Abstract
Aim:
The aim of this study was to analyse the effiacy of HeXue Tongbi Formula in the treatment of oxaliplatin-induced perpheral neuropathy.
Method:
An open randomized, non-blind, controlled study was conducted at the First Affiliated Hospital of Guangzhou University of Traditional Chinese Medicine from September 2019 to December 2020. A total of 78 maligant tumor patients receiving oxaliplatin-containing chemotherapy were recruited, with half of them receiving HeXue Tongbi Formula for 4 cycles of 21 days. The study assessed the incidence and severity of perpheral neuropathy and the safety of HeXue Tongbi Formula.
Result:
After 4 cycles of treatment, the incidence of perpheral neuropathy in the treatment group was significantly lower than that in the control group (30.77% versus 84.62%, P < .05). The severity of perpheral neuropathy in the treatment group increased sligthly and stabilized from the third cycle, whlie it gradually increased in the control group. yhere were no severe adverse reactions to HeXue Tongbi Formula.
Conclusion:
HeXue Tongbi Formula demonstrated good preventive and therapeutic effects on oxaliplatin-induced perpheral neuropathy.
Trial registration:
This trial has been registered with the Chinese Clinical Trial Registry (ChiCTR2000032996).
Keywords
Introduction
Chemotherapy is commonly used in antineoplastic therapy, but all chemotherapy drugs have different degrees of toxicity and side effects. These effects can lead to treatment interruptions, reduce the quality of life of patients, and even cause death. Therefore, addressing the toxic and side effects of chemotherapy is crucial. Chemotherapy-induced peripheral neuropathy (CIPN) is a common side effect, with acute peripheral nerve paresthesia being the most common symptom. Patients may experience abnormal pain, temperature sensations, and touch sensations in their extremities, often described as “glove-stocking” distribution. Even slight touch can cause pain and hypersensitivity, which may be exacerbated by cold stimulation and may even lead to laryngospasm. 1 Chronic neurotoxicity can also occur with prolonged exposure to chemotherapeutic drugs, leading to movement disorders, typically manifested as weakness in the hands and feet. 2 Additionally, it can result in autonomic nerve function disorders, including postural hypotension, arrhythmia, digestive tract dysfunction, and erectile dysfunction. 3 Among commonly used chemotherapeutic drugs, platinum, bortezomib, tubulin inhibitors, and glutamic acid derivatives often induce CIPN, with oxaliplatin being the most common culprit. Approximately 90% of patients receiving oxaliplatin-containing chemotherapy regimens experience peripheral neuropathy, significantly impacting their quality of life. 4 Therefore, effective interventions are urgently needed to alleviate these side effects. In this study, we conducted an open-label randomized controlled trial to observe and analyze the efficacy of HeXue Tongbi Formula in treating oxaliplatin-induced peripheral neuropathy, aiming to provide evidence-based medical support for its clinical application.
Method
Research Design
We conducted an open-label, randomized, non-blinded, controlled study at the First Affiliated Hospital of Guangzhou University of Traditional Chinese Medicine from September 2019 to December 2020.
Research Objects
The study subjects were malignant tumor patients and hospitalized at the First Affiliated Hospital of Guangzhou University of Traditional Chinese Medicine from September 2019 to December 2020. The following were the criteria for inclusion: aged 18 years or older; both male and female; Eastern Cooperative Oncology Group Performance Status (ECOG PS) score of 0 to 2; no history of prior chemotherapy, fulfillment of criteria for chemotherapy; and planned administration of oxaliplatin-containing chemotherapy with a life expectancy longer than 3 months. The following were the criteria for exclusion: patients with other neuropathic diseases; patients with bone and joint diseases and peripheral paresthesia resulting from diabetes; patients with peripheral neuropathy caused by poisoning, infection, or radiotherapy; patients with other concurrent skin diseases; patients receiving other medications that may affect peripheral nervous system symptoms; subjects with mental illness, alcoholism, nicotine dependence, or psychotropic substance abuse that could potentially cause neurological impairment, and those with known allergies to relevant traditional Chinese medicine.
Sample Size
We estimated that a sample size of 60 patients (30 patients in each group) would be sufficient to detect a 40% difference (80% for the control and 40% for the treatment group) in the incidence of CIPN after 4 cycles, with a power of 90% and a 2-sided significance levels of .05 for the analysis of the primary outcome. Accounting for a 20% potential drop-out rate, we planned to enroll a total of 76 patients (38 patients in each group).
Grouping
The groups were divided into a treatment group and a control group with a 1:1 ratio. As we have not found other factors, such as disease severity or age, could markedly influence the incidence of CIPN, apart from the obvious correlation with the cumulative dose of oxaliplatin, we employed a simple randomization method for the random allocation. Randomization concealment was implemented using sealed envelopes.
Interventions
All enrolled patients received a chemotherapy regimen containing intravenous oxaliplatin at 85 mg/m² biweekly or 130 mg/m² triweekly. Additionally, standard CIPN preventive education was provided, including practices such as using warm water for oral hygiene, avoiding cold drinks, consuming warm and soft foods, soaking fruits in hot water before consumption, and maintaining limb movement within the allowable range to prevent muscle atrophy.
The treatment group received Hexue Tongbi formula, an external powder decoction provided by the First Affiliated Hospital of Guangzhou University of Traditional Chinese Medicine. The formula consisted of 10 g Aconiti radix cocta, 10 g Cassia twig, 10 g Manchurian wild ginger, 10 g Armand clematis stem, and 10 g Angelica dahurica. These traditional Chinese medicines were crushed, sieved through a 300-mesh screen, and sterilized to prepare external Chinese medicine powder. For use, the Hexue Tongbi formula powder decoction was mixed with distilled water according to each packet’s instructions (specification: 50 g) in a ratio of 1:20 (powder:water) and boiled. The solution was utilized for the fumigation and soaking of hands and feet, with participants immersing their extremities twice daily for 30 minutes per session at a temperature range of 40°C to 45°C in the temperature-controlled foot bath basin. A total of 4 cycles of traditional Chinese medicine treatment were administered with each cycle lasting 3 weeks. If patients experienced skin hypersensitivity, pruritus, skin chapping, ulceration, or skin rupture, the administration of the formula was discontinued. The control group was given the same topical washing procedure as the treatment group, with the only difference being that the medicinal solution was substituted with warm water at a temperature range of 40°C to 45°C.
Follow-Up Methods
Peripheral neuropathy was evaluated with National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE). Total Neuropathy Score (TNS) and European Organization for Research and Treatment of Cancer Quality of Life Questionnaire–Chemotherapy-Induced Peripheral Neuropathy 20-Item Scale (EORTC QLQ-CIPN20) were also used to evaluate the incidence and severity of CIPN. The EORTC QLQ-CIPN20, which includes sensory, motor, and autonomic subscales, was shown to be responsive to changes in peripheral neuropathy. 5 The subjects were followed up via telephone or face-to-face interviews every 3 weeks by investigators who were unaware of the patients’ treatment group.
Outcomes
The primary outcome was the incidence of peripheral neuropathy after 4 cycles of treatment assessed by NCI CTCAE. NCI CTCAE entailed a grading system, whereby grade 0 denotes an asymptomatic state and grade 1 is characterized by the presence of slight loss of tendon reflexes or paresthesia, without impacting function. Grade 2 is indicative of moderate loss of tendon reflexes or paresthesia, which limits instrumental daily activities. Grade 3 is indicative of severe loss of tendon reflexes or paresthesia, which restricts the ability to perform activities of daily living, while grade 4 is indicative of a loss of function or a life-threatening condition. A grading of 1 to 4 indicated the presence of peripheral neuropathy. The secondary outcomes included changes in peripheral neuropathy scores as evaluated by TNS and EORTC QLQ-CIPN20. Safety was also assessed in the present study.
Statistical Analysis
R software (version 4.0) and EmpowerStats (version 4.0) were utilized for statistical analysis. Clinical features at baseline were presented as mean ± standard deviation (X ± S) for quantitative data, analyzed using the T-test (for normal distribution) or Kruskal-Wallis test (for non-normal distribution). Categorical variables were expressed as frequencies and analyzed using the Chi-square test. The incidence of peripheral neuropathy over time was presented as a percentage and analyzed using the Chi-square test. A repeated-measures analysis of variance (ANOVA) was used to analyze the between-group effects of TNS and EORTC QLQ-CIPN20 at various time points. We also employed generalized estimating equation (GEE) to analyze longitudinal data such as TNS and QLQ-CIPN20 for time effect, with the categorical variables of the cycle being converted to continuous variables. A significance level of P < .05 was considered statistically significant.
Results
Enrollment
A total of 83 patients were screened for the study. Five patients were excluded: two due to allergies to the herbal medicine under investigation, one because of diabetic peripheral neuropathy, and two with neuropathy resulting from radiotherapy. Therefore, 78 patients were enrolled and included in the final analysis with 3 patients dropping out due to tumor progression and 2 patients withdrawing from the study because of hand and foot rash after using the Hexue Tongbi formula (Figure 1).
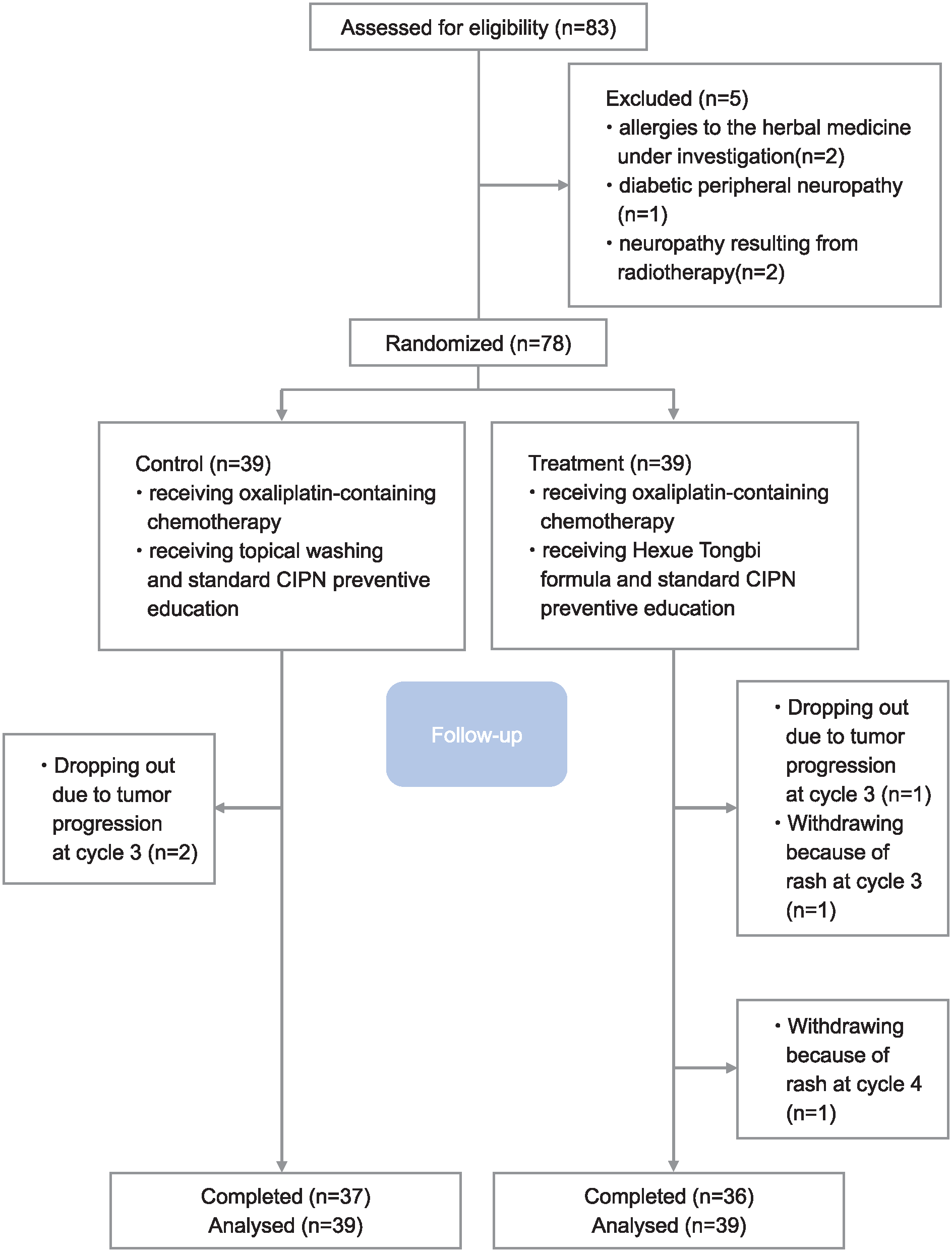
CONSORT flow chart.
Patient Characteristics
This study included a treatment group and a control group, each consisting of 39 individuals. Among all patients included in the study, there were 49 males (62.82%) and 29 females (37.18%). The mean age was 55.22 ± 11.29 years. Regarding tumor types, there were 19 patients with rectal adenocarcinoma, 50 patients with colonic adenocarcinoma, 3 patients with gastric adenocarcinoma, 3 patients with liver cancer, 1 patient with cholangiocellular carcinoma, and 2 patients with esophageal carcinoma. According to tumor stage, 2 patients (2.56%) were in stage II, 18 patients (23.08%) were in stage III, and 58 patients (74.36%) were in stage IV. Additionally, 22 patients (28.21%) received postoperative adjuvant chemotherapy, while 56 patients (71.79%) received palliative chemotherapy. Aside from 3 patients who dropped out due to disease progression, all other patients were administered a nearly full therapeutic dose of oxaliplatin, with a cumulative dose ranging from 500 to 520 mg/m². The baseline characteristics of patients in both groups were balanced (Table 1).
Baseline Characteristics.

Incidence of Peripheral Neuropathy in the 2 Groups
The incidence of peripheral neuropathy in both groups was assessed and presented in Table 2. There was no significant difference in the incidence of peripheral neuropathy between the 2 groups (P > .05) during the first and second cycle. However, starting from the third cycle, the incidence of peripheral neuropathy in the treatment group gradually became lower than that in the control group. Statistical significance was observed from the fourth cycle (P < .05).
Incidence of Peripheral Neuropathy in the 2 Groups.

Degree of Peripheral Neuropathy in the 2 Groups
TNS score
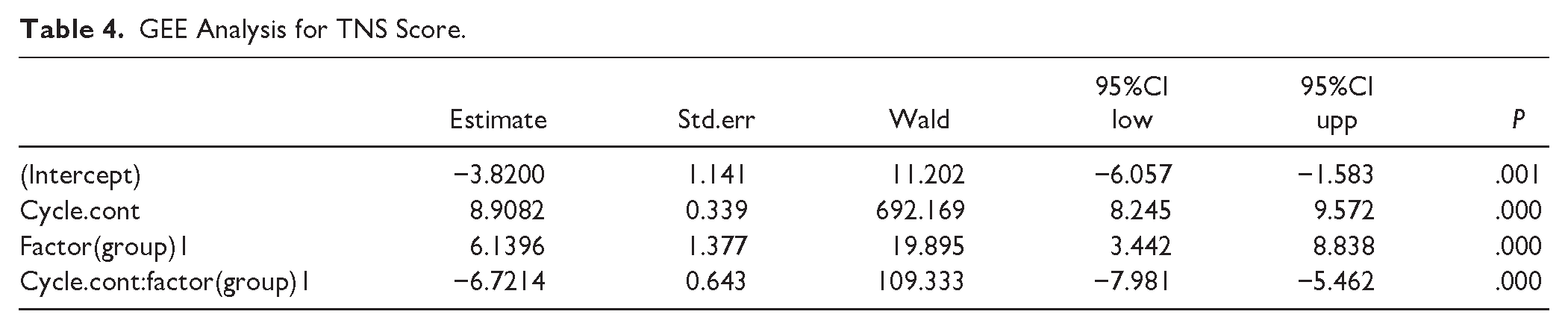
The results of the statistical analysis demonstrated that the TNS scores exhibited notable variability at distinct time points following the administration of the drug, exhibiting a gradual increase with extended measurement intervals (P = .000). Over the course of the study, the TNS scores within the control group demonstrated a consistent upward tendency, indicative of a notable dose-dependent effect. Conversely, the treatment group displayed an initial slow ascent in scores, which was subsequently followed by a downward trend. The results of GEE indicated that the interaction effect of group and cycle was statistically significant (P = .000), suggesting that the changes in TNS scores over time between the treatment group and the control group were significantly different (see Table 4 and Figure 2). Furthermore, according to Table 3 and Figure 2, there was no significant difference in the TNS score between the treatment group and the control group at the first and second cycles (P > .05), whereas at the third and fourth cycles, the TNS scores of the treatment group and the control group were significantly different (P < .05).
Repeated Measures ANOVA Analysis of TNS Score.
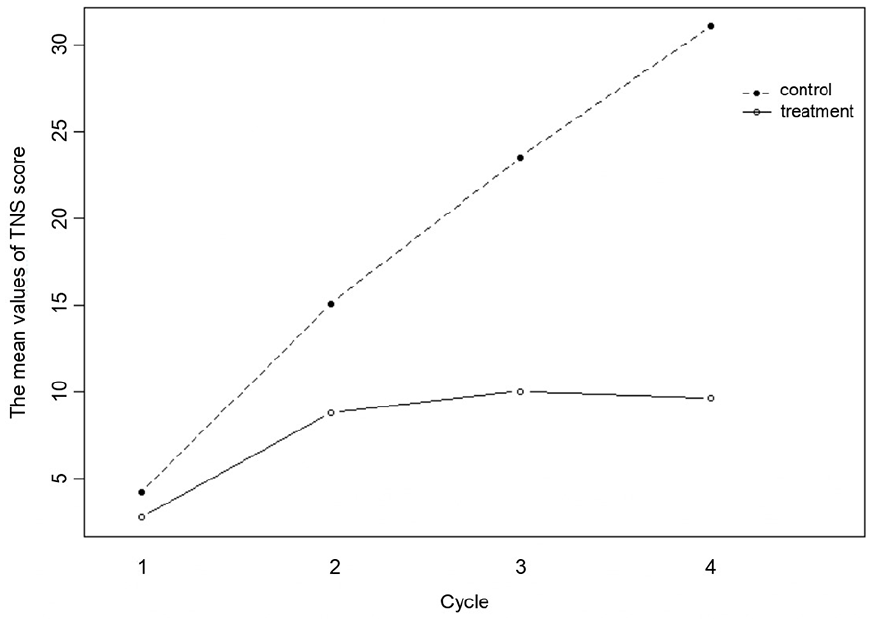
Changes in TNS scores over cycles.
GEE Analysis for TNS Score.

EORTC QLQ-CIPN20 score
The results of the severity of peripheral neuropathy based on the EORTC QLQ-CIPN20 were consistent with the TNS findings mentioned above. According to Table 5 and Figure 3, there was no significant difference in the EORTC QLQ-CIPN scores between the treatment group and the control group at the first and second cycles (P > .05). However, at the third and fourth cycles, there was a significant difference in the EORTC QLQ-CIPN scores between the treatment group and the control group (P < .05). Furthermore, according to the analysis results of GEE, there is an interaction between treatment and time, meaning that as time prolongs, the effect of the treatment group becomes increasingly significant (Table 6). Over time, the EORTC QLQ-CIPN scores in the control group increased gradually, while those in the treatment group increased slowly and then remained stable (Figure 3).
Repeated Measures ANOVA Analysis of EORTC QLQ-CIPN Score.

GEE Analysis for EORTC QLQ-CIPN Score.


Changes in EORTC QLQ-CIPN20 score over cycle.
Adverse Reactions
Among the 78 patients included in the study, no adverse reactions were reported except for 2 patients who developed obvious skin rash and pruritus after medication. The rash in these 2 patients resolved within 3 days after discontinuation of the drug.
Discussion
We conducted this preliminary RCT to explore the efficacy of Hexue Tongbi formula for CIPN. With the accumulation of oxaliplatin dose, the incidence of peripheral neuropathy increased in both groups, from average 5.13% in the first cycle to 30.77% (the treatment group) and 84.62% (the control group) in the last cycle respectively, indicating that the higher the cumulative dose of oxaliplatin, the higher the incidence of neuropathy. A more important discovery is that the incidence of CIPN is significantly lower in the treatment group through 4-course treatments. And an interesting phenomenon is that the incidence of neuropathy in the treatment group did not increase but decreased in the last cycle compared to the third cycle, suggesting that Hexue Tongbi formula powder decoction for external use can improve some of the existing neuropathy. In conclusion, the incidence and severity of oxaliplatin-induced peripheral neuropathy was positively correlated with the cumulative dose of oxaliplatin. And, Hexue Tongbi formula powder decoction for external use can significantly reduce the incidence and severity of peripheral neuropathy. The strength of this study lies in our use of a randomized controlled trial and the adoption of multiple outcome scales for evaluation. And we employed various statistical methods such as ANOVA and GEE to analyze the data. Through multi-angle confirmation of the efficacy of the HexueTongbi Formula, we provide reliable a relatively reliable research conclusion.
The manifestations of CIPN are mainly limb numbness, pain and poor activity. In the traditional Chinese medicine theory system, CIPN is considered to be in Chinese medicine disease “Bi syndrome”. Deficiency of Qi and blood, blood stasis and cold coagulation are the pathogenesis of this disease. 6 Through years of clinical experience, we have gradually developed the methods of promoting blood circulation and removing blood stasis, warming Yang and dredging collaterals to improve CIPN, and formulated Hexue Tongbi formula (Aconiti radix cocta, Cassia twig, Manchurian wild ginger, Armand clematis stem and Angelica dahurica) to prevent and treat CIPN.7 -9
We have previously also conducted in vivo animal experiments to confirm that Hexue Tongbi formula is an effective treatment for oxaliplatin-induced peripheral neuropathy in rats. This efficacy is substantiated by a noticeable decrease in touch- and pain-related hypersensitivity, as well as a marked improvement and mitigation of the peripheral neuronal damage caused by oxaliplatin exposure. 10 Additionally, the formula’s constituents appear to possess analgesic properties. Furanocoumarin found in Angelica dahurica can regulate TRPV1 channel, significantly inhibiting neurotoxic injury in rat plantar tissue induced by formalin and alleviating pain. 11 The coumarin component of Angelica dahurica exhibits skin permeability, 12 making it suitable for topical application. Moreover, sitosterols, which are the primary active ingredients in akebia stem and Cassia twig, possess anti-inflammatory and analgesic attributes. These compounds can be absorbed through the skin to exert an analgesic effect. 13
The quest for effective CIPN treatment is of paramount importance. Although medications like duloxetine, an antidepressant, may be effective, they are not without considerable side effects such as nausea, malaise, headache, and fatigue, which may intensify the suffering of cancer patients. The Hexue Tongbi formula, meticulously crafted from the 5 traditional Chinese medicine components previously discussed, is engineered to leverage a potent synergistic effect that has been found to be highly advantageous in the prevention and treatment of oxaliplatin-induced peripheral neurotoxicity. Additionally, it boasts the benefits of low toxicity and minimal side effects.
Obviously, there was limitation to our study. It was not blinded, and both the participants and implementers were aware of the subgroups and medications, potentially leading to information bias. However, 2 scales (TNS and EORTC QLQ-CIPN20) were utilized to assess the changes in peripheral neuropathy scores, somewhat mitigating the bias.
Conclusion
The results of the present RCT trial indicated that Hexue Tongbi formula effectively reduced the incidence of peripheral neuropathy caused by oxaliplatin. Furthermore, it inhibited the exacerbation of symptoms or alleviated existing peripheral neuropathy symptoms, demonstrating the beneficial preventive and therapeutic effects of Hexue Tongbi formula on oxaliplatin-induced peripheral neuropathy.
Supplemental Material
sj-docx-1-ict-10.1177_15347354241291514 – Supplemental material for Clinical Efficacy of Hexue Tongbi Formula on Oxaliplatin-Induced Peripheral Neuropathy: A Randomized Controlled Study
Supplemental material, sj-docx-1-ict-10.1177_15347354241291514 for Clinical Efficacy of Hexue Tongbi Formula on Oxaliplatin-Induced Peripheral Neuropathy: A Randomized Controlled Study by Lingling Sun, Yiying Lv, Duo Li, Jingyu Feng, Zhuangzhong Chen and Lizhu Lin in Integrative Cancer Therapies
Supplemental Material
sj-docx-2-ict-10.1177_15347354241291514 – Supplemental material for Clinical Efficacy of Hexue Tongbi Formula on Oxaliplatin-Induced Peripheral Neuropathy: A Randomized Controlled Study
Supplemental material, sj-docx-2-ict-10.1177_15347354241291514 for Clinical Efficacy of Hexue Tongbi Formula on Oxaliplatin-Induced Peripheral Neuropathy: A Randomized Controlled Study by Lingling Sun, Yiying Lv, Duo Li, Jingyu Feng, Zhuangzhong Chen and Lizhu Lin in Integrative Cancer Therapies
Footnotes
Author Contributions
Conceptualization: Lingling Sun, Lizhu Lin; Project administration: Lingling Sun, Lizhu Lin; Investigation: Lingling Sun, Duo Li, Jingyu Feng; Recruitment: Lingling Sun, Jingyu Feng, Zhuangzhong Chen, Lizhu Lin; Data analysis: Lingling Sun, Yiying Lv, Jingyu Feng; Writing—original draft: Lingling Sun, Yiying Lv, Jingyu Feng; Writing—review and editing: Lingling Sun, Lv Yiying, Duo Li. All authors read and approved the final manuscript.
Availability of Data and Materials
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Project of The First Affiliated Hospital of Guangzhou University of Chinese Medicine (grant number 2019ZWB10), Project of Guangzhou Municipal Science and Technology Bureau (grant number 2023B01J1016), Project of Administration of Traditional Chinese Medicine of Guangdong Province of China (grant number 2022ZYYJ03) and the Open Project of the Macau Institute for Translational Medicine and Innovation at the University of Macau (grant number SKL-QRCM-MITMI-ORP2303).
Ethics Approval and Consent to Participate
This study was approved by the Ethics Committee of Ethics Committee of the First Affiliated Hospital of Guangzhou University of Traditional Chinese Medicine (No. ZYYECK[2019]077) on November 1st, 2019. This study was conducted ethically in accordance with the World Medical Association Declaration of Helsinki. Informed consents were obtained from all patients before participating in the trial.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
