Abstract
Keywords
Introduction
Globally, the 5-year survival rate of cancer survivors has been improving owing to advancements in cancer screening technologies and treatment strategies, leading to an increase in the number of individuals diagnosed with cancer each year.1,2 Nevertheless, cancer remains refractory, and cancer survivors may continue to experience serious complications following their treatment. Currently, the issue of cancer chemotherapy-related cardiac toxicity (CTRCT) is gradually becoming prominent because of the use of cardiotoxic chemotherapeutic drugs and their sequential therapies, particularly in female survivors of malignant neoplasms, such as breast cancer and endometrial cancer.3,4 CTRCT refers to the impairment of cardiac function caused by cardiotoxic drugs used during chemotherapy, such as anthracycline antibiotics. This damage can either be transient or persistent, and it can manifest as early subclinical myocardial injury and mid-to-late stage cardiorespiratory function (CRF) or ventricular dysfunction, such as decreased cardiorespiratory fitness, impaired cardiac function, elevated serum biomarkers and increased cardiovascular risk events.5,6 In addition, the presence of CTRCT has been found to be closely correlated with the prevalence of cardiovascular risk factors at baseline and during follow-up. 7
As research has shown, cancer survivors may be forced to interrupt chemotherapy due to severe cardiac dysfunction caused by CTRCT, or even have an increased risk of mortality due to non-cancer-related complications such as acute heart failure caused by CTRCT, ultimately leading to a significantly diminished life expectancy for these patients.8,9 Nowadays, therapeutic strategies to mitigate the adverse effects of CTRCT typically involve decreasing the dosage of cardiotoxic chemotherapy drugs and providing symptomatic relief through the use of antidotes and comprehensive supportive care. However, reducing the dose may compromise antineoplastic efficacy, and the effectiveness of multiple drug regimens varies owing to individual patient factors and additional complications.10,11 Searching for safe and effective adjuvant therapies to reduce the risk of CTRCT while potentiating the antineoplastic efficacy of chemotherapeutic agents has thus become imperative for cancer patients receiving chemotherapy.
In recent years, physical activity-based cardio-oncology rehabilitation has garnered considerable interest in both cardiac oncology and oncology rehabilitation research, primarily because of its potential to provide cardiovascular protective effects.6,12,13 Physical activity protects cardiomyocytes from cardiotoxic damage, possibly through a range of cellular mechanisms, such as increasing cardiac antioxidant capacity, promoting mitochondrial biogenesis, and reducing the accumulation of chemotherapeutic agents in myocardial mitochondria. 14 Currently, clinical research suggests that physical activity can be used to effectively combat CRF dysfunction and reduce the likelihood of developing CTRCT.15 -30 However, results in outcome indicators associated with CTRCT have been inconsistent and controversial across studies.
Previous research has demonstrated that physical activity can significantly increase the maximal oxygen uptake (VO2max) and peak oxygen uptake (VO2peak) in cancer survivors, which is beneficial in alleviating CRF dysfunction.15 -19 Moreover, physical activity has been found to be associated with improvements in several other parameters such as left ventricular ejection fraction (LVEF), 15 ratio of early diastolic mitral inflow velocity to mitral annular velocity (E/e’),16,17 ratio of early diastolic mitral inflow velocity to late diastolic mitral inflow velocity (E/A),16,17 high-sensitivity cardiac troponin T (hs-cTn), 17 and N-terminal pro-brain natriuretic peptide (NT-proBNP).15,16 These improvements may help to prevent or mitigate the adverse effects of CTRCT. However, some studies have shown that physical activity does not have a significant impact on LVEF,20,21 global longitudinal strain (GLS),20,22 E/e’,20,22 E/A,20,22 hs-cTn, 20 and NT-proBNP.20,23 In addition, a systematic review conducted by Wang et al 31 found that physical activity was effective in enhancing VO2max but not effective in improving peak heart rate (HRpeak) among breast cancer survivors with cardiovascular toxicity. Their study did not further analyze the intervention intensity of physical activity. Another systematic review conducted by Ma et al 32 revealed that aerobic exercise markedly improved VO2max and E/A in patients with breast cancer, but the effect was not significant for E/e’ and GLS. It is vital to emphasize that their study excluded cancer survivors with CTRCT other than breast cancer.
Therefore, we performed a systematic review and meta-analysis to evaluate the effects of physical activity on CRF and CTRCT among a diverse population of cancer survivors undergoing chemotherapy. Additionally, to investigate whether there are differences in response to physical activity across diverse types of cancer patient with or without reported cardiotoxicity, we meticulously analyzed how different intervention parameters—intensity, duration, and sequence—affect VO2max or VO2peak, LVEF, and GLS, which serve as critical indicators of CTRCT. Our findings may provide recommendations for preventing or reducing CTRCT in various cancer survivors undergoing chemotherapy and support clinicians in making more effectively clinical decision-making when dealing with CTRCT.
Methods
Research Protocol and Registration
This study was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, 33 as detailed in Supplemental Table 1. The protocol was registered in PROSPERO (CRD: 4202123456).
Literature Search Strategy
A literature search was conducted using the China National Knowledge Infrastructure (CNKI), Chinese Scientific Journal Database (VIP), Wang Fang, Chinese Biomedical Literature Database (CBM), PubMed, Cochrane Library, Embase, and Web of Science databases from inception to January 2024. Additional literature was identified through a manual review of the reference lists. We used the search strategy of combining subject terms with free terms, and the search terms were as follows: (1) neoplasms, cancer, cancer survivor, cancer therapy, chemotherapy; (2) cardiotoxicity, cardiotoxic, cardiovascular toxicity, cardiac dysfunction, cardiopulmonary function, cardiorespiratory fitness; and (3) physical activity, exercise, sports, motion, walking, Wuqinxi, Baduanjin, Tai chi, yoga, and traditional sports. Language was limited to Chinese and English. The search strategy in English is presented in Supplemental Material Tables 2–5.
Eligibility Criteria
Inclusion criteria
Study design
Only randomized controlled trials (RCTs) involving physical activity as a means of decreasing CTRCT and improving CRF in the cancer population were considered for inclusion.
Study participants
Patients diagnosed with cancer and who received chemotherapy, regardless of the stage and type of cancer, were included in the study. The participants were required to be at least 18 years of age.
Study interventions
Any type of physical activity-based cardiooncology rehabilitation intervention was included, such as resistance exercise, aerobic exercise (e.g., walking, high-intensity intermittent, moderate-intensity intermittent, and moderate-intensity continuous training), and mind-body exercise (e.g., yoga, Baduanjin, Wuqinxi, and Tai Chi). Of the 3 types of physical activity, the last one is multimodal and of low-to-moderate intensity, often accompanied by conscious breathing, sustained focus, meditation, relaxation or chanting. The intervention duration was at least 4 weeks.
Study controls
The control group did not perform any novel supervised program of exercise and physical activity interventions, including placebo control, usual treatment control, and usual care control (e.g., health education, maintaining the original daily physical activity levels, and exercise recommendations).
Study outcomes
This study included 5 different categories of outcome indicators, as follows: (1) cardio-pulmonary parameters, including VO2peak(VO2max) and HRpeak; (2)cardiac function parameters, including LVEF, stroke volume (SV), and cardiac output (CO); (3) cardiac serum biomarkers, including NT-proBNP and hsTnT; (4) subclinical indicators of cardiotoxicity, including GLS, E/A, and E/e’; and (5) endothelial function parameters, specifically flow-mediated dilatation(FMD). The primary outcomes considered in this study were VO2peak (VO2max), LVEF, GLS, and FMD.
Exclusion Criteria
The following criteria were used to exclude studies:(1) non-RCTs such as cohort and cross-sectional studies; (2) participants under the age of 18, non-cancer patients, non-chemotherapy cancer populations, and unclear whether all participants received chemotherapy or not; (3) ineligible interventions or controls; (4) incomplete data or the full text; (5) poorly designed studies; (6) duplicates, conference papers, dissertations, animal studies, protocols, reviews, and meta-analysis; and (7) low-quality studies with a PEDro score of 0 to 3.
Data extraction and transformation
The process of data extraction was performed by one reviewer (Kang Chen) using a data extraction form, and was then verified by the second reviewer (Hui Guan). Any inconsistencies were resolved by a third (He Zhuang). For each included study, we extracted the following data: general publication details (e.g., author, year of publication, and nation where the study was conducted), participant’ characteristics (e.g., cancer type, staging, chemotherapy regimen, sample size, and mean age), intervention specifics (e.g., exercise type, intensity, duration, frequency, period, and outcomes), and methodological quality (e.g., study design, conduct, blinding, and other relevant information).
Since studies may use inconsistent effect sizes and presentation of results, we transformed and standardized the extracted data. When studies only reported the median along with the upper and lower quartile, we used the method similar to that suggested by Wan et al 34 to calculate the mean and standard deviation. Conversely, when studies exclusively presented statistical graphs that displayed means along with error bars, we used the method developed by Liu et al 35 to estimate the mean and standard deviation. Detailed methods of data conversion are presented in Supplemental Figure 1–3.
Literature bias risk and quality assessment
Assessment of bias risk
Two researchers (Kang Chen and Hui Guan) independently assessed the risk of bias in the included studies according to the Cochrane Risk of Bias Assessment Tool. 36 Any disagreement was resolved by a third (He Zhuang). The assessment of potential bias includes 6 items (i.e., selection, implementation, measurement, follow-up, reporting, and other biases), and each item is categorized as low, uncertain, or high risk.
Assessment of methodological quality
We assessed the methodological quality of the included studies using the Physiotherapy Evidence Database (PEDro) scoring scale. The PEDro scale comprises 11 items, and its total score ranges from 0 to 10, with higher scores indicating studies of higher methodological quality. In accordance with established criteria, studies with a score of 6 to 10 are classified as high-quality, while those with a score of 4 to 5 are considered moderate-quality. Conversely, studies with a score of 0 to 3 are categorized as low-quality. 37 Only moderate- and high-quality studies were included in this systematic review and meta-analysis.
Data Synthesis and statistical analysis
We first used the RevMan software (version 5.3, Cochrane, London, UK) to calculate the combined effect sizes and variability in the between-group meta-analysis. The baseline values of the intervention and control groups in the included studies were comparable; thus, we extracted the outcome data upon completion of the treatment period. As our meta-analysis focused on continuous outcomes, we extracted the mean and standard deviation of the pertinent data. Due to the consistent use of scales and units of measurement in all studies, we calculated the weighted mean difference (WMD) along with the 95% confidence interval (95% CI) to assess the combined effect sizes and its corresponding variability of continuous outcomes.
Subsequently, we assessed statistical heterogeneity in all studies using the I2 statistic and Q test. The values were classified into three levels according to the degree of heterogeneity: low (I² ≤ 50% and P ≥ .1), moderate (50% < I2 < 75% and P < .1), and high (I² ≥ 75% and P < .1). 38 Among of them, both moderate and high levels of heterogeneity were recognized as statistically significant. 38 We then used a fixed-effects model to assess the mean of the effects for the pooled data with low heterogeneity and a random-effects model to evaluate the mean of the effect sizes for the pooled data with moderate to high heterogeneity. 38 Additionally, to address the statistical heterogeneity observed in the meta-analysis, we conducted a sensitivity analysis to assess the robustness of the results and identify sources of heterogeneity that may affect the overall effect estimates.
Furthermore, in order to further comprehensively evaluate the impact of physical activity, comprising various intervention intensities, duration and sequences, on CRF and CTRCT in diverse cancer patient populations, we used Stata software (version 17.0, StataCorp LP, Texas, USA) to perform subgroup analyses on the primary outcomes of cardiorespiratory index (VO2max or VO2peak), cardiac function parameter (LVEF), and cardiotoxicity indicator (GLS). Moreover, we employed the Stata software (version 17.0) to carry out the Egger’s test to assess the presence of publication bias across the primary outcome measures. 31 All statistical significance levels were set at P < .05.
Results
Results of Literature Search and Screening
A total of 1575 articles were retrieved from 8 databases. After deduplication using NoteExpress software and subsequent manual review, 295 duplicates were identified and removed, leaving 1280 publications. Following our initial assessment of titles and abstracts in accordance with the inclusion and exclusion criteria, the number of publications was reduced to 115. After further conducting a full-text review, 16 studies15 -30 were selected for inclusion in the systematic review, and 15 studies15 -23,25 -30 were eligible for meta-analysis. These studies comprised 2 Chinese15,16 and 14 English17 -30 publications. The flow diagram of study selection and screening process is presented in Figure 1.

The flowchart of the process for literature search and screening.
Basic Characteristics of Included Literature
Basic characteristics of participants
The present systematic review included 1042 participants diagnosed with cancer from 10 distinct countries (i.e., the United States, China, France, Thailand, Portugal, Norway, Sweden, Australia, Canada, and Switzerland).15 -30 The mean age of participants varied from 39.1 years 30 to 61 years, 25 and the majority of participants were under the age of 50 years. The cancer stage of the included participants was mainly focused on stages I-III. Moreover, 11 studies reported the effects of physical activity on chemotherapy-related cardiotoxicity in patients with cancer or were conducted based on its correlated research background,15 -17,19,20,22-24,27 -29 while 5 studies reported the effects of physical activity on chemotherapy-related cardiorespiratory dysfunction in patients with cancer.18,25,26,30 In terms of chemotherapeutic agents, patients were administered various chemotherapeutic regimens, including anthracycline antibiotics (e.g., adriamycin and epirubicin), cyclophosphamide, and paclitaxel as single agents, as well as their combination and sequential therapies. Regarding cancer type, the participants primarily focused on female breast cancer patients, especially survivors of stages I-III who had received anthracycline-based regimens and its sequential therapy. Additionally, survivors of lymphoma, smooth muscle sarcoma, testicular cancer, and colorectal cancer were also included in this study. The participants’ basic characteristics are presented in Table 1.
The Basic Characteristics of Included Studies.

Abbreviations: -, no information provided; Tr, the intervention group; Co, the control group; A, anthracycline chemotherapy; C, cyclophosphamide chemotherapy; T, paclitaxel; AC, anthracycline and cyclophosphamide chemotherapy; AC-T, anthracycline and cyclophosphamide chemotherapy followed by Paclitaxel; A-T, anthracycline and paclitaxel; wk, week; ses,one session; AET(AT), aerobic exercise training; RT, resistance training; MICT, moderate-intensity continuous training; HIIT, high-intensity interval training; AT-HIIT, aerobic exercise combined with high-intensity interval training; RT-HIIT, resistance training combined with high-intensity interval training; MBE, mind-body exercise; VO2max, maximum oxygen uptake; VO2peak, peak oxygen uptake; HRmax, maximum heart rate; HRpeak, peak heart rate; LVEF, left ventricular ejection fraction; SV, stroke volume; CO, cardiac output; NT-proBNP, n-terminal pro-brain natriuretic peptide; hs-cTnl, high-sensitivity cardiac troponin l; GLS, global longitudinal strain; E/A, ratio of early to late diastolic filling velocities of the mitral annulus; E/e’, ratio of early mitral inflow velocity to early diastolic mitral annular velocity;FMD, Flow-Mediated Dilation; PPO, Peak Power Output; RPM, revolutions per minute; HRR, heart rate reserve.
Basic characteristics of interventions
The types of physical activity primarily encompassed 5 categories: specifically, 4 studies24,27,28,30 investigated aerobic exercise (AET) among which one study 27 focused on mind-body exercise, 27 only one study 29 focused on high-intensity interval training (HIIT), 2 studies17,25 reported mixed exercise training (mixed ET), 6 studies15,16,19,21,23,26 explored the combination of AET and HIIT (AET + HIIT), and 3 studies18,20,22 evaluated the combination of AET and resistance training (AET + RT). In contrast, the control group received conventional treatment, usual care or recommendation on exercise health education and preserving routine physical activity levels. The basic characteristics of interventions are presented in Table 1.
Basic characteristics of exercise prescription
For the specific classification of exercise intensity, 3 studies18,27,30 reported moderate intensity exercise, 9 studies16,17,20 -26,28 investigated exercise of moderate- to-high intensity, and another 3 studies15,19,29 focused on high intensity exercise. In terms of intervention duration, it commonly ranged from 8 to 16 weeks, with 3 studies19,23,29 setting duration of 8 to 10 weeks, 3 studies17,25,30 being beyond 16 weeks. 10 studies,15,16,18,20 -22,24,26 -28 prescribed an exercise frequency of 2 to 3 sessions per week, with each session duration ranging from 30 to 60 minutes. The basic characteristics of exercise prescriptions are presented in Table 1.
Results of Literature Bias Risk and Quality Assessment
Bias risk of included literature
A total of 16 studies15 -30 reported the randomization process; however, only 5 studies16,20,22 -24 provided detailed information on the specific method of random allocation. The remaining 10 studies15,18,19,21,25 -30 did not disclose sufficient information regarding the concealment of the allocation method, and one study 17 was open-label. Owing to safety concerns associated with the exercise intervention, only one study implemented blinding of either the researchers or participants, whereas 6 studies blinded the outcome assessors. The overall risk of bias assessment for the included studies is shown in Figure 2. A summary graph of the bias risk is shown in Supplemental Figure 4.
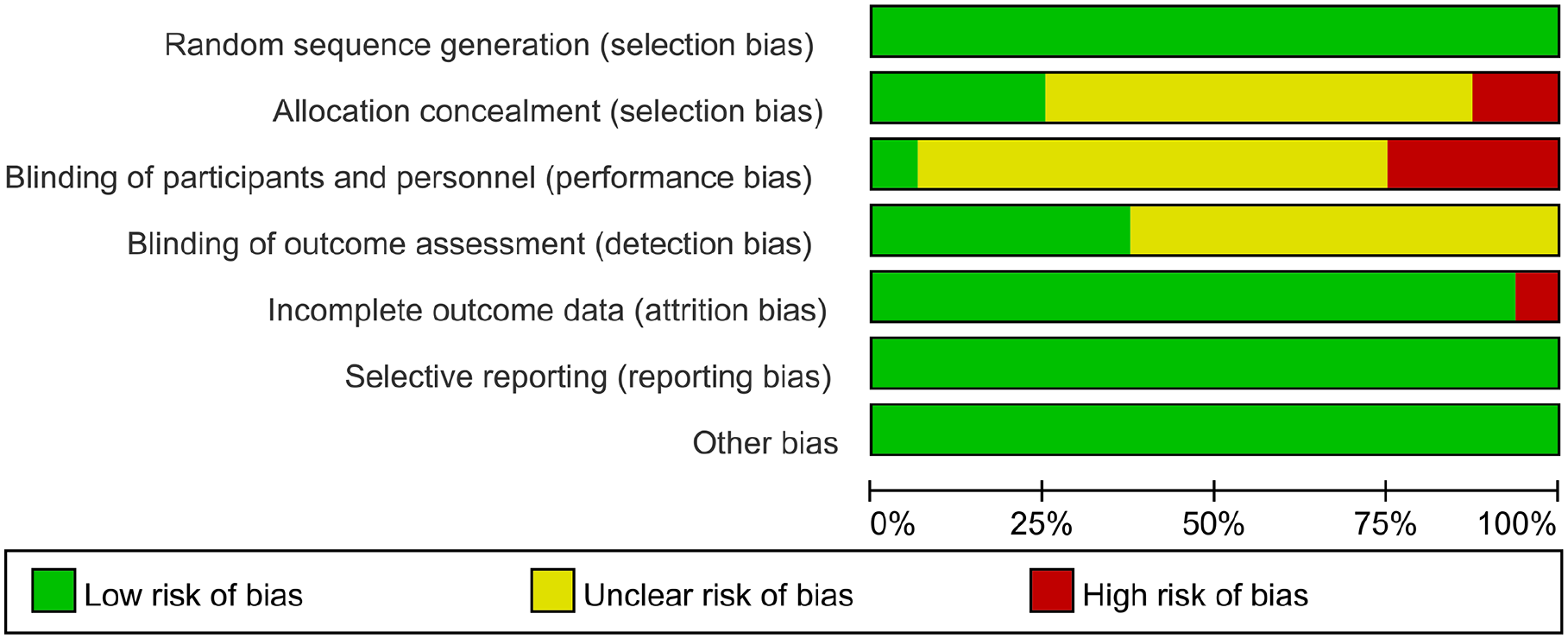
The overall risk bias assessment diagram of included studies.
Methodological quality assessment
The results of the detailed PEDro scale scores are presented in Table 2, with an overall mean score of 6.06. Of the 16 studies, 12 studies16 -24,26,27,29,30 were rated as high quality, while the remaining 4 studies15,25,28,30 were rated as moderate quality.
The PEDro Scores of Included Randomized Controlled Trials.

Abbreviations: M, moderate quality; H, high quality.
Results of Meta-Analysis for Effectiveness Assessment
Meta-analysis of cardiopulmonary parameters
Absolute VO2peak or VO2max (ml/min)
Considering the limitations and safety imposed by physical activity on cancer survivors, as well as the observed proximity of VO2max and VO2peak values in actual clinical measurements, we appropriately integrated these 2 parameters to perform a meta-analysis. Six studies15 -17,19,23,26 reported absolute VO2peak or VO2max in various cancer survivors undergoing chemotherapy, with the intervention and control groups comprising 177 and 160 patients, respectively. Due to the high heterogeneity observed (I 2 = 90%, P < .00001), we used the random-effects model to conduct meta-analysis. The findings revealed that physical activity significantly increased the absolute VO2peak or VO2max (WMD = 292.99, 95% CI: 87.87 to 498.12, P = .005), as illustrated in Figure 3. Subsequently, we performed a sensitivity analysis to identify the sources of heterogeneity. Upon excluding the study by Zhang et al 15 , the level of heterogeneity was significantly reduced to I2 = 35% (P = .19). We then used a fixed-effects model to perform meta-analysis, and its results showed that physical activity can significantly increase absolute VO2peak or VO2max (WMD = 191.21, 95% CI:114.14 to 268.27, P < .001). The results remained robust.
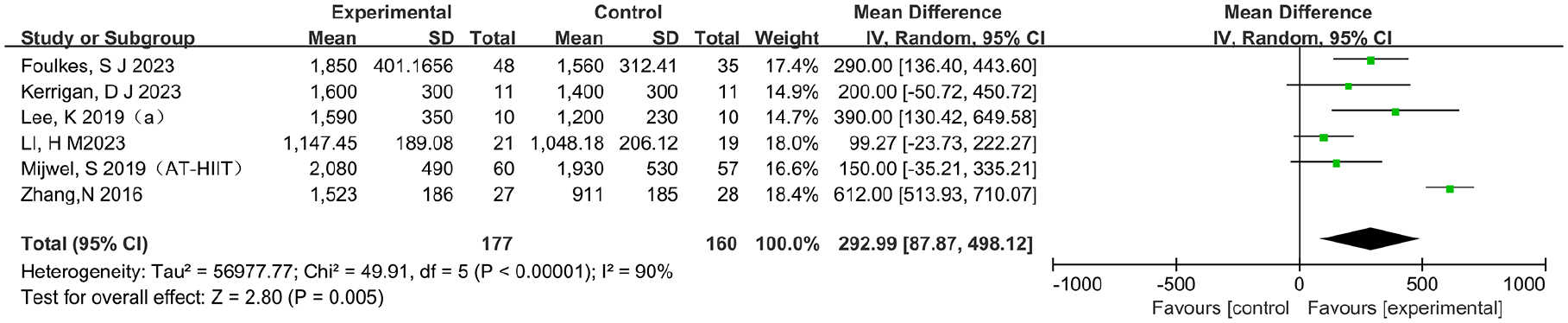
The effects of physical activity on the absolute VO2peak or VO2max (ml/min) for cancer survivors.
Depending on the duration and intensity of the intervention, as well as the type of cancer and the presence of cardiotoxicity, we performed subgroup analysis to investigate the variability of physical activity and the cancer population to absolute VO2peak or VO2max. The results showed that 4 to 10 weeks (P = .02) and more than 16 weeks (P < .01), as well as those who received high-intensity and moderate- high-intensity of physical activity (both P < .01), significantly improved absolute VO2peak or VO2max among cancer patients compared with the control group. Furthermore, physical activity significantly enhanced absolute VO2peak or VO2max in patients with breast cancer (P < .0001) and those experiencing cardiotoxicity (P < .00001). However, interventions lasting 10 to 16 weeks and targeting the mixed cancer population as well as patients not reporting cardiotoxicity failed to show any significant effects (all P > .05). Although some trends were observed, the findings should be interpreted with caution because of the limited number of studies analyzed in the subgroups. The details can be found in Table 3 and Supplemental Figure 5.
The Subgroup Analysis of Absolute VO2peak or VO2max (ml/min).

Relative VO2peak or VO2max (ml/min/kg)
A total of 14 studies15 -23,25-28,30 reported the relative VO2peak or VO2max, with 435 cancer patients in the intervention group and 396 in the control group. Because of the moderate heterogeneity (I² = 67%, P = .0001), we utilized a random-effects model to conduct the meta-analysis. The results showed that the relative VO2peak or VO2max was markedly higher in the intervention group among various cancer patients receiving chemotherapy compared to the control group (WMD = 3.30, 95% CI: 2.02 to 4.58, P < .00001), as shown in Figure 4. Subsequently, we performed a sensitivity analysis to identify the source of heterogeneity. After excluding the study by Zhang et al 15 the heterogeneity was significantly reduced to I² = 24% (P = .19). We then used a fixed-effects model to conduct a meta-analysis, and its results indicated that physical activity significantly advanced relative VO2peak or VO2max (WMD = 2.87, 95% CI: 2.17 to 3.58, P < .001). The results remained robust. Additionally, to investigate whether mind-body exercises exerted an influence on the overall result, we conducted a sensitivity analysis. After excluding the study regarding walking meditation exercise intervention by Siripanya et al, 27 we utilized a random-effects model to perform the meta-analysis. The results demonstrated that exercise-based physical activity continued to significantly enhance the relative VO2peak or VO2max. (WMD = 3.12, 95% CI: 1.80 to 4.45, P < .00001), with moderate heterogeneity (I2 = 68%, P = .0001), as shown in Supplemental Table 6.

The effects of physical activity on the relative VO2peak or VO2max (ml/min/kg) for cancer survivors.
Based on the duration, intensity, and sequence of interventions, as well as the type of cancer and the presence of cardiotoxicity, we conducted subgroup analysis to assess the variability of physical activity and the cancer population to absolute VO2peak or VO2max. The findings showed that cancer patients who underwent 4 to 10 weeks (P = .02) and more than 16 weeks (P < .01) of physical activity, as well as those who received high-intensity and moderate-high-intensity (both P < .01) of physical activity, exhibited a statistically significant improvement in absolute VO2peak or VO2max compared to the control group. Moreover, the implementation of physical activity during the chemotherapy period (P < .01) demonstrated a substantial rise in absolute VO2peak or VO2max among cancer patients. And physical activity significantly enhanced absolute VO2peak or VO2max among patients with breast cancer (P < .001) and those with (P < .001) or without (P = .007) cardiotoxicity. However, interventions lasting 4 to 10 weeks and patients with colon cancer or mixed types of cancer failed to show any significant effects (all P > .05). Detailed information is presented in Table 4 and Supplemental Figure 6.
The Subgroup Analysis of the Relative VO2peak or VO2max (ml/min/kg) for Cancer Survivors.

HRpeak or HRmax (bpm)
Five studies15,16,21,23,27 measured HRpeak or HRmax using treadmill exercise test or cardiopulmonary exercise test, with 81 patients in the intervention group and 78 in the control group. Due to the high heterogeneity (I2 = 76%, P = .002), we used a random effects model to conduct the meta-analysis. The result showed that there was no significant effect of physical activity on HRpeak or HRmax (WMD = −3.26, 95% CI: −12.08 to 5.56, P = .47), as presented in Supplemental Figure 7. Subsequently, we performed a sensitivity analysis to identify potential sources of heterogeneity. After removing 2 studies by Li et al 16 and Hornsby et al 21 , the level of heterogeneity decreased to I2 = 18% (P = .29). We then used a fixed-effects model to perform a meta-analysis, and the result revealed that physical activity significantly reduced the HR peak or HRmax (WMD = −9.22, 95% CI: −13.74 to −4.70, P < .001). Conversely, while sensitivity analysis excluded another 3 studies by Zhang et al, 15 Siripanya et al, 27 and Kerrigan et al, 23 the level of heterogeneity reduced to I2 = 0% (P = .81). We then used a fixed-effects model to perform meta-analysis, and its result revealed that physical activity tended to increase the HRpeak or HRmax (WMD = 7.38, 95% CI:−0.01 to 14.76, P = .05). The results of the meta-analysis were not robust and should be interpreted with caution.
Moreover, to further explore whether mind-body exercises exerted the impact on the overall result, we conducted a sensitivity analysis. After excluding the study regarding walking meditation exercise intervention, 27 we used a random-effects model to perform the meta-analysis. The findings indicated that exercise-based physical activity persistently showed a trend toward reducing the HRpeak or HRmax (WMD = −1.42, 95% CI: −13.92 to 11.07, P = .82), with high heterogeneity (I2 = 79%, P = .002), as shown in Supplemental Table 6.
Meta-Analysis of Cardiac Function Parameters
LVEF(%)
Five studies15 -17,20,21 assessed LVEF only among breast cancer survivors, with 148 patients in the intervention group and 135 in the control group. Due to the high heterogeneity (I2 = 98%, P < .00001), we utilized a random-effects model to conduct the meta-analysis. The findings suggested that there was no significant improvement of physical activity on LVEF compared to the control group (WMD = 2.89, 95% CI: −3.28 to 9.06, P = .36), as shown in Figure 5. Following sensitivity analysis, excluding the study by Zhang et al, 16 the level of heterogeneity decreased to I2 = 0% (P = .86). Subsequently, we used a fixed-effects model to perform the meta-analysis. The result showed that physical activity did not significantly increase LVEF (WMD = −0.21, 95% CI: −1.13 to 0.71, P = .65), and the findings remained robust.

The effects of physical activity on the LVEF (%) for breast cancer survivors.
According to the duration, intensity of the interventions, and reporting of cardiotoxicity, we conducted subgroup analysis to investigate the response of physical activity and cardiotoxicity to LVEF. The findings revealed that in patients with breast cancer, neither physical activity lasting 10 to 16 weeks nor more than 16 weeks, as well as taking moderate-to-high intensity (all P > .05) had a significant effect on LVEF. Additionally, this effect was not affected by cardiotoxicity(P > .05). Details are presented in Table 5 and Supplemental Figure 8.
The Subgroup Analysis of the LVEF(%) for Breast Cancer Survivors.

SV (ml/beat) and CO (L/min)
All 4 studies16,20,21,27 measured SV and CO in cancer survivors, with 85 patients in the intervention group and 81 in the control group. For measured SV, because no heterogeneity was observed (I2 = 0%, P = .44), we used a fixed-effects model to conduct the meta-analysis. The findings indicated that there was no significant effect of physical activity on SV when compared to the control group (WMD = 0.01, 95% CI: −3.56 to 3.57, P = 1.00), as illustrated in Supplemental Figure 9. To further explore whether mind-body exercises exerted the impact on the overall result, we conducted a sensitivity analysis. After excluding the study regarding walking meditation exercise intervention, 27 we used a fixed-effects model to perform the meta-analysis. The findings persistently indicated that there was no significant effect of exercise-based physical activity on SV (WMD = −0.31, 95% CI: −4.05 to 3.43, P = .87), with low heterogeneity (I2 = 16%, P = .31), as shown in Supplemental Table 6.
For measured CO, however, due to high heterogeneity (I2 = 80%, P = .002), we utilized a random effects model to conduct the meta-analysis. The result revealed that physical activity did not significantly enhance CO when compared to the control group (WMD = −0.52, 95% CI: −1.74 to 0.71, P = .41), as illustrated in Supplemental Figure 10. We then performed sensitivity analysis to identify the source of heterogeneity. After excluding the study by Li et al, 16 the level of heterogeneity decreased to I2 = 42% (P = .18). The result of the fixed-effects model indicated that physical activity did not significantly affect CO WMD = 0.11, 95% CI: −0.20 to 0.41, P = .49), revealing robust and consistent results. Additionally, to further explore whether mind-body exercises exerted an impact on the overall result, we conducted a sensitivity analysis. After excluding the study regarding walking meditation exercise intervention, 27 we used a random-effects model to perform the meta-analysis. The findings persistently indicated that there was no significant effect of exercise-based physical activity on CO (WMD = −1.52, 95% CI: −2.87 to 0.57, P = .19), with low heterogeneity (I2 = 85%, P = .002), as shown in Supplemental Table 6.
Meta-Analysis of Cardiac Serum Biomarkers
NT-proBNP (ng/L)
Three studies15,16,22 reported NT-proBNP levels with 69 and 70 patients in the intervention and intervention groups, respectively. Owing to the high heterogeneity observed (I2 = 97%, P < .00001), we used a random effects model to perform the meta-analysis. The result indicated that physical activity tended to decrease the level of NT-proBNP (WMD = −131.02, 95% CI: −295.55 to 35.52, P = .12), as presented in Supplemental Figure 11. Further sensitivity analysis revealed significant heterogeneity across the studies, regardless of which ones were excluded (I2 > 50% and P < .05). Nevertheless, there was a positive trend toward a reduction in NT-proBNP levels in response to physical activity, although this difference was not statistically significant ( P > .05).
Hs-cTn (ng/L)
Two studies22,23 reported hs-cTn, with 32 patients in the intervention group and 33 patients in the control group. Due to the lack of heterogeneity (I 2 = 0%, P = .50), we used a fixed-effects model to perform meta-analysis. The result showed that there was no significant effect of physical activity on hsTnT compared with the control group (WMD = −0.55, 95% CI:−4.93 to 3.57, P = 3.83), as shown in Supplemental Figure 12.
Meta-Analysis of Subclinical Indicators of Cardiotoxicity
GLS(%)
Four studies17,20,22,23 measured the GLS in cancer survivors, with 113 patients in the intervention group and 104 in the control group. Due to the absence of heterogeneity (I 2 = 0%, P = .63), we used a fixed-effects model to conduct meta-analysis. The results showed that physical activity performed during chemotherapy period of moderate-to-high intensity had no significant effect on GLS compared to the control group (WMD = 0.37, 95% CI: −0.20 to 0.94, P = .20), as shown in Figure 6. Subsequently, based on intervention duration and cancer type, we performed subgroup meta-analysis. The findings indicated that neither the duration of the intervention nor the type of cancer had a significant impact on the effects of physical activity, as shown in Table 6 and Supplemental Figure 13.

The effects of physical activity on the GLS (%) for cancer survivors.
The Subgroup Analysis of the GLS (%).
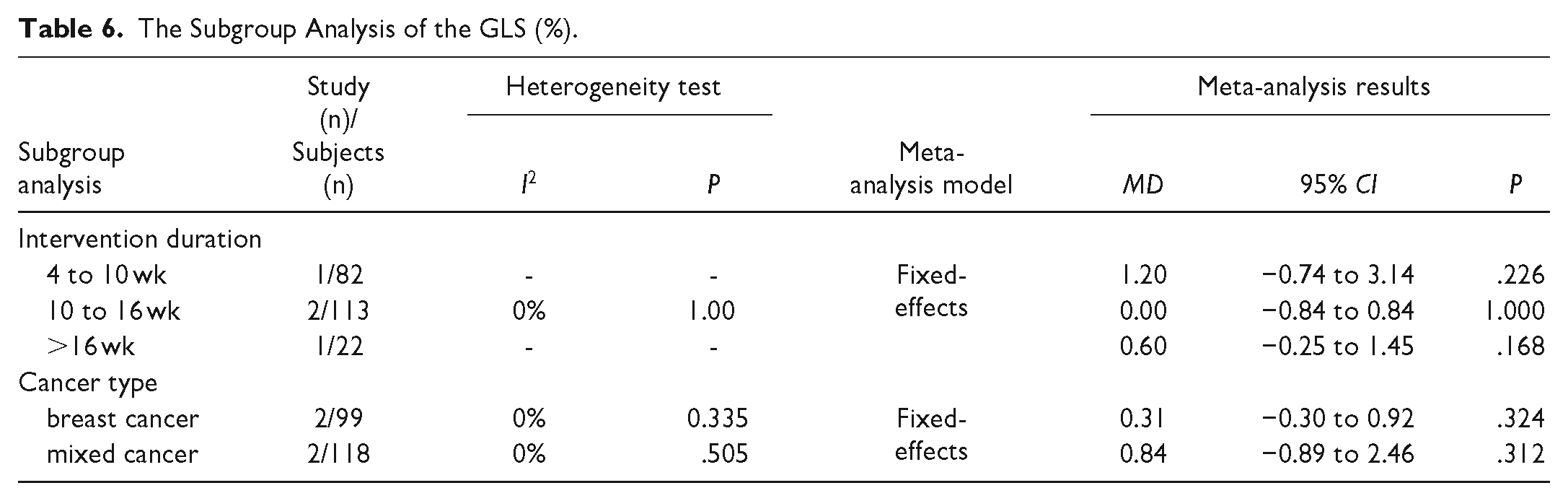
E/A(cm/s)
Five studies15 -17,20,22 reported E/A values, with 166 patients in the intervention group and 155 in the control group. Given the lack of heterogeneity (I 2 = 0%, P = .60), we used a fixed-effects model to conduct meta-analysis. The result revealed that physical activity significantly improved E/A values compared to the control group (WMD = 0.11, 95% CI: 0.03 to 0.18, P = .007), as presented in Figure 7.
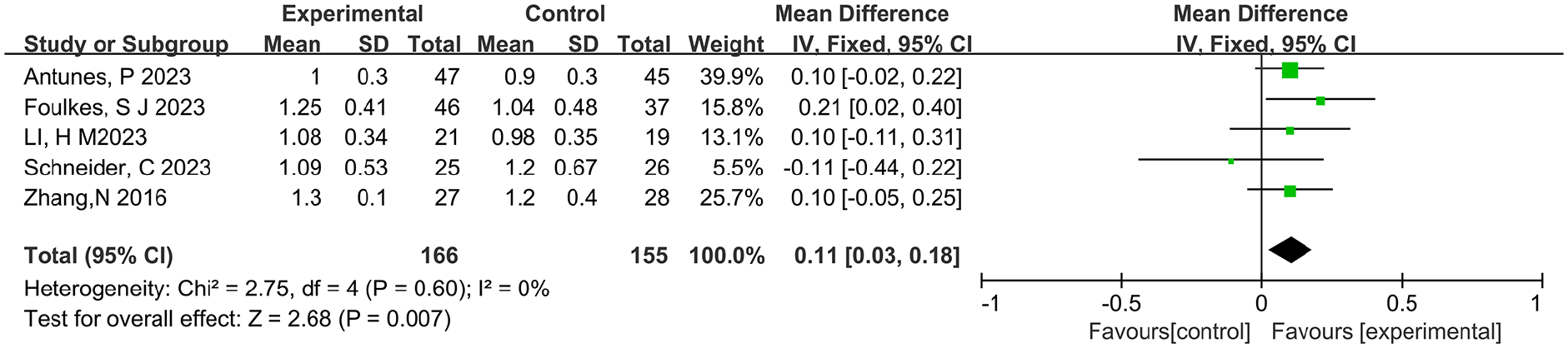
The effects of physical activity on the E/A (cm/s) values for cancer survivors.
E/e’(cm/s)
Four studies16,17,20,22 assessed E/e’ values, with 136 and 126 patients in the intervention and control groups, respectively. Due to moderate heterogeneity (I2 = 72%, P = .01), we used a random-effects model to perform the meta-analysis. The findings indicated that physical activity did not have a significant effect on E/e’ values compared to the control group (WMD = 0.35, 95% CI:−0.68 to 1.39, P = .50), as shown in Figure 8. Following a sensitivity analysis that excluded the study by Li et al, 16 the level of heterogeneity disappeared (I2 = 0%, P = .68). Subsequently, we utilized a fixed effects model for meta-analysis, and the results showed no significant effect of physical activity on E/e’ values (P = .62).

The effects of physical activity on the E/e’ (cm/s) for cancer survivors.
Meta-Analysis of Endothelial Function Parameters
FMD(%)
Two studies27,29 investigated the effect of physical activity on FMD among breast cancer survivors during chemotherapy, with 26 patients in the intervention group and 26 in the control group. Owing to the moderate heterogeneity (I2 = 72%, P = .06), we used a random-effects model to conduct the meta-analysis. The findings demonstrated that physical activity notably enhanced FMD compared with the control group (WMD = 2.71, 95% CI: 1.49 to 3.94, P < .0001), as depicted in Figure 9.

The effects of physical activity on the FMD (%) for cancer survivors.
Results of Publication Bias
Publication bias was analyzed using funnel plots and Egger’s test for the primary outcome, which revealed that there was no publication bias in absolute VO2peak or VO2max, relative VO2peak or VO2max, LVEF, or GLS (all P > .05). Details are presented in Supplemental Figure 14–17.
Discussion
Chemotherapy-Related Cardiotoxicity (CTRCT) and Exploring Physical Activity as a Therapeutic Intervention for Cancer Survivors
Chemotherapeutic agents, such as anthracycline, cyclophosphamide and paclitaxel, have been widely demonstrated to have significant inhibitory effects on a variety of tumor cells. Nonetheless, each chemotherapeutic regimen has inherent limitations, particularly in terms of limited efficacy and severe complications.39 -41 These limitations have prompted researchers to seek alternative strategies to improve patient outcomes, especially in the context of oncological rehabilitation. In response to the challenges posed by the side effects of chemotherapy, previous research focused on alleviating cancer-related complications such as cancer-related fatigue, lymphedema, anxiety, and depression.42 -44 In recent years, anthracycline-based chemotherapeutic agents and its related sequential therapies have shown significant efficacy in the treatment of breast, ovarian and lymphoma cancers. However, there is a growing concern that their use may lead to the development of severe CTRCT in cancer survivors.10,45,46
Nowadays, studies have found that the effective approach to prevent or reduce the risk of CTRCT is to implement strategies that improve CRF and the ability to resist cardiotoxicity during chemotherapy, while also closely monitoring cardiorespiratory markers, cardiac function, and subclinical indicators of cardiotoxicity.47,48 While 2 meta-analyses have specifically evaluated the impact of exercise on cardiovascular toxicity among breast cancer patients, no systematic reviews have yet assessed the potential benefits of physical activity in mitigating cardiotoxicity across a wider range of cancer types.31,32 This gap in the literature emphasizes the need for a comprehensive evaluation of physical activity as a therapeutic intervention for diverse cancer populations. Therefore, to fill the gap on this topic, this study is the first systematic review of physical activity in improving cardiotoxicity among various cancer survivors. Notably, of the studies included in our study, only one study involved mind-body exercise—specifically, home-based Buddhist walking meditation. Walking meditation training, while incorporating meditative elements characterized by the recitation of “Budd” and “Dha,” primarily emphasized the walking component. Essentially, it constituted a form of low to moderate intensity physical activity. 27 Therefore, this study included walking meditation intervention, in order to comprehensively assess the impact of various forms of physical activity on cardiotoxicity in chemotherapy patients.
Effects of Physical Activity on CRF and CTRCT
Changes in cardiorespiratory parameters, especially a decrease in VO2peak or VO2max, may be a sensitive indicator for detecting occurrence of CTRCT in cancer survivors. 48 Studies have revealed that anthracycline, cyclophosphamide, and paclitaxel chemotherapy lead to a significant decrease in VO2peak or VO2max, and that this downward trend is closely linked to the occurrence of CTRCT.15 -17,22,23 This may be because chemotherapy agents damage myocardial cells, thereby interfering with VO2peak or VO2max-dependent physiological processes, such as cardiac output and oxygen-carrying capacity, which in turn affects the individual’s ability to exchange oxygen at maximum loads.15,16,48 Therefore, a lower VO2peak indicates not only a direct reduction of overall CRF but also myocardial injury associated with potential CTRCT.
We found that physical activity significantly increased absolute and relative VO2peak or VO2max in various cancer survivors undergoing chemotherapy, which in turn improved the capacity of prevention and resistance to the risk of CTRCT. Subgroup analyses found that absolute VO2peak or VO2max was significantly enhanced by moderate-to-high intensity training and short-to-medium duration training, particularly in patients with breast cancer and cardiotoxicity; relative VO2peak or VO2max was more significantly increased in medium- to long-term training, particularly in patients with breast cancer and physical activity performed during chemotherapy. Moreover, the positive reaction to physical activity among individuals with cardiomyotoxicity showed the potential benefits of exercise in cardio-protection. However, the results were influenced by substantial heterogeneity, which may be attributed to the inherent physiological limitations of cancer patients and differences in intervention protocols and chemotherapy regimens across studies as well as the selection of data conversion. In addition, this result found that physical activity did not significantly improve the HRpeak. Following sensitivity analysis, it was determined that the effect on HRpeak was not consistent; therefore, interpreting the results should be approached with caution.
Effects of Physical Activity on Cardiac Function and CTRCT
LVEF is widely regarded as an optimal indicator for assessing left ventricular contraction and cardiac ejection function. Given its role in measuring cardiac function, a decrease in LVEF of more than 10% or a LVEF value falling below 53% (with a more conservative threshold of 50%) can be used to predict or diagnose intermediate to late CTRCT. 49 A cohort study conducted by Cardinale et al 50 evaluated 2625 cancer survivors who received anthracycline treatment. Their findings found that the median duration for a 10% or greater reduction in LVEF was 3.5 months (3-6) following chemotherapy, and cardiotoxicity occurred most frequently in the first year after chemotherapy. However, the median length of time for recovery from cardiotoxicity was reported to be as long as 5.2 years (6-8.0). Consequently, it is crucial to closely monitor, prevent, and improve the left ventricular ejection fraction (LVEF) in cancer patients in order to prevent and alleviate the occurrence of CTRCT in patients with cancer.
In the present meta-analysis, a positive trend was observed, indicating that physical activity had a favorable impact on enhancing LVEF when compared to the control group; however, this difference was not statistically significant. Similarly, our result was consistent with the findings of a meta-analysis conducted by Ma et al 32 Our subgroup analysis further demonstrated that high-intensity exercise for 10 to 16 weeks during chemotherapy drove a potential improvement of LVEF among breast cancer patients with CTRCT. In addition, consistent with the findings of LVEF, physical activity did not significantly enhance SV and CO in patients with breast cancer. Considering the limited number of studies and the small sample sizes, future studies with larger sample sizes are required to explore the potential benefits of physical activity on cardiac function.
Effects of Physical Activity on Subclinical Indicators of Cardiotoxicity
While LVEF is a crucial indicator for diagnosing and predicting CTRCT, its accuracy and sensitivity in identifying the development of early CTRCT are reduced in patients with mild left ventricular (LV) systolic dysfunction or preserved ejection fraction.51,52 It has been reported that subclinical indicators of cardiotoxicity, namely GLS, E/A, and E/e’, serve as effective predictive factors for identifying early CTRCT.47,53,54 GLS is a noninvasive index obtained by measuring global longitudinal strain during myocardial contraction. GLS is more sensitive than LVEF in identifying changes in myocardial contractile function, and its decline indicates subclinical myocardial injury (i.e., myocardial damage could have occurred prior to a significant decline in cardiac function). In addition, E/A primarily reflects the flow dynamics of LV early diastole and atrial systole, whereas E/e’ reflects LV filling pressures and diastolic function. As such, E/A and E/e’ are crucial indices to assess diastolic dysfunction with a preserved ejection fraction. Consequently, GLS, E/A, and E/e’ ratio can detect minor alterations and subclinical injury to LV myocardial function prior to a significant decline in LVEF.47,53,54 This has led to the possibility of early identifying and managing the risk of cardiotoxicity in cancer survivors receiving chemotherapy.15 -17,20,22,23
We found that in contrast to the anticipated cardioprotective effects of physical activity, there was a non-significant, but negative impact of physical activity on GLS (WMD = 0.37, 95% CI:−0.20 to 0.94). This may be due to the limited number of included RCTs, diversity of chemotherapeutic regimens, and combinations of cardioprotective drugs. However, we observed that physical activity markedly improved E/A, and that there was a positive trend toward improvement in E/e’ with physical activity. Based on the results of meta-analysis for subclinical indicators of cardiotoxicity, various cancer survivors can improve subclinical indicators of cardiotoxicity and reduce the risk of developing CTRCT through a potentially cardioprotective effect of physical activity.
Effects of Physical Activity on Cardiac Serum Biomarkers and CTRCT
In our meta-analysis, we assessed the effects of physical activity on cardiac serum biomarker levels. Elevated NT-proBNP levels are typically associated with an increased risk of ventricular dysfunction and heart failure, whereas elevated hs-cTn levels reflect damage or necrosis of cardiomyocytes.15,16,22,23 Therefore, NT-proBNP and hscTn levels may serve as sensitive serological indicators of cardiotoxicity, which can reflect myocardial injury from chemotherapeutic agents in cancer survivors receiving chemotherapy.
Consistent with the results of Ma et al, 32 we found that physical activity did not significantly reduce NT-proBNP and hs-cTn levels. This may be attributable to various factors, including the limited number of studies, heterogeneity of the interventions, and baseline biomarker levels. Nevertheless, the observed downward trend in cardiac serum biomarker levels remains clinically relevant, suggesting that physical activity may mitigate cardiotoxicity to some extent by promoting cardiomyocyte repair and enhancing cardiac adaptations.
Effects of Physical Activity on and Vascular Endothelial Function and CTRCT
We additionally evaluated FMD, a crucial indicator of vascular endothelial function, in individuals who have undergone chemotherapy for cancer. FMD can assess endothelial diastolic function by measuring the vasodilatory effect of blood vessels in response to physiological blood flow stimuli. This index reflects the ability of endothelial cells to release nitric oxide (NO), which is a crucial vasodilatory factor.27,29
Studies have shown that vascular endothelial diastolic dysfunction is strongly associated with anthracycline-induced cardiotoxicity.55 -58 On the one hand, anthracycline-based chemotherapeutic agents can directly damage vascular endothelial cells, affecting their dependent vasodilatory function (e.g., brachial and coronary arteries). This vascular endothelial dysfunction further increases the risk of developing cardiotoxicity, as it leads to impaired coronary vasodilatory function, which ultimately affects blood supply to the heart. On the other hand, after the occurrence of cardiotoxicity, CTRCT further impairs the pumping function of the heart and reduces the shear force of blood on the vascular endothelium, which in turn drives a reduction in the release of vasodilatory factors of the vascular endothelium.55 -58 Therefore, FMD can be used as a screening tool to detect anthracycline-induced cardiotoxicity. In the present meta-analysis, we found that physical activity markedly increased FMD values compared to the control group. This indicated that physical activity could help to counteract the impaired vascular endothelial function, which provided a potential novel mechanism for enhancing CTRCT.
Study Limitations and Perspectives
Based on our understanding, despite this study is the first systematic review and meta-analysis of physical activity aimed at reducing cardiotoxicity among cancer survivors undergoing chemotherapy, we do recognize several limitations. First, the number of included studies was limited, especially those that measured primary outcomes (GLS, LVEF, and FMD). And the heterogeneity observed in the study results may be attributed to various interventions and the diversity of cancer chemotherapy regimens, potentially influencing the reliability of the meta-analysis.
Second, due to the individual needs of physical activity in cancer patients, there is variability in the reporting of exercise intervention specifics (e.g., intensity and duration), and its variability could affect the comparability and reproducibility of the results, which in turn potentially affects the conclusions of the meta-analysis. Therefore, we recommend that future studies strictly follow the guidelines for exercise prescription among various cancer patients receiving chemotherapy when reporting the effects of exercise interventions, thereby ensuring the scientific validity and efficacy of exercise intervention protocols.
In addition, most studies included focused on short-to-medium term follow-up, and there was a lack of long-term follow-up studies (only 3 studies were identified17,18,25). This limits the understanding of the long-term effects of physical activity on CTRCT and CRF. Three studies reported relative VO2peak or VO2max, and one study reported E/A, both of which showed significant improvement during long-term follow-up (>16 weeks). Therefore, we recommend that future researchers could consider investigating physical activity (such as 16-48 weeks of progressive aerobic exercise combined with resistance training) on CTRCT and CRF among stage I-III cancer survivors undergoing chemotherapy so that the long-term effects are more well-understood.
Moreover, only 6 studies reported explicit blinding of outcome assessors, and one study reported explicit blinding of the participants or therapists, which may potentially introduce performance and detection bias. Therefore, we recommend that future researchers consider performing and reporting blinding of the participants and therapists as much as possible when exploring the effects of physical activity on cancer survivors undergoing chemotherapy with or without cardiotoxicity. In addition, the blinding of outcome assessors should be maximized to reduce detection bias.
Furthermore, only one study explored the impact of mind-body exercise on cardiotoxicity in cancer patients undergoing chemotherapy, which may limit the comprehensive understanding of physical activity affecting cardiotoxicity. In sensitivity analysis, including the Buddhist walking meditation study did not change the results of assessing the effects of exercise. The results provided additional support; however, incorporating a greater number of studies on mind-body exercises in the future could enhance the reliability of the findings. Hence, we suggest that future research should continue to delve into this area to achieve a more comprehensive understanding.
Finally, some of the studies required data transformation for some of the outcome indicators due to differences in data statistics and presentation methods, which could potentially introduce bias.
Conclusion
Overall, physical activity may be an effective and safe complementary therapy for various cancer survivors undergoing chemotherapy, particularly for improving cardiorespiratory function and reducing cardiotoxicity. Medium-to-long term and medium-to-high intensity aerobic exercise combined with resistance training may be more beneficial for cardiorespiratory function, subclinical indicators of cardiotoxicity, cardiac function, and vascular endothelial function. However, the study was limited by the limited number of studies and sample sizes, as well as the heterogeneity of the interventions and chemotherapy regimens. Future studies should optimize the design, expand the sample sizes, and control for confounding variables to more accurately assess the effects of physical activity on cardiopulmonary function and cardiotoxicity in various cancer syrvivoes following chemotherapy.
Supplemental Material
sj-docx-1-ict-10.1177_15347354241291176 – Supplemental material for Effects of Physical Activity on Cardiotoxicity and Cardio respiratory Function in Cancer Survivors Undergoing Chemotherapy: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-ict-10.1177_15347354241291176 for Effects of Physical Activity on Cardiotoxicity and Cardio respiratory Function in Cancer Survivors Undergoing Chemotherapy: A Systematic Review and Meta-Analysis by Kang Chen, Hui Guan, Meixia Sun, Yukun Zhang, Wenwen Zhong, Xiaonan Guo, Anqi Zuo and He Zhuang in Integrative Cancer Therapies
Footnotes
Author Contributions
He Zhuang and Kang Chen designed the study. Kang Chen and Hui Guan contributed to the publications search, data extraction and data assessment. Kang Chen completed the entire manuscript writing. Meixia Sun, Yukun Zhang, Wenwen Zhong, Anqi Zuo and Xiaonan Guo helped with manuscript revision. All authors read and approved the final version of the manuscript. Kang Chen is the first author. He Zhuang is the leader of this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Shandong Provincial Administration of Traditional Chinese Medicine funding project (2021Q081).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
