prolonged use of anti-angiogenesis drugs often leads to drug resistance, a phenomenon closely related to Vasculogenic Mimicry (VM) in tumors. 4 VM, first proposed by Maniotis et al in 1999, refers to the formation of vessel-like structures by tumor cells through self-deformation and extracellular matrix remodeling, thereby providing an alternative blood supply to support tumor growth and metastasis. 5 Furthermore, Epithelial-Mesenchymal Transition (EMT) is another key factor promoting tumor cell migration, invasion, and drug resistance. 6 The interaction between VM and EMT presents new challenges for HCC treatment. Therefore, exploring the molecular mechanisms of VM and EMT, as well as developing therapeutic strategies targeting both, holds significant scientific and clinical value.
Jianpi Huayu Formula (JPHY), composed of Bai Shu (Atractylodes macrocephala), E Shu (Curcuma zedoaria), Fu Ling (Poria cocos), Fo Shou (Citrus medica), Ku Shen (Sophora flavescens) and She She Cao (Hedyotis diffusa), is derived from the experience of Professor Qian Baiwen. It has spleen-invigorating, qi-tonifying, and blood-activating effects. The formula contains various anti-cancer components 7 and demonstrates significant anti-tumor effects. Our previous research demonstrated that JPHY inhibits tumor angiogenesis by modulating the Jagged1/Notch signaling pathway and also suppresses the expression of transforming growth factor-β (TGF-β).8,9 Although JPHY has been demonstrated to inhibit angiogenesis, its role in vasculogenic mimicry remains unclear. Also, it is well known that the activation of TGF-β-Smad2/3 signaling pathway is a major driver of EMT,10,11 suggesting the potential of JPHY to inhibit EMT.
C-X-C Chemokine Receptor Type 4 (CXCR4) is a G protein-coupled receptor with 7 transmembrane domains, 12 highly expressed in various malignant tumors.13 -15 It promotes tumor proliferation, migration, and invasion and is associated with poor prognosis.16,17 Studies have shown that Cisatracurium targets CXCR4 and regulates the TGF-β-Smad2/3 signaling pathway to inhibit the migration of colon cancer cells. 18 The use of the CXCR4 inhibitor AMD3465 effectively suppresses the activation of the TGF-β-Smad2/3 pathway, thereby reducing cardiac fibrosis induced by this pathway. 19 Furthermore, CXCR4 plays a key role in the EMT process, and can promote liver metastasis in breast cancer by inducing the FUS/circEZH2/KLF5 feedback loop. 20 In lung adenocarcinoma, CXCR4 is upregulated by SDF-1 to promote EMT and generate poor prognosis. 21 In addition to EMT, CXCR4 is also closely associated with the formation of tumor VM. 22 However, whether CXCR4 mediates VM and EMT in HCC through Smad2/3, and whether JPHY inhibits VM and EMT in HCC by suppressing the CXCR4-Smad2/3 signaling pathway, remains to be further investigated. Therefore, we hypothesize that JPHY may exert therapeutic effects on HCC by inhibiting the CXCR4-Smad2/3 signaling pathway to suppress EMT and VM formation, and we conducted studies to investigate this hypothesis.
Method
Preparation of JPHY Herbal Extract
Step 1: Combine 960 mL purified water with 12 g each of Bai Shu (batch: 221102), E Shu (batch: 220601), and Fo Shou (batch: 220901) in a round-bottom flask. Soak for 30 minutes, then boil at 90°C to 100°C for 3 hours. Add 20 g each of Fu Ling (batch: 221130), Ku Shen (batch: 220801), and She She Cao (batch: 221001), soak for 30 minutes, and boil for 1.5 hours, collecting decoction and volatile oils.
Step 2: Add 960 mL purified water to the residue, boil for 1.5 hours, and collect decoction and oils. Repeat extraction once.
Step 3: Combine all decoctions, filter, concentrate, and add volatile oils to a final concentration of 2.4 g/mL. All herbs were sourced from Guangzhou Zhixin Traditional Chinese Medicine Decoction Pieces Company.
Preparation of JPHY-Medicated Serum
Twenty male SD rats (SPF grade, 190-220 g) were randomized into control and experimental groups (n = 10 each). The experimental group received 17.28 g/kg JPHY (rat equivalent dose) via oral gavage twice daily for 4 days, while the control group received equal volumes of saline. One hour post-final gavage, rats were anesthetized with 4% sodium pentobarbital (intraperitoneal), and blood was collected via abdominal aorta puncture. After 1 hour coagulation, samples were centrifuged at 3000 rpm (20 minutes, 4°C). Serum was heat-inactivated (56°C, 30 minutes), sterilized by filtration, aliquoted, and stored at −80°C.
Rats were procured from Guangzhou Ruige Biological Technology Co., Ltd. (License: SYXK [Yue] 2023-0259).
Construction of CXCR4 Overexpressing Cell Line
Sk-Hep-1 human hepatocellular carcinoma cells (Catalog No.: CL-0212, CVCL_0525; Ruiqian Biotechnology Co., Ltd.) at 20% to 30% confluency were transfected with lentiviral vectors containing green fluorescent label (GFP) and either a CXCR4 overexpression plasmid or an empty plasmid (Catalog No.: GXDL0397387; Shanghai Jikai Gene Technology Co., Ltd.) following the lentivirus protocol under serum-free conditions. After 16 hour, the medium was replaced with complete culture medium (Catalog No.: C11995500BT; Gibco) supplemented with 10% fetal bovine serum (FBS; Catalog No.: 164210-50; Procell). Cells were cultured for 24 to 48 hours until robust fluorescence was observed. Stable transfectants were selected using 5 μg/mL puromycin (Catalog No.: BS111-25mg; Biosharp) and expanded to establish stable cell lines.
Molecular Docking
Molecular docking of the 8 core components of JPHY 7 with the target protein CXCR4 was performed using AutoDock Vina 1.1.2. Protein structures were preprocessed using PyMol 2.4, and compound structures were energy-minimized using ChemDraw 20.0. Docking simulations were prepared as PDBQT files using AutoDock Tools 1.5.6. Binding interactions between ligands and the receptor were visualized and analyzed using PyMol 2.4 and Discovery Studio 2019 to evaluate complex stability and interaction patterns. Compound structures were retrieved from PubChem (http://pubchem.ncbi.nlm.nih.gov), and protein models were obtained from the Protein Data Bank (PDB; https://www.rcsb.org).
Animal Experiments
Animal Modeling, Drug Administration, and Sample Collection
Twenty-four male BALB/C-nu mice (4 weeks old, 18-22 g) were housed in an SPF-grade facility with a 12 hour light-dark cycle and provided ad libitum access to food and water. After a 3-day acclimatization period, 6 mice were randomly assigned as the blank control group (CON), while the remaining 18 mice were subcutaneously injected with 200 μL of HCCLM3 cell suspension (1 × 107 cells; Catalog No.: CTCC-400-0193, CVCL_6832; Meisen Cell Technology Co., Ltd.) into the dorsal hind limb. When tumor volume reached ~5 mm in diameter, mice were randomized into 4 groups (n = 6): CON, model (MOD), low-dose JPHY (JPHY-L, 6.24 g/kg), and high-dose JPHY (JPHY-H, 24.96 g/kg). MOD and CON received saline, while JPHY-L and JPHY-H were administered JPHY extract daily by gavage. The dosage of mice was based on previous studies. 23
Tumor volume (calculated as length × short diameter²/2) and body weight were measured every 3 days. When tumor volume approached 1500 mm³, mice were euthanized via cervical dislocation under 2% sodium pentobarbital anesthesia. Tumors were excised, photographed for documentation, and divided for processing: one portion was fixed in 4% paraformaldehyde solution (Catalog No.: BL539A; Biosharp), while the remaining tissue was stored at −80°C for subsequent analysis.
Mice were procured from the Guangdong Medical Laboratory Animal Center (Production License: SCXK [Yue] 2023-0035; Use License: SYXK [Yue] 2023-0342).
Immunohistochemical Staining and PAS Staining
Tumor tissues were paraffin-embedded, sectioned, and processed for deparaffinization, rehydration, and antigen retrieval. Sections were blocked with 3% BSA (Catalog No.: FB29904-100; FEIMOBIO LIFE SCIENCES) at room temperature (RT) for 30 minutes to prevent non-specific binding, followed by incubation with primary antibodies at 4°C overnight. After washing, sections were incubated with secondary antibodies at RT for 60 minutes, and color development was performed using DAB chromogen (Catalog No.: ZLI-9034; ZSGB-BIO). For PAS staining (Catalog No.: C0142S; BEYOTIME), sections were treated with periodic acid solution (Catalog No.: PH1143; Phygene) in the dark for 10 minutes, washed, and incubated with Schiff’s reagent (Catalog No.: G1286; Solarbio) at 37°C for 30 minutes (light-protected). Then Sections were counterstained with hematoxylin (Catalog No.: C0107; BEYOTIME), differentiated, and blued. Finally, sections were dehydrated, mounted, air-dried, and imaged using a Nikon 80i microscope (Nikon, Japan). Antibody details are provided in Supplemental Table S1.
Cell Viability Assay
Sk-Hep-1 cells were seeded in a 96-well plate at 5000 cells/well and treated with 200 µL of complete medium containing 5%, 10%, or 20% JPHY-medicated serum (JPHY-MS) for 24, 48, or 72 hours. At each time point, 10 µL of CCK-8 solution (Catalog No.: GK10001-5; GLPBIO) was added to each well and incubated for 60 minutes. Absorbance at 450 nm was measured to determine cell viability.
Quantitative Real-Time PCR (qRT-PCR)
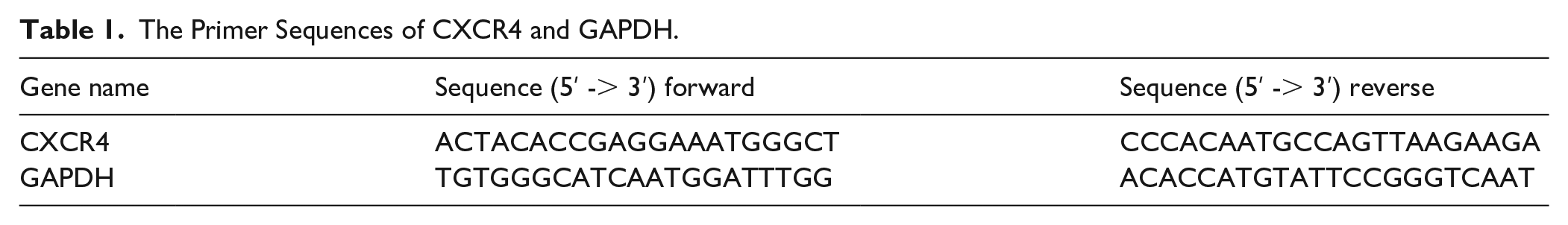
Total RNA was extracted using an RNA extraction kit (Catalog No.: RC112-01; Vazyme) following the manufacturer’s instructions. cDNA was synthesized from RNA using Evo M-MLV RT Premix (Catalog No.: AG11706; AG). qRT-PCR was performed using 2X SYBR Green Master Mix (Catalog No.: AG11702; AG) with gene-specific primers. GAPDH served as the internal control for normalization. Primer sequences are listed in Table 1.
The Primer Sequences of CXCR4 and GAPDH.
Transwell Assay
Sk-Hep-1 cells (4 × 105 cells/mL) were seeded in the upper chamber of a Transwell insert (200 µL/well), while 600 µL of complete medium supplemented with 10% FBS was added to the lower chamber. Cells were incubated at 37°C with 5% CO₂ for 24 hours. After incubation, cells were fixed with 4% paraformaldehyde for 30 minutes and stained with 0.1% crystal violet (Catalog No.: R27575; Biosharp) for 20 minutes. Non-migrated cells on the upper membrane surface were gently removed with a cotton swab. Inserts were air-dried, and migrated cells were imaged under a microscope.
Vasculogenic Mimicry Formation Assay
Matrigel (Catalog No.: 082724; CORNING) was thawed overnight at 4°C and maintained on ice during use. Using precooled pipette tips, 10 µL of Matrigel was added to each well of angiogenesis slides (Catalog No.: 81506; Ibidi). Slides were incubated at 37°C with 5% CO₂ for 1 hour to allow gel polymerization. Sk-Hep-1 cells (3.5 × 104 cells/well) were seeded onto the Matrigel and treated with designated compounds for 4 hours. Tube formation was observed and imaged using an inverted microscope.
Western Blotting
Cells were lysed in RIPA buffer (Catalog No.: P0013C; Beyotime) supplemented with protease inhibitors (Catalog No.: HY-K0010; MCE) and phosphatase inhibitors (Catalog No.: HY-K0021, HY-K0022; MCE). For the prepared small pieces of tumor tissue, add the aforementioned lysis buffer and homogenize using a pre-chilled ultrasonic homogenizer. Lysates (including those from tumor tissues and cells) were centrifuged at 4°C for 10 minutes, and protein concentrations were quantified using a BCA assay kit (Catalog No.: GK10009; Glpbio) with absorbance measured at 562 nm. Equal protein amounts were denatured at 100°C for 10 minutes. Proteins were separated by SDS-PAGE (120 V) using a 180 kDa marker (Catalog No.: MP102; Vazyme) and transferred to PVDF membranes (Catalog No.: IPVH00010; Merck Millipore) at 250 mA for 90 minutes. Membranes were blocked and incubated with primary antibodies overnight at 4°C with gentle shaking, followed by incubation with HRP-conjugated secondary antibodies. Chemiluminescent signals were detected using a Bio-Rad ChemiDoc imaging system (Bio-Rad, USA), and band intensities were quantified using ImageJ software. Antibody details are provided in Supplemental Table S1.
Immunofluorescence Staining
Cells treated in confocal dishes (No. BS-15-GJM, Bioshap) were washed and fixed with 4% paraformaldehyde, followed by additional washing. Each dish was permeabilized with 500 μL of 0.5% Triton-X-100 (No. R27575, Yuanye) at room temperature for 10 minutes and then washed again. Blocking was performed with 500 μL of 5% BSA for 1 hour. Primary antibodies were incubated at 4°C overnight. After washing, cells were incubated with secondary antibodies in the dark for 1 hour. Finally, DAPI (No. F6057-20ML, Sigma-Aldrich) was added for fluorescence staining for 30 minutes. Images were captured using a confocal microscope (AX NIS Elements, Nikon, Japan). Details of antibodies used are listed in Supplemental Table S1.
Wound-Healing Assay
7 × 106 cells were seeded in a 6-well plate. At confluence, a straight scratch was created in the monolayer using a sterile 200 µL pipette tip. Detached cells were gently washed away with PBS (Catalog No.: C10010500BT; Gibco). Cells were treated with the designated compounds for 24 h, and migration was monitored at 0 and 24 hours using a Nikon TI-S microscope (Nikon, Japan).
Statistical Analysis
Statistical analysis and graphing were performed using GraphPad Prism 9.0 software. The Shapiro-Wilk test was employed to assess normality distribution, while the Brown-Forsythe test was used to evaluate homogeneity of variance. Normally distributed data were expressed as mean ± standard deviation (Mean ± SD), whereas non-normally distributed data were presented as median (interquartile range, M [P25, P75]). For normally distributed data with homogeneous variance, one-way ANOVA was conducted for multiple group comparisons, followed by Dunnett’s post hoc test when statistical significance was observed. Non-parametric tests were applied when data violated either normality or homogeneity of variance assumptions. A P-value < .05 was considered statistically significant.
Results
JPHY Inhibits EMT and VM In Vitro
In the CCK-8 assay, Sk-hep-1 cells were exposed to varying concentrations of JPHY-MS for 24, 48, and 72 hours. The findings revealed that 5% JPHY-MS (JPHY-Medicated Serum) did not significantly impact cell viability at any time point (P > .05). Conversely, 10% and 20% JPHY-MS markedly reduced cell viability (P < .05; Figure 1A). In wound-healing and vasculogenic mimicry (VM) formation assays, 5% JPHY-MS exhibited no inhibitory effect on Sk-hep-1 cell migration or tube formation (P > .05). In contrast, 10% and 20% JPHY-MS significantly suppressed cell migration and tube formation, with the 20% JPHY-MS group demonstrating more pronounced inhibitory effects (P < .05; Figure 1B-F). Western blot analysis indicated that, compared to the control(Ctrl) group, the protein expression levels of VE-cadherin and Vimentin were significantly reduced in the 10% and 20% JPHY-MS groups (P < .05). Moreover, E-cadherin protein expression was significantly upregulated in the 5%, 10%, and 20% JPHY-MS groups (P < .05; Figure 1G-J). These results collectively demonstrate that JPHY effectively inhibits hepatocellular carcinoma cell proliferation, migration, epithelial-mesenchymal transition (EMT), and VM formation, with higher concentrations exerting more potent effects.

In vitro, JPHY-MS inhibits cell proliferation, migration, and VM formation while regulating EMT and VM. Ctrl: Control group; 5%, 10%, 20% JPHY-MS: different concentrations of JPHY-MS (JPHY-Medicated Serum). (A) CCK8 assay. (B-C) Wound-healing assay, with migration area measured. Magnification: 100×. (D-F) VM formation assay, with branch and node points quantified. Magnification: 100×. (G-J) Western blotting, showing the expression of EMT and VM-related proteins (E-cadherin, Vimentin, VE-cadherin) under different concentrations. Results are expressed as mean ± SD, with experiments repeated three times (n = 3).
Correlation Between the JPHY and CXCR4
Using molecular docking techniques, we systematically analyzed the binding interactions between the 8 core components of JPHY and CXCR4. All 8 components demonstrated substantial binding affinity with CXCR4, as evidenced by binding energies below −5 kcal/mol, where more negative values indicate stronger binding interactions. Notably, Hesperidin and Chlorogenic acid emerged as the most potent binders, exhibiting the lowest binding energies of −9.2 and −8.8 kcal/mol, respectively (Figure 2A and B). These findings suggest that these 2 components may play crucial roles in mediating the pharmacological effects of JPHY through CXCR4 interaction.

Correlation between JPHY and CXCR4. (A) Heatmap of docking binding energies. (B) Molecular docking of the eight core components with the key target CXCR4.
CXCR4 Modulates Smad2/3 Signaling to Promote EMT and VM
Previous studies have identified an association between JPHY and CXCR4, yet the underlying molecular mechanisms involving CXCR4 remain unclear. To address this knowledge gap, we established a stable CXCR4-overexpressing SK-Hep-1 cell line via lentiviral transduction to specifically investigate this aspect of the mechanism. Transfection efficiency was rigorously validated using both Western blot analysis and quantitative real-time PCR (qPCR). The results showed no significant differences in CXCR4 expression between the control (Ctrl) and negative control (NC) groups (P > .05). In contrast, the overexpression (OE) group exhibited significantly higher CXCR4 expression levels compared to both control groups (P < .01; Figure 3A-C). The successful establishment of CXCR4-overexpressing SK-Hep-1 cells provides a reliable model system for subsequent mechanistic investigations.

CXCR4 regulates the Smad2/3 signaling pathway to promote EMT and VM. Ctrl: Control group; NC: Model group; OE: CXCR4 overexpression group; NC+Sb: SB431542 inhibitor group; OE+Sb: CXCR4 overexpression with SB431542 inhibitor group. (A-C) Construction of CXCR4-overexpressing stable cell lines and verification via Western blotting and qPCR. (D-H) Western blotting analysis of VM, EMT, and Smad2/3 signaling protein expression.Results are expressed as mean ± SD from three independent experiments (n = 3).
To investigate whether CXCR4 mediates EMT and VM through the Smad2/3 signaling pathway, we employed SB431542, a specific inhibitor of the Smad2/3 pathway, for functional intervention studies. Western blot analysis revealed distinct molecular patterns across experimental groups. Compared to the NC group, SB431542 treatment (NC + Sb) significantly reduced the expression of VE-cadherin, p-Smad2/3, and Vimentin (P < .05, P < .05, P < .05), while increasing E-cadherin expression (P < .01). The OE group exhibited the opposite pattern, with upregulation of VE-cadherin, p-Smad2/3, and Vimentin, and downregulation of E-cadherin (P < .001, P < .01, P < .05, P < .05). Importantly, SB431542 treatment in the OE + Sb group reversed these changes, significantly decreasing mesenchymal markers and increasing epithelial marker expression. Notably, total Smad2/3 protein levels remained constant across all groups, confirming the specificity of SB431542’s action on Smad2/3 phosphorylation (Figure 3D-H). These findings provide compelling evidence that CXCR4, functioning upstream of Smad2/3, promotes EMT and VM by activating the Smad2/3 signaling cascade. The ability of SB431542 to antagonize CXCR4-mediated effects further underscores the pivotal role of Smad2/3 signaling in these processes.
JPHY Inhibits EMT and VM via the CXCR4-Smad2/3 Signaling Pathway
To further elucidate the molecular mechanism underlying JPHY’s action, we investigated its effects on the CXCR4 and Smad2/3 signaling pathways in Sk-hep-1 cells. Cells were treated with varying concentrations of JPHY-MS, followed by evaluation of CXCR4 expression and Smad2/3 signaling activity. Western blot analysis revealed that compared to the control group, both 10% and 20% JPHY-MS treatments significantly downregulated CXCR4 protein expression and reduced Smad2/3 phosphorylation levels (Figure 4A-C). These results demonstrate that JPHY exerts its pharmacological effects, at least in part, through inhibition of the CXCR4-mediated Smad2/3 signaling pathway.

JPHY regulates the CXCR4-Smad2/3 signaling pathway to inhibit EMT and VM formation. Ctrl: Control group; NC: Model group; OE: CXCR4 overexpression group; OE+JPHY-MS: CXCR4 overexpression treated with 20% JPHY-MS. (A-C) Protein changes in CXCR4, Smad2/3, and p-Smad2/3 under different JPHY-MS concentrations. (D-E) Transwell assay and quantificationof migrating cells. (F-H) VM formation in different groups, with branchand node points quantified. (I) Immunofluorescence observation of VE-cadherin expression changes. (J-O) Western blotting analysis of JPHY’s effects on CXCR4-Smad2/3 signaling and EMT/VM-related protein expression in CXCR4-overexpressing cells. Results are expressed as mean ± SD from three independent experiments (n = 3).
Subsequently, we treated the CXCR4-overexpressing cells with 20% JPHY-MS to further investigate its regulatory effects. In Transwell migration and vasculogenic mimicry (VM) formation assays, the OE group cells demonstrated significantly enhanced migratory capacity and tube formation compared to both Ctrl and NC groups. However, treatment with 20% JPHY-MS effectively suppressed these oncogenic phenotypes, significantly reducing both migration and VM formation capabilities (Figure 4D-H). To further investigate the underlying mechanisms, we assessed VE-cadherin expression patterns using immunofluorescence staining. The OE group exhibited significantly higher VE-cadherin fluorescence intensity compared to control groups, consistent with its enhanced metastatic potential. Notably, 20% JPHY-MS treatment substantially reduced VE-cadherin expression in CXCR4-overexpressing cells (Figure 4I). These findings collectively demonstrate that JPHY exerts its anti-metastatic effects by modulating CXCR4-mediated pathways, thereby inhibiting hepatocellular carcinoma cell migration and VM formation.
To further elucidate the molecular mechanisms underlying JPHY’s effects, we conducted comprehensive Western blot analysis of key signaling molecules. No significant differences were observed in the expression levels of VE-cadherin, p-Smad2/3, Vimentin, and E-cadherin between the Ctrl and NC groups (P > .05). Notably, while total Smad2/3 protein levels remained constant, the OE group significantly enhanced Smad2/3 phosphorylation compared to the Ctrl groups (P < .05). This activation was effectively reversed by JPHY-MS treatment, which significantly reduced Smad2/3 phosphorylation levels (P < .01). Furthermore, the OE group exhibited significant upregulation of mesenchymal markers VE-cadherin and Vimentin (P < .05, P < .01), accompanied by downregulation of the epithelial marker E-cadherin (P < .05). Importantly, JPHY-MS treatment reversed these expression patterns in the OE+JPHY-MS group (Figure 4J-O). These findings collectively demonstrate that JPHY exerts its anti-tumor effects by modulating the CXCR4-Smad2/3 signaling axis, thereby suppressing EMT and VM formation in HCC.
In Xenograft Models, JPHY Suppresses EMT and VM by Modulating the CXCR4-Smad2/3 Pathway
Finally, we evaluated the antitumor efficacy of JPHY using an HCCLM3 hepatocellular carcinoma xenograft model, tumor volumes in both the JPHY-L (low-dose) and JPHY-H (high-dose) groups were significantly reduced compared to the model (MOD) group. On the final day of observation, the body weights of mice in the MOD and JPHY-L groups were significantly lower than those in the control (CON) group. Notably, no significant weight difference was observed between the JPHY-H and CON groups. Furthermore, the body weights in both JPHY treatment groups were significantly higher than those in the MOD group (Figure 5A-C). These findings demonstrate that JPHY effectively inhibits HCC growth while improving the physical condition of mice in a dose-dependent manner. Immunohistochemical analysis of tumor sections revealed distinct patterns of protein expression. In the MOD group, Vimentin protein exhibited intense reddish-brown staining predominantly localized around cell nuclei, with staining intensity markedly stronger than that observed in both JPHY treatment groups. Conversely, E-cadherin expression showed dark brown staining in the JPHY-H group, with significantly higher intensity compared to both the MOD and JPHY-L groups (Figure 5D and E). These observations suggest that JPHY modulates the expression of EMT-related proteins in tumor tissues. CD31/PAS dual staining provided further insights into vascular patterns. The MOD group displayed normal brown-stained CD31-positive blood vessels accompanied by abundant irregular PAS-positive/CD31-negative vasculogenic mimicry (VM) structures, some of which contained adherent red blood cells (as indicated by arrows). The JPHY-L group showed scattered VM structures, while the JPHY-H group exhibited significantly reduced VM formation with observable CD31-stained blood vessels (Figure 5F). Western blot analysis revealed that, compared to the MOD group, JPHY treatment significantly downregulated protein levels of VE-cadherin, Vimentin, P-Smad2/3, and CXCR4 while upregulating E-cadherin expression. Notably, total Smad2/3 protein levels remained unchanged throughout the intervention, suggesting that JPHY may partially exert its therapeutic effects by suppressing EMT and VM through the CXCR4-Smad2/3 pathway (Figure 5G-L). Collectively, JPHY demonstrated remarkable anti-tumor efficacy in the subcutaneous xenograft model, with the high-dose treatment (JPHY-H) exhibiting superior tumor growth suppression and physiological improvement, highlighting its potential as a therapeutic agent for HCC.

In vivo, JPHY inhibits tumor growth and regulates EMT and VM by CXCR4-Smad2/3 pathway. CON: Control group; MOD: Model group; JPHY-L: JPHY low-dose group; JPHY-H: JPHY high-dose group. (A) Tumor images from mice (n = 6). (B) Tumor volume measurements in mice. (C) Body weight changes in mice. (D-E)Immunohistochemical staining to detect changes in EMT markers (n = 3). (F) CD31 and PAS dual staining to observe VM formation (n = 3) . Magnification: 20×/40×. (G-L)Western blot analysis was performed to examine the protein expression changes in CXCR4, Smad2/3 signaling pathway components, and EMT/VM markers. All values are expressed as mean ± SD. Experiments were repeated three times (n = 3).
Discussion
Hepatocellular carcinoma (HCC), one of the leading causes of cancer-related mortality worldwide, faces significant therapeutic challenges due to chemotherapy resistance and the complexity of the tumor microenvironment, prompting increased interest in complementary and alternative therapeutic strategies. 24 In this context, Traditional Chinese Medicine (TCM) emerges as a highly promising complementary strategy, owing to its multi-target regulatory properties. Studies have confirmed that TCM exerts anti-tumor effects through multiple mechanisms, including inhibition of tumor cell proliferation and migration, suppression of angiogenesis, enhancement of immune function, and modulation of the tumor microenvironment. 25 These findings underscore the potential of TCM resource development as a pivotal direction in future HCC research. Our previous studies have shown that the Jianpi Huayu Formula (JPHY) exerts anti-cancer effects by inhibiting HCC cell proliferation and inducing apoptosis, 26 alleviating immune suppression to promote HCC dormancy, 9 preventing postoperative recurrence, and improving patients’ postoperative quality of life.27,28 However, the exact mechanism of the JPHY’s anti-HCC effects remains unclear. Preliminary studies have revealed that JPHY can modulate the expression of transforming growth factor-beta (TGF-β), a critical regulator of epithelial-mesenchymal transition (EMT) through the TGF-β-Smad2/3 signaling pathway.10,11 This suggests that JPHY exerts its anti-tumor effects, at least partially, through EMT inhibition and potentially involves an association with the Smad2/3 signaling pathway. Furthermore, our previous research has demonstrated JPHY’s ability to inhibit tumor angiogenesis, indicating its potential role in suppressing vasculogenic mimicry (VM) formation 8 Based on these observations, we hypothesize that JPHY may mediate its anti-HCC effects through dual modulation of EMT and VM processes. Further investigation of these mechanisms may provide novel insights into the therapeutic potential of JPHY in HCC management.
In this study, intervention with graded concentrations of JPHY-MS on Sk-hep-1 cells effectively suppressed tumor proliferation and migration, while concurrently modulating the expression of EMT- and VM-associated proteins. These in vitro findings demonstrate the tumor-suppressive effects of the Jianpi Huayu Formula and suggest its potential involvement in regulating EMT and VM processes. To elucidate the underlying mechanism, our previous study demonstrated that JPHY can downregulate TGF-β expression against HCC, suggesting its potential association with the TGF-β-Smad2/3 pathway. Meanwhile, the chemokine receptor CXCR4 was identified as a molecule closely related to the TGF-β-Smad2/3 pathway. Based on these observations, we hypothesize that there may be a potential link between JPHY and CXCR4.
CXCR4, a chemokine receptor, plays a pivotal regulatory role in the progression of various cancers. Previous studies have demonstrated that CXCR4 interacts with its ligands to activate downstream signaling pathways, including the PI3K/AKT/mTOR and JAK/STAT signaling pathways, which promote tumor cell migration, invasion, and EMT.29 -32 Additionally, CXCR4 is closely associated with tumor angiogenesis, immune evasion, and chemotherapy resistance. Its high expression often promotes tumor progression, significantly reduces patient survival rates, and leads to poor prognosis. 17 To explore whether the anti-tumor effects of JPHY are mediated through CXCR4, we performed molecular docking analysis between CXCR4 and the 8 core components of JPHY. The results demonstrated that all 8 core components exhibited effective docking with CXCR4, with hesperidin and chlorogenic acid displaying particularly strong binding energies of −9.2 and −8.8 kcal/mol, respectively. These findings suggest a significant binding affinity between JPHY and CXCR4, providing preliminary evidence for a potential mechanistic link between JPHY and CXCR4 in its anti-tumor activity.
CXCR4 not only directly regulates tumor cell behavior but also exhibits synergistic interactions with other signaling pathways. For instance, Chen et al revealed a bidirectional regulatory relationship between CXCR4 and the TGF-β/Smad2/3 signaling pathway, which plays a dual role in promoting tumor angiogenesis and conferring chemotherapy resistance. 33 In HCC studies, the TGF-β-TGF-βR1-Smad2/3 and CXCL12-CXCR4 signaling pathways have been shown to synergistically drive tumor progression by enhancing EMT and VM. 34 Nevertheless, the precise mechanistic relationship between the CXCR4-Smad2/3 signaling axis and EMT/VM processes remains poorly understood, underscoring a critical gap in current knowledge that warrants further investigation. To investigate this mechanism, we established CXCR4-overexpressing Sk-hep-1 cells and employed SB431542, a specific Smad2/3 pathway inhibitor, to examine whether CXCR4 promotes EMT and VM through Smad2/3 activation. Notably, while total Smad2/3 protein levels remained unchanged, CXCR4 overexpression significantly upregulated phosphorylated Smad2/3 (p-Smad2/3) and enhanced expression of EMT/VM-associated proteins. These effects were markedly attenuated upon SB431542 treatment. These findings demonstrate that CXCR4 likely regulates EMT and VM processes through the Smad2/3 signaling pathway.
Previously, we proposed that JPHY might be associated with the TGF-β-Smad2/3 signaling pathway. Building on the established relationships between CXCR4, JPHY, and the TGF-β-Smad2/3 signaling pathway, we further hypothesized that JPHY exerts its anti-tumor effects by modulating the CXCR4-Smad2/3 signaling axis. To investigate whether JPHY inhibits EMT and VM through this pathway, Sk-hep-1 cells were treated with 3 different concentrations of JPHY-MS. Western blot analysis revealed that the expression levels of CXCR4 and p-Smad2/3 decreased in a concentration-dependent manner with increasing JPHY-MS concentrations. Notably, 20% JPHY-MS demonstrated the most pronounced inhibitory effect and was therefore selected for subsequent mechanistic studies. These findings suggest that JPHY is functionally linked to the CXCR4 and Smad2/3 signaling pathways, providing a potential mechanistic basis for its anti-tumor activity.
To further validate these findings, we treated Sk-hep-1 cells, a human HCC cell line with high CXCR4 expression, with 20% JPHY-MS. We observed that while high CXCR4 expression significantly enhanced cell migration and VM formation, these effects were markedly suppressed following JPHY treatment. This suggests a strong association between the anti-tumor effects of JPHY and CXCR4. Further Western blot analysis demonstrated that the expression of p-Smad2/3 increased with elevated CXCR4 levels but was significantly reduced after treatment with 20% JPHY-MS. These results indicate that CXCR4, as a key upstream effector molecule of Smad2/3, potentially regulates the Smad2/3 signaling pathway. Moreover, our findings suggest that JPHY exerts its anti-tumor effects, at least in part, by inhibiting the CXCR4-Smad2/3 signaling pathway.
In our HCC xenograft models, JPHY administration significantly suppressed tumor growth and increased body weight in HCCLM3 tumor-bearing mice. Immunohistochemical and Western blot analyses further confirmed its inhibitory effects on both EMT and VM processes, while demonstrating concurrent modulation of the CXCR4-Smad2/3 pathway. These results are consistent with previous findings and reinforce both the pharmacological efficacy and mechanistic foundation of JPHY. Importantly, our study proposes innovative therapeutic strategies targeting EMT and VM in cancer treatment, thereby establishing a novel theoretical framework for clinical translation.
In conclusion, this study demonstrates that JPHY inhibits EMT and VM in HCC by targeting the CXCR4-Smad2/3 signaling pathway, offering a novel therapeutic strategy for HCC treatment (Figure 6). Additionally, Smad2/3 was identified as a key mediator of CXCR4-driven EMT and VM. However, this study still has several limitations. Since anti-angiogenic drugs such as sunitinib and sorafenib in HCC have shown that drug resistance is closely associated with VM formation,4,35 and no definitive anti-VM drugs currently exist, our research lacks a positive control drug. In future studies, we will investigate the combined application of JPHY with anti-angiogenic agents to potentially improve the efficacy of HCC treatment. Additionally, CXCR4 as an upstream regulator of Smad2/3, CXCR4 may participate in EMT and VM through modulation of the Smad2/3 pathway. However, it remains unclear whether CXCR4 interacts directly with Smad2/3 signaling or functions indirectly via TGF-β-dependent mechanisms as suggested by existing studies. Whether this interaction depends on intermediate molecules such as TGF-β (eg, TGF-β receptor involvement) is still unknown. In subsequent investigations, we will employ TGF-β receptor inhibitors or immunofluorescence co-localization experiments to elucidate the precise mechanistic relationship between CXCR4 and Smad2/3 signaling. Furthermore, although molecular docking results suggest potential interactions between the main active components of JPHY and CXCR4, several limitations should be noted. Most importantly, the computationally predicted binding affinities require experimental validation through techniques such as surface plasmon resonance (SPR) or isothermal titration calorimetry (ITC). In our subsequent research, we plan to systematically validate these key predicted interactions through rigorous experimental approaches. Then, we only employed a single cell line and one animal model system the mechanistic investigations were primarily conducted at the cellular level, and we lacked genetically engineered mouse models to validate these mechanisms in vivo. Future studies should incorporate multiple HCC cell lines, patient-derived xenografts, and conditional knockout mouse models to strengthen our findings. More rigorous experimental designs will be implemented to address these limitations in our subsequent research.

Mechanism Diagram. JPHY inhibits EMT and VM formation by regulating the CXCR4-Smad2/3 signaling pathway.
Conclusion
Our study demonstrates that JPHY inhibits the proliferation, migration, and tube formation of Sk-Hep-1 cells, suppresses tumor growth in HCCLM3 mouse models, and improves mouse body weight in a concentration-dependent manner. Mechanistically, JPHY inhibits EMT and VM by targeting the CXCR4-Smad2/3 signaling pathway, with Smad2/3 playing a key role in CXCR4-driven EMT and VM. These findings highlight the potential of JPHY as a therapeutic agent for HCC and provide new insights into integrating traditional Chinese medicine with Western medicine for future cancer treatment strategies.
Supplemental Material
sj-docx-1-ict-10.1177_15347354251366580 – Supplemental material for Anti-Hepatocellular Carcinoma Effect of Jianpi Huayu Formula: Revealing the Molecular Mechanisms of the CXCR4 - Smad2/3 Pathway in Regulating Vasculogenic Mimicry Formation and Epithelial-Mesenchymal Transition
Supplemental material, sj-docx-1-ict-10.1177_15347354251366580 for Anti-Hepatocellular Carcinoma Effect of Jianpi Huayu Formula: Revealing the Molecular Mechanisms of the CXCR4 - Smad2/3 Pathway in Regulating Vasculogenic Mimicry Formation and Epithelial-Mesenchymal Transition by Enling Xu, Juze Lin, Qiuhong Yong, Xinhao Lu, Qiang Zhang, Xiaojuan Huang, Chaoyuan Huang and Changjun Wang in Integrative Cancer Therapies
Supplemental Material
sj-xls-1-ict-10.1177_15347354251366580 – Supplemental material for Anti-Hepatocellular Carcinoma Effect of Jianpi Huayu Formula: Revealing the Molecular Mechanisms of the CXCR4 - Smad2/3 Pathway in Regulating Vasculogenic Mimicry Formation and Epithelial-Mesenchymal Transition
Supplemental material, sj-xls-1-ict-10.1177_15347354251366580 for Anti-Hepatocellular Carcinoma Effect of Jianpi Huayu Formula: Revealing the Molecular Mechanisms of the CXCR4 - Smad2/3 Pathway in Regulating Vasculogenic Mimicry Formation and Epithelial-Mesenchymal Transition by Enling Xu, Juze Lin, Qiuhong Yong, Xinhao Lu, Qiang Zhang, Xiaojuan Huang, Chaoyuan Huang and Changjun Wang in Integrative Cancer Therapies
Footnotes
Acknowledgements
We sincerely thank the Guangdong Provincial People’s Hospital for providing the public laboratory platform.
Ethical Considerations
The study was approved by the Ruige Biological Laboratory Animal Ethics Committee (Approval No.: 20231112-001; Date: November 12, 2023) and the Animal Ethics Committee of Guangzhou University of Chinese Medicine (Approval No.: 20240307011; Date: March 7, 2024).
Author Contributions
Enling Xu: Writing—Original draft, Validation, Visualization, Data curation, Formal analysis, Investigation, Methodology, Project administration and Conceptualization. Juze Lin: Writing—Review and editing, Original draft, Investigation, Conceptualization, Funding acquisition. Qiuhong Yong: Review and editing, Investigation. Qiang Zhang: Investigation. Xiaojuan Huang: Investigation. Xinhao Lu: Investigation. Chaoyuan Huang: Review and editing, Conceptualization. Changjun Wang: Review and editing, Funding acquisition, Supervision.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by key project of Science and Technology Planning Project of Jiangxi Provincial Administration of Traditional Chinese Medicine: To explore the mechanism of Jianpi Huayu Recipe in inhibiting the formation of vasculogenic mimicry in liver cancer cells based on the CXCR4-Smad2/3 signaling pathway (No.2022Z026) and Ganzhou Science and Technology Plan Project: Jianpi-Huayu Decoction promotes the Inhibition of Liver Cancer Stem Cell Transformation by Antagonizing Wnt/β-catenin Pathway and Reducing Postoperative Recurrence Mechanism of Liver Cancer (Number: 2023LNS27059).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sets used & analyzed during previous study are available from author Enling Xu.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
