Abstract
Background:
Hepatocellular carcinoma (HCC) is the most prevalent form of primary liver cancer, characterized by high incidence and mortality rates. Yangzheng-Xiaoji capsules (YXC) have been widely used in Traditional Chinese Medicine for the treatment of HCC in China; however, the composition of compounds in YXC and its underlying anti-tumor mechanisms remain unclear.
Methods:
High-performance liquid chromatography quadrupole time-of-flight mass spectrometry (HPLC-Q-TOF-MS/MS) was used to analyze the composition of YXC. An MHCC97H orthotopic mouse model of HCC was used to assess the therapeutic efficacy of YXC and its impact on the p53-induced apoptotic pathway and the PI3K/Akt pathway. YXC-mediated serum was prepared, and its optimal intervention concentration was determined using the MTT assay. The effects of YXC-mediated serum on apoptosis in MHCC97H cells, as well as on the p53-induced apoptotic pathway and the PI3K/Akt pathway, were investigated and verified through the TUNEL assay.
Results:
HPLC-Q-TOF-MS/MS analysis identified 58 components in YXC. In vivo, YXC significantly improved body weight, dietary status, survival rate, and tissue pathology, inhibited xenografts growth, promoted apoptosis of HCC cells, activated the p53-induced apoptotic pathway, and inhibited the PI3K/Akt signaling pathway with good safety. In vitro, YXC-mediated serum showed similar efficacy. When 20% YXC-mediated serum was added to MHCC97H cells, the increased apoptosis was significantly rescued by the addition of p53 inhibitor (PFT-α) or PI3K activator (740Y-P).
Conclusion:
The results of this study indicate that YXC exhibits anti-proliferative and pro-apoptotic pharmacological activity via activating the p53-induced apoptotic pathway and suppressing the PI3K/Akt pathway.
Introduction
Liver cancer is the fourth leading cause of cancer-related deaths worldwide, with approximately 630 000 new cases diagnosed annually, half of which occur in China. 1 Hepatocellular carcinoma (HCC), the most common form of liver cancer, is associated with high incidence and mortality rates. Despite advances in chemotherapy, radiotherapy, and surgery, the recurrence and mortality rates of HCC remain alarmingly high. 2 These treatments are often limited in their effectiveness and can cause significant side effects, severely impacting patients’ quality of life. 3 Therefore, it is crucial to identify therapies that not only inhibit the progression of HCC but also effectively and safely alleviate symptoms while extending patient survival.
Traditional Chinese Medicine (TCM) is widely used as an adjunctive to chemotherapy in advanced liver cancer. It can help reduce chemotherapy-related side effects, alleviate clinical symptoms, and improve patients’ quality of life. Compared to Western medicine, TCM is associated with low toxicity, higher safety and less resistance. 4 Yangzheng-Xiaoji Capsules (YXC), a traditional TCM formulation composed of 16 herbal ingredients, are commonly prescribed to treat HCC linked to spleen-kidney deficiency and blood stasis. Recent research has highlighted the broad pharmacological activities of YXC, including anti-tumor, anti-inflammatory, and immune-boosting effects. 5 Clinical studies have reported its positive impact on various cancers, including liver, gastrointestinal, and lung cancers.6,7 However, the exact composition and underlying biological mechanisms of YXC remain unclear.
Apoptosis is a form of programed cell death that regulates cell division to maintain tissue homeostasis and support development. Malignant tumors often arise when cells are persistently stimulated by proliferative signals and become resistant to apoptosis. 8 Consequently, selectively killing tumor cells by promoting apoptosis has become a critical strategy in cancer treatment. Apoptosis mainly occurs through 2 pathways: the endogenous mitochondrial pathway and the exogenous death receptor pathway. 9 The p53 protein plays a central role in regulating apoptosis, acting as a key regulator of the endogenous apoptotic pathway. It triggers caspase activation and induces apoptosis in tumor cells by controlling downstream target genes. 10 Additionally, as a tumor suppressor, p53 also interacts with the PI3K/Akt pathway, where activation of p53 can inhibit the pathway and promote apoptosis. 11 The PI3K/Akt pathway is a key intracellular signaling cascade that regulates various cell processes and activates downstream molecules, influencing apoptosis, transcription, translation, and metabolism in tumor cells. Abnormal activation of Akt is a hallmark of poor prognosis in many cancers, including HCC. 12
This study aimed to identify the primary components of YXC and establish an MHCC97H orthotopic HCC model in nude mice. We assessed the therapeutic efficacy of YXC in nude mice with HCC, focusing on its impact on apoptosis and potential anti-tumor mechanisms. These results provide a certain basis for further exploration of the clinical applications of YXC.
Materials and Methods
Preparation Process and Ingredients Identification
YXC was provided by Shijiazhuang Yiling Pharmaceutical Co., Ltd. (Shijiazhuang, China; Batch No. B2106002). A detailed list of the herbs used in YXC is provided in Table 1. The HPLC-Q-TOF-MS/MS procedures were performed according to the standard method. 13 The YXC powder was dissolved in water (1 g/mL). A 0.1 mL aliquot was mixed with 0.4 mL methanol, vortexed, and then centrifuged at 12 000 rpm for 10 minutes at 4ºC. The supernatant was further diluted with 0.1 mL of 50% methanol and analyzed by HPLC using an Agilent ZorBax SB-C18 column (1.8 µm, 4.6 × 150 mm). Data were processed using Peakview Software™ 1.2, and components were identified by molecular weight, retention time, and MS/MS spectra.
Specific Information of YXC.

Medicated Serum Preparation
The protocol for the preparation of drug-containing serum was adapted from the previously published approach. 13 Animal experiments were conducted on 30 male Sprague-Dawley (SD) rats (220-300 g), sourced from the Experimental Animal Center of Nanjing University of Chinese Medicine. The study was approved by the Animal Ethics Committee of the university in accordance with the “Guidelines for Ethical Review of Laboratory Animal Welfare” (Approval Number: 202108A010). After a 1-week acclimation, the rats were randomly divided into 2 groups: YXC group (n = 20) and control group (n = 10). The YXC group was administered YXC by gavage at a dose of 2 g/kg for 7 days, while the control group received an equivalent volume of saline. 14 Two hours after the final administration, blood samples were collected from the abdominal aorta. Serum from each group was pooled, centrifuged, filtered through a 0.22 μm membrane, heat-inactivated at 56ºC for 30 minutes, and stored at −80ºC until further use.
Cell Culture
The MHCC97H cell line, obtained from the Chinese Academy of Sciences (Shanghai, China), was cultured in DMEM supplemented with 10% fetal bovine serum and 1% penicillin-streptomycin. The culture medium was refreshed every 2 days. Cells were passaged upon reaching 90% confluence and maintained at 37°C in a 5% CO2 environment.
Model Establishment
The animal model was successfully established based on a previous study. 15 Male BALB/c nude mice, 4 weeks old and weighing 18 ± 2 g, were purchased from WeiTongLiHua Experimental Animal Technical Co., Ltd (Zhejiang, China, Certificate No. SYXK [Zhe] 2019-0001). The mice were housed in the SPF animal experiment center at Nanjing University of Chinese Medicine. Ethical guidelines were followed to minimize animal use, ensuring statistical validity with the smallest number of mice required. The mice were randomly assigned to 2 groups: the negative control (NC, n = 6) and the negative model (NM, n = 36). To establish an orthotopic HCC model, MHCC97H cells (1 × 107) were injected into the livers of the NM group. One week after injection, the NM group was subdivided into 3 groups: model, Oxaliplatin (OXA), and YXC (n = 12 per group). The YXC dosage was calculated using a human-to-animal dose conversion and prepared fresh daily to maintain its pharmacological activity. The NC and model groups received equivalent volumes of saline. The OXA group was administered 8 mg/kg of OXA every other day via intraperitoneal injection. 16 The YXC group received 1 g/kg of YXC through daily gavage. 17 The intervention lasted for 4 weeks.
Body Weight and Food Intake
The weight of the nude mice was measured weekly. Cages were also weighed to monitor food consumption, and the average daily food intake was then calculated. After 4 weeks of treatment, the mice were sacrificed, and their livers, lungs, and spleens were weighed. Liver and lung tissues were fixed in 4% paraformaldehyde, embedded in paraffin, and sectioned into 5 μm slices for further analysis. The sections were stained with HE for examination.
Western Blotting
Western blotting was performed as previously described. 18 Total protein was extracted from fresh liver tissues using pre-cooled reagents. The tissues were minced, lysed in tissue lysis buffer, and centrifuged at 12 000 rpm for 15 minutes at 4°C. Protein concentration was measured with a BCA protein assay kit (Biyuntian, China). The supernatant was mixed with 4× loading buffer and boiled at 100°C for 10 minutes. Equal protein amounts (25 μg) were separated on 10% or 12% polyacrylamide gels, then transferred to PVDF membranes. The membranes were blocked with 5% skimmed milk for 2 hours at room temperature, washed with TBST, and incubated overnight at 4°C with primary antibodies (p53, Bcl-2, Bax, cleaved Caspase-3, Caspase-3, cleaved Caspase-8, Caspase-8, cleaved Caspase-9, Caspase-9, p-PI3K, PI3K, p-Akt, and Akt). After washing with PBST, they were incubated with secondary antibodies for 40 minutes at room temperature, followed by 3 washes with PBST. Protein levels were quantified using the Bradford assay.
Immunohistochemisty (IHC)
Western blotting was performed as previously described. 18 Tissue sections were blocked with 3% normal goat or horse serum in PBS for 30 minutes, then incubated overnight at 4°C with diluted primary antibodies (p53, p-PI3K, and p-Akt) in blocking serum. After washing with PBS, the slides were treated with the avidin-biotin-peroxidase complex (ABC) and developed using diaminobenzidine (DAB) with hydrogen peroxide. Finally, the slides were mounted with Aqua-Mount and examined under a light microscope.
MTT Assay
MTT assay was performed following the protocol with minor modifications. 13 Adherent MHCC97H cells were treated with varying concentrations of YXC-medicated serum for 24, 48, and 72 hours. After incubation, 20 μL of MTT (5 mg/mL) was added to each well and incubated at 37°C for 4 hours. DMSO was then added to dissolve the formazan crystals, and the absorbance was measured at 565 nm using a BioRad microplate reader.
TUNEL Staining
The TUNEL staining protocol was based on the manufacturer’s instruction (Kaiji, China). Adherent MHCC97H cells were pre-incubated with the p53 inhibitor PFT-α (10 µM), the PI3K activator 740Y-P (15 µM), or the PI3K inhibitor LY294002 (10 µM) for 24 h. Subsequently, the cells were co-cultured with 20% YXC-containing serum for an additional 48 h. After treatment, the cells were washed with PBS, fixed in 4% paraformaldehyde for 30 minutes, and permeabilized with 1% Triton X-100 for 5 minutes. Apoptosis was observed under a fluorescence microscope, and the percentage was calculated from the ratio of apoptotic cells to total cells.
Statistical Analysis
A 1-way ANOVA and Student’s t-test were used to assess the differences between groups. A P-value of less than .05 was considered statistically significant.
Results
Identification of Components in YXC
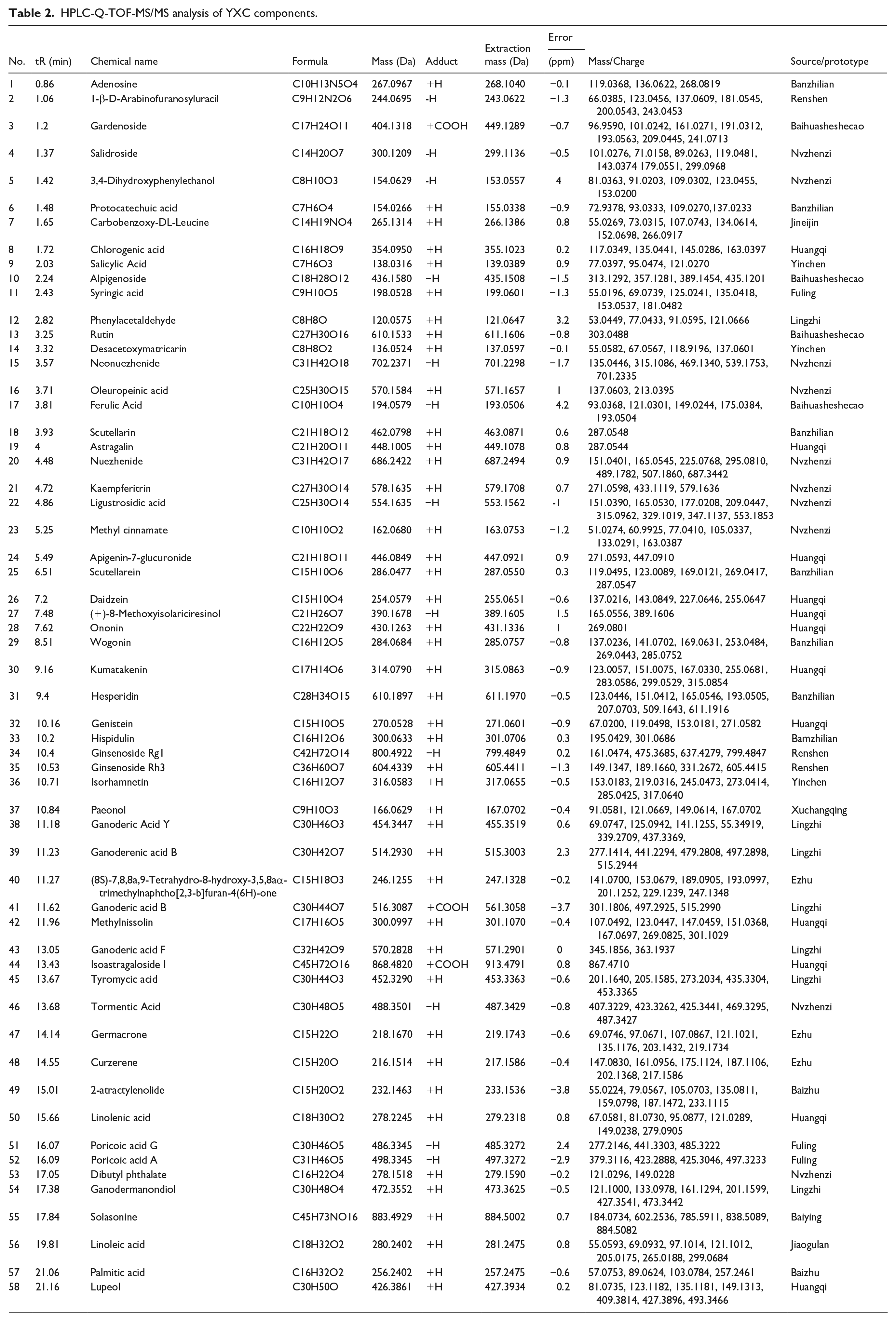
To analyze the components of YXC, Peakview 1.2 Software was employed to generate the base peak chromatogram (BPC) of YXC samples processed in both positive and negative ion modes (Figure 1). A total of 58 compounds were identified by HPLC-Q-TOF-MS/MS, with further details provided in Table 2.

Analytical HPLC-Q-TOF-MS/MS BPC chromatogram.
HPLC-Q-TOF-MS/MS analysis of YXC components.
YXC Treatment Improved the Physical Condition
To investigate the effect of YXC on the physical condition of HCC in vivo, we established an MHCC97H orthotopic mouse model. Before the study, all mice in the 4 groups exhibited normal health and behavior. After 28 days of treatment, the nude mice in the model group showed significant reductions in both body weight and food intake compared to the NC group (P < .05). In the YXC group, both body weight and food intake changed little, whereas in the OXA group, these parameters decreased significantly (P < .05, Figure 2A and B). The Kaplan-Meier curve indicated a longer overall survival in the YXC group compared to the model and OXA groups (Figure 2C). Liver and lung weights were significantly reduced in both the YXC and OXA groups compared to the model group (P < .01, Figure 2D and E). Furthermore, spleen weight in the model group was significantly lower than in the NC group (P < .001, Figure 2F). While the spleen weight in the model group was higher than in the OXA group, it remained lower than that in the YXC group (P < .05, P < .01, Figure 2F).

YXC treatment enhances physical condition in vivo. (A-B) Changes in mouse body weight and food intake over time. *P<.05 compared with the NC group. (C) Kaplan-Meier survival curve of nude mice from each group. (D-F) Liver weight, lung weight, and spleen weight of nude mice on 28 day. ##P<.01, ###P<.001 compared with the NC group; *P<.05, **P<.01, ***P<.001 compared with the model group.
YXC Treatment Improved the Liver and Lung Pathology
To investigate the anti-HCC activity of YXC in vivo, HE staining was performed to observe the livers and lungs of mice with HCC. In the model group, tumor cells displayed typical heteromorphism, characterized by high density and significant variation in size and shape, contrasting sharply with normal liver cells. The YXC and OXA groups, on the other hand, showed a notable reduction in tumor cell numbers. The cells in these groups were more loosely arranged, with lower density and diminished mitotic activity, indicating reduced cellular heteromorphism. Additionally, lung tissue sections revealed fewer tumor cells in the lungs of nude mice treated with YXC and OXA compared to those in the model group (Figure 3).
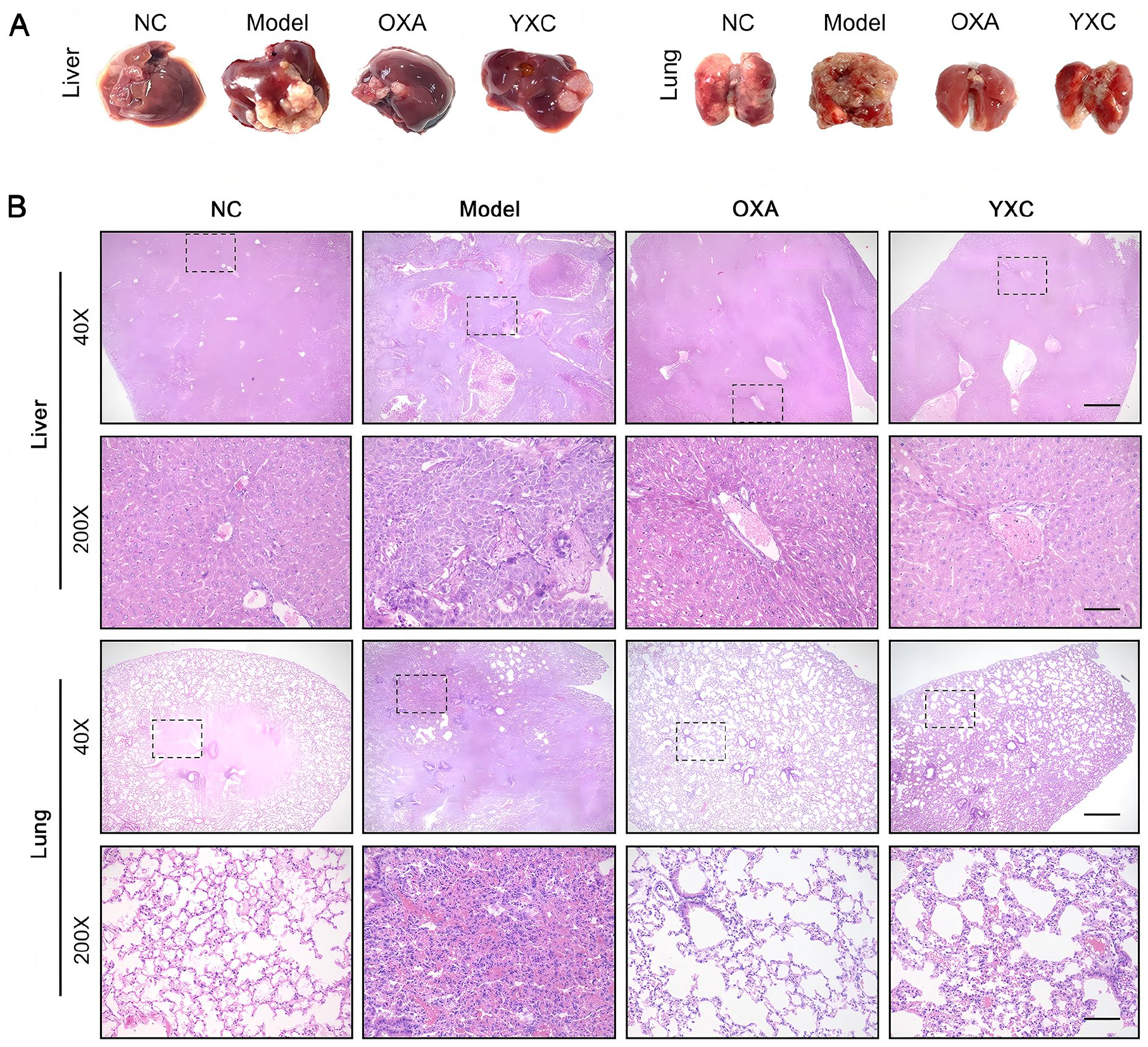
YXC treatment restrains HCC growth in vivo. (A) Representative images of the liver and lung from each group. (B) H&E staining of liver and lung sections from each group. (magnification, ×40), scale bars=80 µM; (magnification, ×200), scale bars=400 µM.
YXC Treatment Activated the p53-Induced Apoptotic Pathway
To investigate the effect of YXC on apoptosis in HCC cells, we assessed the expression of apoptosis-related proteins in mouse liver tissue samples by Western blot. The results showed that, compared to the model group, the YXC and OXA groups exhibited significantly increased protein expression of Bax (P < .01), cleaved Caspase-3/Caspase-3 (P < .05), cleaved Caspase-8/Caspase-8 (P < .01), and cleaved Caspase-9/Caspase-9 (P < .001). In contrast, Bcl-2 expression was significantly reduced (P < .001) in these groups. Additionally, the expression level of p53 protein was significantly upregulated after YXC and OXA (P < .01, Figure 4A and B). P53 is closely associated with apoptosis induction. However, when YXC was co-treated with p53 inhibitor PFT-α, the number of YXC-induced apoptotic cells was significantly decreased (P < .001, Figure 6N). Based on these findings, we propose that YXC inhibits the progression of HCC in the MHCC97H orthotopic mouse model through the p53-induced apoptotic pathway.

YXC treatment activates the p53-induced apoptotic pathway and inhibits the PI3K/Akt signaling pathway in vivo. (A-B) Western blotting analysis to detect the expression levels of p53, Bcl-2, Bax, cleaved Caspase-9, Caspase-9, cleaved Caspase-8, Caspase-8, cleaved Caspase-3, and Caspase-3 in mouse liver tissues, subjected to varying concentrations of OXA (8 mg/kg) and YXC (1 g/kg). (C) Western blotting analysis to detect the expression levels of p-PI3K, PI3K, p-Akt and Akt in mouse liver tissues, subjected to varying concentrations of OXA (8 mg/kg) and YXC (1 g/kg). ##P<.01, ###P<.001 compared with the NC group; *P<.05, **P<.01, ***P<.001 compared with the model group.
YXC Treatment Inhibited the PI3K/Akt Signaling Pathway
As the PI3K/Akt signaling pathway plays a pivotal role in the pathogenesis of HCC, 32 we investigated whether YXC affects this pathway. Western Blot analysis showed that, in both the YXC and OXA groups, the protein expression levels of p-PI3K and p-Akt were significantly lower than those in the model group (P< .01, Figure 4C and D). These findings were further validated by IHC in 3 independent experiments (P< .01, Figure 5). Additionally, MHCC97H cell lines were treated with various concentrations of YXC-medicated serum to evaluate its cytotoxic effects. As shown in Figure 6A, cell viability was significantly reduced after treatment with YXC-medicated serum. After 48 hours, 20% and 30% YXC-medicated serum significantly reduced cell viability (P < .001, Figure 6B). To examine the pro-apoptotic effect of YXC-medicated serum on MHCC97H cells, TUNEL staining was employed to measure apoptotic cells. As exhibited in Figure 6C-F and M, YXC-medicated serum significantly increased the number of TUNEL-positive cells in a dose-dependent manner, indicating that YXC could promote apoptosis in MHCC97H cells. However, when YXC was co-treated with the PI3K activator 740Y-P, the number of YXC-induced apoptotic cells was significantly reduced (P < .001). No significant difference was observed between the YXC group and the combination of YXC with PI3K inhibitor LY294002 (Figure 6E, G-L, and N). These results suggest that YXC could exert a pro-apoptotic effect on MHCC97H cells via the PI3K/Akt signaling pathway in liver cancer.
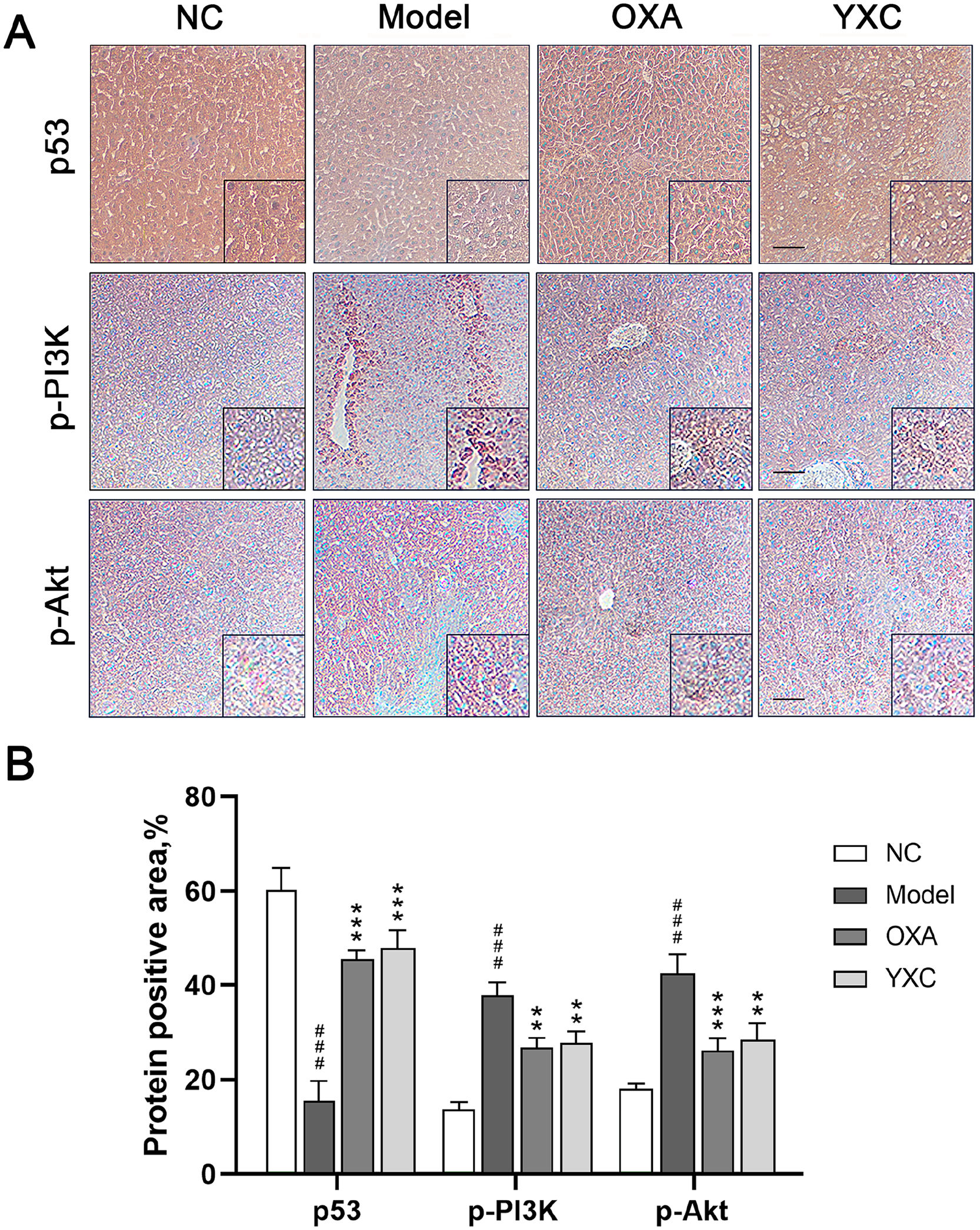
Effects of YXC treatment on Expression of p53, p-PI3K and p-Akt. (A-B) IHC analysis to detect the expression levels of p53, p-PI3K, and p-Akt in mouse liver tissues, subjected to varying concentrations of OXA (8 mg/kg) and YXC (1 g/kg). (magnification, ×100), scale bars=200 µM. ###P<.001 compared with the NC group; **P<.01, ***P<.001 compared with the model group.

YXC treatment promotes apoptosis in MHCC97H cells via the p53-induced apoptotic pathway and inhibits the PI3K/Akt signaling pathway. (A) Cell viability after treatment with different concentrations of YXC-medicated serum (5%, 10%, 20%, and 30%) at 24, 48, and 72 h. (B) Cell viability after different concentrations of YXC-medicated serum for 48 h. TUNEL staining images (C-L) and quantification (M, N). (C) Control group. (D-F) Cells treated with YXC(L), (M), and (H) doses for 48 h. (G) Cells treated with 10 µM of PFT-α for 24 h. (H) Cells pre-cultured with 10 µM PFT-α followed by YXC (M) treatment for 48 h. (I) Cells treated with 10 µM LY294002 for 24 h. (J) Cells pre-cultured with 10 µM LY294002 followed by YXC (M) treatment for 48 h. (K) Cells treated with 15 µM 740Y-P for 24 h. (L) Cells pre-cultured with 15 µM 740Y-P followed by YXC (M) treatment for 48 h. (magnification, ×100), scale bars=200 µM. (M-N) Quantification of TUNEL positive cells. ***P<.001 compared with the control group; ###P<.001 compared with the YXC(M) group.
Discussion
Liver cancer is one of the most common malignant tumors worldwide. In 2020, approximately 906 000 new cases and 830 000 deaths were reported globally, with China accounting for about 360 000 new cases and 350 000 deaths annually. 19 HCC, the most common subtype, represents 80% of all primary liver cancers, and its incidence is projected to reach 1 million cases per year by 2025. 20 The main treatments for HCC include chemotherapy, radiotherapy, and surgery. However, despite the effectiveness of these therapies in targeting tumor cells, they are associated with significant cytotoxicity and side effects, which limit their clinical application. Therefore, there is a critical need for more effective and less toxic treatments for HCC. This study aims to investigate the therapeutic effects and underlying mechanisms of YXC in the treatment of HCC, providing insights into potential strategies for improving HCC therapy.
Compound Chinese Traditional Medicine (CCTM), which combines multiple herbs based on principles of TCM, has been used for centuries in China to treat various diseases. 21 Collateral disease theory, a modern branch of TCM rooted in the Huangdi Neijing, is widely applied in the clinical treatment of malignant tumors. YXC, a nationally recognized anti-tumor Chinese patent medicine developed by Dr. Wu Yiling, is based on this theory. It is believed to strengthen the spleen, tonify the kidney, eliminate toxins, remove stagnation, and unblock meridians. When used appropriately, YXC has been shown to significantly inhibit tumor progression and reduce the side effects of radiotherapy and chemotherapy in HCC treatment. 22 HPLC-Q-TOF-MS/MS has proven to be an effective, fast, accurate, and cost-efficient method for analyzing the complex chemical constituents of Chinese medicinal preparations. 23 In this study, 58 primary chemical components of YXC were identified using HPLC-Q-TOF-MS/MS.
Oxaliplatin (OXA), a third-generation platinum-based chemotherapy drug, induces cell apoptosis by forming platinum-DNA adducts, contributing to its relatively favorable toxicity profile. 24 Due to its potent anti-tumor effects, OXA is widely used in the treatment of HCC and has become a standard therapeutic option. 25 However, its use is often associated with significant side effects, such as peripheral neuropathy, gastrointestinal damage, and reduced blood cell counts during cancer treatment. 26
In this study, nude mice treated with OXA exhibited poor appetite, sluggish reflexes, and significant reductions in body and liver weight. In contrast, mice in the YXC group showed slightly higher body weights than those in the model group and maintained generally good health, with liver weights similar to those in the NC group. Both the OXA and YXC groups showed significantly reduced liver and lung weights compared to the model group, indicating that YXC effectively inhibits the progression of HCC in nude mice. This finding was further confirmed by HE staining. Additionally, the YXC group exhibited a higher overall survival rate than the OXA group, suggesting YXC treatment may result in fewer side effects. The spleen, as the body’s largest immune organ, plays a crucial role in systemic immunity. 27 The results demonstrated that the spleen weight was significantly reduced in the OXA group compared to the model group, likely due to chemotherapy-induced side effects, which caused overall weight loss in the mice. In contrast, YXC treatment significantly increased spleen weight, suggesting a potential of immune function in the spleen of cancer-affected mice.
The p53, a potent tumor suppressor, plays a pivotal role in nearly all human cancers, including HCC. As a transcription factor, p53 regulates essential cellular processes such as apoptosis, cell differentiation, DNA repair, and energy metabolism. 28 Notably, p53 mutations are present in at least half of all liver cancer cases. 29 In our study, p53 expression was significantly lower in the model group than in the NC group, suggesting p53 mutations in HCC nude mice. However, treatment with YXC and OXA notably increased p53 expression, indicating that YXC may enhance p53-mediated pathways. This observation is further supported by IHC staining results. Apoptosis, a form of programed cell death, is a vital mechanism for eliminating damaged cells and is a key target for anti-tumor therapies. 30 As a key regulator of apoptosis, p53 facilitates the removal of defective cells by influencing apoptosis-related proteins, including Bax and Bcl-2. Studies have shown that p53 activation promotes the release of pro-apoptotic factors by altering the balance between these proteins. 31 In our findings, YXC treatment significantly elevated the expression levels of Bax, cleaved Caspase-9/Caspase-9, cleaved Caspase-8/Caspase-8, and cleaved Caspase-3/Caspase-3, while reducing Bcl-2 expression. These results indicate that YXC activates the caspase-dependent signaling pathway, thereby enhancing apoptosis in tumor cells. Furthermore, the addition of p53 inhibitors substantially reduced apoptosis in MHCC97H cells, reinforcing the notion that YXC promotes cell death via the p53-induced apoptotic pathway. Collectively, these findings underscore the potential of YXC as a therapeutic agent that leverages the p53 pathway to suppress tumor growth in HCC.
The PI3K/Akt pathway is a well-established signaling pathway known for promoting cell survival and inhibiting apoptosis, playing a critical role in tumor development, progression, and resistance to chemotherapy. 32 As the central component of this pathway, Akt regulates key downstream apoptosis-related signals, including the p53/caspase-3 pathway. 33 This pathway is highly expressed in various human tumors, including HCC. Consequently, targeting the PI3K/Akt pathway with specific inhibitors has emerged as a promising therapeutic strategy for HCC treatment. In this study, we investigated the mechanism by which YXC inhibits tumor progression by analyzing the expression levels of p-PI3K, PI3K, p-Akt, and Akt proteins. Our findings showed a significant reduction in p-PI3K and p-Akt expression in the YXC-treated group, compared to the model group, indicating that YXC effectively inhibits the activation of the PI3K/Akt pathway. As expected, IHC staining results showed a similar result in tumor tissues. Moreover, the activator of the PI3K/Akt pathway can significantly reduce YXC-induced apoptosis in MHCC97H cells. Interestingly, the PI3K/Akt pathway inhibitor had little impact on the number of apoptotic cells induced by YXC. This may be attributed to YXC’s complete suppression of the activity, potentially masking the pro-apoptotic effects of the inhibitor LY294002. These findings suggest that YXC’s anti-tumor effects in HCC may be achieved by inhibiting the PI3K/Akt pathway.
Conclusion
YXC, a traditional Chinese medicinal formula, has been used in clinical practice for years to treat HCC in China. In this study, we identified the main components of YXC and confirmed its ability to inhibit HCC proliferation and progression in an orthotopic nude mouse model. The results indicate that YXC induces tumor cell apoptosis by regulating the p53-mediated apoptotic pathway and suppressing the PI3K/Akt signaling pathway.
Footnotes
Acknowledgements
Author’s Note
Yu-han Wang is also affiliated to Wuxi Hospital of Traditional Chinese Medicine, Wuxi, China.
Ethical Considerations
This study was approved by the Ethics Committee of Nanjing University of Chinese Medicine Authority (Approval Number: 202108A010).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was performed without financial support from the industry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during the current study will be available from the corresponding author upon reasonable request.
