Abstract
Background:
Hepatocellular carcinoma (HCC) is a common clinical malignant tumor of the digestive system. Hu-Qi-Zheng-Xiao (HQZX) decoction has been clinically found to prolong the survival of patients with hepatocellular carcinoma and improve the quality of patients’ survival, but its antitumor biological mechanism is still unclear.
Methods:
A nude mouse hollow fiber hepatocellular carcinoma model was constructed to analyze the in vivo efficacy of HQZX decoction against 7 different hepatocellular carcinoma cells. The subcutaneous graft tumor model was again validated. In vitro, the effect of HQZX decoction on the growth and metastasis of the cell line with the highest growth inhibition was evaluated. The cell line with the best efficacy response screened was again used to construct a hollow fiber hepatocellular carcinoma model and hollow fiber conduit cells were extracted to detect the expression of HIF-1α, VEGF, EMT-related molecules, LCSCs-related molecules, and to observe the density of the subcutaneous vascular network of hollow fiber conduits. The liver metastasis model of splenic injection was constructed to observe the effect of HQZX decoction on tumor metastasis.
Results:
The hollow fiber hepatocellular carcinoma model was evaluated for the efficacy of HQZX decoction, and it was found to have the highest growth inhibition of LM3-luc cells. In vitro, the CCK8 assay revealed that HQZX decoction could inhibit tumor migration and invasion and promote apoptosis. In addition, the mechanism study of extracting cells from hollow fiber tubes found that HQZX decoction could inhibit metastasis-associated HIF-1α, VEGF, EMT-related molecules, and LCSCs-related molecules expression. capillary network around subcutaneous fiber tubes was reduced in the HQZX decoction gavage group of mice. It inhibited tumor metastasis in nude mice.
Conclusions:
HQZX decoction inhibited the growth of a variety of hepatocellular carcinoma cells. HQZX decoction suppressed the expression of metastasis-associated VEGF, EMT-related molecules, and LCSCs-related molecules and inhibited tumor angiogenesis and growth and metastasis, which may be related to the inhibition of the HIF-1α signaling pathway. It reveals that HQZX decoction may be a promising herbal compound for anti-HCC therapy, and also reveals the accurate feasibility of the hollow fiber hepatocellular carcinoma model for in vivo pharmacodynamic evaluation and mechanism study.
Keywords
Introduction
Hepatocellular carcinoma is one of the common malignant tumors of the digestive system, and the common clinical histological type is hepatocellular carcinoma (HCC), which accounts for 90% of primary liver cancers. According to GLOBOCAN2020 global cancer statistics, hepatocellular carcinoma is the third leading cause of cancer-related deaths worldwide, with high morbidity and mortality rates, with the highest incidence in Asian populations. 1 Early-stage HCC patients are mostly treated with liver transplantation or surgical resection, which has a high cure rate. However, due to the untimely monitoring of HCC and the difficulty of early diagnosis, most of them are already in advanced stages or have metastasis at the time of diagnosis, and the therapies of liver transplantation or surgical resection are not applicable.2,3 Currently, the first-line drug therapy for advanced HCC patients is mainly immunotherapy and gene therapy, which has greater side effects on patients over a long period. The surgical treatment options such as Radiofrequency Ablation (RFA) and Transarterial Chemoembolization TACE, on the other hand, have higher requirements on patients’ liver function and still have the risk of recurrence and metastasis after surgery.4,5
In recent years, the use of traditional Chinese medicine (TCM) adjuvant therapy to limit tumor progression has gradually become the focus of complementary and alternative therapies for the treatment of HCC. For patients with advanced HCC, clinically, TCM-assisted TACE, RFA, and targeted therapy can alleviate adverse effects, improve patients’ clinical symptoms, improve patients’ quality of survival, reduce resistance to targeted drugs and radiotherapy, improve immune level, and reduce the risk of recurrence and metastasis in patients.6 -8 Due to the rapid growth of hepatocellular carcinoma cells, HCC commonly has the high expression of hypoxia-inducible factor-1α (HIF-1α), and the tumor tissues show pathologically dense blood vessels. 9 Chinese herbs can regulate the microenvironment and inhibit tumor cell growth and metastasis through multi-targets and multi-pathways, and their pathways of action mainly include promoting apoptosis, inhibiting the process of cellular epithelial-mesenchymal transition (EMT), and regulating the expression of liver cancer stem cell (LCSC), which have demonstrated an important value in the prevention and treatment of HCC development and recurrence and metastasis. 10
Hu-Qi-Zheng-Xiao (HQZX) decoction is composed of 10 Chinese herbs, including mistletoe, astragalus, scutellaria baicalensis, white-flowered snakeroot, bitter leaf hypodermis, Panax ginseng, tulip, trichosanthes, xiakoucao, and bitter ginseng (Table 1). It is a modified version of Hu-Qi-San, a basic formula for the treatment of hepatocellular carcinoma and precancerous lesions by Prof. Qian Ying, a master of national medicine, which enhances the power of detoxification, clearing heat, and dispersing blood stasis. In the previous study, it was shown that HQZX decoction has the effects of promoting apoptosis of hepatocellular carcinoma cells, inhibiting telomerase activity, inhibiting migration of hepatocellular carcinoma cells, and immunomodulation, 11 and clinical studies found that its adjuvant TACE treatment could improve the survival of patients with advanced hepatocellular carcinoma. This study further investigated the efficacy and mechanism of HQZX decoction in controlling hepatocellular carcinoma and tumor recurrence and metastasis resistance.
Medicinal Composition of HQZX Decoction.

The hollow fiber tumor model is an in vivo antitumor drug screening model established by Hollingshead in 1995, which has the advantages of full fidelity, high throughput, and universal applicability for the pharmacodynamic evaluation of antitumor drugs.12,13 In this article, we applied a hollow fiber hepatocellular carcinoma model for the screening of the anti-hepatocellular carcinoma efficacy of HQZX decoction and verified the efficacy with a transplanted tumor model. In addition, we also investigated the possible mechanism of action of HQZX against hepatocellular carcinoma metastasis, and its anti-tumor growth and metastasis effects may be related to the down-regulation of HIF-1α, which regulates the expression of EMT-related molecules, LCSC-related molecules, and the inhibition of tumor angiogenesis. The present study will provide more laboratory evidence for its clinical application against recurrence and metastasis of hepatocellular carcinoma.
Materials and Methods
Experimental Drugs
HQZX water decoction was prepared by the Beijing You’an Hospital Chinese herbal pharmacy and boiled according to the preparation method and production specification standard for patients’ clinical use; quality control was fully guaranteed. The method was as follows: cold water was added over 2 to 3 cm above the drug surface before decoction, soaked for 30 minutes, continued to decoct for 20 minutes after the first decoction was boiled, and then decocted for 15 minutes after the second decoction was boiled, and the herbal juice from both times was mixed and concentrated in a water bath to the drug concentration required for the experiment. The decoction was filtered through a 0.22 m microporous membrane and stored below 4℃ for use. The main components of HQZX decoction were detected by liquid chromatography-mass spectrometry (LC-MS) to control the quality of different batches.
According to the Basic Operation Technique Manual of Animal Experiments, the conversion factor of body weight dose for the conversion of 20 g mice and 60 kg adult dose is 9.01. When the modification is made according to the body weight of 20 g per nude mouse and the gavage dose of 0.2 ml per nude mouse, the drug concentration of the Chinese medicine group is 1.3515 g/ml for the small dose group, 2.703 g/ml for the medium dose group, and 5.406 g/ml for the large dose group. Huachansu (HCS) capsule as a positive control drug, 14 from Shanxi Dongtai Pharmaceutical Co., Ltd (State Pharmaceutical License Z20050846), a daily adult dosage of 1.5 g, according to the above body weight dosage conversion coefficient, nude mice daily dosage of 4.505 mg, the use of dissolved in distilled water to set the volume.
Hollow Fiber Assay
HCC cells were resuspended with serum-free DMEM medium, the concentration of cells was adjusted to 1 × 107/ml, cells were injected into hollow fiber tubes with a 10 ml syringe, and the hollow fiber tubes were sealed at both ends with a heat sealer first, and then separately by 2 cm length, separated with sterile scissors, and placed in 15% DMEM medium containing antibiotics for use. Four hollow fiber tubes were taken as blank control for each cell line, and the remaining hollow fiber tubes were implanted into the subcutaneous and abdominal cavities of nude mice until the final determination of OD value. The animals were divided into the control (CON) group, HQZX large-dose (HQZXL) group, HQZX medium-dose (HQZXM) group, HQZX small-dose (HQZXS) group, and HCS group, with 4 animals in each group. The drug was started on the third day after the implantation of the hollow fiber tubes. After 7 days of drug administration, the hollow fiber tubes were removed and incubated with fisetin at 4°C for 4 hours. The tubes were discarded and reincubated with fisetin for at least 24 hours. The tubes were completely dried in the air, the hollow fiber tubes were clipped, and 5 mg/ml of MTT was added and incubated for 4 hours. The OD values were measured, and the OD values were used to calculate the effects of the HQZX decoction and HCS on the different hepatocellular carcinoma cells. Relative inhibition rate = 1 − (OD value of administered group − OD value of blank control)/(OD value of control group − OD value of blank control).
Animal Experiments
In vivo metastasis assay
Five-week-old nude mice were randomly divided into 2 groups of 4 mice each. After the mice were anesthetized with sodium pentobarbital, the abdomen was disinfected with 75% alcohol, sterile scissors were cut under the skin first then a small cut in the peritoneum, the spleen was pulled out with forceps, and then 100 μl of HCC cell suspension (5 × 106/each)was slowly injected into the spleen with a 1 ml syringe, and the peritoneum of the spleen was seen to swell and turn white after the injection was completed, the needle hole was pressed with a 75% alcohol cotton ball for 3 minutes, and then the spleen was returned to its original position, and the peritoneum and skin were sutured in turn. Approximately 7 days later, when optical density was observed in the liver area by in vivo imager, gavage administration of HQZX decoction was started. Twenty-one days after gavage, nude mice were executed and the liver and spleen were removed. The livers and spleens were counted, weighed, and photographed.
Subcutaneous graft tumor experiment
Five-week-old male mice were randomly divided into 3 groups, and then 1 × 107 HCC cells were resuspended in 150 μl serum-free DMEM and then injected subcutaneously into the axilla of each mouse forelimb. When the tumors took shape, we started to administer HQZX decoction by gavage at 0.2 ml/mg daily with a gavage needle. Three weeks after administration, the mice were executed and the subcutaneous tumors were harvested, weighed, and photographed. For PDX, the PDX tumor blocks were first divided into small pieces of 2 mm × 2 mm × 2 mm, and then attached to the axils of the forelimbs of the mice separately, and the rest were as above.
Cell Culture
Human hepatocellular carcinoma cells LM3-luc, Huh7, HepG2, Hep3B, MHCC-97H, MHCC-97L, and PDXC were provided by the Beijing Institute of Liver Diseases. The cells were resuscitated with DMEM medium containing 10% fetal bovine serum (FBS) and placed in a cell culture incubator with 5% CO2 and 37°C Bohol humidity.
CCK8 Assay
The cell concentrations were adjusted according to the different administration times, the cells were inoculated into 96-well plates, and the cells were administered according to the different concentrations on the second day after cell walling. After incubation in the cell incubator for 24, 48, and 72 h, 10 μl of CCK-8 (Abcam) was incubated with the cells for another 4 hours and the OD values were measured.
Wound Healing Assay
The cells were inoculated onto a 6-well plate, and after the cells grew to a suitable density, the bottom of the well plate was scored vertically with the tip of a 10 ml pipette, washed 2 to 3 times with PBS, photographed after 24 hours, 48 hours, and analyzed with ImageJ software.
Transwell Assay
For migration assay, the cell concentration was adjusted to 5 × 105/ml, 100 ml of cell suspension was added to the upper chamber and 600 ml of medium containing 20% FBS was added to the lower chamber, incubated for 24 hours, and the non-migrated cells in the upper chamber were removed with a cotton swab, and the migrated cells were fixed with 4% paraformaldehyde for 30 minutes and then stained with 0.1% crystal violet for 20 minutes, randomly imaged under a bright field microscope for 5 fields, and the number of cells was counted. For the detection of invasion, 100 ml of Matrigel (BD, USA) was added to the upper chamber and incubated at 37°C for 4 hours to become gelatinous, and other operations were performed as in the migration assay.
Flow Cytometry Assay
The samples were processed according to the manufacturer’s protocol using Annexin-V/PI Apoptosis Kit (Yisheng Technology, Shanghai, China), detected using a FACS Calibur flow cytometer (BD, USA), and the data were processed with FlowJo software.
For hepatocellular carcinoma stem cell expression assay, the cells were incubated with CD44 (BD, USA), and CD133 (BD, USA) for 20 minutes at room temperature and protected from light, and the cell suspensions were filtered with a 40 mm single-cell sieve before going on the machine and detected using a FACS Calibur flow cytometer (BD, USA), and the data were processed with FlowJo software.
Quantitative Real-time PCR
Total RNA was extracted from cells using TRIzol reagent (Thermo Fisher, USA), cDNA was synthesized by reverse transcription using cDNA kit (Applied Biosystems, USA), and qRT-PCR was performed using TB Green kit (TAKARA, Japan), and CT values were measured on a ViiA 7 Dx real-time fluorescence quantitative PCR instrument (Applied Biosystems, USA) to determine CT values. GAPDH was used as an endogenous reference and relative mRNA expression was determined using the 2-ΔΔCt method. All primer sequences are shown in Table S1.
Western Blot
Cellular proteins were extracted with a mixture of RIPA lysis buffer (Solarbio, Beijing, China) and protease inhibitor (Solarbio, Beijing, China). Protein concentrations were determined using the BCA protein quantification kit (Yeasen, Shanghai, China). Proteins were separated by SDS-PAGE and transferred to polyvinylidene difluoride (PVDF) membranes (Millipore, USA). The membranes were incubated with primary antibodies at 4°C overnight using the following primary antibodies: GAPDH (CST, USA), E-cadherin (CST, USA), N-cadherin (CST, USA), Vimentin (CST, USA), HIF-1α (CST, USA), VEGF-A (CST, USA) and incubated with horseradish peroxidase-linked secondary antibody (CST, USA) for 1 to 2 hours at room temperature, followed by 3 washes with TBST for 5 minutes. The signals were detected using the ECL chemiluminescence system and the grayscale values of each target band were analyzed by ImageJ software.
Hematoxylin and Eosin (H&E) Staining
The fixed tumor tissue was embedded in paraffin and cut into 4 -μm-thick sections. Sections were dewaxed with xylene and rehydrated with different gradients of ethanol. The sections were then stained with hematoxylin for 5 minutes, rinsed with running water, and then stained with eosin for 2 minutes. Sections were then dehydrated with ethanol and treated with xylene. Images were acquired under a microscope for analysis.
Immunohistochemical (IHC) Staining
The tissue was fixed in 4% paraformaldehyde and then embedded in paraffin, cut into 4 -μm-thick sections, and the sections were dewaxed with xylene and rehydrated with different gradients of ethanol. The sections were incubated with 3% BSA for 30 minutes at room temperature for closure, and after antigen repair, the primary antibody was added and incubated overnight at 4°C. The sections were washed 3 times with PBS for 5 minutes each time, and then horseradish peroxidase-linked secondary antibody was added and incubated for 50 minutes at room temperature avoiding light. Finally, the samples were incubated with diaminobenzidine (DAB) for color development reaction and then restained with hematoxylin. The results were analyzed by ImageJ software.
Immunofluorescence Assay
Cells were fixed by cell shaking, sections were washed, antigen repair was performed, sections were incubated with 3% hydrogen peroxide solution for 25 minutes at room temperature and protected from light, endogenous peroxidase was blocked, the primary antibody was added and incubated overnight at 4°C, PBS was washed, secondary antibody was incubated for 50 minutes at room temperature and protected from light, followed by tyrosine fluorescein incubation for 20 minutes at room temperature, DAPI re-staining of nuclei, blocking of sections, and microscopic examination for photographs.
Statistical Analysis
All data were analyzed by GraphPad Prism 9 software and expressed as mean ± SD. Data were analyzed using Student’s t-test or ANOVA. For all results, P < .05 was considered statistically significant (*).
Results
HQZX Decoction Inhibits the Growth of Multiple Human Hepatocellular Carcinoma Cells in an in vivo Hollow Fiber Hepatocellular Carcinoma Model
To investigate the pharmacodynamic effects of HQZX decoction on the in vivo growth of different hepatocellular carcinoma cells, a nude mouse hollow fiber hepatocellular carcinoma model was constructed, and the pharmacodynamic effects of HQZX decoction and a positive control drug, HCS, were evaluated in high throughput in a short period on 6 different human hepatocellular carcinoma cell lines, as well as on PDXC (Figure 1). The heat maps in Figure 2A and B represent the OD values of subcutaneously implanted and peritoneally implanted hollow fiber tubes, respectively. Inspecting OD values of all hollow fiber tubes, it can be seen that the growth rates of different liver cancer cell lines in vivo are different. Figure 2C shows that after implantation of the hollow fiber tubes, the weight of the mice decreased slightly, but after the administration of HQZX decoction and HCS by gavage starting on the third day, respectively, the weight of the mice in each group showed an increasing trend, which indicated that the mice could gradually tolerate the hollow fiber tubes and that HQZX decoction had no obvious toxic side effects on mice. Figure 2D and E then calculated the inhibition rate of HQZX decoction and HCS on the cells of each tube in the subcutaneous and peritoneal cavity respectively, and it could be seen that the peritoneal efficacy was better than the subcutaneous efficacy. The number of hollow fiber tubes with an efficacy rate of >30% of the intraperitoneal implantation was more than that of the subcutaneous implantation by HQZX decoction (Table S2). The inhibition rates of HQZX decoction and HCS on cells were calculated by OD value, as in Figure 2F. We can see that HQZX decoction had different degrees of inhibitory effects on LM3-luc, Huh7, HepG2, and PDXC, and the efficacy of HQZX decoction was shown as LM3-luc > PDXC > HepG2 > Huh7, which was dose-dependent, with high doses > higher doses > Huh7. For Hep3B and MHCC97H, HQZX decoction had an inhibitory effect on peritoneally implanted cells, and only the high dose of HQZX decoction had an inhibitory effect on subcutaneously implanted cells. For MHCC97L, only medium- and high-dose HQZX decoction was effective on peritoneally implanted cells, but not on subcutaneously implanted cells.

A schematic diagram for evaluating the efficacy of HQZX decoction on different human hepatocellular carcinoma cells based on the hollow fiber liver cancer model.

HQZX inhibited the growth of multiple human hepatocellular carcinoma cells in vivo. (A) The heat map made with the OD value of all hollow fiber tubes implanted subcutaneously (s.c.). (B) The heat map made with the OD value of all hollow fiber tubes implanted intraperitoneally (i.p.). (C) The changes in body weight of mice after implantation of hollow fiber tubes. (D) The efficacy of different doses of HQZX and HCS on cells within subcutaneously implanted hollow fiber tubes. (E) The efficacy of different doses of HQZX and HCS on cells within intraperitoneally implanted hollow fiber tubes. (F) The inhibition rates of different doses of HQZX and HCS on different hepatocellular carcinoma cells implanted subcutaneously and intraperitoneally were calculated by OD values. Error bars indicate SD.
Subcutaneous Transplantation Tumor Model Validates the Efficacy of HQZX Decoction in an in vivo Hollow Fiber Hepatocellular Carcinoma Model
To verify the accuracy of the efficacy of HQZX decoction in the hollow fiber liver cancer model, for LM3-luc and PDXC, which have the most pronounced efficacy of HQZX decoction, the subcutaneous transplantation tumor model of nude mice was applied for the validation of the efficacy of HQZX decoction. 15 After 21 days of high-dose HQZX decoction administration by gavage, the LM3-luc tumor mass and PDXC tumor mass in the HQZX group were smaller than the tumor mass size in the control group (Figure 3A and F), Tumor tissues of the 2 groups were taken for pathological comparison, as shown in Figure 3E and J. The control group had the most serious damage to the tumor tissues, abnormal structure of the cancer tissues, pathological nuclear division phase of some cancer cells, and a small amount of inflammatory cell infiltration in the tissues, which was dominated by neutrophils; while tumor tissues of the HQZX group had less serious damage, the normal structure of the cancer tissues, and the cancer cells did not have a pathological nuclear division phase, and the tissues did not have an obvious inflammatory cell infiltration. cell infiltration. As shown in Figure 3B to D, HQZX decoction reduced the weight, tumor weight-to-mouse body weight ratio, and volume of LM3-luc tumor blocks, and similarly, HQZX decoction reduced the weight, tumor weight-to-mouse body weight ratio, and volume of PDXC tumor blocks (Figure 3G-I).

HQZX inhibits the growth of hepatocellular carcinoma subcutaneous graft tumors in nude mice. (A) The representative pictures of each group of LM3-luc tumor masses. (B) The LM3-luc tumor weight of each group of nude mice. (C) The ratio of LM3-luc tumor weight to body weight in each group of nude mice. (D) The volume of LM3-luc tumor masses in each group of nude mice. (E) The representative pictures of the LM3-luc tumor masses were analyzed by H&E staining. Scale bar, 50 μm. (F) The representative pictures of each group of PDXC tumor masses. (G) The PDXC tumor weight of each group of nude mice. (H) The ratio of PDXC tumor weight to body weight in each group of nude mice. (I) The volume of PDXC tumor masses in each group of nude mice. (J) The representative pictures of the PDXC tumor masses were analyzed by H&E staining. Scale bar, 50 μm. Continuous variables are presented as mean and standard deviation as examined by independent samples t-test (*P < .05, **P < .01, ***P < .001); error bars indicate SD.
HQZX Decoction Inhibited Metastasis and Invasion of LM3-luc Cells in vitro
Comprehensive pharmacodynamic evaluation of HQZX decoction in hollow fiber hepatocellular carcinoma models showed that HQZX decoction had the best inhibitory effect on the growth of the human hepatocellular carcinoma cell line LM3-luc, which is a highly metastatic hepatocellular carcinoma cell line, and thus, we investigated the metastatic invasive effect of HQZX decoction on LM3-luc cells in vitro. effect. As Figure 4A indicates the cell viability after gradient administration of different concentrations of HQZX decoction, in vitro intervention of cells after 24, 48, and 72 hours, the IC50 concentration of 3 mg/ml after 24 hours of intervention, as well as 1/2 IC50 concentration of 1.5 mg/ml, were selected as the subsequent concentration of administration. As Figure 4B and C cell scratch experiments can be concluded, HQZX decoction administration inhibited wound healing, indicating the inhibition of cell migration. Transwell experiments showed that HQZX decoction intervention for 24 hours inhibited cell invasion and migration (Figure 4D-F). In addition, apoptosis is also one of the key factors regulating the efficiency of tumor metastasis. The effect of apoptosis was detected by flow cytometry after HQZX intervention for 24 h. The results showed that HQZX promoted cells to undergo early apoptosis, and a high concentration of intervention resulted in a high rate of early apoptosis (Figure 4G and H).
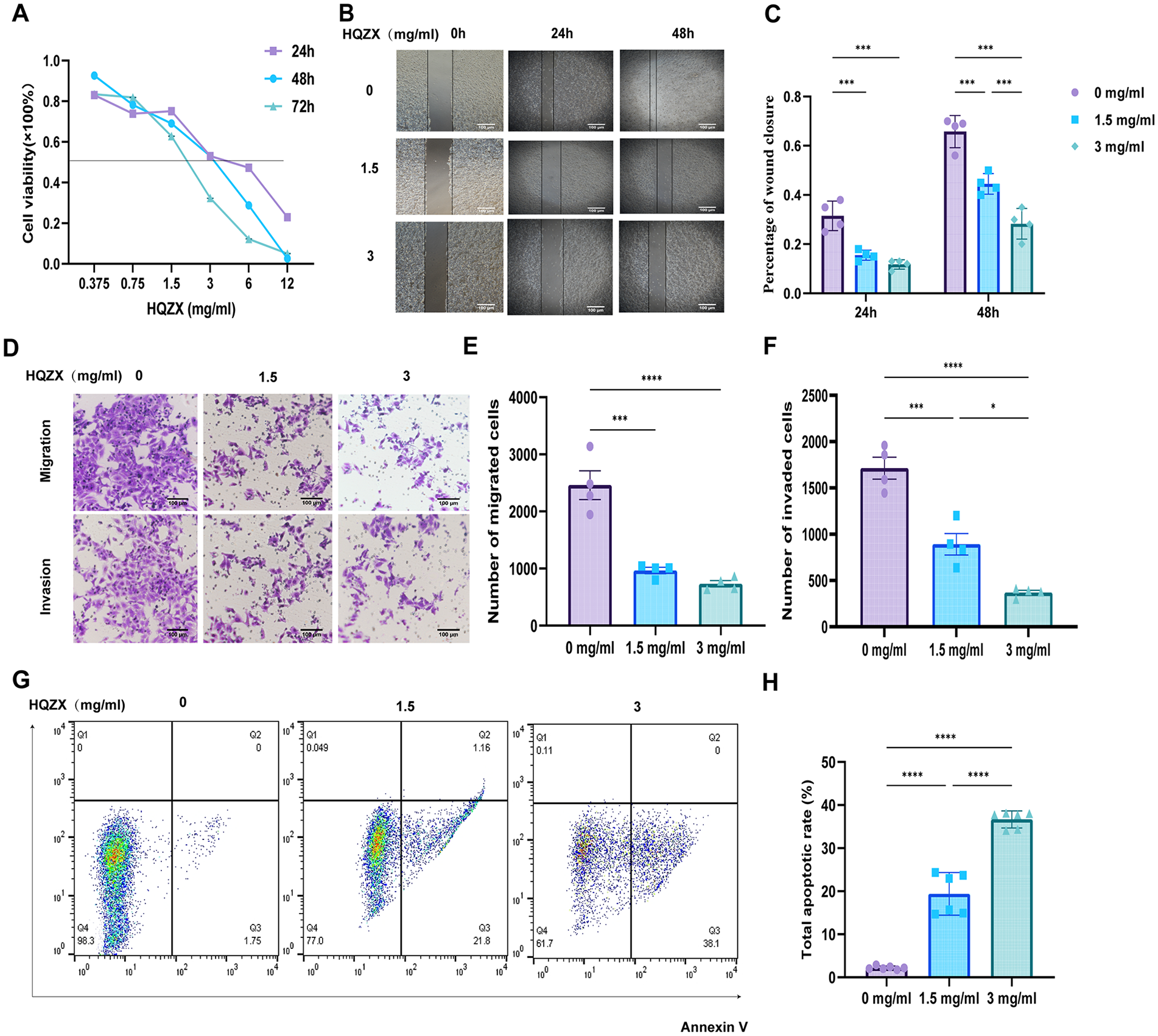
HQZX inhibits the growth and metastasis of human hepatoma cells LM3-luc in vitro. (A) The cell viability was measured by CCK8 assay after 24, 48, and 72 hours administration of HQZX at different concentrations. (B and C) The cell migration ability of different concentrations of HQZX was measured by scratch wound healing assay after 24 and 48 hours of intervention. Scale bar, 100 μm. (D-F) The cell migration and invasion ability were detected by transwell migration and matrigel invasion assays after 24 h intervention with different concentrations of HQZX. Scale bar, 100 μm. (G and H) The apoptosis rate after different concentrations of HQZX intervention was detected by flow cytometry. Student’s t-test and two-way ANOVA was used (*P < .05, ***P < .001, ****P < .0001); error bars indicate SD.
HQZX Decoction in vivo Inhibited the Expression of Molecules Related to EMT and LCSC
To analyze the mechanism of metastasis inhibition by HQZX decoction, we also relied on the hollow fiber hepatocellular carcinoma model, and extracted cells after drug intervention for cellular phenotyping. 16 Cells in the fibrous ducts of the 2 groups were extracted separately and counted, as shown in Figure 5A and B. It can be seen that the number of cells in the drug administration group was significantly less than that in the control group. The expression of HIF-1α can directly or indirectly regulate the expression of related molecules, such as EMT and LCSC, that are related to metastasis.17,18 Promoting tumorigenesis and metastasis, Figure 5C to E By qrt-PCR and WB experiments, it can be seen that HQZX decoction promotes the mRNA and protein expression of the epithelial cell marker molecule E-cadherin, and inhibits the expression of the mesenchymal cell marker molecules N-cadherin and Vimentin. Hepatocellular carcinoma stem cells are also important factors promoting metastasis and can be inter-transformed with EMT. 19 HQZX decoction down-regulated the expression levels of CD133 and CD44 in hepatocellular carcinoma stem cells, as shown by flow cytometry analysis.

HQZX regulates the expression of EMT, CD44 in vivo. (A) The representative pictures of H&E staining of cells extracted in hollow fiber tubes. Scale bar, 25 μm. (B) The number of cells extracted from the hollow fiber tubes. (C-E) The expression levels of E-cadherin, N-cadherin, and Vimentin were detected by Western blot and qRT-PCR assay. (F and G) The expression of CD44 and CD133 was detected by flow cytometry. Student’s t-test and two-way ANOVA were used (*P < .05, **P < .01, ***P < .001, ****P < .0001); error bars indicate SD.
HQZX Decoction Inhibits the HIF-1α Signaling Pathway in vivo and Suppresses Angiogenesis
Multiple studies have shown that several medicinal constituents in HQZX decoction can down-regulate the expression of HIF-1α and VEGF. Thus, to further investigate the mechanism of tumor metastasis inhibition by HQZX decoction, we analyzed the expression of HIF-1α and VEGF. Cells were extracted from a hollow fiber hepatocellular carcinoma model, and the intensity of cellular molecular expression was analyzed by immunofluorescence and immunohistochemistry staining, as can be seen in Figure 6A to E. In the HQZX group, the HIF-1α and VEGF expression of the fluorescence intensity and cell positivity were decreased. In Figure 6F and G, as shown by qrt-PCR and WB experiments, mRNA and protein expression of HIF-1α and VEGF were down-regulated in the HQZX group. Figure 6B shows by immunohistochemical staining of CD31 that treatment with traditional Chinese medicines significantly inhibited peripheral angiogenesis. In Figure 6I, for the mice subcutaneous hollow fibrous tubes, surrounding skin tissues were photographed, and it was visualized that the vascular network around the hollow fibrous tubes in the HQZX group was finer in contrast, lighter in color, and with less angiogenesis.

HQZX inhibits the expression of the HIF-1α/VEGF signaling pathway and suppresses angiogenesis in vivo. (A, C, and D) The expression of HIF-1α and VEGF were detected by cell immunofluorescence assay. Scale bar, 50 μm. (B and E) The expression of HIF-1α, VEGF, and CD31 were detected by IHC staining assay. Scale bar, 50 μm. (F) The expression of HIF-1α and VEGF were detected by qRT-PCR assay. (G and H) The expression of HIF-1α and VEGF were detected by Western blot assay. (I) The representative pictures of the blood vessels around the periphery of the hollow fiber tubes. Two-way ANOVA was used (*P < .05, **P < .01, ***P < .001, ****P < .0001); error bars indicate SD.
Splenic Injection Liver Metastasis Model Further Verified That HQZX Decoction Inhibited Tumor Metastasis in vivo in Nude Mice
To verify whether HQZX decoction inhibited tumor metastasis in vivo, a nude mice splenic injection liver metastasis model was constructed, and the liver metastasis was observed to determine whether there was liver metastasis or not after 21 days of drug administration. As shown in Figure 7A, the liver tumor mass in the control group existed diffusely, with a large number of tumor masses and the spleen became larger; the liver in the HQZX group was smoother, and the number of tumor masses was significantly smaller. The fluorescence intensity of liver tumor masses was observed by in vivo imaging in mice as in Figure 7B, and HQZX decoction treatment significantly reduced the fluorescence intensity of liver tumor masses in mice. The weight of the spleen, number of liver tumors, and weight of the liver were compared between the 2 groups, respectively, as shown in Figure 7C-E. After HQZX decoction treatment, the number of liver tumors was reduced, and the weights of the liver and spleen were smaller than those of the control group. This indicates that HQZX decoction administration inhibited tumor metastasis in mice.

HQZX inhibits liver cancer metastasis in vivo. (A) The representative pictures of the liver and spleen of each group of nude mice. (B) Detection of liver metastases from spleen-injected hepatocellular carcinoma cells by in vivo live imaging assay. (C) The weight of the spleen of each group of nude mice. (D) The number of tumor metastases in each group of nude mice. (E) The weight of the liver of each group of nude mice. Continuous variables are presented as mean and standard deviation as examined by independent samples t-test (**P < .01, ***P < .001); error bars indicate SD.
Discussion
Recurrence and metastasis have become one of the main reasons for the low long-term survival and poor prognosis of HCC patients. Inhibition of tumor angiogenesis plays an important role in inhibiting tumor growth and metastasis. Angiogenesis, the growth of new blood vessels from pre-existing ones through a process known as germination, is one of the hallmarks of cancer. VEGF, which is overexpressed in the vast majority of solid tumors, is widely regarded as a key player in mediating tumor angiogenesis, and anti-VEGF therapy has even led to a significant reduction in metastasis. 20 Epithelial-mesenchymal transition, which causes epithelial cells to lose their original gene expression signature and gain a mesenchymal gene expression signature, is a key event in cancer recurrence and metastasis and positively correlates with the stem cell potential of tumor cells. EMT causes cells to lose their ability to adhere to neighboring cells and appear to have enhanced migratory properties, 21 and in a cohort of 150 patients, E-cadherin down-regulation was correlated with vascular invasion and extrahepatic recurrence. 22
In addition, studies have confirmed that there are multiple subpopulations of cancer cells in HCC tumors that exhibit intrinsic differences in tumor behavior and that liver cancer stem cells are a special subpopulation of tumor cells, 23 which have the potential for continuous self-renewal and multidirectional differentiation, and can contribute to hepatocellular carcinoma development and postoperative recurrence. CD44 and CD133, which have been demonstrated to be several markers common to liver cancer stem cells, are more effective in liver cancer stem cell development than other tumor cell types. LCSCs can circulate in vivo, significantly promoting distant metastasis and homing ability. In other words, LCSCs promote tumor growth of primary cancer cells and metastasis of transplanted secondary tumors, leading to recurrence of HCC, 24 and are at the root of tumor recurrence and metastasis.
HIF-1α is a key transcription factor involved in the hypoxia response of cancer cells, and HIF-1α overexpression has been detected in HCC,25,26 which is closely related to the growth and metastasis of HCC. 27 HIF-1α activation drives vascular endothelial growth factor (VEGF) production and angiogenesis, inhibits hypoxia-induced HIF-1α protein synthesis, and blocks the HIF-1α/VEGF pathway. Clinical reduction of HIF-1α, VEGF expression and microvessel density by drug therapy can prolong the time to recurrence. 28 HIF-1α prolongs the time to recurrence by regulating EMT-related signaling pathways (TGF-β, Notch, Wnt/β-catenin, and NF-κB),29 -32 modulation of EMT-TF expression33,34 and EMT-associated miRNA and lncRNA networks in tumors 35 to stimulate EMT. In addition, Stem cell-like spherocytes have higher levels of HIF-1α mRNA expression.17,36,37 Several medicines and active ingredients in HQZX decoction played a role in inhibiting tumor metastasis. 38
The study was completed based on a hollow fiber hepatocellular carcinoma model. The hollow fiber assay has multiple advantages for antitumor drug screening, such as high throughput, all-fidelity, efficiency, savings, and universal applicability. Compared with the in vitro assay, it can simulate the full-fidelity in vivo environment, and the preliminary screening of compounds can be performed in high throughput (multiple cell lines in one mouse), which greatly improves the speed of in vivo drug testing. And it has a good correlation with the xenograft test in drug screening, overcoming the shortcomings of xenograft experiments such as their time-consuming and costly nature, consumption of experimental animals, and consumption of experimental consumables. In this study, hepatocellular carcinoma cells were injected into hollow fiber tubes, a hollow fiber hepatocellular carcinoma model was constructed, and the efficacy of HQZX decoction was evaluated against 6 medium hepatocellular carcinoma cell lines and a PDXC. The results were confirmed to be relevant by transplantation tumor experiments.
In the current process of antitumor drug development, HFA can be applied not only to the evaluation of antitumor natural drugs and the pharmacodynamic evaluation of antitumor compounds 39 but also to the study of drug molecular mechanism based on genomics and proteomics. 40 In addition, HFA shows important application value in the in vivo evaluation of new drugs. In vivo preclinical analysis needs to screen potential drugs targeting tumor vasculature. This study extracted cells inside hollow fiber tubes for WB experiments, qRT-PCR experiments, immunofluorescence, immunohistochemical staining, and HE staining experiments, which confirmed that the mechanism of metastasis inhibition by Hu-Qi-Zheng-Xiao down-regulated the expression of metastasis-related molecules, EMT and LCSC, which was related to the inhibition of HIF-1α, and the inhibition of the HIF-1 α/VEGF signaling pathway, inhibited peripheral vasculogenesis of hollow fiber tubes and had the effect of inhibiting tumor growth and metastasis. The study demonstrated that it was feasible to conduct mechanism studies with this method. And because of the in vivo culture mode of hollow fiber tubes, the results were more reliable than those of in vitro experiments.
Conclusion
In summary, this study confirmed the anti-tumor growth effect and metastasis inhibition of HQZX decoction on a variety of hepatocellular carcinoma cell lines based on the hollow fiber hepatocellular carcinoma model, which inhibited the HIF-1α signaling pathway, suppressed the expression of EMT-related and LCSC-related molecules, and inhibited angiogenesis related to the inhibition of the HIF-1α/VEGF signaling pathway. This may explain the clinical prolongation of patient survival with this complex formula, suggesting that HQZX decoction is a clinically promising herbal treatment option for HCC. However, given the complex composition of the herbal formula, more research is needed to identify the specific active compounds in HQZX decoction. This study had some limitations and did not target HIF-1α, suggesting a direct correlation between the antitumor effects of HQZX and its. In future studies, we will look for the upstream of HIF-1α that HQZX decoction acts on, aiming to elucidate the mechanism by which HQZX targets the HIF-1α signaling pathway to inhibit hepatocellular carcinoma growth and metastasis.
Supplemental Material
sj-docx-1-ict-10.1177_15347354231226126 – Supplemental material for Hu-Qi-Zheng-Xiao Decoction Inhibits the Metastasis of Hepatocellular Carcinoma Cells by Suppressing the HIF-1α Signaling Pathway to Inhibit EMT, LCSC, and Angiogenic Process
Supplemental material, sj-docx-1-ict-10.1177_15347354231226126 for Hu-Qi-Zheng-Xiao Decoction Inhibits the Metastasis of Hepatocellular Carcinoma Cells by Suppressing the HIF-1α Signaling Pathway to Inhibit EMT, LCSC, and Angiogenic Process by Xuejing Wang, Ling Yin, Mengyin Chai, Buxin Kou, Xiaoni Liu and Xiaojun Wang in Integrative Cancer Therapies
Footnotes
Acknowledgements
Xuejing Wang proposed the research ideas and was responsible for the experimental design, all the experimental contents and the completion of the manuscript; Ling Yin, Buxin Kou and Chai Mengyin helped to purchase nude mice; Xiaoni Liu provided experimental consumables; Xiaojun Wang provided financial support.
Date Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Special scientific research project of Capital Health Development (No. 2022-2-2186) and the Clinical Technology Innovation Project of “Yangfan” Program of Beijing Hospital Management Center (ZLRK202332).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
