Abstract
Objectives:
Hepatocellular carcinoma (HCC) represents the third-most prevalent cancer in humans worldwide. The current study’s objective is to search for the potentiality of Washingtonia robusta H. Wendl (W. robusta) leaf extract in a nanoemulsion (NE) form in enhancing radiotherapy against HCC induced in rats using diethylnitrosamine (DEN).
Material and methods:
The metabolic composition of the bioactive extract of W. robusta leaves was investigated by LC-MS. Oral epithelial (OEC) and liver carcinoma (HepG2) cell lines were used to examine the safety and anticancer activity of the NE, respectively. In the in vivo study, HCC was induced in male albino rats through administration of DEN in drinking water for 8 weeks, then treatment with NE (100 mg/kg) until the experiment’s ending. Rats were irradiated by a fractionated dose of 2Gy*4.
Results:
NE exerted remarkable cytotoxicity in comparison to the parent extract and the standard doxorubicin on the HepG2 cell line. Besides, the NE administration and/or γ-irradiation (IRR) significantly reduced the elevated alanine aminotransferase (ALT), total proteins, and albumin levels in HCC-induced rats. Likewise, the tumor markers alpha-fetoprotein (AFP) and gamma-glutamyl transferase (GGT) levels were considerably reduced in HCC rats. In addition, NE treatment before IRR significantly decreased the expression of the poly ADP ribose polymerase-1
Conclusion:
NE of W. robusta extract may possess a radiosensitizing novel impact and provide a new strategy to combat HCC in clinical practices.
The most frequent liver epithelial cancer, HCC, is the third-most prevalent cancer worldwide and fifth among cancers in general. 1 Over the last few years, the incidence rate of HCC has risen, increasing the frequency with which it spreads in Egypt. The development of HCC may be influenced by a number of factors such as cigarette smoking, occupational chemical exposure as pesticides and paints, and endemic illness in the neighborhood, including schistosomiasis. 2
One of the most significant cancer-causing agents in animal models is DEN. It is known to modify the enzymes required for DNA replication and repair. Through the development of the pre-neoplastic localized lesions, DEN influences the initiation stage of carcinogenesis during a time of enhanced cell proliferation coupled with hepatic necrosis, resulting in DNA strand breakage, DNA carcinogen adducts, and HCC without cirrhosis. 3
AFP is formed by embryonic endoderm cells during the fetal stage. Following birth, AFP production is significantly lowered. An increase in AFP is used as one of the metrics for screening HCC in chronic hepatitis patients, as well as one of the diagnostic criteria for HCC. 4 Aside from AFP, GGT has been shown to be a valuable biomarker for tumor prognosis and tumor assertiveness characteristics indicator, notably in case of small tumors or people with low serum AFP levels who have HCC. 5 HCC is usually treated with radiation, chemotherapy, and surgical resection. The therapeutic impact of ionizing radiation therapy is dependent on DNA damage and repairing signaling pathways which cause HCC resistance to radiation-based therapy. 6 A considerable subset of cancers may benefit from the management of poly ADP ribose polymerase (PARP) enzyme inhibition. Targeting therapy to molecularly defined tumor subtypes is effectively demonstrated by the discovery of novel PARP inhibitors that selectively target and cause cell death to cancer cells.7,8 The PARP enzymes control DNA damage repair, as well as tumor development and growth.9,10
Herbal treatments have shown promising radiosensitizing and hepatoprotective benefits, especially against HCC.11,12 W. robusta leaf extract was chosen because of our previous research (in vitro and in vivo) that showed its bioactivities as a hepatoprotective against hepatotoxicity and inflammation produced by gamma radiation. W. robusta has effectively acclimatized in Egypt and comprises significant amounts of lipoids, polyphenols (flavonoids and phenolic acids), and saponins as primary and secondary metabolites. Its economic importance is allocated to their edible fruits as well as their antioxidant abilities. 13 Due to their long-term stability, simplicity in synthesis (spontaneous emulsification), high solubilization of drug molecules, and improved bio-accessibility; the inclusion of natural bioactive elements into microemulsion systems has become a popular trend. Moreover, bioactive medications can be protected, have their release controlled, and have their functional qualities improved by using microemulsion formulations. 14 The kinetic and thermodynamic stability of microemulsions means that they rarely show signs of aggregation and coalescence. 15 Due to the tiny size of their droplets, microemulsions are also known as nanoemulsions.
The goal of this work is to develop a novel radiosensitizer approach for cancer patients by exploring the mechanism of nanoemulsion dosage form of an ethanolic extract of W. robusta leaf in DEN-initiated HCC. This was accomplished by analyzing the NE’s antiproliferative, DNA repair, oxidative stress status, and in vitro cytotoxic characteristics against the HepG2 cell line. The metabolic profile of this extract was examined by Ultra-Performance Liquid Chromatography-Tandem Mass Spectrometry (UPLC/MS-MS) before its use in NE pharmaceutical formulation.
Material and Methods
Plant Source and Identification
The harvesting of fresh W. robusta leaves from palm trees planted in the Zoological Garden (Giza, Egypt) was in the summer of 2020 (August to September). Dr. Rim Samir Hamdy, plant taxonomy professor in the Botany Department, Faculty of Science, Cairo University, Egypt, identified the sample.
Extraction and Samples Preparation
The extraction of the herbal leaves followed the method of Benahmed-Bouhafsoun et al. with some modifications. 16 The chosen species’ air-dried, powdered leaves (2.5 kg each) were extracted using petroleum ether (40-60°C, 6 L) then by ethanol (90%, 6 L). A rotary evaporator (Büchi, Switzerland) was used to evaporate the extracts at a lower pressure. The dried ethanolic extracts of Washingtonia filifera and Washingtonia robusta (encoded EWF and EWR) were weighed, and their yield percentages were computed (Table 1).
Annotated Metabolites From W. robusta Leaf Ethanolic Extract.

Preparation and Characterization of the Nanoemulsion
Leaves of W. robusta were extracted using ethanol, evaporated under decreased pressure, and weighed. For the preparation of nanoemulsion, the oil phase used was isopropyl myristate. PEG 400 (polyethylene glycol 400) was employed as a co-surfactant, and Tween 80 was used as a surfactant. By combining the surfactant and co-surfactant, Smix was created. The desired component ratios for the nanoemulsion formulation (10% isopropyl myristate and 55% Smix having equal parts Tween 80 and PEG 400 in addition to 35% water) were created. A final concentration of 1 g/10 ml of dist. H2O containing the tested extract was attained and homogenized using a bath sonicator (Branson 2510E-DHT, Richmond, VA, USA), and kept at room temperature. A digital pH meter (JENWAY, 3510, Staffordshire, UK) was used to measure the pH values of the generated nanoemulsion at 25°C. 29 The Nano Zetasizer (Nano-ZS) was used to measure the formulation’s globule size and dispersion, represented by the polydispersity index (PDI) (Malvern Instruments, Worcestershire, UK). By using a Transmission Electron Microscope (TEM) (JEM-1230, JEOL, Tokyo, Japan), operating at 200 kV, the morphology of the produced nanoemulsion was examined.
UPLC-MS/MS Profiling
According to Haggag et al., 30 metabolic profiling of the extract of W. robusta leaves in ethanol was performed using a Synapt G2 HDMS quadrupole time-of-flight hybrid mass spectrometer linked to an Acquity Ultra-Performance Liquid Chromatography system (Waters, Milford, USA). Chromatographic separation was performed using a guard column (2.15 mm, 1.7 m particle size) and an Ethylene Bridged Hybrid reversed phase column (BEH C18 column) (2.1100 mm, 1.7 m particle size; Waters, Milford, USA). Acetonitrile was used as solvent B and 0.1% formic acid in water (v/v) as solvent A during the 6-minute elution process, which was carried out at a flow rate of 0.3 ml per minute. The injection volume was 2 µl, and the column temperature was 40°C. MS Convert software was used to convert the raw data into separate positive and negative ionization files. After that, it was loaded into the data mining application MZmine 2.10 for formula prediction 11, alignment, deconvolution, peak selection, and deisotope removal. The database used to identify the chemicals was the 2015 Dictionary of Natural Products.
In Vitro Cytotoxic Activity
Sulforhodamine B (SRB) colorimetric assay 31 was adopted for screening the cytotoxic activity of NE of W. robusta leaves (90%) alongside the whole extract, which were evaluated against OEC cell line and HepG2 cell line in comparison to the standard drug (doxorubicin). OEC isolated from human oral mucosa was obtained from AcceGen Biotech (Cat.# ABC-TC4365, Fairfield, NJ, USA), and HepG2 cells were acquired from ATCC (Cat.# HB-8065, RRID:CVCL_0027, Manassas, VA, USA). All cells tested negative for mycoplasma, bacteria, yeast, and fungi. GibcoTM (Waltham, MA, USA) supplied the Dulbecco’s Modified Eagle Medium and high-glucose DMEM, where cells were cultured at 37°C and 5% CO2 and supplemented with 10% fetal bovine serum. Samples ranging in concentration from 10 to 100 g/ml were employed. The data were presented as mean ± SD (n = 3), and the IC50s were computed.
In Vivo Study
Animals and irradiation
The experimental protocol adopted was accepted by the Institutional Animal Care and Use Committee (CU-IACUC), Cairo University, Egypt, with approval code# CU III F 84 19. Adult male albino rats (150-170 g) and Swiss albino mice (22-25 g) were supplied from the National Center for Radiation Research and Technology (NCRRT), Cairo, Egypt. The animals were kept in typical laboratory settings in plastic cages. At the animal house, the temperature was kept at 23 ± 3°C. Before the investigation, the animals were kept for a week to acclimatize to environmental factors such as temperature, pressure, humidity (60% ± 10%), good ventilation, and a 12-hour light-dark cycle. Animals were offered free availability of water and a standard nutritious diet (obtained from EL-Nasr Chemical Co., Cairo, Egypt).
The Canadian Gamma Cell-40 biological radiator (137 Cesium), manufactured by the Atomic Energy of Canada Limited, Ontario, Canada, was used for animal gamma-irradiation at the NCRRT, Egyptian Atomic Energy Authority (EAEA), Cairo, Egypt. At the moment of exposure, the dose rate of the gamma radiation was 0.613 rad/sec.
Lethal dose estimation and experimental design
The lethal dose and the potential toxicity of the NE were estimated using Swiss albino mice following the Proposed method of Chinedu et al., 32 illustrated in Table 3. As NE showed no toxicity at higher doses (5000 mg/kg), the administered dosage was chosen according to our previous study on the same plant extract. 13
Sixty male rats were allocated into six groups (10 rats, each).
Complete blood picture estimation
From the orbital sinus, blood was withdrawn from all groups and collected in ethylene diamine tetraacetic acid (EDTA)-coated tubes for the estimation of the complete blood count (CBC) using a hematology fully automated analyzer (Zybio-31, China).
Liver function and tumor marker estimation
Sera were used for the estimation of liver function; ALT, 33 albumin (ALB), 34 total protein (TP) 35 and GGT through kinetic technique 36 using particular diagnostic kits (ALT and ALB by Biodiagnostic, Cairo, Egypt; TP by Biomed Labordiagnostik GmbH, Oberschleißheim, Germany; GGT by Spectrum, Hannover, Germany). An ELISA kit was used to measure AFP (Cloud-Clone Corp., Katy, Texas, USA; Cat.# SEA153Ra).
Oxidative stress marker estimation
The homogenized liver tissues were used for measuring reactive oxygen species (ROS), 37 lipid peroxidation (malondialdehyde; MDA formation) 38 , and nitric oxide (NO) 39 contents through the referenced chemical methods.
Estimation of PARP1 expression by western blot analysis
RIPA lysis buffer was used to homogenize liver tissues (Sigma-Aldrich Co., Saint Louis, Missouri, USA). The Bradford technique was used to ascertain the protein content (Bio-Rad, CA, USA). Rat PARP1 anti-body was used (Ab-Cat.# 1753958, MyBioSource, CA, USA). Monoclonal antibody against beta-actin, membrane rehybridization was used to assess loading accuracy (1:500; Ab-Cat.# 3598-100, BioVision Inc., CA, USA). The bands were densitometrically examined by Diana 95.1 camera, Raytest Isotopenmebgeräte GmbH and Aida 2.1 software program (Straubenhardt, Germany).
Immunohistochemistry (IHC) and histopathology evaluations
Liver sections were taken from the same location and preserved in 10% formalin immediately after sacrifice. The sections were cleaned in xylene, embedded in paraffin wax, and dehydrated in increasing degrees of ethanol. 4 µm-thick paraffin sections were put on sterile slides and stained with Ehrlich’s hematoxylin-eosin (H&E) then, viewed byOlympus microscope (BX41, Hamburg, Germany). 40 The evaluation of the histopathological alterations was made through the valuation of necrosis, the presence of nodules, chronic hepatitis, and stage of fibrosis. 41
For determination of the IHC labeling of Ki67, 4 μm-thick liver sections were fixed on positively charged slides. Monoclonal antibodies against rabbits and rats were used to stain the Ki67 antigen (Ab-Cat.# 275R-16, Cell Marque, Darmstadt, Germany), according to the manufacturer’s instructions. For IHC evaluation, specimens were scored for 5 high power fields, as a minimum. Positivity was the number of the stained nuclei. 42 The examination was done by an expert histologist. To prevent bias, the sample key was blinded.
Statistical Analysis and Software
Data were analyzed statistically, and the figures were composed by means of GraphPad Prism, version 5 software. The normality of data was verified by the Shapiro-Wilk’s test (not significant at P > .05). In addition, homogeneity of variance was assumed by Levene’s test (not significant at P > .05). All the biochemical parameters were analyzed by one-way analysis of variance (ANOVA), followed by Bonferroni’s multiple comparison test as a post-hoc test to compare differences between means. The ANOVA assumptions were made and the independence observations were measured on a scale level. Findings were presented as a mean ± standard error (SE). At P < .05, differences were deemed significant. Data regarding the histological scoring was analyzed (non-parametric) using the Kruskal-Wallis test with Dunns post-hoc for comparing the medians of different groups. The references and citations were placed in by the EndNote X7 software.
Results
Nanoemulsion Characterization
The prepared microemulsion system had globules that were 65 nm in size and had a uniform globule size distribution (PDI = 0.4) as seen in Figure 1. The prepared nanoemulsion’s morphology was studied using transmission electron microscopy. The generated nanoemulsion globules were spherical, distinct, and had a homogeneous droplet size distribution, as is evident. The pH of the created nanoemulsion (6.4) was nearly physiological (Figure 2).

Globule size and distribution of the NE represented by the PDI using Nano-ZS.

Transmission electron micrograph of the W. robusta nanoemulsion showing the particle diameter. Scale bar represents 200 nm.
UPLC- MS/MS Profiling
Several types of chemicals were annotated as a result of the dereplication of the secondary metabolites from the ethanolic extract of W. robusta leaves. The bulk of the dereplicated chemicals were in the positive mode, which also showed the presence of numerous phenolics, flavonoids, taxane diterpenoids, drimanic sesquiterpenes, and alkaloids. The results are shown in Table 1.
In Vitro Study
The cytotoxic activities against HepG2 and OEC cell lines were assessed by adopting the same SRB protocol. In this respect, NE of W. robusta leaf extract exerted a remarkable in vitro cytotoxicity against HepG2 cell line with IC50 18.1 ± 0.5 μg/ml in comparison to the parent extract (IC50 53.8 ± 4.97 μg/ml) and the standard drug, doxorubicin (IC50 1.2 ± 0.0094 μg/ml). Besides, it was inactive on normal cell line (OEC), confirming its safety (Table 2). These results support the selection of NE for further in vivo trials aiming to confirm its ability to treat liver cancer in comparison to radiotherapy or as a radiosensitizer.
IC50s of the NE and the Ethanol Extract on the Cell Line HepG2, and Their Effects on the Normal OEC.

Data were expressed as mean ± standard deviation (SD) (n = 3)
Abbreviations: IC50, the concentration that caused 50% inhibition of the cancer cells growth; viability, surviving cancer cells.
In Vivo Study
Estimation of in vivo safety and lethal dose
The NE was found to be safe at levels as high as 5000 mg/kg b.wt. (Table 3). (no behavioral abnormalities or toxicity). The therapeutic dose of 100 mg/kg b.wt. was chosen in order to further evaluate the cytotoxic effectiveness against DEN-induced liver cancer in rats.
Schedule for Determination of the Lethal Dose of the NE on Male Swiss albino Mice.

Physical markers (survival rate, body weight change, and liver weight index) and tumor nodule incidence in different DEN-treated groups
Survival rate in Figure 3 indicates that no deaths occurred in the control and NE groups (n = 10), while the maximum percentage of deaths was in the DEN-treated group (n = 4). On the contrary, HCC-induced rats treated with NE the surviving rats were increased (n = 9), while IRR alone or NE/IRR increases the survival rate compared to DEN group (n = 6). The b.wt. and liver weight index are signs of HCC progression. As compared to the normal rats (50.9% increase in b.wt.), after 17 weeks the group treated with DEN in drinking water showed a slight increase in b.wt. (23.9%) and a significant increase in the liver index indicating HCC induction. Administration of NE increases the b.wt. significantly, while it did not affect the liver index compared to the control rat. Treatment of HCC-induced rats with NE and/ or IRR ameliorated the liver index in comparison to the DEN group. This enhancement in the liver index was more noticeable in the group pretreated with NE and followed by IRR, which suggests that NE potentiates the effect of gamma irradiation. However, this treatment failed to counteract the decrease in b.wt. (Figure 4). Livers from the HCC group displayed abnormal morphological features with several macroscopic tumor nodules (Figure 5), with a nodule incidence equal to 50%. Quite the reverse, the liver of the HCC-group treated with NE showed a relatively ameliorative morphological appearance with very few micro-nodules and 17% nodule incidence, while the most ameliorative morphological result was in the HCC-group treated with NE before IRR with no nodule incidence in all treated animals.

Effect of NE and IRR on the survival rate in DEN-intoxicated rats.

Effect of NE and IRR on liver/body weight index and body weight change in DEN-intoxicated rats.

A photograph of liver xenografts after sacrifice showing the morphologic features and the HCC nodule incidence in the liver of different treated groups.
Complete blood picture estimation
The DEN-treated groups at 18 weeks recorded a highly significant decrease in the hemoglobin (HGB) concentration, platelets (PLT) and red blood cells (RBCs) count with an increase in the white blood cells (WBCs) count recording percentage changes 15%, 25%, 10%, and 56% respectively from their analogous control values. Exposure to gamma radiation intensifies the hematological alteration caused by DEN as compared to the DEN group. Treatment of HCC rats with the NE before irradiation caused a significant reduction in the elevated WBCs compared to the DEN group and restored HGB to normal levels (Table 4).
Effect of NE and IRR on HGB Concentration, PLT, RBCs, and WBCs Counts in DEN-intoxicated Rats.

Data are expressed as mean ± SE. Differences between means were analyzed using one-way ANOVA followed by Bonferroni’s multiple comparison test.
Significantly different from the control group.
Significantly different from the DEN group.
Significantly different from the DEN+IRR group.
Liver function and tumor marker estimation
Accumulating evidence found that liver biomarkers assume a crucial role in the HCC etiology. DEN administration significantly raises the levels of serum ALT (53%) with a decrease in the TP (27.8%) and ALB (37.2%) levels in comparison with the control group (Table 5). DEN-induced HCC treated with NE ethanolic extract of W. robusta either separately or in combination with IRR significantly improves the level of ALT, TP, and ALB levels in contrast to the DEN group. DEN administration significantly elevates the levels of serum AFP (395%) and GGT (351%) in comparison with control rats (Figure 6). DEN-induced HCC treated with NE and/or IRR significantly decreased the level of GGT and AFP in contrast to the DEN group. Yet again, the more marked effect seen in the group treated with NE followed by gamma irradiation supports that NE may possess radiosensitizer activity.
Effect of NE and IRR on the Liver Function, ALT, TP, and ALB Levels in DEN-intoxicated Rats.

Data are expressed as mean ± SE. Differences between means were analyzed using one-way ANOVA, followed by Bonferroni’s multiple comparison test.
Significantly different from the control group.
Significantly different from the DEN group.

Effect of NE and IRR on serum AFP level and GGT activity in DEN-intoxicated rats.
Oxidative stress marker estimation
Oxidative stress is a good indicator in evaluating the efficacy of cancer therapy. Treatment that could elevate the levels of the reactive oxygen and nitrogen species, leads to an increase in apoptosis and a decrease in proliferation. HCC-induced rats treated with NE followed by IRR showed the most increase in ROS, NO, and MDA levels (Figure 7), which reflects that NE may potentiate the effect of gamma irradiation.

Effect of NE and IRR on MDA, ROS, and NO levels in DEN-intoxicated rats.
Estimation of PARP1 expression by western blot analysis
The liver homogenate of DEN-treated groups at 17 weeks revealed a significant increase (P = .001) in the PARP1 relative expression by 322% when compared with values in the sham group. HCC-induced rats treated with either NE separately or in combination with IRR showed significant inhibition in PARP1 relative expression compared with DEN. However, HCC rats injected with co-therapy (NE before IRR) showed the greatest PARP1 inhibition, which may indicate an impairment in the DNA repairing mechanism, reflecting a radiosensitizing effect of NE (Figure 8).
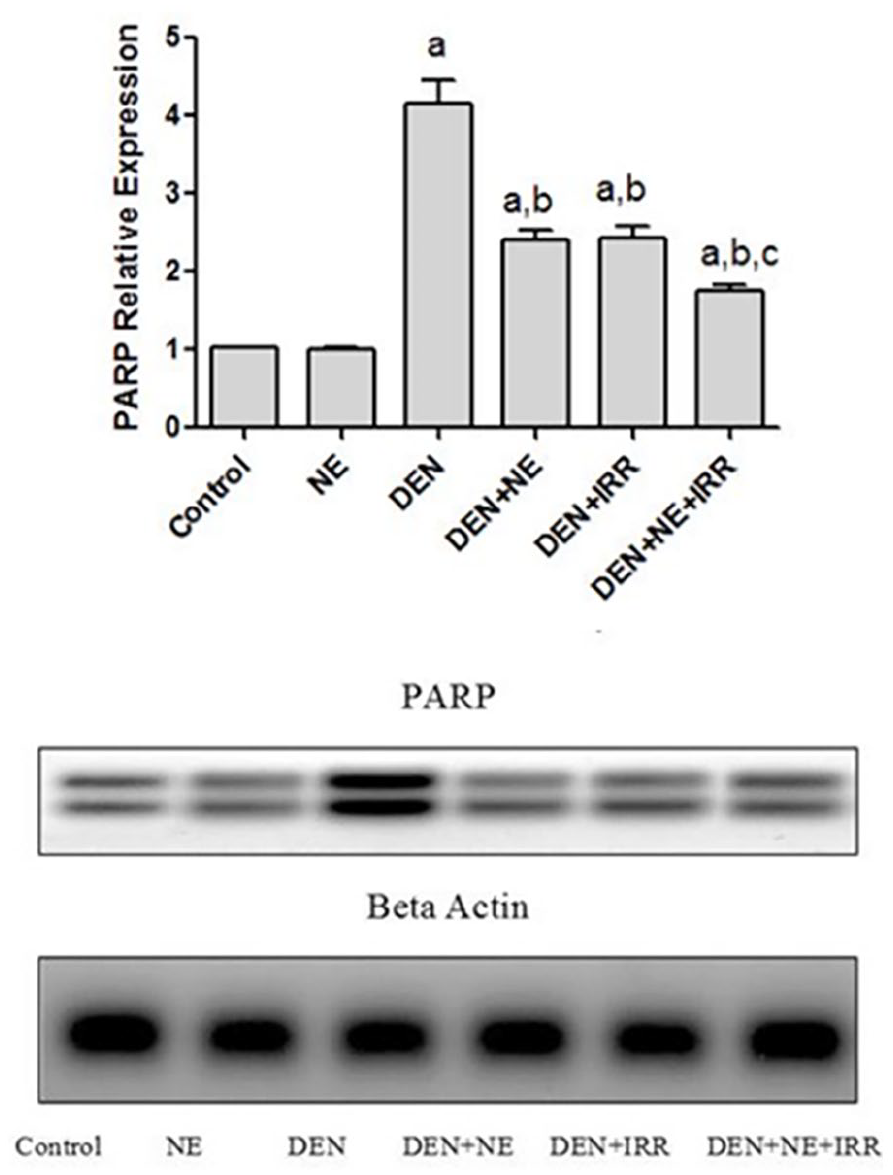
Effect of NE and IRR on liver PARP1 relative expression in DEN-intoxicated rats.
Histopathology and immunohistochemistry evaluations
The expression of the proliferation marker Ki67 shown in Figure 9 reflected that the HCC group induced by DEN showed very high nuclear positivity (3.86 folds) of Ki67 with P = .006 compared with the control, reflecting excessive proliferation in the HCC group. Treatment of HCC-rats with NE of W. robusta leaves revealed moderate positivity in all taken photos and some possessed high nuclear positivity. On the other hand, exposure of HCC-groups to γ-radiation either alone or pretreated with NE expressed low to no nuclear positivity of Ki67 compared to the HCC induced group. It seemed that the radiotherapy or combined treatment might have an anti-proliferative effect on the HCC-rats.

Effect of NE and IRR on liver Ki67 immunohistochemical expression in DEN-intoxicated rats.
The histopathological changes, the grading of chronic hepatitis, and the stage of fibrosis in the hepatic tissues of HCC-induced rats treated with NE and IRR were illustrated in Figures 10 and 11. DEN group revealed the maximum chronic hepatitis and liver fibrosis. In addition to dilated portal tract (PT), moderate portal and peri-portal inflammatory infiltrate, marked apoptosis and ballooning of hepatocytes with bile stasis, multiple complete nodules, and a large nodule of acinar HCC (grade I), and some slides show a large nodule of trabecular HCC (grade II). However, DEN groups treated with NE then exposed to gamma radiation showed mildly edematous and fibrotic PT with short fibrous septa, incomplete nodules, mild to moderate macro- and micro-vesicular steatosis, moderate apoptosis, and bile stasis.

Histopathological view of the hepatic tissues in different treated HCC-induced rat model. Normal control: slide a (X 200): liver showing average portal tracts (black arrow), average central vein (blue arrow), and average hepatocytes (yellow arrow); slide b (X 400) showing average central vein (CV) and average hepatocytes arranged in single-cell cords (yellow arrow) with average intervening blood sinusoids (blue arrow). NE: slide c (X 200): liver showing average portal tracts (black arrow), average central vein (blue arrow), and average hepatocytes (yellow arrow); slide d (X 400): showing average CV and hepatocytes in the peri-venular area (yellow arrow) and bile duct (red arrow). DEN: slide e (X 200): showing mildly expanded portal tract with a mild portal and peri-portal inflammatory infiltrate (black arrow), marked apoptosis (blue arrow) and complete nodules (red arrow), and large nodule of acinar HCC (yellow arrow); slide f (X 400): showing acinar HCC composed of markedly pleomorphic cells with prominent nucleoli (black arrow), scattered mitotic figures (blue arrow), scattered apoptosis (yellow arrow), and thin-walled congested blood vessels (red arrow). DEN+NE: slide g (X 200): liver showing average portal tracts (black arrow) with few complete nodules (N), and scattered apoptosis (red arrow); slide h (X 400): another view showing average portal tracts (black arrow) with the average portal vein (blue arrow), and scattered apoptotic hepatocytes in the peri-portal area (red arrow). DEN+IRR: slide i (X 100): liver showing mildly dilated congested portal vein (black arrow), and markedly dilated central vein (CV) with detached lining (blue arrow), slide j (X 400): high power view showing portal tracts (black arrow) with mildly dilated congested portal veins (PV) and short fibrous septa (blue arrow), and scattered apoptosis (yellow arrow). DEN+NE+IRR: slide k (X 200): liver showing average portal tracts (black arrow), with few incomplete nodules with moderate macro- and micro-vesicular steatosis (blue arrow), scattered apoptosis (red arrow); slide l (X 400): another view showing fairly edematous portal tracts (black arrow) with mildly dilated portal veins (PV), average bile duct (red arrow), and scattered apoptotic hepatocytes in the peri-portal area (blue arrow).

Grading of chronic hepatitis and the stage of fibrosis of DEN-induced HCC in rats treated with NE and IRR.
Discussion
HCC is a significant problem in both worldwide and, to a greater extent, in developing countries. 43 Poisonous contemporary chemicals, air pollutants, food additives, fungal toxins, and viral infections are the major causes of HCC. 44 Through oral gavage and drinking water, the hepatotoxicity and carcinogenicity of DEN have been methodically demonstrated in rats. 45 For thousands of years, medicinal herbs have been utilized to treat a range of liver-related maladies, and more recently, studies have shown that many of their bioactive chemicals may interact with targets linked to liver carcinoma. 46 NE was almost inert on normal cell lines (OEC), which further supports its safety; it also has a hepatoprotective effect. These findings justify the choice of this herbal extract for additional in vivo studies. According to the results of the current investigation, rats given DEN in drinking water developed HCC, which was demonstrated by pathological, morphological, cellular, and biochemical abnormalities. It’s possible that the aberrant liver function brought on by exposure to hepato-carcinogens, which resulted in a decrease in appetite and food consumption, is what caused the b.wt. loss in DEN-treated rats. 44 After being exposed to DEN, the weight of the animals’ livers increased, which may have been caused by the formation of liver nodules and tumors or inflammation of the hepatocytes. 47 Moreover, our data indicated that the DEN administration led to an impairment of CBC parameters. The key liver-specific markers like ALT, ALB, and TP, which serve as diagnostic indicators of hepatic function, increased noticeably abnormally in DEN-treated rats. 48 The gold standard for treating HCC is thought to be AFP. Comparing with the normal rats, the DEN group exhibits increased levels of AFP and GGT. The biochemical findings are supported by the histopathological results, which displayed a total breakdown of the hepatocellular architecture along with many hyperplastic nodules that may be HCC formation; the same results were shown previously by studies of Sadek et al.12,44 On the other hand, rats that were treated with NE and/or IRR showed an improvement in liver wt index, ALT, ALB, TP, AFP, and GGT, along with an enhancement in some CBC parameters comparing with the DEN group.
Radiation resistance of tumors is influenced by the reactive oxygen/nitrogen species and MDA levels. 49 In the present study, HCC-induced rats that received NE first and were then gamma irradiated have shown an elevation in the ROS, MDA, and NO generation, suggesting that NE could potentiate the effects of gamma irradiation. Histopathological examination revealed apoptosis in HCC-rats treated with NE+IRR, which may be due to the increased levels of ROS, MDA, and NO. In the course of radiotherapy, ROS produced during mitochondrial respiration exacerbates DNA damage. 50 LPO has been employed as a marker for cell membrane damage brought on by ROS. 51 Nitric oxide is said to have a tumoricidal effect; it has been proposed to alter a number of cancer-related processes, such as invasion, metastasis, apoptosis, angiogenesis, and the cell cycle. 52 Increased ROS leads to GSH content depletion; this decreases the radioprotective response, making tumor cells more sensitive to radiation. 53
Numerous intracellular functions, such as transcription, signaling, and DNA repair, are regulated by the PARP enzyme. PARP expression is elevated in both malignant and non-cancerous cells that are growing. 54 PARP inhibitors competitively bind with NAD+ and inhibit its catalytic activity. They cause cytotoxicity via irreparable damage to DNA by inhibiting repair protein and they also block cellular replication by inducing collapsing of the replication fork. This results in continuous single-strand breaks (SSBs) due to the dissociation of PARP from DNA, which in turn leads to cell death. 12 PARP inhibitors potentiate ionizing radiation via inhibition of base excision repair. Yet, they promote the action of radiation-induced ROS production in DNA damage. 55 As well, double-strand breaks accumulate in cells with defective PARP function after exposure to radiation. Additionally, they may also enhance radiation therapy by impairing of NF-κB. 56 A powerful targeted therapy is the creation of novel PARP inhibitors that can specifically destroy cancer cells. The current research revealed the inhibitory impact of NE on PARP1 expression in HCC-rats compared to their corresponding rats (DEN group). However, an obvious synergism appeared in the irradiated-HCC group pretreated with NE, reflecting the radiosensitizing effect of the NE. In fact, PARP inhibitors have successfully sensitized tumors to radiation in both in vitro and in vivo models. 57 It was shown previously that increased oxidative stress (ROS, MDA, and NO as shown in our results) facilitates the antitumor effect of PARP inhibitors in cancerous tissues. 58 Through vasoactivation, PARP inhibitors may alter the amount of hypoxia present in tumors. It may be beneficial to reoxygenate hypoxic tumors because hypoxic cells are more radioresistant than oxygenated cells. 59 It may be possible to enhance radiotherapy by combining PARP inhibitors with ionizing radiation.
Ki67 is a non-histone cellular proliferation marker; its expression was reported previously to be associated with liver cancer tissue and clinical outcomes of HCC patients. 60 When evaluating the biological behavior of carcinoma and forecasting its clinicopathological and prognostic relevance, the proliferative activity of cancer cells is a crucial component. To differentiate between tumors with low and high proliferative activity, Ki67 is frequently utilized. 61 The current study has evaluated Ki67 since the progression of the majority of malignancies is partially reliant on the proliferation of the tumor cells. Low or no nuclear positivity of Ki67 was seen in HCC-rats treated with NE and then gamma irradiated compared to HCC-induced rats. Prior work demonstrated that down-regulating NF-κB and Ki67 expressions could decrease the activity of tumors removed from mice. 62 Hence, we could postulate that the co-therapy may have an antiproliferative effect against HCC. Washigtonia robusta leaf extract in nanoemulsion form may have promise as a radiosensitizer since radiosensitizing medications should limit proliferation. 63
Conclusion
Our results have shown that NE has biological safety on normal cells and possesses an anticancer effect (cytotoxic activity) on HepG2 in vitro. Additionally, the pretreatment of NE to the irradiated HCC-induced rats encouraged the radiation action by the induction of the oxidative stress biomarkers as well as inhibition of DNA repair mechanisms and cell proliferation. Also, NE administration decreased the tumor markers AFP and GGT; at the same time, it succeeded in ameliorating liver function. To conclude, our findings could suggest a novel usage of NE as a radiosensitizer through the journey of cancer radiotherapy. Despite the W. robusta leaf extract’s safety profile, some restrictions on its application may be ascribed to adverse effects that could make it incompatible with other cancer treatments. Additionally, other radiosensitization hallmarks like evaluation of DNA damage, apoptosis, angiogenesis, and metastasis should be investigated in further studies.
Supplemental Material
sj-docx-1-ict-10.1177_15347354241308858 – Supplemental material for Washingtonia robusta H. Wendl Leaf Metabolites Potentiate the Radiosensitivity of Hepatocellular Carcinoma Through Ki67 and PARP Inhibition
Supplemental material, sj-docx-1-ict-10.1177_15347354241308858 for Washingtonia robusta H. Wendl Leaf Metabolites Potentiate the Radiosensitivity of Hepatocellular Carcinoma Through Ki67 and PARP Inhibition by Nabil M. Selim, Seham S. El-Hawary, Soheir M. El Zalabani, Rehab Nabil Shamma, Nariman El Sayed Mahdy, Noheir H. Sherif, Khayrya A. Youssif, Usama Ramadan Abdelmohsen, Mai H. Mekkawy and Hanan A. Fahmy in Integrative Cancer Therapies
Footnotes
Acknowledgements
The authors are thankful to Prof. Dr. Rim Samir Hamdy, Professor of Plant Taxonomy, Department of Botany, Faculty of Science, Cairo University, Cairo, Egypt, for approving the taxonomic identity of the plant. Additionally, the authors are very grateful to the gamma irradiation unit (NCRRT) staff member for their precious cooperation in carrying out the irradiation process.
Author Contributions
Conceptualization, S.S.E.-H., S.M.E.Z., R.N.S., N.M.S., N.E.S.M., M.H.M., H.A.F., N.H.S., K.A.Y. and U.R.A.; methodology, N.M.S., R.N.S., N.E.S.M., H.A.F., M.H.M., and N.H.S.; software, N.M.S., U.R.A., K.A.Y H.A.F., and M.H.M.; validation, N.E.S.M., M.H.M., H.A.F., and N.H.S.; formal analysis, N.E.S.M., H.A.F., M.H.M., and U.R.A.; investigation, N.M.S., N.E.S.M., H.A.F., and M.H.M.; resources, N.M.S., N.E.S.M., K.A.Y., M.H.M., H.A.F., and U.R.A..; data curation, N.E.S.M., M.H.M. and H.A.F.; writing—original draft preparation, N.E.S.M., M.H.M., H.A.F., N.H.S., and S.M.E.Z.; writing—review and editing, all authors. All authors have read and agreed to the published version of the manuscript.
Availability of Data and Materials
The authors attested that all necessary information is contained in the publication and that raw data is available upon request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration of Generative AI in Scientific Writing
AI-assisted technologies and generative artificial intelligence were not used by the writers during the writing process.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Every technique carried out in animal-based research complied with Cairo University’s Institutional Animal Care and Use Committee (CU-IACUC), approval code # CU III F 84 19. The care and use of animals was conducted in accordance with all relevant national, international, and/or institutional guidelines.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
