Abstract
Objective
To compare the therapeutic effects and adverse reactions of pemetrexed and docetaxel as salvage chemotherapy in patients with nonsmall-cell lung cancer (NSCLC) after the failure of epidermal growth factor receptor-tyrosine kinase inhibitors (EGFR-TKI).
Methods
In this randomized Phase 2 trial, patients with NSCLC who had previously failed EGFR-TKI therapy were randomized to receive intravenous pemetrexed (500 mg/m2 for 21 days [one cycle]) or docetaxel (75 mg/m2 for 21 days [one cycle]). Therapeutic effects were evaluated according to Response Evaluation Criteria in Solid Tumours standards and adverse effects were evaluated according to the US National Cancer Institute Common Terminology Criteria for Adverse Events.
Results
There was no statistically significant difference in disease control rate, response rate, median survival and 1-year survival between treatment groups. Rates of nausea, myelosuppression, renal damage and hair loss were significantly higher in the docetaxel group than the pemetrexed group.
Conclusion
Pemetrexed is effective and well tolerated as salvage chemotherapy in patients with NSCLC after EGFR-TKI failure and may be a suitable therapeutic option in these patients.
Keywords
Introduction
Nonsmall-cell lung cancer (NSCLC) accounts for approximately 80% of all lung cancers. 1 Platinum-based dual-drug chemotherapy is widely used in clinical practice to treat NSCLC but rates of tumour resistance and recurrence are high, the response rate is only 20–40% and the median survival time is 8–10 months.2–4 The use of conventional drugs as second- or third-line chemotherapy can improve survival quality and extend progression-free survival (PFS) in advanced NSCLC.5,6 Drug resistance has been observed in patients with NSCLC treated with epidermal growth factor-tyrosine kinase inhibitors (EGFR-TKIs), 7 and the optimum chemotherapeutic regimen in these patients is unclear. Docetaxel and pemetrexed are currently used in clinical practice as second-line chemotherapy in patients with NSCLC. 4
In order to provide a basis for their use in clinical practice, the aim of the present study was to compare the therapeutic effects and adverse reactions associated with pemetrexed and docetaxel as salvage chemotherapy in patients with NSCLC in whom EGFR-TKIs had failed to work.
Patients and methods
Study population
Patients with NSCLC treated at The First Affiliated Hospital of Zhengzhou University, Zhengzhou, Henan, China between January 2008 and January 2012 were randomized to receive pemetrexed or docetaxel according to a computer-generated randomization schedule. The study was a randomized Phase 2 trial. The primary endpoint was survival and the secondary endpoint was response rate. Inclusion criteria were: (i) a diagnosis of NSCLC, as determined by exfoliative cytology, percutaneous lung biopsy or bronchofiberscope biopsy, consistent with the World Health Organization 2011 histological classification; 8 (ii) EGFR-TKI failure (defined as evidence of disease progression while on continuous EGFR-TKI therapy); (iii) age ≥ 18 years; (iv) Eastern Co-operative Oncology Group score <3; (v) no evidence of severe hepatic and renal dysfunction; (vi) grade III or IV carcinoma; (vii) adenocarcinoma; (viii) presence of a measurable lesion. Patients were excluded from study participation if they had a history of pemetrexed or docetaxel treatment, or if they had a contraindication for treatment with either agent.
Prior to drug treatment, all patients underwent blood analyses, assessment of hepatic and renal function, an electrocardiogram and chest computed tomography (CT) scan. Patients underwent a chest CT every two cycles of treatment during the study.
The study protocol was approved by the Ethics Committee of The First Affiliated Hospital of Zhengzhou University and all enrolled patients gave written informed consent to participate. The clinical trial registration number is chinadrugtrials.ord.cn CTR20100054.
Salvage chemotherapy regimens
Patients in the pemetrexed group received 500 mg/m2 pemetrexed intravenously once daily over a 60–70 min time-period for 21 days (one cycle of treatment). In addition, patients received: (i) 0.4 mg/day folic acid from 1 week before pemetrexed administration to 3 weeks after the end of treatment; (ii) 1 mg vitamin B12 intramuscularly every three cycles of treatment; (iii) 4 mg dexamethasone twice daily for 3 days from 1 day before pemetrexed administration. Patients in the docetaxel group received 75 mg/m2 docetaxel intravenously once daily for 21 days (one cycle of treatment). They also received 7.5–8 mg dexamethasone twice daily for 3 days from 1 day before docetaxel administration. The time interval between cycles of pemetrexed or docetaxel was 21 days. During treatment patients were carefully observed for any evidence of bone pain or myelosupppression (routine blood test performed once per week) and, if necessary, analgesics were used for pain relief (non-steroidal anti-inflammatory drugs). In the event of grade III or IV myelosuppression, the drug dose was decreased by 20%. They also completed questionnaires 9 to collect data on adverse reactions before each cycle of chemotherapy.
Evaluation of treatment response
The therapeutic effects of pemetrexed or docetaxel were evaluated according to Response Evaluation Criteria In Solid Tumours (RECIST) standards every two cycles of treatment. 10 Complete response (CR) was defined as the complete disappearance of all lesions lasting more than 4 weeks. Partial response (PR) was defined as a reduction in the size of all lesions of more than 30% lasting at least 4 weeks. Stable disease (SD) was defined as a reduction in lesion size of up to 30% or by no increase in lesion size and no new lesions. Progressive disease (PD) was defined as an increase in lesion size of ≥20% or the appearance of new lesions. Response rate (RR) was calculated as the sum of CR and PR and disease control rate (DCR) was calculated as the sum of CR, PR and SD. The median survival time was defined as the interval between the start of treatment and the date of death. Adverse effects of treatment were evaluated according to the US National Cancer Institute Common Terminology Criteria for Adverse Events (version 4.0). 11 Patients were followed-up for 2 years after treatment; all patients had died before the end of the follow-up.
Statistical analyses
Statistical analyses were performed with SPSS® statistical software, version 19.0 (SPSS, Inc. Chicago, IL, USA) for Windows®. Categorical data were expressed as a percentage and between-group comparisons were performed by χ2-test. Continuous data were expressed as mean ± SD and between-group comparisons were performed by Student’s t-test. Statistical significance was established at P < 0.05.
Results
Clinical characteristics of patients with nonsmall-cell lung cancer who had failed previous treatment with epidermal growth factor receptor-tyrosine kinase inhibitors (EGFR-TKIs).
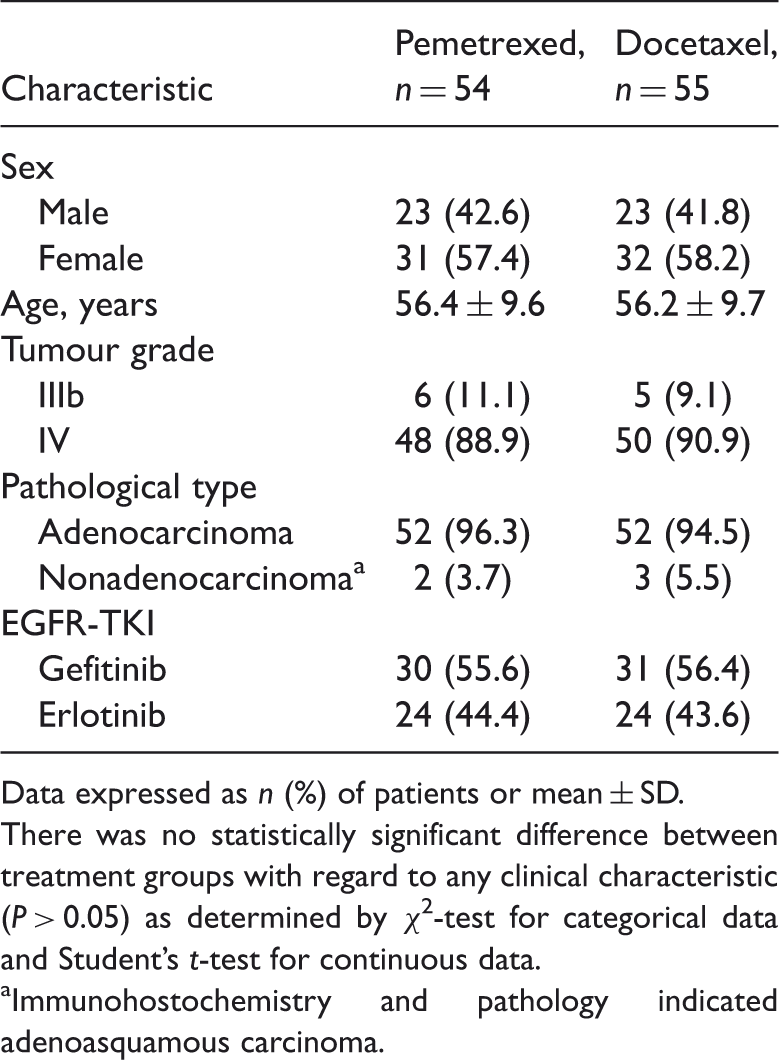
Data expressed as n (%) of patients or mean ± SD.
There was no statistically significant difference between treatment groups with regard to any clinical characteristic (P > 0.05) as determined by χ2-test for categorical data and Student’s t-test for continuous data.
Immunohostochemistry and pathology indicated adenoasquamous carcinoma.
Comparison of response rates after treatment with pemetrexed or docetaxel in patients with nonsmall-cell lung cancer who had failed previous treatment with epidermal growth factor receptor-tyrosine kinase inhibitors.

Data are expressed as n (%) of patients.
There was no statistically significant difference between treatment groups with regard to any response parameter (P > 0.05) as determined by Student’s t-test.
The median survival time was 8.5 ± 1.1 months and 8.4 ± 1.1 months in the pemetrexed and docetaxel groups, respectively, and the corresponding 1-year survival rates were 25.9% (14/54) and 25.5% (14/55). There was no statistically significant difference between treatment groups with regard to median survival time or 1-year survival rate.
Adverse events graded by severity following treatment with pemetrexed or docetaxel in patients with nonsmall-cell lung cancer who had failed previous treatment with epidermal growth factor receptor-tyrosine kinase inhibitors.

Data are presented as n of patients.
Adverse event were graded according to the US National Cancer Institute Common Terminology Criteria for Adverse Events. 11
Statistically significant difference between treatment groups (P < 0.05) as determined by Student’s t-test.
Discussion
The pathogenesis of NSCLC, a common glandular epithelial cancer, is unclear, although it appears to be strongly associated with long-term smoking, air pollution, vocational factors, chronic lung diseases, familial inheritance and endocrine disorders. 12 Early symptoms of NSCLC include mainly extra-pulmonary manifestations involving the joints, such as clubbed finger or hypertrophic pulmonary osteoarthropathy; later symptoms include cough, dyspnoea, weight loss and chest pain. 13 It has been shown that conventional second- or third-line chemotherapy can improve survival quality and extend PFS in patients with NSCLC,5,6 but elderly patients in particular have a low tolerance for the adverse effects associated with treatment as their organ reserve capacity is low. 14 Selection of an efficacious and well tolerated chemotherapy regimen is, therefore, especially challenging in elderly patients with NSCLC. 15
Advanced NSCLC is commonly treated with pemetrexed and docetaxel. Docetaxel is unique among the taxane anticancer drugs in that it is administered intravenously. It promotes tubulin polymerization and inhibits microtubule depolymerization such that nonfunctional microtubules are formed, which subsequently restrict tumour cell mitosis.16–18 Research has suggested that docetaxel is more effective in the treatment of cancer than paclitaxel: the concentration of docetaxel has been shown to be four times higher than that of paclitaxel in somatocytes and its affinity for microtubules is twice that of paclitaxel.19,20 In addition, docetaxel has been found to persist in tumour cells longer than paclitaxel. 21 Docetaxel is used as second-line therapy in anthracycline-resistant breast cancer 22 and has been used alone or in combination with other drugs in chemotherapeutic regimens for NSCLC, prolonging patient survival. 23 The pharmacokinetic profile of docetaxel is consistent with a three-compartment model and faecal excretion is the main route of elimination. 24 Docetaxel exhibits a plasma protein binding ratio of more than 95% and cytochrome P540 3A4 (CYP3A4) is involved in its metabolism, meaning that docetaxel is suitable as salvage treatment after failure of cisplatin chemotherapy. 25
Pemetrexed is a multi-target antifolic acid preparation containing pyrrole pyrimidine that can inhibit key enzymes in the folate-dependent metabolic pathways thereby affecting tumour growth.26,27 It inhibits the activities of dihydrofolate synthetase, thymidylate synthetase and glycinamide ribonucleotide formyltransferase. In addition, it transports folate carrier and the cell membrane-folate binding protein transport system into cells to synthesize glutamic acid, which inhibits enzyme activity to prevent tumour growth. 28 Pemetrexed is eliminated from the body via the urine with a creatinine clearance rate of 90 ml/min. 29 The plasma protein binding ratio of pemetrexed is approximately 81%, which is independent of renal function. 30 After achieving effective maintenance therapy, pemetrexed has been shown to extend overall survival, especially in NSCLC. 31
The present study indicated that there was no statistically significant difference in DCR, RR, median survival time and 1-year survival rate between pemetrexed and docetaxel in patients with NSCLC who had previously failed EGFR-TKIs. Rates of nausea, myelosuppression, renal damage and hair loss were, however, significantly higher in patients treated with docetaxel compared with pemetrexed. These findings suggest that the therapeutic effect of pemetrexed is similar to that of docetaxel in patients with NSCLC after EGFR-TKI failure, but that pemetrexed may be better tolerated than docetaxel.
This study has several limitations. The number of participants was small, the median follow-up time was only 1 year, and there was no analysis of subgroups according to EGFR or ALK markers. These factors should addressed in further studies. The optimum salvage chemotherapy regimen for NCLC after TKI failure remains to be further investigated.
In conclusion, the present study demonstrated the pemetrexed was effective as salvage chemotherapy in patients with NSCLC after EGFR-TKI failure and that it was associated with a lower incidence of adverse reactions than docetaxel. These data indicate that pemetrexed may be a suitable chemotherapeutic option for patients with NSCLC after EGFR-TKI failure.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
