Abstract
Screening for pulmonary nodules (PN) using low-dose CT has proven effective in reducing lung cancer (LC) mortality. However, current treatments relying on follow-up and surgical excision fail to fully address clinical needs. Pathological angiogenesis plays a pivotal role in supplying oxygen necessary for the progression of PN to LC. The interplay between hypoxia and angiogenesis establishes a vicious cycle, rendering anti-angiogenesis therapy alone insufficient to prevent PN to LC transformation. In traditional Chinese medicine (TCM), PN is referred to as “Feiji,” which is mainly attributed to Qi and blood deficiency, correspondingly, the most commonly prescribed medicines are Astragalus membranaceus (Fisch.) Bge. var. mongholicus (Bge.) Hsiao (huang qi) (AR) and Angelica sinensis (Oliv.) Diels (dang gui) (ARS). Modern pharmacological studies have demonstrated that AR and ARS possess immune-enhancing, anti-tumor, anti-inflammatory, and anti-angiogenic properties. However, the precise mechanisms through which AR and ARS exert anti-angiogenic effects to delay PN progression to LC remain inadequately understood. This review explores the critical roles of hypoxia and angiogenesis in the transition from PN to LC. It emphasizes that, compared to therapies targeting angiogenic growth factors alone, AR, ARS, and their compound-based prescriptions offer additional benefits. These include ameliorating hypoxia by restoring blood composition, enhancing vascular structure, accelerating circulation, promoting vascular normalization, and blocking or inhibiting various pro-angiogenic expressions and receptor interactions. Collectively, these actions inhibit angiogenesis and delay the PN-to-LC transformation. Finally, this review summarizes recent advancements in related research, identifies existing limitations and gaps in knowledge, and proposes potential strategies and recommendations to address these challenges.
Introduction
Lung cancer (LC) is a severe malignancy originating in the bronchial mucosa or lung glands. LC accounts for approximately 2 million fatalities annually worldwide, with both incidence and mortality rates continuing to rise.1,2 Early diagnosis and intervention are critical for improving cure rates, reducing mortality, and lowering treatment costs.3 -5 Pulmonary nodules (PN), specific precursors of LC, are defined radiologically as localized, round, denser solid, or subsolid shadows ≤30 mm in diameter, without associated lung collapse, mediastinal lymph node enlargement, or pleural effusion. 6 The 2022 National Comprehensive Cancer Network (NCCN) guidelines classify PN based on their number, diameter, and density. 7 However, The NCCN further emphasizes that the risk of malignancy increases with nodule diameter and number. 8 With the widespread adoption of low-dose computed tomography (LDCT), the detection rate of PN has dramatically increased, with millions of new cases identified annually.
Pathological angiogenesis plays a central role in the progression of PN to LC. 9 In the early stages of PN, new vasculature is generally absent. However, when PN is switched on to LC, the blood and oxygen supply needs to be increased through additional vessels. 10 While PN enlargement may not immediately result in pathological changes, elevated pro-angiogenic factors and microvascular density strongly indicate angiogenesis. 11 Therefore, vascular convergence or extramural vascular invasion, as seen on LDCT, is often a consequence of pathological angiogenesis. 12 Since the concept of targeting angiogenesis to “starve” tumors was introduced in the last century, anti-angiogenic therapies have become a significant focus of cancer treatment. 13 But, anti-angiogenic drugs, which target angiogenic growth factors, do not adequately meet clinical needs. This is because on the one hand, anti-angiogenic drugs have obvious side effects, such as pulmonary hypertension, proteinuria, hypertension.14,15 On the other hand, angiogenesis plays a protective role in myocardial infarction and cerebral infarction. 16 In addition, the high price also hinders the long-term use of PN patients. Consequently, current strategies for PN management rely on long-term monitoring and, if necessary, surgical, ablative, or radiotherapeutic interventions to remove nodulesv.17 -19 Unfortunately, these approaches fail to prevent angiogenesis or reduce microvessel density, and in some cases, the removal of localized lesions may even trigger the formation of new nodules.
Traditional Chinese Medicine (TCM) has demonstrated effectiveness in reducing nodule diameter, nodule number, and improving density.20,21 According to TCM, PN, known as “Feiji,” is attributed to Qi and blood deficiency combined with external stimuli. 22 The expert consensus on the diagnosis and treatment of pulmonary multiple ground-glass nodules by integrated traditional Chinese and Western medicine (2020) states that Qi and blood deficiency is a central factor in the development of PN and that patients with PN should be prescribed Astragalus membranaceus (Fisch.) Bge. var. mongholicus (Bge.) Hsiao (huang qi) (黄芪) (AR) and Angelica sinensis (Oliv.) Diels (dang gui) (当归) (ARS)-based herbal medicines to avoid deterioration.23 -25 In addition, physicians can also add medications based on the specific symptoms of patients.26 -28 Figure 1 illustrates the recognition of PN diseases in TCM. Figure 2 labels the origins, plants, and sections of the AR and ARS. It is encouraging to note that a considerable body of clinical research has demonstrated the efficacy of prescriptions based on AR and ARS in the prevention of PN development.29,30 Furthermore, these prescriptions have been proven to alleviate symptoms such as coughing, wheezing, fatigue, and insomnia in patients with PN.31 -33 As the most commonly employed and extensively studied natural medicine in TCM, AR, and ARS possess immune-enhancing, anti-tumor, anti-inflammatory, and antioxidant properties. AR and AR-based prescriptions have been utilized extensively in the treatment of cardiovascular diseases, diabetes, tumors, and stroke.34 -36 However, at present, no manuscript has summarized the mechanism of AR and ARS anti-angiogenesis delaying PN. Therefore, this paper aims to discuss the research progress of Chinese medicine AR and ARS in delaying the transformation of PN. By elaborating the key roles of hypoxia and angiogenesis in the process of PN development, it reveals the mechanism and potential of AR and ARS in tonifying Qi and blood to mitigate hypoxia and inhibit angiogenesis of PN. This provides the necessary theoretical support for the further development and utilization of TCM resources in the treatment of PN.

Qi and blood deficiency is the core reason for the emergence of PN. The depletion of Qi and blood in the body, often triggered by mood swings and external stimuli, serves as a prerequisite for disease development. TCM aims to restore the patient’s constitution by replenishing Qi and blood through the use of AR and AR-based formulas, thereby ameliorating lung symptoms and impeding PN-LC progression.

(A and B) show the plant information of AR and ARS. The images were sourced from the website: https://www.gbif.org; http://www.iplant.cn/frps. (C) Identification component stipulated in Pharmacopeia of the People’s Republic of China 2020, A for AR and B for ARS.
Vascular Invasion is the Key to PN Worsening
The progression of PN is critically dependent on the availability of sufficient blood and oxygen, with pathological angiogenesis serving as a pivotal driver in PN transformation. 37 Under normal physiological conditions, the body mitigates hypoxia by initiating new blood transport systems or by accelerating blood circulation, ceasing these processes once oxygen homeostasis is achieved. In contrast, the growth dynamics of PN result in a persistent and escalating demand for oxygen and blood flow. Chronic hypoxia, caused by an imbalance between oxygen supply and demand, and acute hypoxia, driven by fluctuations in blood flow, collectively contribute to PN growth under worsening hypoxic conditions (pO2 < 8 mmHg). 38 This sustained hypoxic state disrupts the body’s natural angiogenic regulation, triggering pathological neovascularization—a key factor in the rapid malignant transformation of PN.
The human circulatory system comprises a complex network of vessels, including arteries, veins, capillaries, and the aorta, responsible for transporting vital substances throughout the body. Capillaries, the smallest and most abundant vessels, consist of endothelial cells (EC), pericytes, and a basement membrane. These capillaries serve as the primary site for tissue-blood exchange and act as the origin of angiogenesis. 39 The formation of human vasculature occurs through 2 primary mechanisms: vasculogenesis and angiogenesis. Vasculogenesis, which predominantly takes place during embryogenesis, involves the direct differentiation of blood vessels from vascular EC. Angiogenesis, by contrast, entails the proliferation and migration of EC to form new vessels branching from existing capillaries or arteries. 40 Angiogenesis is categorized into several distinct types: (1) Sprouting angiogenesis: Stem cells follow migratory tip cells to form lumens, which connect to other vessels through anastomosis. 41 (2) Intussusceptive angiogenesis: Tissue pillars form within pre-existing vessels, fuse, and thicken until the vessel divides into 2. 42 (3) Vasculogenesis: Endothelial precursor cells induce the formation of embryonic blood vessels through differentiation and binding, representing a distinct mechanism of angiogenesis. 43 (4) Vascular mimicry: Tumor cells interact with the extracellular matrix, deforming to simulate vascular structures and forming microcirculatory channels. 44 (5) Differentiation of tumor stem cells: Tumor stem cells differentiate into EC, generating new blood vessels within the tumor microenvironment. 45
Under typical physiological conditions, angiogenesis is a localized process initiated in response to injury or hypoxia and terminates once tissue repair is achieved. However, in a hypoxic microenvironment, angiogenic factor expression is significantly upregulated, while the degradation of these factors is concurrently suppressed.46,47 Mature blood vessels are stable, flexible, and efficient; by contrast, pathological neovascularization leads to structurally immature and deficient vessels. 48 These pathological blood vessels cause several: (1) Aggravated blood flow disorders: Resulting in sustained or intermittent regional hypoxia. 49 (2) Increased vascular permeability and interstitial fluid pressure: Leading to edema and an elevated risk of bleeding. 50 (3) Impaired drug delivery: Dysfunctional vessels hinder the distribution of therapeutic agents, exacerbate local hypoxia, and trigger another cycle of angiogenesis. 51 The circulation established by these immature pathological vessels perpetuates oxygen deficiency, inducing a vicious cycle of hypoxia-driven angiogenesis. This cycle results in a rapid increase in microvascular density at the nodule site, which manifests as vascular invasion—a hallmark feature observable on LDCT imaging. Figure 3 illustrates the vascular changes during the PN-LC process.
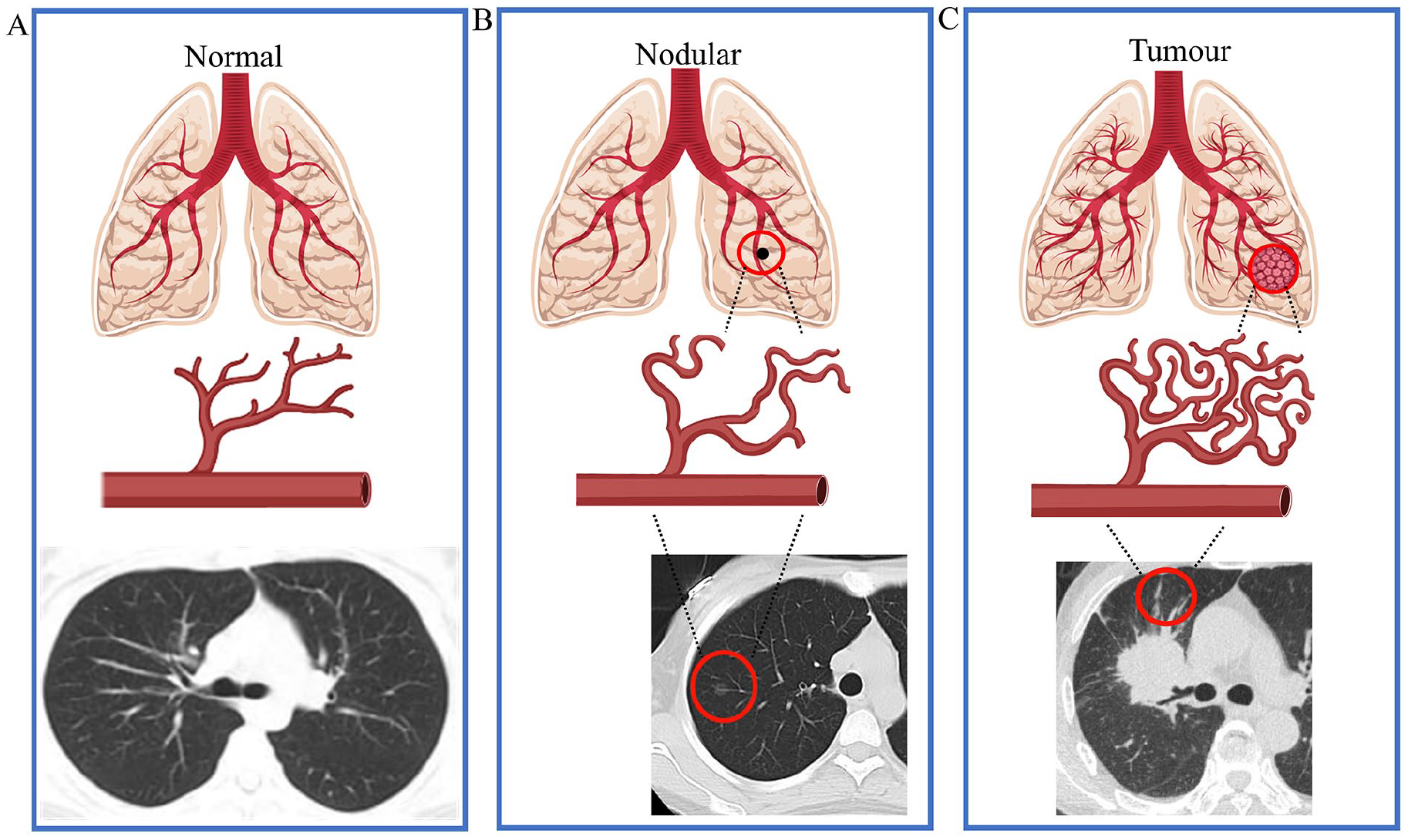
Angiogenesis provides nutrition and oxygen for the growth of PN, and local microvessel density increases with the development of PN. Under hypoxic conditions, diseased tissue necessitates increased vascularization to support rapid growth and oxygen supply. Continuous angiogenesis, orchestrated by pro-vascular growth factors, facilitates the transformation of nodules into malignant tumors. Panel A depicts lung tissue in a normal state with vascular and CT effects, Panel B represents PN, and Panel C portrays LC. CT images were obtained from Google Images.
Vascular Invasion Results From the Interplay of Various Pro-angiogenic Growth Factors
The process of angiogenesis is regulated by a variety of vascular growth factors that are induced by hypoxia. Under physiological conditions, elevated concentrations of angiogenic growth factors are triggered by tissue hypoxia, which stimulates the formation of new blood vessels. Once local blood flow and oxygen circulation are restored, and tissue oxygenation improves, these growth factors are rapidly degraded to maintain homeostasis. 52 But, the progressive transformation of PN results in a persistent and progressive lag of oxygen supply behind consumption. This results in the sustained deterioration of the hypoxic environment and the persistent activation of angiogenic growth factors. 53
Hypoxia-inducible factors (HIFs) are heterodimeric transcription factors consisting of an oxygen-sensitive α subunit and a constitutively expressed β subunit. The 3 known α subunits—HIF-1α, HIF-2α, and HIF-3α, with HIF-1α being the most important for regulating oxygen metabolism. Under normoxic conditions, the α subunit undergoes hydroxylation by HIF hydroxylases, followed by ubiquitination and degradation, maintaining HIF-1α levels at very low concentrations in healthy tissues. Conversely, during hypoxia, this hydroxylation-ubiquitination pathway is disrupted, leading to a rapid increase in HIF-1α levels. A significant elevation in HIF-1α can activate various pro-angiogenic factors. 54 These factors include Vascular Endothelial Growth Factor (VEGF), Fibroblast Growth Factor (FGF), Platelet-Derived Growth Factor (PDGF), and Angiopoietins (Ang). These molecules, along with their receptors, play critical roles in different stages of angiogenesis. The role played by pro-angiogenic factors in the mechanism of angiogenesis is shown in Figure 4.

AR and ARS inhibit angiogenesis through multiple pathways. In oxygen-deficient environments, the excessive binding of VEGF, PDGF, FGF, Ang, and their respective receptors triggers the proliferation, migration, invasion, and permeability of blood vessels. AR and ARS counteract angiogenesis by enhancing vascular structure repair, optimizing blood composition, and inhibiting the expression of pro-vascular growth factors and receptors through various mechanisms. Figure 4 was created using Figdraw.
The VEGF family consists of 5 members: VEGF-A to VEGF-D and Placenta Growth Factor, with VEGF-A being the most well-characterized. VEGF is known for promoting vascular permeability and extracellular matrix degradation, and is also referred to as a vascular permeability factor. 55 Produced by tumor cells and the extracellular matrix, VEGF enhances the density of new blood vessels by inducing EC proliferation and migration, thereby supporting tumor growth, proliferation, invasion, and recurrence. 56 VEGF promotes angiogenesis through several mechanisms. By binding to VEGF receptor 2 (VEGFR-2), VEGF activates extracellular signal-regulated kinase (ERK) and phosphatidylinositol 3-kinase (PI3K)/protein kinase B (Akt) pathways, leading to sustained EC proliferation and migration. 57 Additionally, VEGF can induce inflammation via 2 pathways: Plcγ/calcineurin/nuclear factor of activated T cells and nuclear factor kappa B, both downstream of Akt, thereby recruiting leukocytes and facilitating angiogenesis. 58 Notably, VEGF increases vascular permeability through junction remodeling and fenestrae formation, which can lead to leakage, elevated interstitial fluid pressure, and extravasation of cancer cells. 59
PDGF, initially identified in platelets, can be released upon platelet degranulation and synthesized by various cells in response to tissue injury. It is also secreted by hepatic stellate cells and inflammatory cells following liver injury. The PDGF family consists of 4 members: PDGF-A to PDGF-D. 60 PDGF promotes pericyte recruitment and vascular maturation by upregulating VEGF and binding to its 2 tyrosine kinase receptors, PDGF receptor-α (PDGFR-α) and PDGF receptor-β (PDGFR-β). 61 In the tumor microenvironment, PDGF, along with PDGFR, promotes EC proliferation, migration, sprouting, and lumen formation via autocrine and paracrine signaling. 62
The FGF family comprises 22 members, from FGF-1 to FGF-22. Among them, acidic FGF (aFGF) and basic FGF (bFGF), also known as FGF-1 and FGF-2, respectively, with FGF-2 being the most potent angiogenic factor. 63 When bound to fibroblast growth factor receptor (FGFR), FGF-2 directly stimulates EC proliferation and migration. 64 Additionally, FGF contributes to the induction of Matrix Metalloproteinase-9 (MMP-9), Plasminogen Activator, and Collagenase secretion in collaboration with VEGF. 65 Interestingly, during anti-VEGF therapy, tumor cells activate FGF signaling to promote angiogenesis. Inhibiting FGF not only restores tumor sensitivity to anti-VEGF drugs but also effectively inhibits angiogenesis. 66
The Ang family consists of 4 members, Ang-1 to Ang-4, which are crucial in regulating vascular development, stability, and remodeling. Ang-1 and Ang-2 are particularly important for angiogenesis, transmitting signals through endothelial tyrosine kinase receptors. Binding to Tyrosine Kinase Receptor 2 (Tie2), Ang-1 enhances EC activity via the Akt/Survivin pathway, promoting vascular maturation and stability. 67 In contrast, overexpression of Ang-2 leads to unstable, immature blood vessels with increased permeability. Ang-2 is synthesized and secreted by endothelial cells at sites of vascular remodeling in response to hypoxia, inflammation, and other angiogenic signals. It is highly expressed in the tumor inflammatory microenvironment, especially in tumors with high VEGF expression. 68 This reduces the efficacy of anti-VEGF therapies and contributes to resistance. Therefore, dual inhibition of Ang-2 and VEGFR-2 demonstrates enhanced anti-angiogenic effects in various cancer treatments. 69
Various factors within the human body contribute to angiogenesis by influencing the expression of pro-angiogenic factors. For example, the MMP family plays a significant role in breaking down extracellular matrix components, thus dismantling barriers to tumor invasion and migration. Notably, MMP-2 and MMP-9 can simultaneously upregulate the expression of VEGF and PDGF by activating TGF-β, thereby promoting vascular regeneration.70,71 Additionally, tumor necrosis factor-alpha (TNF-α), an inflammatory cytokine released by macrophages, mast cells, and T lymphocytes, contributes to angiogenesis by upregulating the expression of VEGFR-2. 72 Furthermore, human macrophages, neutrophils, and lymphocytes indirectly regulate pro-angiogenic factors, thus participating in angiogenesis. 73
It has been over 50 years since Judah Folkman proposed the concept of anti-angiogenic therapy. Currently, anti-angiogenic drugs are categorized into 2 main types: large molecular antibodies, such as bevacizumab, which primarily target VEGF/VEGFR-2; and small molecule inhibitors, such as anlotinib, which have multiple targets, including dual inhibition of VEGFR-2/3 and PDGFR/FGFR.74,75 Despite these advancements, the clinical application of anti-angiogenic drugs still faces challenges, including elevated recurrence rates, unaltered overall survival rates, drug resistance, and limited therapeutic scope.76,77 These challenges may arise from the inability of inhibitors targeting angiogenic factors to modify the hypoxic environment, which allows for accelerated tumor growth and evasion of immune surveillance. 78 The exclusive reliance on anti-angiogenic drugs may have adverse effects on physiological functions, diminishing their benefits. Studies have shown that angiogenic growth factors play diverse protective roles in maintaining normal bodily functions and in repairing diseased tissue. For instance, increased VEGF expression during ischemic stroke has been shown to protect neural networks in the brain. 79 From a broader perspective, the long-term use of anti-angiogenic drugs may not represent a beneficial strategy for PN. Similar to the challenges encountered in treating various types of tumors, drugs targeting specific angiogenic factors may not effectively prevent or treat complex diseases that alter the surrounding microenvironment. 80 It is promising, however, that the combination of AR and ARS has been shown to exert an inhibitory effect on angiogenesis, offering potential therapeutic applications for PN.81 -83
In conclusion, a more effective approach to delaying vascular invasion in PN may involve combining various effective drugs with low side effects and low cost to improve overall body function, addressing both hypoxia and angiogenesis. TCM natural medicines, such as AR and ARS, have shown fewer side effects, lower costs, and ease of use. Long-term clinical administration (up to 60 days) of these drugs has demonstrated effective inhibition of angiogenesis without significant toxicity. 84 Interestingly, AR and ARS have also been shown to promote angiogenesis in conditions like stroke, myocardial infarction, and diabetes.85,86 Given that indiscriminate use of angiogenesis inhibitors may accelerate the progression of normal tissues to malignancy, the unique dual regulatory effect of AR and ARS could be key to preventing PN. 87
Effect and Mechanism of AR and ARS in Ameliorating Hypoxia and Inhibiting Angiogenesis
Improvement in Hypoxia
Pathological angiogenesis begins with hypoxia induced, which leads to the subsequent upregulation of HIF-1α. Although studies have demonstrated the inhibitory effects of both AR and ARS on HIF-1α, the sole inhibition of HIF-1α does not fully elucidate the therapeutic mechanisms of these drugs.88,89 The integration of AR and ARS may aim to enhance spontaneity by modifying blood composition, restoring blood flow, alleviating hypoxia, enhancing immune function, and inhibiting inflammatory factors. This method of regulating the physiological functions of the human body is frequently referred to as “invigorating Qi and producing blood” in TCM.90,91 Figure 5 illustrates the potential mechanism of action of AR and ARS in alleviating hypoxia and inhibiting pro-vascular growth factors.

Schematic representation depicts the potential mechanisms by which AR and ARS may alleviate hypoxia and inhibit angiogenic factors.
Repair blood composition
Chronic hypoxia can lead to immune dysfunction, damaging immune cells in the blood and inducing chronic inflammation. A previous study reported that the combination of AR + ARS administered to mice at a dose of 12 g/kg for 8 weeks effectively improved the number of T cells, CD3+, CD4+, and CD8+, and adjusted the CD4+/CD8+ ratio. 92 Additionally, T cells exert an immunosuppressive effect by secreting cytokines such as TGF-β, IL-4, IL-6, IL-10, IL-35, and Th17, inhibiting and regulating the proliferation and activation of effector T cells, thereby slowing pathological immune responses and reducing vascular inflammatory injury. 93 The combination of AR and ARS (100/200 mg/kg), administered for 4 weeks, has been observed to modulate the Th17/Treg cell balance in a concentration-dependent manner while inhibiting the expression of vascular inflammatory factors. 94 Furthermore, the combination of AR and ARS has shown potential to inhibit vascular inflammation by modulating the CD4+/CD8+ ratio and reducing the production of IL-4, IL-6, and IL-10 in clinical therapy. 95 RBCs are the primary means of oxygen transport in the blood. Chronic inflammation results in the increased consumption of RBCs and hemoglobin (Hb). A relative deficiency in RBCs inevitably leads to the upregulation of HIF-1α expression. The dynamic regulatory interaction between HIF-1α and RBCs has been observed in various pathological conditions.96,97 Clinical studies have shown that the combination of AR and ARS stimulates bone marrow hematopoiesis, resulting in an increase in red blood cell production in patients. Encouragingly, multiple studies suggest that this pro-hematopoietic effect can be utilized to address a range of pulmonary disorders and alleviate symptoms associated with RBC deficiency.98 -100 Similarly, animal studies have demonstrated that AR and ARS (3.6 and 10 g/kg, 14d) promote hematopoietic function in a concentration-dependent manner, as well as enhance the recovery of physical function.101 -103 Importantly, both clinical and animal studies have shown that long-term use of AR and ARS does not result in significant adverse effects.
Promoting vascular structure normalization
Pathological studies have confirmed that the triad of hypoxia, vascular inflammation, and abnormalities in vascular structure tends to catalyze a vicious cycle. 104 AR and ARS can further ameliorate hypoxia and inhibit angiogenesis by promoting vascular normalization and improving the efficiency of oxygen delivery. Hypoxia-induced increases in HIF-1α levels lead to elevated VEGF expression, which increases vascular permeability, resulting in vascular damage, edema, and leakage in the pathological state.105,106 Additionally, MMP-9, another factor involved in vascular permeability, shows increased expression alongside HIF-1α. 107 The combined action of VEGF and MMP-9 leads to the destruction of the vascular basal membrane. These minor vascular injuries not only mark the onset of pathological blood vessels but also represent common sites of vascular inflammation. MMP-9-induced vascular damage is a significant contributing factor to tumor metastasis. 108 Thus, inhibiting MMP-9 expression is important for promoting vascular repair.109,110 The expression of MMP-9 typically increases in the presence of elevated inflammatory factors, which is why it is considered an inflammatory mediator. Notably, the combination of AR and ARS (9.36 g/kg) can inhibit the expression of MMP-9 as well as the levels of IL-6 and IL-10. 111
Normalization of pathological blood vessels, which involves restoring vascular morphology and function, and further repairing the immune microenvironment by balancing pro-angiogenic and anti-angiogenic processes, has proven to be an effective method for treating various chronic and complex diseases. 112 A substantial body of research indicates that AR and ARS facilitate angiogenesis and promote skin repair in experimental animals via the PI3K/AKT/VEGF pathway. 113 Promoting VEGFR-2 expression induces an increase in microvascular density, thereby protecting cardiac function following myocardial infarction. 114 Nevertheless, it has been shown that the combination of AR and ARS can inhibit the proliferation and migration of vascular smooth muscle cells, thus preventing tumor growth and migration by inhibiting angiogenesis.115,116
These seemingly contradictory results highlight the dual regulatory role of AR and ARS in angiogenesis under different conditions. This may be because the combination of AR and ARS does not merely inhibit angiogenic growth factors, but also promotes vascular normalization by stimulating the body’s intrinsic hypoxia response mechanism. This process alleviates hypoxia by inhibiting inflammation, improving the immune environment, and increasing blood supply efficiency, thereby promoting the cessation of angiogenesis and the healing of vascular damage. It is possible that this process involves competition between VEGFR-1 and VEGFR-2, as well as angiogenesis inhibition via the Notch signaling ligand Dll-1. However, there remains a lack of relevant studies to further elucidate the therapeutic mechanisms of AR and ARS.117,118
Accelerating blood circulation
Blood flow shear stress (FSS) is the force per unit area exerted on the vessel wall in the direction of blood flow. The combination of AR and ARS regulates FSS to alleviate hypoxia. In pathological angiogenesis, blood vessels often lack normal microvascular regulatory mechanisms, including the abnormal expression of endothelial growth factors such as VEGF and PDGF, which disrupt vascular smooth muscle structure and vascular tension. 119 Furthermore, hypoxia-induced overexpression of angiogenic factors such as PDGF disrupts the coagulation-fibrinolysis system, resulting in a hypercoagulable state that impedes blood flow and exacerbates hypoxia, significantly contributing to increased whole-blood viscosity, plasma viscosity, and fibrinogen levels in tumor patients.120 -122 Therefore, regulating vascular tension to accelerate blood flow is crucial to alleviating acute hypoxia. Encouragingly, the combination of AR and ARS has been shown to improve intimal hyperplasia, repair lumen structure, and enhance blood flow velocity. 123 Our team found that the modulatory effects of AR and ARS on blood flow were not evenly distributed but selectively controlled. For example, while both drugs accelerated blood flow, AR had a stronger effect on cerebral blood flow, whereas ARS was more effective in accelerating renal artery blood flow. 124 To understand the underlying causes of this phenomenon, another study suggests that the impact of AR and ARS on blood flow velocity may be linked to vasodilatory processes and a reduction in blood resistance. 125 This effect may also be associated with improved lipid metabolism, reduced blood viscosity, and enhanced peripheral blood EPC function. 126
The optimal functioning of the blood transport system depends on maintaining healthy blood, intact blood vessels, and a moderate blood flow rate. By enhancing these 3 factors, AR and ARS can effectively improve the hypoxic microenvironment and terminate the vicious cycle of hypoxia/angiogenesis. However, given the varying regulatory effects of AR and ARS on different blood vessels, more direct and detailed evidence is needed to achieve more convincing outcomes. High-resolution scanning of lesion sites and immunofluorescence techniques can be employed to analyze and demonstrate alterations at the lesion site before and after treatment. Additionally, the small animal LDCT technique has been used to observe the pathological process in experimental animals.127,128
Inhibiting Angiogenesis Factor
The processes of angiogenesis and hypoxia are closely intertwined in a cause-and-effect relationship. Under normal physiological conditions, angiogenesis in the human body is resolved once local tissue damage is repaired or oxygen deficiency is alleviated. However, during carcinogenesis induced by PN, excessive expression of pro-angiogenic factors driven by hypoxia leads to the transformation of the body’s repair mechanisms into facilitators of disease progression. This highlights the potential for further investigation into therapeutic strategies that combine the inhibition of pro-angiogenic factors with the improvement of hypoxia. The role of AR and ARS in alleviating hypoxia has been well-documented in the literature. Interestingly, by leveraging the multi-component nature of TCM, these compounds can exert anti-angiogenic effects by reducing microvascular density through the suppression of various angiogenic factors.
AR and ARS inhibit VEGF
The VEGF family plays a pivotal role in human angiogenesis, with VEGF being the most potent inducer of angiogenesis and a key regulator of vascular permeability. It is crucial that AR and ARS exert their anti-angiogenic effects by suppressing the over-expression of VEGF, thereby mitigating subsequent vascular damage and inflammation.129,130 In hypoxic conditions, VEGF triggers responses in EC by binding to its receptor, VEGFR-2. This results in increased vascular permeability, disruption of existing vessels, vascular inflammation, and the proliferation, migration, and invasion of EC. 131 The anti-angiogenic properties of AR and ARS were first reported in 200. 132 Subsequent research from our team revealed that these effects are mediated through the inhibition of the VEGF/VEGFR-2 pathway. 133 These effects extend beyond PN, as numerous studies have demonstrated that the therapeutic effects of AR and ARS on pulmonary fibrosis, diabetic retinopathy, and atherosclerosis are closely linked to their inhibition of VEGF. 134 Importantly, VEGF also plays a crucial role in regulating vascular permeability. 135 During pathological angiogenesis, the increase in vascular permeability not only renders blood vessels more fragile but also exposes them to chronic inflammatory infiltration, thereby accelerating tissue cancer progression. 136 Although some evidence suggests that various AR and ARS compounds, such as astragaloside, astragalus polysaccharide, mononin, angelica polysaccharide, and ferulic acid, may reduce vascular damage by inhibiting MMP-2 and MMP-9, further studies are needed to substantiate the efficacy of AR and ARS in inhibiting these matrix metalloproteinases.137 -139 Recent studies have reported that the inhibitory effects of AR and ARS on NF-κB, STAT3, and VEGF were significantly weaker in nude mice compared to C57BL/6 mice, suggesting that the anti-angiogenic effects of AR and ARS are not solely due to VEGF inhibition but may also involve the suppression of local inflammation and immune modulation. 140 The PI3K/Akt pathway, which is activated by receptor tyrosine kinases and linked to classic phosphatidylinositol signaling, has been shown to effectively inhibit tumor cell growth and promote apoptosis by blocking Akt phosphorylation. 141 VEGF-mediated modulation of the PI3K/Akt pathway may represent a key mechanism regulating endothelial cell proliferation and inflammatory responses. Studies have demonstrated that AR and ARS can exert anti-inflammatory effects and protect vascular structures via the PI3K/Akt pathway.142,143 Furthermore, the Ras/ERK pathway, known to activate transcription factors and enhance VEGF transcription and translation, plays a pivotal role in angiogenesis. 144 Fortunately, AR and ARS (3.6 g/kg, 14d) can inhibit angiogenesis by suppressing the activation of the Ras/ERK pathway. 145
It can be concluded that the combined use of AR and ARS has the potential to not only inhibit the VEGF-induced increase in microvessel density, but also to prevent the development of secondary inflammation and vascular structural damage. Moreover, the principal objective of AR and ARS appears to be the attainment of anti-angiogenic effects through the modulation of immune function and the alleviation of local inflammatory infiltration.
AR and ARS inhibit PDGF
AR and ARS have the potential to mitigate angiogenesis-related alterations in blood flow and reduce vascular inflammation by inhibiting PDGF. PDGF is stored in platelets as α-granules and is secreted by various cell types, including endothelial cells, epithelial cells, glial cells, and inflammatory cells. It promotes vascular maturation, recruits pericytes, and induces VEGF expression, thus enhancing angiogenesis through interaction with its receptors PDGFR-α and PDGFR-β. 146 Within the PDGF family, PDGF-AA induces STAT3 phosphorylation Rb inactivation, accelerating angiogenesis. 147 PDGF-CC mediates angiogenesis through cancer-associated fibroblasts, while PDGF-BB, one of the most studied factors, regulates endothelial cell proliferation and migration via downstream signaling pathways such as MAPK/ERK, PI3K/Akt, and JNK.148,149 Excitingly, AR and ARS have demonstrated anti-angiogenic effects by inhibiting PDGF production. 150 PDGF is primarily stored in platelets, and excessive accumulation of PDGF due to inflammatory factors can increase blood viscosity and reduce blood flow velocity. 151 The combination of AR and ARS has been shown to exert anti-PDGF effects. 152 Moreover, AR and ARS can inhibit PDGF expression, improving blood flow retardation. 153 NF-κB, a critical nuclear transcription factor in cellular inflammatory and immune responses, regulates the expression of PDGF-B by binding to the PDGF-B promoter response element. 154 Studies have shown that AR effectively inhibits NF-κB and PDGF-B-induced angiogenesis. 155 Unfortunately, while there is evidence suggesting that the active ingredients in ARS may inhibit NF-κB, compelling evidence to support ARS’s efficacy is still lacking.156,157 Some studies have suggested that the high expression of PDGF may be the cause of resistance to anti-angiogenic drugs. 158
The current evidence indicates that VEGF-induced angiogenesis primarily occurs in the endothelial cells of vessels, whereas PDGF-induced angiogenesis involves the participation of smooth muscle cells and fibroblasts. Although both factors are involved in angiogenesis, their specific mechanisms, receptor preferences, and induction sites differ. 79 Unfortunately, majority of studies did not differentiate between the anti-angiogenesis sites of AR and ARS. Furthermore, due to the structural similarity between PDGF and VEGF, the majority of studies only utilized PDGF as an alternative target of VEGF. However, elucidating the specific action sites and intricate mechanisms of drugs in the complex angiogenesis process can provide a deeper understanding of the pathogenesis of PDGF, which may be the focus of relevant research in the future.
AR and ARS inhibit FGF
AR and ARS have been shown to inhibit angiogenesis induced by high FGF expression, although detailed studies in this area remain sparse. FGF, a peptide secreted by the hypothalamus and pituitary, is known for its mitogenic properties and its influence on wound healing through both mitogenic effects and non-mitogenic hormone-like activities, earning it the nickname “wound hormone.” 159 FGF is secreted by vascular endothelial cells, stem cells, and damaged cardiomyocytes, regulating angiogenesis through the synergistic action of FGFR, heparan sulfate polysaccharides, and αvβ integrins. 160 FGF-2 (bFGF) is a key pro-angiogenic factor within the FGF family, modulating collagenase, protease, urokinase-type plasminogen activator, and integrins to degrade the vascular basement membrane. This degradation allows cells to invade the surrounding matrix, migrate, proliferate, and differentiate, ultimately forming new capillary networks. 161 There is evidence suggesting that both AR and ARS inhibit bFGF, with their combined effects being more pronounced.130,162 The FGFR receptor plays a pivotal role in FGF-mediated angiogenesis by phosphorylating itself and activating downstream pathways such as Src family kinases, PLCγ/DAG/PKC, Ras/Raf-MAPK, and PI3K/Akt.163,164 Importantly, FGF and VEGF collaborate to regulate MMP-9, and clinical findings suggest that inhibiting FGF can enhance sensitivity to VEGF inhibitors, highlighting the close relationship between these angiogenic factors.165,166 Although research has indicated that AR and ARS can inhibit angiogenesis induced by FGF and suggested a close association with VEGF, unfortunately, there is currently a lack of in-depth studies on this topic.
AR and ARS inhibit Ang
AR and ARS have been demonstrated to effectively inhibit pathological angiogenesis and vascular inflammation induced by elevated expressions of Tie-2 and Ang-2. Ang-2, primarily located in endothelial cells, is the member of the Ang family most closely associated with angiogenesis. It can be detected in tissues at an early stage of angiogenesis in solid tumors, even before the formation of pathological blood vessels is complete, suggesting that Ang-2 may be one of the earliest active pro-angiogenic growth factors. 167 Interestingly, another family member, Ang-1, binds to the Tie-2 receptor to promote vascular maturation and stability. Conversely, Ang-2 antagonizes Tie-2, resulting in vessel destabilization and facilitating angiogenesis. 168 AR and ARS (3 g/kg, 14d) exert their anti-angiogenic effects by inhibiting Ang-2-induced angiogenesis and by stabilizing blood vessels through modulation of the Tie-2/Ang-2 axis.169,170 Notably, Ang-2 is also considered a critical factor in regulating vascular permeability. Inhibiting Ang-2 to maintain vascular integrity represents a promising strategy for therapeutic intervention. 171 Similar to VEGF, FGF can induce vascular inflammation through the PI3K/Akt pathway. 172 Although the inhibitory effects of AR and ARS on VEGF-mediated inflammation via the PI3K/Akt pathway have been previously discussed, the specific similarities and differences in the mechanisms of Ang-2/VEGF-induced vascular inflammation by AR and ARS remain unexplored. It is also important to note that studies have suggested that high Ang-2 expression may reduce the efficacy of anti-VEGF therapies. While dual inhibition of Ang-2/VEGF may more effectively suppress angiogenesis, it could also lead to more severe vascular structural deficits, potentially hindering the effectiveness of other therapeutic drugs.173,174 This underscores the urgency and necessity of exploring effective natural anti-angiogenic compounds. The compounded use of anti-angiogenic drugs, while beneficial, also implies the acceptance of potential consequences.
The core of pathological angiogenesis lies in the hypoxia-induced abnormal overexpression of VEGF. Various vascular growth-promoting factors contribute to angiogenesis through VEGF, either directly or indirectly. Encouragingly, a growing body of clinical evidence has demonstrated the efficacy of multi-target anti-angiogenesis drugs in inhibiting angiogenesis. However, this evidence also indicates that anti-angiogenesis therapy alone can only delay, but not completely halt, the angiogenesis process. This does not diminish the importance of anti-angiogenesis therapy; rather, it underscores the need for a more holistic approach to pathological angiogenesis. TCM, with its multi-component, multi-target characteristics, holds great promise. Prior research has demonstrated that AR and ARS can not only effectively inhibit angiogenic growth factors, but also mitigate related complications in pathological angiogenesis, including vascular injury, inflammation, and immune suppression. One significant limitation in the majority of studies on AR and ARS is that they tend to focus on VEGF at a single time point, which limits our understanding of the entire angiogenesis process.175,176 A significant limitation of the majority of studies on AR and ARS is that they examine VEGF at a specific point in time, thereby failing to provide a comprehensive understanding of the entire process. While VEGF is undeniably crucial in pathological angiogenesis, studying other pro-angiogenic factors is essential to fully understand the disease development process. Particularly important, water decoction remains the most prevalent therapeutic modality for AR and ARS. However, the lack of standardization in drug sources and combinations continues to impede progress in this field. Integrating TCM components and targets through proteomics and metabolomics could enhance the efficacy of TCM, leveraging its multi-component, multi-target approach. The potential for advancement in these related fields is highly promising.
Compounds
The effects of AR and ARS in improving hypoxia and inhibiting angiogenesis have been previously described. In order to enhance the efficacy of the treatment and gain deeper insight into their mechanism of action, it is essential to elucidate the therapeutic effects of their principal components. Astragaloside IV (AS-IV) and Ferulic acid (Fa) represent the core components of AR and ARS as outlined in the Pharmacopeia of the People’s Republic of China 2020.177 -179 Additionally, they are currently regarded as the most pivotal therapeutic components. Consequently, the anti-angiogenesis mechanisms of these 2 components will be discussed in depth in the following sections.
Mechanism of neovascularisation inhibition by AS-IV
Normal vascular structure and function are essential for maintaining optimal blood flow. In contrast, pathological angiogenesis depletes blood components and reduces blood flow. 180 Previous studies have shown that 40 mg/kg AS-IV can improve pulmonary perfusion and cardiac ejection, related to the reduction of vascular resistance and the repair of vascular structures. 181 Additionally, inflammatory anemia, a common complication of pathological angiogenesis, can be indirectly ameliorated by AS-IV through improving iron content, ferritin, and transferrin expression in liver tissue. 182 AS-IV also has the function of accelerating bone marrow hematopoiesis. 183 Moreover, RBC and Hb levels can be increased through the MAPK and estrogen receptor signaling pathways. 184 By improving blood flow, AS-IV alleviates local hypoxia, as it has been shown to reduce vasospasm.185,186 Elevated expression of HIF-1α under hypoxic conditions induces pathological angiogenesis, and 10 mg/kg AS-IV impedes this process by targeting the HIF-1α/VEGF pathway.187 -189 Furthermore, during hypoxia, the imbalance between oxygen demand and supply results in anaerobic metabolism and an increase in lactic acid accumulation. This metabolic shift is associated with elevated expression of monocarboxylate transporters (MCTs), particularly MCT-1 and MCT-4, mediated by HIF-1α. 190 Interestingly, 50 mg/kg AS-IV inhibits the expression of HIF-1α, MCT-1, and MCT-4, thereby reducing abnormalities in lactic acid metabolism. 191 This indicates the possibility of AS-IV in mitigating hypoxia-induced metabolic dysregulation. Immune escape mechanisms contribute to treatment resistance in anti-angiogenic therapies. The ILT4-PI3K/Akt-B7-H3 pathway, in addition to VEGF, has been identified as a crucial mechanism. The inhibition of VEGF expression through this pathway may underlie AS-IV’s anti-angiogenic efficacy 192 The process of pathological angiogenesis involves multiple pro-vascular growth factors working synergistically. Notably, recent evidence indicates that AS-IV can inhibit the activity of PDGF, FGF, and Ang. About 5 mg/kg AS-IV has been shown to inhibit the proliferation of smooth muscle cells and endothelial cells induced by bFGF in a concentration-dependent manner. 193 Additionally, AS-IV suppresses PDGF expression and secondary inflammation through modulation of NF-κB, IL-6, and TNF-α. 194 AS-IV also impedes Ang II-induced neovascularization by regulating the Ca2+/PI3K/Akt/eNOS/NO pathway. 195 MMP-2 and MMP-9, common downstream targets of VEGF and PDGF, are significantly inhibited by AS-IV, which also enhances the anti-neovascular effects of bevacizumab.196 -198
Mechanism of neovascularisation inhibition by Fa
Fa is the main medicinal component of ARS and holds significant research and application potential for ameliorating hypoxia and inhibiting pathological angiogenesis. 199 Direct evidence shows that Fa inhibits HIF-1α, thereby delaying hypoxia-induced cellular damage via the HIF-1α/BNIP3 pathway. 200 Studies have demonstrated that low concentrations (40 mg/l) of Fa accelerate angiogenesis in the chorioallantois membrane, while high concentrations (120 mg/l) of Fa exert the opposite effect, suggesting that Fa may exhibit bidirectional regulatory effects on VEGF under certain conditions. 201 Furthermore, Fa’s bidirectional regulation of endothelial cells has been documented, although the precise mechanism remains unclear. Molecular docking results showed that Fa binds strongly to key amino acid residues, including ASP1046 and CYS919, within the VEGFR protein, though additional direct evidence is required.202,203
Combination of AS-IV and Fa inhibits angiogenesis
The concept of modifying the ratio of herbs to enhance the release of active ingredients is a fundamental principle in TCM. Extensive research has demonstrated that adjusting the ratio of AR and ARS alters their impact on blood vessels.204,205 Studies have shown that aqueous decoctions of AR and ARS, with varying ratios, exhibit distinct effects. 206 Interestingly, the effects of these components on angiogenesis may be opposite, with 1:1 and 5:1 ratios of AR to ARS being optimal, as these ratios most effectively release AS-IV and Fa.207,208 Furthermore, it has been shown that aqueous decoctions from different parts of AR and ARS possess distinct compositions.209,210 Remarkably, a study employing 4 extraction methods (acetone, phenol, methanol, hexane, and chloroform) found that extracts from ARS co-cultured with the GBM 8401 glioma cell line inhibited microvessel growth through concentration-dependent suppression of VEGF expression, while the other 2 extracts did not. 211 These findings highlight that variations in extraction methods and herbal sources lead to differences in the clinical applications of AR and ARS. It can be inferred that the observed effects of AR, ARS, AS-IV, and Fa in promoting or inhibiting angiogenesis to alleviate symptoms and treat diseases are attributed to these differences in composition and extraction methods.212,213
Both AR and ARS are commonly used in TCM clinics. Their combined use is a key aspect of TCM, emphasizing the dual replenishment of Qi and blood to strengthen the body’s resistance and eliminate pathogenic factors. However, despite favorable clinical outcomes, research on the individual components, particularly ARS, is relatively limited. Additionally, TCM is predominantly practiced in East Asia, with its research mainly confined to Chinese-related websites, which remains insufficient in terms of depth and scope. Moreover, studies demonstrate that AR, ARS, AS-IV, and Fa exhibit dual regulation of angiogenesis. This outcome may be attributed to differences in the extraction methods and drug formulations, although there is a lack of relevant studies. It is increasingly important to explore, utilize, monitor, and evaluate the long-term therapeutic effects of herbal multicomponent combination therapies. Cutting-edge methodologies such as Panoramic Pathological Tissue Detection, High Performance Liquid Chromatography-Mass Spectrometry (HPLC-MS), and Blood Component Analysis will provide deeper insights into the active constituents of TCM and the overall physiological processes involved. Despite the current constraints of these technologies, their development promises a more comprehensive, multidimensional assessment of TCM.
Conclusions and Discussion
TCM extensively used in East Asia, especially in China, has considerable clinical experience in treating PN. Especially, the therapeutic mechanisms of AR and ARS, the most commonly used drugs for treating PN, have been a focus of research. AR and ARS can delay the conversion of PN to LC by inhibiting vascular neogenesis and decreasing microvessel density. Besides, they differ from chemical drugs designed to inhibit a single pro-angiogenic growth factor; AR, ARS, and related prescriptions block the hypoxia-vascular normalization cycle by promoting hematopoiesis, accelerating circulation, modulating immunity, inhibiting the expression of pro-angiogenic growth factors, suppressing vascular inflammation, and promoting vascular normalization. These multifaceted actions effectively delay the development of PN to LC. In this article, we have clearly explained the great potential of TCM, including AR and ARS, in both PN clinical and scientific research.
However, considering the need for long-term development, several issues require attention. Firstly, PN to LC transformation occurs gradually over years, emphasizing the importance of monitoring potential cumulative and progressive damage to human metabolism and organs from long-term drug usage, despite the generally low toxicity of natural drugs. 25 Novel administration methods such as enemas, intermittent dosing, and medicated baths strongly compensate for traditional delivery modalities. Additionally, AR and ARS ameliorate anemia and metabolic disorders through modulation of intestinal flora.214,215 However, there is still a lack of reports indicating whether alternating multiple modes of drug delivery in PN treatment can further reduce the side effects of AR and ARS. Furthermore, it is challenging for natural drugs to achieve the consistency of chemical drugs. Different extraction methods can lead to variations in the active ingredients, which sometimes act in contradiction to each other. This inconsistency greatly hampers continued research in the field of natural drugs. Techniques such as high-performance liquid chromatography, microfluidic chip models, and real-time drug entry component analysis offer substantial potential for researching the composition and concentration of these drugs. 216 Angiogenesis is a multilevel process involving a variety of pro-vascular growth factors and immune factors. However, current studies have mostly focused on key factors such as VEGF, overlooking other important elements. Similarly, it has been shown that cardiac and intestinal pathologies can trigger respiratory diseases, highlighting the potential value in exploring whether PN angiogenesis interacts with other organs. The emergence of metabolomics and proteomics offers a new, safe, comprehensive, and efficient approach to studying the long-term, dynamic transformative process of PN. Fourth, LDCT and pathological examination remain reliable diagnostic indicators, emphasizing the importance of regular testing for controlling nodule cancerous progression. However, it’s important to note that there is currently no universally recognized animal or cell model for PN. Fifthly, the transition from PN to LC involves various complex physiological and pathological mechanisms, and a single animal or cell model cannot fully simulate this process. Therefore, combine the above 2 points, more advanced animal models and mechanism studies may be more helpful in clarifying the essence of PN and evaluating the efficacy of TCM. 217 In summary, the inhibition of angiogenesis by ARS and AR, along with their active components, proves effective in the clinical treatment of PN. The study of their specific mechanisms serves as a potent driving force for advancing the prevention and treatment of the malignant progression of PN and LC in TCM.
Footnotes
Author Contributions
Fengming You and Sanyin Zhang conceived the study. Ying Guo and Peng Yang wrote the manuscript. Zihong Wu draw the Figures. Sanyin Zhang and Fengming You supervised the manuscript preparation and directed the final version of the manuscript. The final version of the manuscript was read and approved by all authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the Department of Science and Technology, Chengdu University of Traditional Chinese Medicine (030041042).
