Abstract
Background:
Evidence regarding Chinese herbal medicine for facial rash related to epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs) is limited. Therefore, this study aimed to investigate whether ex ternal application of a Xiaozhen zhiyang (XZZY) decoction is effective for EGFR-TKI-related facial rash in Chinese patients.
Methods:
In this prospective cohort study, 123 participants with EGFR-TKI-related facial rash were recruited from July 18, 2019 to May 20, 2021. The intervention was external application of the XZZY decoction twice daily for 2 weeks using a facial mask. Treatment efficacy, change in the WoMo score, and quality of life were evaluated as endpoints.
Results:
The average age of the 123 enrolled participants was 54.83 ± 12.45 years, and 48.78% were female. The total efficiency of treatment was 93.5%, including 12 (10%) cases were cured, 20 (16.26%) cases were markedly improved and 83 (67.48%) cases were improved. The generalized estimating equations showed decreased WoMo scores, itching, and pain, as well as an improved quality of life after the treatment. Only 1 participant reported skin allergies, and no other adverse effects were reported.
Conclusion:
This study showed the effectiveness and safety of a XZZY decoction for EGFR-TKI-related rash.
Keywords
Introduction
According to the data released by the National Cancer Center in 2022, the incidence rate of lung cancer in China in 2016 was 59.89 per 100 000 population. Among them, the rate was 77.64 per 100 000 for males and 41.26 per 100 000 for females, ranking first among malignant tumors. 1 The mortality rate of lung cancer reached 47.51 per 100 000, also ranking first among malignant tumors. 2 The epidermal growth factor receptor (EGFR) has become a critical molecular target for non-small cell lung cancer (NSCLC), and EGFR tyrosine kinase inhibitors (TKIs) are widely used to treat NSCLC with EGFR mutations. 3
However, EGFR-TKIs cause specific skin-related side effects, including skin toxicities. EGFR-TKI-related skin toxicities include acne-like rash, paronychia, abnormalities in hair growth, itching, and dry skin, known as PRIDE syndrome. 4 The incidence of rash/acne-like rash in a phase III clinical study of EGFR-TKIs was between 54% and 89%. 5 Because the face, chest, and back have abundant sebaceous glands, itching and painful maculopapular and impetigo appear 1 to 2 weeks after starting EGFR-TKI treatment. One to third of patients experience significant itching and burning symptoms, and 38.0% of patients have secondary skin infections, which significantly reduce quality of life.6 -8 These rashes have adverse effects that increase the patient’s risk for additional infections and lead to inconsistent EGFR-TKI administration. 9 While trials have demonstrated that antibiotics, such as tetracycline and minocycline, can prevent papulopustular rash,10 -15 evidence for the effective treatment of such rashes remains limited. 16
External application of Chinese herbal medicine (CHM) is commonly used in China for various skin conditions. However, previous studies have shown that oral Traditional Chinese Medicine combined with external application treatments has demonstrated effectiveness. However, research on using external Chinese herbal medicine treatments alone is still relatively limited. 17 Xiaozhen zhiyang (XZZY) decoction is an empirical formula formulated based on Traditional Chinese Medicine theory to address the main pathological mechanism of EGFR-TKI-related rash. This prescription represents the cumulative wisdom of traditional clinical practice, where repeated clinical applications have gradually refined the herbal combination into a systematically optimized formula.18,19 The formula includes herbs such as Lonicerae japonicae flos (Jin Yin Hua), Dandelion (Pu Gong Ying), Sophorae flavescentis radix (Ku Shen), Kochiae fructus (Di Fu Zi), Dictamni cortex (Bai Xian Pi), Cortex moutan (Mu Dan Pi), Phellodendri amurensis cortex (Guan Huang Bai), and Menthae herba (Bo He). The entire formula has the effects of clearing heat, cooling blood, drying dampness, and relieving itching. This study is essential to further justify the use of XZZY decoction as a treatment option, potentially improving the quality of life for patients undergoing EGFR-TKI therapy.
Patients and Methods
Patients
Eligible patients were 18 to 75 years old and had histologically or cytologically confirmed NSCLC with unresectable stage IIIB or IV disease, according to the seventh edition of the International Association for the Study of Lung Cancer staging system. All patients had EGFR-sensitive mutations, received EGFR-TKI therapy, and developed a facial rash. Inclusion criteria encompass informed consent, life expectancy ≥3 months, good compliance, ECOG PS score 0 to 3, and meeting laboratory requirements including hemoglobin ≥90 g/L, ANC ≥1.5 × 109/L, PLT ≥100 × 109/L, and normal liver and kidney function. Exclusion criteria include non-EGFR-TKI-related facial rash during screening, recent treatments potentially affecting study results, pregnancy or breastfeeding, uncontrolled comorbidities, allergies to Chinese medicine, mental illness or substance abuse, and any condition that may confound study results or compromise patient safety. Investigators may exclude patients based on medical history or current evidence that could interfere with study procedures or outcomes. Participants were recruited from the First Affiliated Hospital of Guangzhou University of Chinese Medicine and Sun Yat-sen University Cancer Center.
The study was approved by an independent ethics committee and was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. The Ethics Committee of the First Affiliated Hospital of Guangzhou University of Chinese Medicine approved this trial (NO.K【2019】026). Each participant signed informed consent before enrollment.
Intervention
The XZZY decoction contained 8 CHMs: Lonicerae japonicae flos (Jinyinhua), Dandelion (Pugongying), Sophorae flavescentis radix (Kushen), Kochiae fructus (Difuzi), Dictamni cortex (Baixianpi), Cortex moutan (Mudanpi), Phellodendri amurensis cortex (Guanhuanbai), and Menthae herba (Bohe; see Table 1). The XZZY decoction was prepared by the Department of Pharmacy at the First Affiliated Hospital of Guangzhou University of Chinese Medicine and stored at 4°C. A total of 1.88 g XZZY decoction was added to 50 ml bottles. Each patient used a dry mask soaked in the XZZY formula to create a traditional Chinese medicine (TCM) mask. The patients covered their face with the mask for 30 minutes, twice a day for 2 weeks (14 days). Routine care included applying skin moisturizer to the face, hands, feet, neck, back, and chest daily in the morning, followed by sunscreen (PABA free, SPF 15, UVA, and UVB protection) to exposed skin areas before going outdoors. Researchers provided an instructional video on preparing the TCM mask and routine care to patients before enrollment.
The Composition of the Externally Applied Xiaozhen Zhiyang (XZZY) Decoction.

Evaluation
The primary endpoint was the efficacy rate after 2 weeks intervention based on the Guiding Principles for Clinical Research of New Chinese Medicines. 20 The assessment criteria were as follows: the condition was cured (skin rash reduced by at least 95%), markedly improved (skin lesion area reduced by 70%-94%), improved (skin lesion area reduced by 50%-70%), or not improved (skin lesion area reduced by <50%).
Digital photography, was performed at baseline, week 1, and week 2, using the same standard poses (photo poses: frontal face, profile face, Figure 1). The secondary endpoints were the score on the scale developed by Wollenberg et al (WoMo), 21 itching visual analog scale (VAS) score, pain VAS score, safety, and quality of life. The secondary outcome was a change in the WoMo score. The WoMo scale is suitable for monitoring the severity of skin lesions over time and consists of 3 parts: part A—body involvement, part B—facial involvement, and part C—clinical grading of skin items (erythema, papulation, pustulation, and scaling/crusts). The final score ranges from 0 to 100, with higher scores indicating worse rashes.

Photo poses: frontal face, profile face.
As the study aimed to evaluate the efficacy of the CHM face mask for facial rash, only the WoMo score based on parts B and C was used, with the total score ranging from 0 to 75. A generalized estimating equation was employed to estimate the change in the WoMo score from baseline (week 0) to week 2. Itching VAS and pain VAS were used to assess itching and pain, with scores ranging from 0 to 10. Skin-related quality of life was measured using the Skindex-16 and Dermatology Quality of Life Index (DLQI). The Skindex-16 score 22 is the most frequently used dermatology-specific health-related quality of life measurement, with the total scale ranging from 0 to 100. The DLQI was analyzed as the total score across 6 domains: symptoms and feelings, daily activities, leisure, work and school, personal relationships, and treatment. The total scale ranges from 0 to 30. 23 Higher scores on the Skindex-16 and DLQI indicate a worse quality of life.
All side effects were recorded. If the rash worsened, the investigator determined whether it was necessary to manage emergent skin toxicity during the intervention. Rescue therapy included topical steroids (1% hydrocortisone cream) and doxycycline 100 mg twice daily.
Statistical Analysis
In this prospective single-arm observational study, we calculated the sample size based on methods from published studies and data previously collected from our institution. Published studies have shown that the efficacy of external application of Chinese medicines for EGFR-TKIs related rash is 63.3% based on the measurement of the Guiding Principles for Clinical Research of New Chinese Medicines (p0). 24 In our previous clinical observation, the efficacy of the XZZY formula was 75% (p1). We set α to 0.05 and power (1−β) to 0.8 using a 2-sided test, and determined that a sample size of 123 participants was needed, accounting for a 20% dropout rate. Analyses were conducted using PASS 15 Power Analysis and Sample Size Software (2017). NCSS, LLC. Kaysville, Utah, USA). Continuous variables with a normal distribution are expressed as means ± standard deviation, while those with a skewed distribution are reported as medians (min, max). Categorical variables are expressed as frequencies or as numbers (percentage).
The efficacy is reported as a percentage comparing the baseline and Week 2. The efficiencies of different generations of EGFR-TKIs were compared using the chi-squared test. To further assess differences in WoMo, Skindex-16, DLQI, pain VAS, and itch VAS scores according to the study time points (baseline, week 1, and week 2), linear regression models with generalized estimating equations were employed. The last observation carried forward (LOCF) method was used for imputation of missing values.
All analyses were performed using the statistical software packages R (http://www.R-project.org, The R Foundation) and EmpowerStats (http://www.empowerstats.com, X&Y Solutions, Inc, Boston, MA). A P-value < .05 (2-sided) was considered statistically significant.
Results
Baseline Characteristics of the Participants
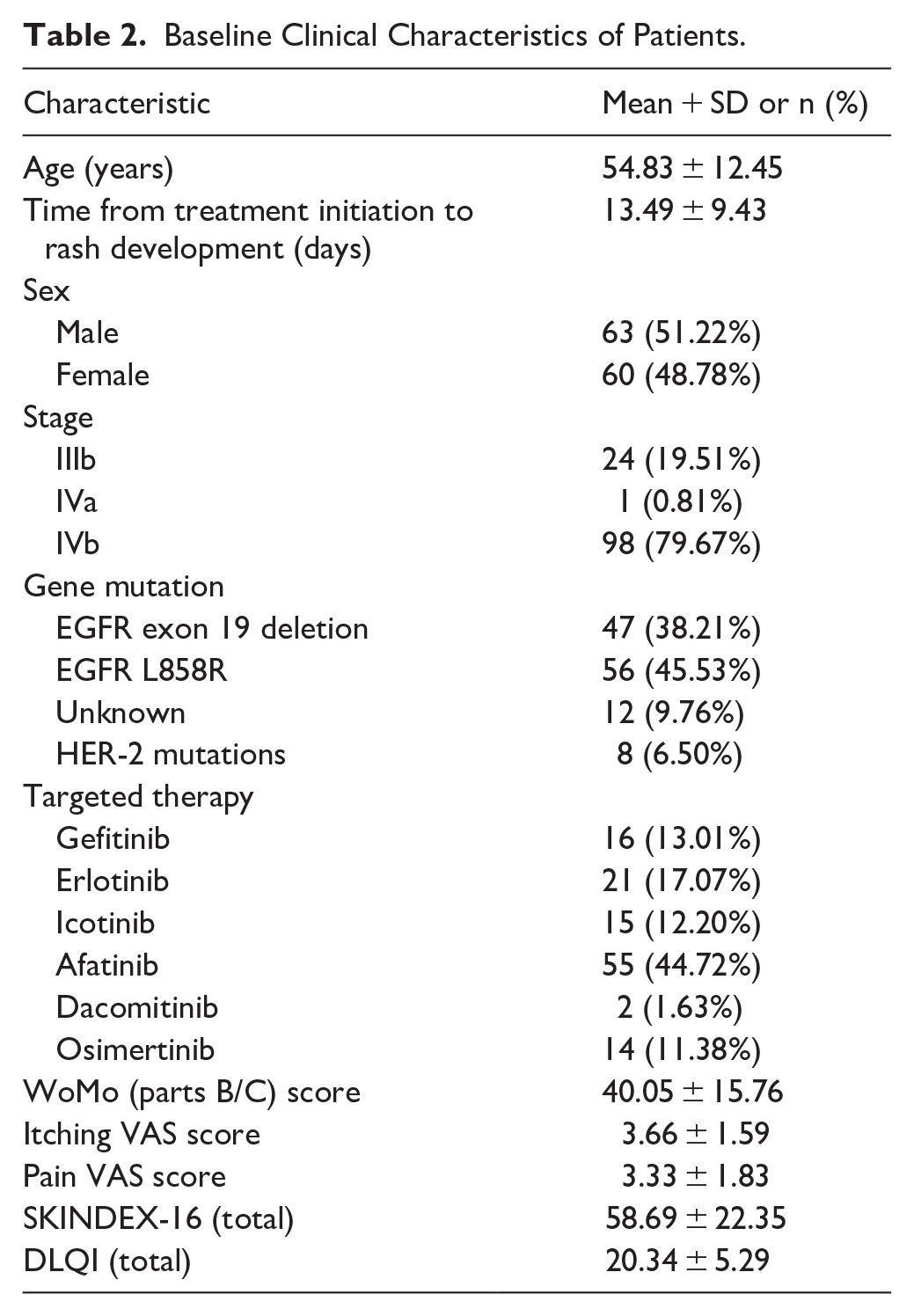
A total of 170 patients were initially assessed for eligibility. Of these, 20 patients were excluded due to absence of facial rash, leaving 150 patients for screening. During enrollment, 16 patients were excluded due to concurrent antibiotic treatment, and 124 patients were enrolled in the study. Among the enrolled patients, 1 patient was excluded due to skin allergies. At week 1 (W1), 123 patients completed the evaluation. By week 2 (W2), 110 patients completed the evaluation, with 13 patients lost to follow-up. A total of 123 patients were included in the final analysis. (see Figure 2 for a flowchart). The baseline characteristics of these participants are presented in Table 1. The average age of the 123 participants was 55.91 ± 13.15 years, and 51.52% were male. The study included 31 stage IIIb cases (23.48%), 31 stage IVa cases (23.48%) and 70 stage IVb cases (53.03%). There were 48 (36.36%) cases with EGFR exon 19 deletion, 58 (43.94%) with the EGFR L858R mutation, 7 (5.30%) with unknown mutations, and 19 (14.39%) with HER-2 mutations. The severity of facial rash was assessed using the WoMo scale (parts B/C) and yielded an average score of 40.05 ± 15.76. Participants reported mild itching and discomfort, with average scores of 3.66 ± 1.59 and 3.33 ± 1.83, respectively. Quality of life was evaluated through the SKINDEX-16 and DLQI, resulting in average scores of 58.69 ± 22.35 and 20.34 ± 5.29, respectively (refer to Table 2).

Flow chart.
Baseline Clinical Characteristics of Patients.
The Efficacy of XZZY Formula for EGFR-TKI-Related Rash
Dermatologists evaluated all images of the rashes. According to the Guiding Principles for Clinical Research of New Chinese Medicines, 12 (10%) cases were cured, 20 (16.26%) were markedly improved, 83 (67.48%) were improved, and 8 (6.50%) were not improved. The total efficacy (control rate = cured + markedly improved cases (%) + improved case (%) ) was 93.5%. Statistical analysis revealed no significant differences in treatment efficacy across patient characteristics including age (P = .219), gender (P = .462), disease stage (P = .179), genetic mutation type (P = .861), targeted drug used (P = .615), and rash type (P = .289). All patients presented with acneiform rash, with some also exhibiting pustular (48.78%) or maculopapular rash (39.84%). The consistent efficacy across diverse patient subgroups suggests that this TCM formula may be broadly applicable for managing EGFR-TKI-related rash (see Table 3).
The Efficacy of Treatment of Xiaozhen zhiyang (XZZY) Decoction.
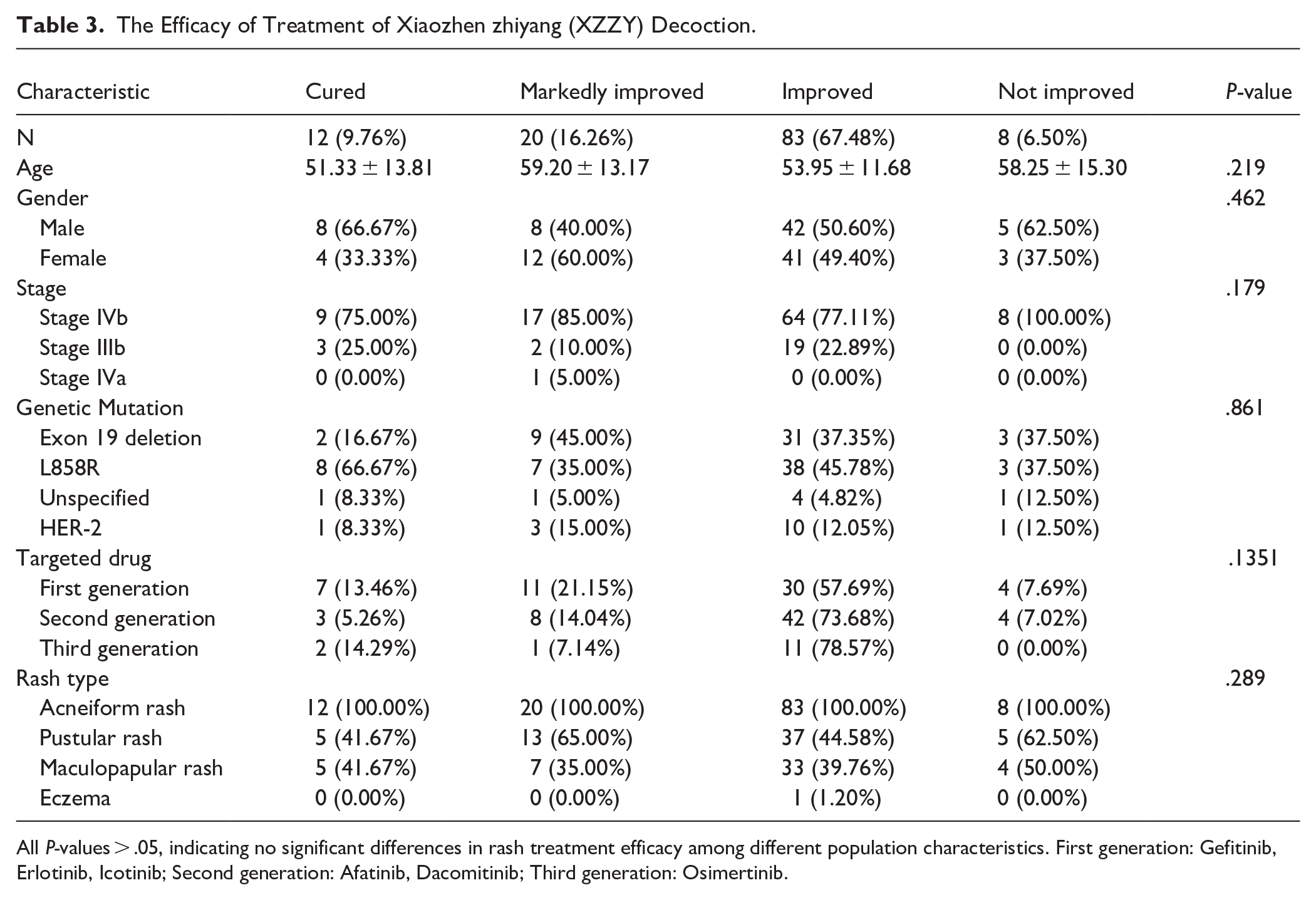
All P-values > .05, indicating no significant differences in rash treatment efficacy among different population characteristics. First generation: Gefitinib, Erlotinib, Icotinib; Second generation: Afatinib, Dacomitinib; Third generation: Osimertinib.
A linear regression model was used to compare changes in the WoMo score before and after treatment. The WoMo parts B/C score was calculated using the following formula: (1/4) part B score + (10/3) part C score. For the dependent variables in the analysis, baseline (week 0) was used as the reference, and comparisons between weeks 1 and 2 were made. The partial regression coefficients at weeks 1 and 2 were −8.3085 and −10.3847, respectively, both of which were statistically significant (P < .0001). This demonstrated that over time, the WoMo part C score decreased significantly, suggesting that the treatment was effective. The itching and pain VAS scores followed the same trend. (Supplemental Table 1) Both Skindex-16 and DLQI were used to measure quality of life. The generalized estimating equations showed a significant difference in these measures from baseline to week 2 (Supplemental Table 2).
The Safety
Only 1 participant reported skin allergies. The patient developed pruritus and erythema 10 minutes after using XZZY formula. Clinically diagnosed as contact dermatitis, prompting immediate discontinuation. Dexamethasone acetate ointment applied, improving symptoms with no complications. No other adverse effects were reported.
Discussion
EGFR-TKI-induced rash likely results from abnormal follicular keratinization and enhanced immune response due to EGFR blockade.25 -27 The management of EGFR-TKI-related rashes lacks sufficient evidence from therapeutic research. Current treatments, mainly antibiotics and hormonal symptomatic treatments, have unclear clinical effects and may lead to dose reduction or treatment discontinuation in moderate-to-severe cases.8,28,29 In traditional Chinese medicine (TCM) theory, external treatment methods are an important therapeutic approach, particularly effective in treating skin diseases. Our team conducted a meta-analysis on TCM treatments for EGFR-TKI-related skin rashes, and the results showed that external application of Chinese herbal medicine can effectively treat EGFR-TKI-related skin rashes. 17 Therefore, we chose to use external Chinese herbal medicine treatment for EGFR-TKI-related skin rashes.
In our study, the XZZY decoction effectively reduced the severity of EGFR-TKI-related rash, improved quality of life, and proved safe for patients. The XZZY decoction addresses these issues through its carefully selected components:
This study demonstrates that the standardized TCM topical regimen achieved a 93.5% overall efficacy rate (10% cured, 16.26% markedly improved, 67.48% improved) in managing EGFR-TKI-related rashes, surpassing conventional antibiotic/steroid therapies (65%-78% efficacy). Critically, efficacy remained consistent across all subgroups (age, genetic mutation, cancer stage; P > .05), supporting broad applicability without biomarker screening. The regimen effectively addressed multiple rash morphologies (acneiform, pustular, maculopapular) through a unified protocol, potentially streamlining clinical workflows. We believe these positive outcomes can be attributed to several factors: (1) Comprehensive formulation. The XZZY decoction combines herbs with anti-inflammatory, antimicrobial, and antipruritic properties, addressing multiple aspects of EGFR-TKI-related rash simultaneously. (2) Topical application. By applying the decoction directly to the affected area, we potentially achieved higher local concentrations of active compounds compared to systemic treatments. (3) Consistent application. The use of facial mask paper and standardized application time ensured consistent drug delivery, which may have contributed to the observed efficacy.
Comparing our results with similar studies, we found both similarities and differences. For instance, a study by Chen et al. 17 also reported positive effects of Chinese herbal medicine on EGFR-TKI-related rash. However, their study used oral administration, while ours focused on topical application. Our approach may offer advantages in terms of reduced systemic side effects and potentially higher local drug concentrations.
The reduction in WoMo scores observed in our study (mean difference of −10.3847 at week 2) is comparable to or better than those reported in studies using conventional treatments. In the EVITA trial, the WoMo part C score effectively detected changes during treatment intervention as a secondary endpoint. 35 Therefore, we used WoMo part B and C scores as the secondary endpoint to evaluate the change in rash severity. We found that both WoMo scores decreased during the treatment course. The WoMo scale may be a better method to evaluate efficacy, and further exploration is needed. Our study achieved a similar level of improvement using natural topical treatment. The improvement in quality-of-life measures (Skindex-16 and DLQI) in our study is particularly noteworthy. Previous studies have highlighted the significant impact of EGFR-TKI-related rash on patients’ quality of life. 9 Our results suggest that the XZZY decoction not only reduces rash severity but also meaningfully improves patients’ well-being.
We acknowledge several limitations in our study. The absence of a control group limits our ability to definitively attribute the observed improvements to the XZZY decoction. The single-arm design was chosen for several reasons. Firstly, as an initial exploratory study investigating the novel topical application of the XZZY formula for EGFR-TKI-related rash, this approach allowed for a preliminary assessment of safety and potential efficacy. Ethical considerations led us to provide all participants with potentially beneficial treatment. Resource constraints also influenced the study design. Furthermore, this approach provides valuable preliminary data to inform future larger-scale randomized controlled trials, including comparisons with established Western treatments. Additionally, the single-arm design may have introduced observational bias. Despite these limitations, our study provides valuable preliminary evidence for the efficacy of topical XZZY decoction in managing EGFR-TKI-related rash.
There are 2 limitations. Firstly, we don’t use the individualized treatment. While Traditional Chinese Medicine (TCM) emphasizes individualized treatment based on pattern differentiation, the development of standardized preparations is crucial for clinical application and research reproducibility. In this study, we focused on creating a fixed-composition topical formula targeting the primary TCM-identified pathogenesis of EGFR-TKI-related rash. This approach balances the core principles of TCM with the requirements of modern pharmaceutical standards. By concentrating on the predominant pattern associated with EGFR-TKI-related rash, we aimed to develop a broadly applicable yet TCM-principled treatment. This standardization not only facilitates consistent production and application but also lays the groundwork for future large-scale clinical trials and potential integration into conventional treatment protocols. Secondly, this is a single-arm observational study, which may lead to various biases such as selection bias and information bias, despite our efforts to minimize them. However, the results of this study are encouraging, and we will conduct a randomized controlled trial to further confirm the exact therapeutic efficacy of this formula.
In conclusion, our small-scale single-arm clinical trial demonstrates that topical XZZY decoction may improve EGFR-TKI-related rash, offering a promising alternative or complementary treatment to conventional approaches. These findings provide a solid foundation for future large-scale randomized controlled trials to further investigate the efficacy and safety of this treatment approach.
Supplemental Material
sj-docx-1-ict-10.1177_15347354251335598 – Supplemental material for External Application of a Xiaozhen zhiyang Decoction for Facial Rash Related to Epidermal Growth Factor Receptor Tyrosine Kinase Inhibitors in Chinese Patients: A Prospective Single-Arm Observational Study
Supplemental material, sj-docx-1-ict-10.1177_15347354251335598 for External Application of a Xiaozhen zhiyang Decoction for Facial Rash Related to Epidermal Growth Factor Receptor Tyrosine Kinase Inhibitors in Chinese Patients: A Prospective Single-Arm Observational Study by Jietao Lin, Lingling Sun, Jing Zhang, Ke Zhu, Hanrui Chen, Xinting Zhen, Yang Cao, Siyu Wang and Lizhu Lin in Integrative Cancer Therapies
Footnotes
Acknowledgements
The authors would like to thank all the staff members in our institution.
Author Contributions
JTL and LZL designed the study and wrote the manuscript. JZ, XTZ, and YC recruited the volunteers. KZ assessed the efficacy. LLS analyzed the data. SYW and LZL coordinated and designed the entire clinical study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grant support or other sources of funding:This work was supported by Guangzhou MunicipalScience and Technology project (2023A03J0300); Key R&D Plan of the Ministry of Science and Technology of China (2022YFC3500203); Guangdong Provincial Basic and Applied Basic Research Fund (2022B1515230003); The Project of Traditional Chinese Medicine and Western Medicine Clinical Collaboration for Major Difficult Diseases: Lung Cancer; National Administration of Traditional Chinese Medicine: 2019 Project of Building Evidence-Based Practice Capacity for TCM (No. ZXZL-4) [Project BEBPC-TCM (No. ZXZL-4)]; Guangzhou Science and Technology Plan project (No. 202102010488); Guangzhou Science and Technology Project (No. 202201020387).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Trial Registration Number
ChiCTR1900024461
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
