Abstract
Keywords
Background
Patients with advanced cancer exhibit a wide range of agonizing physical and psychological signs and symptoms, including pain, fatigue, insomnia, and distress. Consequently, an individual’s capacity to perform their daily tasks and the effectiveness of cancer therapy may both be compromised by the burden of signs and symptoms. 1 Moreover, in patients with advanced cancer, the intensity of the symptoms is a proven predictor of physical functionality. Furthermore, distressing cancer symptoms have a substantial effect on the patient’s emotional well-being and have been associated with a poor quality of life among patients with advanced cancer.2 -5
Cancer-related fatigue is a broad concept that is challenging to define appropriately. Moreover, the multidimensional nature of this condition is exemplified by its incorporation of numerous factors, including the environment, physical health, emotional well-being, cognitive functioning, social relationships, and even spiritual aspects. Nonetheless, it has become increasingly essential to develop a comprehensive understanding and clear definition of cancer-related fatigue to consistently diagnose, manage, and alleviate the accompanying signs and symptoms.6-8,20 In addition, cancer-related fatigue is characterized by a significant decrease in energy levels and an increased requirement for rest that is unrelated to previous physical exertion. As well, cancer-related fatigue also has a detrimental impact on the overall quality of life as it impairs both mental and physical functioning, disrupts mood stability, and reduces the ability to carry out daily tasks.10,11 Therefore, considering the multidimensionality associated with the concept of cancer-related fatigue, there have been multiple interpretations and definitions, resulting in a wide variety of understanding.
The definition of cancer-related fatigue has been proposed by several international healthcare institutions. To illustrate, one of the earlier and most used definitions of cancer-related fatigue was established by the North American Nursing Diagnosis Association (NANDA). According to NANDA in 1998, cancer-related fatigue is “An overwhelming, sustained sense of exhaustion and reduced capacity for physical and mental work.” This definition emphasizes the overwhelming and long-lasting nature of fatigue experienced by individuals affected by cancer. 12 Furthermore, the National Comprehensive Cancer Network (NCCN) in 2023, defined cancer-related fatigue as “a distressing persistent subjective sense of physical, emotional, and/or cognitive tiredness or exhaustion related to cancer or cancer treatment that is not proportional to recent activity and interferes with usual functioning.” 13
Cancer-related fatigue affects nearly all cancer patients at a certain point throughout their illness and treatment regimen, in addition, it is one of the most frequently noticed detrimental consequences of cancer progression.14 -16 To illustrate, according to previous studies, about 50% to 90% of cancer patients experience fatigue regardless of their age, gender, malignancy type, stage of the disease, as well as cancer medical treatment. Furthermore, it is worth noting that approximately 85% of individuals who have metastatic tumors additionally experience significant levels of cancer-related fatigue.6,17
Fatigue has a negative impact on the overall quality of life and the ability to perform everyday tasks. As a result, individuals experiencing fatigue are compelled to adjust their regular habits, and it further hinders their participation in various social and physical activities. Therefore, the debilitating consequences of fatigue extend beyond tiredness and significantly disrupt various aspects of an individual’s life. 42 Previous literature consistently emphasizes the profound influence of fatigue on individuals’ well-being and highlights the necessity for interventions in order to relieve cancer-related fatigue effectively.17-20
According to the National Comprehensive Cancer Network’s (NCCN) recommendations, fatigue should be examined, evaluated, and treated in accordance with standard clinical practice. However, a number of variables are linked with cancer-related fatigue across different stages of the disease. Therefore, assessment and identification continue to be complicated and frequently overlooked. As a result, the diagnosis and management of this common condition are still challenging, despite the substantial prevalence of cancer-related fatigue. Early management and enhanced awareness and focus on cancer-related fatigue are thus particularly crucial. Pain, emotional issues, and sleep issues are all treatable causes of cancer-related fatigue. These treatable issues may necessitate a combination of nonpharmacological and pharmaceutical therapies. 13 Furthermore, due to the limited effectiveness of pharmacological, there is a growing interest in a non-pharmacological approach as an effective way to manage chronic cancer-related fatigue. 21
Several patient-centered technologies developed and employed recently seem to promote the present shifts in healthcare paradigms by optimizing and integrating services as required in accordance with patient needs. For instance, smartphone technology, virtual reality, video games, and smartphone applications are a few of the innovative interventions employed for alleviating cancer-related fatigue. Additionally, this technology provides opportunities for psychological techniques including guided imagery and relaxation exercises in order to improve the management of symptoms. Moreover, evidence from the literature showed a positive effect of various technology procedures when combined with traditional standards as an option for effectively managing these symptoms. In addition, all of these interventions foster the empowerment of patients and individualized care. Recently, greater emphasis has been directed toward technologies embracing virtual reality for producing the most substantial advance in the field.22 -24
Specifically, virtual reality technologies are considered as a novel strategy and have acquired prominence in recent years as non-pharmacological symptom management approaches. Furthermore, virtual reality can serve as an advanced alternative suggestion for traditional psychological techniques by distracting patients from unpleasant sensory stimuli and shifting their attention to the virtual world.25 -27 Virtual reality is the computer-generated simulation of a three-dimensional image or environment that allows users to interact with it in a way that feels real or physical using highly specialized electronic equipment, such as a head-mounted display or a cave with stereoscopic projections and various input devices.28,29 Moreover, virtual reality technologies can serve as an advanced alternative suggestion for traditional psychological techniques by distracting patients from sensory stimuli and shifting their attention to the virtual world.25 -27 Furthermore, virtual reality technology is increasingly becoming accessible, affordable, and effective in achieving a higher degree of realism attributable to rapid technical advancements in cancer symptom management.30,31
However, there is a lack of awareness and availability of alternative nonpharmacological treatments, which can be attributed to the inadequacies of cancer symptom management therapies across patients with cancer. 32 In addition, despite the fact that virtual reality has many advantages, there is little information at the national and international levels about how it relieves cancer-related fatigue. In addition, despite the progress and utilization of virtual reality, there remains a significant gap in understanding the potential side effects and negative consequences of this technology among cancer patients, both on a national and international level. Hence, conducting this study will contribute substantially to the existing body of knowledge regarding the adverse effects of virtual reality, which is a highly needed knowledge.
In the scope of virtual reality protocols and guidelines, there is still a variation in the protocol for employing virtual reality since there is no standardized protocol or guideline worldwide; therefore, this review study aims to identify the most common and effective protocol for utilizing the virtual reality in managing fatigue by thorough and comprehensive review of previous literature. Thus, this systematic review aimed to evaluate the effectiveness of immersive virtual reality in cancer-related fatigue management, and to explore the main side effects of immersive virtual reality among patients with cancer.
Method
Protocol and Registration
This systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) standard. The protocol for this study has been registered in the prospective international register of systematic reviews “PROSPERO,” with the registration ID CRD42024507044.
Searching Strategies
The search process targeted articles published in peer-reviewed journals relevant to the study keywords in the period from 2014 to 2024. The following electronic databases were used in the search: MEDLINE, PubMed, Embase, CINAHL (Cumulative Index to Nursing and Allied Health Literature), EBSCO, SAGE Journals, Google Scholar, and ScienceDirect. In addition, an extensive search was conducted for gray literature in order to identify articles that were relevant to the topic (Figure 1). The PICO framework was used to form clinical queries and facilitate a literature search (Table 1). Relevant keywords were identified through a primary search on various electronic databases. The keywords used in the search were: “cancer-related fatigue,” “virtual reality,” “cancer distressing symptoms,” “patients with cancer” and "virtual reality related side effects;” Boolean operators (AND, OR) have been used alone, and combined with keywords to expand, combine, or exclude keywords in search to filter the results. Before including any study in this systematic review, the relevant articles underwent a thorough critical appraisal process. This appraisal was conducted according to the guidelines recommended by the Critical Appraisal Skills Program (CASP). Each study was carefully assessed using a set of questions: was the study conducted in a valid manner? What were the outcomes reported? And how useful are these outcomes in addressing the research question? The evaluation of all articles retrieved during the search aimed to identify any weaknesses and strengths, thus minimizing any potential threats to the validity of the findings.

PRISMA flow diagram of study selection.
PICO.

Eligibility
The inclusion criteria for considering relevant articles in this review were: (1) Patients diagnosed with any type of cancer, (2) patients suffering from cancer-related fatigue, (3) patients older than 18 years, (4) systematic reviews or clinical trials (randomized control trials and quasi-experimental studies), had virtual reality with or without cancer-related fatigue medications as study intervention, (5) and published in English. However, studies were excluded if the population of interest was receiving more than 1 complementary therapy to manage cancer-related fatigue.
Study Selection
Two researchers independently screened retrieved studies by titles and abstracts to identify relevant articles that meet inclusion criteria. Any disagreement was resolved by a third researcher. Studies identified as relevant and eligible were reviewed by the full text.
Study Synthesis
The study’s data synthesis was a collaborative effort involving 3 reviewers who meticulously examined and evaluated the gathered data. To ensure the accuracy and trustworthiness of the findings, any discrepancies in opinions or interpretations were resolved through consensus among the reviewers. Only data that received unanimous approval from all 3 reviewers were incorporated into the final analysis. A comprehensive narrative-descriptive synthesis was created to provide an overarching view of the evidence, which included a summary table. This table showcased the primary findings from each study and also examined the consistency of these findings across the different research studies.
Data Extraction and Management
The process of reviewing research records was carried out by 2 reviewers who worked independently. In the event of any disagreement between the 2 reviewers, a third researcher was involved to help resolve it. To ensure comprehensive data collection, various relevant information was included in a data extraction form. This information encompassed details such as the names of the authors, the year of publication, the study design, the characteristics of the participants, any additional interventions used alongside virtual reality, the size of the sample, the specific type of virtual reality intervention employed, and the method of measuring outcomes were provided in a data extraction form.
Quality Assessment
The quality of clinical trials was evaluated by using the Effective Public Health Practice Project (EPHPP) Quality Assessment Tool. Each study was evaluated in 6 domains (study bias, study design, control of confounders, blinding, data collection method, and withdrawals and dropouts of participants from the study). Each article’s quality was classified as strong, moderate, or weak based on the scores given to each dimension. The results from each domain were then summed to provide an overall score. The studies were graded as poor quality (two or more elements were rated as weak), moderate quality (one element was rated as weak), or high quality (no element was rated as weak). Two researchers independently assessed the studies’ quality.
The quality of systematic reviews was assessed using the measurement tool to assess systematic reviews (AMSTAR2). The tool has 16 elements that have to be rated as critically low, low, moderate, or high. The overall confidence rating was derived for each systematic review based on a combination of scores on 7 critical and 9 noncritical items. Therefore, each article could obtain a score between 0 (no criteria) and 16 (all criteria). Disagreements and questions that emerged throughout the review process were resolved through discussions and agreement between researchers.
Risk of Bias
The risk of bias in the included quantitative studies was evaluated using the Cochrane risk of bias tool/version 2 (ROB 2). Accordingly, each study was evaluated through the different domains included in ROB 2, which reflect several aspects of the result of each included clinical trial. These domains include random sequence generation, allocation concealment, degree of blinding, sample drop, and non-selective reporting. Each domain of ROB 2 is evaluated in relation to the risk of bias found in 1 of the 3 major assessments: Low risk of bias, some concerns of bias, or high risk of bias. The overall risk of bias judgment is evaluated by assessing the different domains of ROB 2 for each study and deciding based on those domains’ evaluations of the risk of bias.
Assessment of bias was conducted using the ROBIS tool, which was designed specifically to assess the risk of bias in systematic reviews. Accordingly, the bias tool evaluated each systematic review as a “high,” “low,” or “unclear” risk. The tool is completed in 3 phases: relevancy assessment (optional), identification with the review process, and risk judgment of bias in the review study. The risk of bias assessment includes signaling questions to assist in analyzing specific concerns regarding potential biases. The evaluations from these signaling questions assist assessors in determining the overall risk of bias.
Result
Characteristics of Included Studies
Ten articles were included in this review study, all studies published between 2019 and 2023 and written in English. As well as in all included studies immersive virtual reality was utilized as a non-pharmacological innovational intervention for cancer-related fatigue among patients with different types of cancer. In total, 6 out of 10 articles were clinical trial studies, and the other 4 were review studies. A summary of the study’s characteristics is shown in (Table 2), which includes study title, design, and major findings.
Study Characteristics Based on Authors, Publication Year, Study Title, Design, and Major Findings.

Result of Quality Assessment
Quality of included clinical trial studies
The quality of 6 included clinical trial studies (50% of studies were randomized control trials and 50% were quasi-experimental studies) in this review was assessed using the Effective Public Health Practice Project (EPHPP) quality assessment tool for quantitative studies (Table 3). Based on the result, in all studies the researchers used the voluntary approach of participant recruitment, therefore, all participants agreed to participate in the study prior any intervention and the authors stated that the consents were signed by all participants before the intervention. Furthermore, the data in all included studies (100%) were collected using a tool shown to be reliable and valid, all authors of included studies (100%) reported the number of withdrawals and dropouts of participants in their study. In addition, the percentage of participants who completed the study in all articles was above 80%. However, one study used the unblinded approach. 32 Thus, the overall result after both reviewer discussions showed that 30% of the studies were rated with moderate quality, no study was rated with low quality, and (70%) were rated as high-quality studies. Hence, the results emphasize the high quality of the clinical trials incorporated in this review. This indicates that the studies included in this analysis were conducted with robust methodologies and adhered to rigorous standards. Consequently, the evidence synthesized in this review can be considered trustworthy.
Effective Public Health Practice Project (EPHPP) Quality Assessment Tool for Quantitative Studies (n = 6).

Total rating score (1 = strong, 2 = moderate, 3 = weak).
Quality of included review studies
The quality of all review studies in this review was assessed using a quality assessment checklist for systematic reviews (AMSTAR 2). According to this review, 2 systematic review and meta-analysis studies fulfilled all the AMSTAR 2 scale requirements. Indeed, the majority of included systematic reviews showed a low risk of bias and high to moderate power of evidence (Table 4). The score varied from (12) to (16) points, with an average of 14 out of 16 possible total points. Items 1, 2, 3, 6 to 13, and 16 were scored as positive for all included reviews.
Quality Assessment Checklist for Systematic Reviews (AMSTAR 2).

Result of Risk of Bias Assessment
Risk of bias assessment of the included clinical trials
The risk of bias for included trials was evaluated by the Cochrane Risk of Bias tool (ROB). Each domain of ROB2 was evaluated concerning the risk of bias found in 1 of the 3 major assessments: Low risk of bias, some concerns of bias, or high risk of bias. The overall risk of bias judgment was evaluated by assessing the different domains of ROB 2 for each study. The decision was made based on those domains’ evaluations of the risk of bias. Among the included clinical trials, 3 studies (50%) were considered to have a low risk of bias after assessment with ROB 2. As well as 3 studies (50%) exhibited only minor concerns of bias. Thus, these findings signify the methodological robustness observed in the majority of the included clinical trials. As well as the significant proportion of studies demonstrating a low risk of bias underscores the adherence to rigorous research practices and standards within the field of clinical trial conduct. Conversely, the identification of some bias concerns in the remaining studies suggests areas for potential improvement in future research endeavors. The information in Table 5 presents a comprehensive overview of the evaluation of each domain in the ROB 2 tool for each study that was included, along with an overall risk of bias assessment.
Cochrane Risk of Bias Tool for Quantitative Studies (n = 6).

Reported (Some concerns of bias).
Reported in full (low risk of bias).
–Not reported (High risk of bias).
Risk bias for systematic review
The evaluation results obtained using the ROBIS tool indicated that there was a low overall risk of bias in all 3 reviews that were included in the analysis Figure 2. The systematic review and meta-analysis that were included in the assessment framed their research questions in a manner that was consistent with the scope of their review. Additionally, all of the studies provided comprehensive details regarding their eligibility criteria, and they clearly defined the PICO components. However, it is worth noting that none of the reviews reported the registration of a previous protocol, and all of the studies imposed restrictions on including studies based on language without providing a justification for doing so. The risk of bias in the included systematic reviews, which was evaluated using the ROBIS tool, is presented in Table 6.

Risk of bias assessment with ROBIS tool.
Risk of Bias Assessment With ROBIS Tool.

?, unclear risk.
T = the effectiveness of virtual reality in managing cancer-related fatigue based on included clinical trials
Several randomized control trials have been conducted among patients with cancer to assess the effectiveness of virtual reality in managing multiple distressing cancer-related symptoms such as fatigue and were included in this review study Table 7. Out of all the trials that were included, 2 clinical trials successfully enrolled participants who had been diagnosed with different types of cancers, without any limitations on the specific type of cancer. To illustrate, a recent RCT was conducted in the United States to compare the level of fatigue pre- and post-virtual reality application among patients diagnosed with various cancers. As well, the participants were asked to rate their fatigue levels before and after the intervention by selecting a number from 1 to 5 based on their perceived level of fatigue. For instance, a rating of 5 corresponded to feeling “Very Good,” while a rating of 1 indicated feeling “Very Bad.” Consequently, the results of this research study demonstrate that the integration of virtual reality technology with meditation techniques has a significant impact on reducing fatigue among patients diagnosed with cancer. Furthermore, the data obtained strongly suggests that the inclusion of virtual reality as an adjuvant to conventional therapies has yielded positive outcomes for patients, resulting in a sense of calm, heightened relaxation, refreshment, and a strengthened sense of self-empowerment among the individuals undergoing cancer treatment. 33
Characteristics of Included Trials Based on Country, Number of Participants and Type of Cancer.
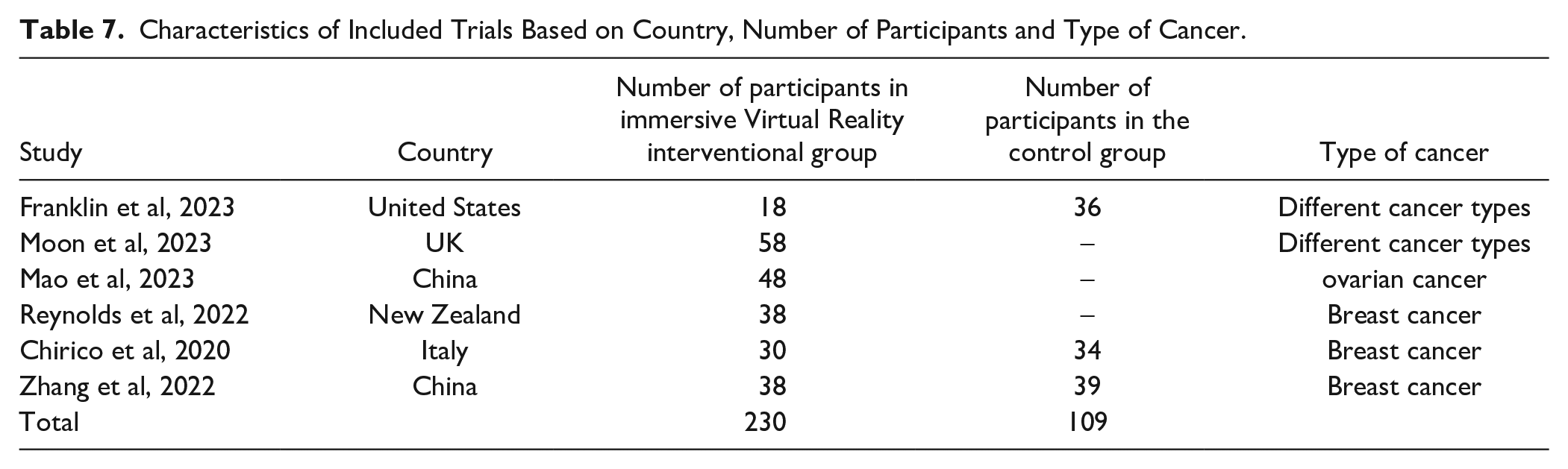
The second trial included in this review was conducted in the UK with the purpose of investigating whether the use of virtual reality had an impact on the revised Edmonton Symptom and Assessment System (ESAS-r) score, which is a measure of symptom control and well-being. The results of this study revealed that virtual reality led to a significant improvement in symptom control, particularly in reducing tiredness. Furthermore, there was a notable enhancement in overall well-being, which was both statistically and clinically significant. Specifically, the level of tiredness experienced by the participants decreased from an average score of 3.17 (with a standard deviation of 2.62) to 2.16 (with a standard deviation of 2.46) after the Virtual Reality intervention. 34
In the specific context of investigating the efficacy of using virtual reality as an intervention to alleviate cancer-related fatigue and other symptoms among individuals diagnosed with ovarian cancer, it is important to note that only a single clinical trial was discovered and subsequently included in this comprehensive review study. This clinical trial was conducted in China, to investigate the effects of a unique training approach that combines virtual reality technology and mindfulness on cancer-related fatigue among patients with ovarian cancer undergoing chemotherapy. The fatigue level was assessed using a cancer-related fatigue scale. The results of the study revealed that the use of mindfulness training based on virtual reality technology resulted in a significant reduction in cancer-related fatigue among ovarian cancer patients undergoing chemotherapy, with a statistically significant difference (P < .001). 35
In addition, numerous clinical studies have been carried out to assess the efficacy of virtual reality in managing cancer-related fatigue and other cancer-related symptoms among patients with breast cancer. Thus, within the scope of this review, 2 clinical trials were identified that specifically investigated the impact of virtual reality on cancer-related fatigue among patients with breast cancer. To illustrate, in a recent study conducted in New Zealand, researchers aimed to explore the potential effectiveness and acceptability of virtual reality as a supplementary therapy for women with metastatic breast cancer (MBC). The study involved a total of thirty-eight women diagnosed with MBC who underwent virtual reality interventions and were included in the analysis. Throughout the study, all participants engaged in 2 separate week-long virtual reality experiences, and their symptoms were evaluated at various points in time: before the intervention, after 1 week, and 48 hours later. The fatigue level was assessed using the FACIT fatigue scale. The findings of the study revealed notable improvements in fatigue levels immediately after the intervention and 48 hours later. Furthermore, the participants expressed positive emotions such as relaxation, comfort, and enjoyment during the virtual reality experiences and indicated a strong inclination to continue utilizing these interventions in the future. 36 In addition, a second trial was carried out in Italy. The purpose of this study was to evaluate the effectiveness of immersive and interactive virtual reality as a distraction approach to alleviate various symptoms associated with cancer treatment, particularly fatigue. Additionally, the study aimed to compare the effects of virtual reality intervention with a music therapy-based intervention in a group of individuals diagnosed with breast cancer. This trial involved a total of 94 patients, with 30 patients assigned to the virtual reality group, 30 patients assigned to the music therapy (MT) group, and 34 patients assigned to the control group, which received standard care while undergoing chemotherapy for breast cancer. The findings of this study indicate that both virtual reality and music therapy interventions show promise in reducing fatigue levels among breast cancer patients undergoing chemotherapy. Additionally, the results suggest that virtual reality may be more effective than music therapy in alleviating symptoms of anxiety, depression, and fatigue. 32
Furthermore, the utilization of a virtual reality-based intervention also proved to be effective in alleviating fatigue in breast cancer survivors. For instance, recent research conducted in China recruited a total of 98 breast cancer patients for the study. These patients were randomly divided into 2 groups: the VR-CALM group and the care-as-usual (CAU) group. Before and following the application of VR-CALM or CAU, all patients underwent evaluations using the Piper Fatigue Scale (PFS). The findings from this study indicated that individuals in the VR-CALM group experienced a significant reduction in fatigue levels and reported a higher quality of life compared to those in the CAU group post-intervention. 37
The effectiveness of virtual reality in managing cancer-related fatigue based on included review studies
A comprehensive search was performed to identify relevant systematic reviews and meta-analysis studies. As a result, a total of 3 relevant reviews were included in this analysis Table 8. Each of these reviews specifically focused on investigating the effectiveness of virtual reality as a therapeutic intervention for addressing cancer-related fatigue and various other cancer-related symptoms. All the review studies that are included in this review have consistently shown that the use of virtual reality interventions has a notable and statistically significant effect in reducing cancer-related fatigue among individuals with various types of cancer. To illustrate, a comprehensive review conducted by Zeng et al 38 focused on investigating the latest studies that utilized virtual reality-based interventions for managing symptoms in patients diagnosed with cancer. The primary objective of this study was to quantitatively assess the effectiveness of virtual reality-based interventions in alleviating cancer-related symptoms. In order to achieve this goal, a total of 6 empirical studies were carefully selected, analyzed, and included in the review. The findings of this study revealed that virtual reality-based interventions exhibited statistically significant effects in reducing fatigue symptoms among cancer patients. 38 In accordance with these findings, also there is a previous systematic review conducted to assess the effectiveness of virtual reality on symptoms such as anxiety, depression, fatigue, and pain. Consequently, the findings of this systematic review provide strong support for the hypothesis that virtual reality has a significant positive impact on reducing symptoms of anxiety, depression, fatigue, and pain when compared to standard care. Overall, this study contributes to the growing body of research on the therapeutic benefits of virtual reality in the field of healthcare, particularly in improving the well-being of individuals facing various physical and psychological challenges, such as those associated with cancer. 39
Characteristics of Included Systematic Reviews and Meta-Analysis Based on the Number of Included Studies, Type of Cancer and Assessed Variables After Virtual Reality Sessions.

Furthermore, according to the findings of a recent systematic review conducted by Yazdipour et al, 39 virtual reality has the potential to offer a multitude of benefits to individuals who are diagnosed with breast cancer. As well as utilizing this advanced innovation technology, patients can experience a reduction in anxiety levels, pain, fatigue, distress, and depression. Additionally, virtual reality has proven to enhance the range of motion, strength, and overall functionality of these patients. Therefore, introducing virtual reality intervention into the treatment of breast cancer burden symptoms presents an opportunity for patients. This novel technology has demonstrated remarkable effectiveness in aiding rehabilitation efforts and addressing several symptoms associated with the disease. Moreover, virtual reality interventions can be implemented at various stages of therapy, all with the ultimate goal of significantly improving the overall condition and well-being of patients with breast cancer. 40 In accordance with the findings of the included studies, a recent study conducted by Gautama et al 40 aimed to evaluate the impact of immersive virtual reality on alleviating various symptoms experienced by adult and pediatric cancer patients undergoing chemotherapy. This systematic review and meta-analysis included a total of fifteen trials, with a combined participation of 607 adult patients and 257 pediatric patients. Thus, the findings of the study revealed that the use of immersive virtual reality resulted in a significant decrease in symptoms such as anxiety, depression, fatigue, and systolic blood pressure in the participants 41
Side Effects of Immersive Virtual Reality
After analyzing data from numerous clinical trials, it was revealed that a significant majority of the studies documented and assessed the adverse effects experienced by participants following the use of virtual reality interventions. These side effects were consistently reported by researchers after the participants completed their virtual reality sessions.32,34 -36 However, there is still a significant variation in the measures used to assess these side effects across the different studies. Nevertheless, virtual reality is generally considered to be a safe intervention with minimal or temporary side effects that typically resolve within 24 hours without the need for management. For example, in a study conducted by Mao et al, 35 a combination of quantitative clinician and patient questionnaires (using a scoring system of 0-10) as well as a qualitative questionnaire on negative effects were used. The results of this study indicated that all participants believed that the virtual reality model was safe and did not experience any uncomfortable symptoms or adverse events such as dizziness or nausea. Another study by Chirico et al 32 assessed the negative side effects of virtual reality using the Virtual Reality Symptom Questionnaire (VRSQ). The results from this questionnaire showed that all symptoms occurred with a frequency of less than 20%, which the authors of the scale considered to be negligible, except for a slight difficulty in concentrating which was reported by 21.4% of the participants. 32
On the other hand, in a study conducted by Moon et al, 34 it was found that only a single participant, representing 1.7% of the total, had to discontinue their virtual reality experience due to a negative reaction. This individual cited the smell of the headset plastic as the reason for stopping. The most frequently reported negative effect among participants was feelings of nausea, with other complaints including feeling overheated, experiencing visual strain, and finding the headset to be cumbersome or uncomfortable. 34 Furthermore, as indicated in previous research conducted by Reynolds et al, 36 few individuals who took part in the study shared experiences of encountering negative effects while utilizing virtual reality headsets. These adverse effects included reporting feelings of being “claustrophobic” and expressing that during their initial exposure to virtual reality, they experienced dizziness and nausea after engaging with various interactive elements within the virtual environment.
Virtual Reality Implementation Protocols
In the scope of virtual reality protocols for cancer-related fatigue management among patients with cancer, the examination of implemented protocols within the studies incorporated in this review discovered heterogeneity in the application of virtual reality-based interventions Table 9. This diversity refers to the number of virtual reality sessions, which vary from a minimum of one session to a maximum of twenty-eight sessions. Additionally, there was considerable variation in the duration of virtual reality interventions across the included clinical trials. Notably, two of the included studies32,33 implemented virtual reality interventions within 1 day, encompassing both the virtual reality session and the subsequent follow-up period. In contrast, in a recent study conducted by Zhang et al,30,37 the virtual reality intervention extended over 3 months, comprising a total of six separate sessions. This prolonged intervention timeframe, spanning multiple months, represents the most extended duration observed among the studies included in this review. 37 Furthermore, 2 of the included studies reported that the virtual reality interventions were completed within a 2-week timeframe in their respective trials. Thus, this duration suggests a more moderate approach for virtual reality implementation.34,36 Lastly, in a study conducted by Mao et al, 35 the intervention was conducted and completed over 4 weeks. However, it is crucial to note that this variability in implemented protocols aligns closely with the specific aims of each study, particularly in addressing various burden symptoms beyond cancer-related fatigue. These additional symptoms may include depression, anxiety, and the enhancement of quality of life. Notably, the analysis of included studies suggests that the management of these clinical outcomes may necessitate a higher number of virtual reality sessions. This finding underscores the nuanced nature of treatment protocols, wherein the tailored adjustment of session frequency and duration is essential to effectively address the multifaceted therapeutic objectives associated with Virtual reality-based interventions in oncology settings.
Virtual Reality Protocols.

Abbreviation: VR, Virtual Reality.
Discussion
Cancer-related fatigue is a highly prevalent symptom that is commonly observed among patients both before and after undergoing medical treatment. Numerous studies have consistently demonstrated that cancer patients often consider fatigue to be the most distressing symptom, significantly impacting their overall quality of life when compared to other symptoms such as pain and depression that are also associated with cancer. Consequently, individuals who experience severe fatigue are often less inclined to participate in social interactions and physical activities.17 -20 In recent years, there has been a growing recognition of the limitations of pharmacological methods in effectively managing chronic cancer-related fatigue. As a result, there is a growing interest in exploring non-pharmacological approaches that can provide significant relief for individuals experiencing this debilitating condition. However, there is still a lack of existing literature regarding the effectiveness of immersive virtual reality in cancer-related fatigue. Thus, this review study has been conducted to assess the effectiveness of immersive virtual reality as an innovative technology for managing cancer-related fatigue. Additionally, this research aims to investigate any potential side effects associated with immersive virtual reality among patients with cancer.
Virtual reality represents an innovative and interactive approach to distraction, wherein individuals actively engage with virtual environments to immerse themselves in alternate realities. 32 This immersive experience is further enhanced in immersive virtual reality, where users are equipped with head-mounted displays facilitating three-dimensional interaction with computer-generated environments. 32 By simulating realistic sensory experiences, immersive virtual reality enables users to feel fully present within the virtual environment, blurring the boundaries between physical reality and digital simulations. Consistently, all of the included clinical trials in this review used head-mounted display devices and other supported virtual reality equipment to enhance the immersion experience and shift patient attention effectively to the virtual environment among participants during virtual reality sessions.32 -37
Cancer-related fatigue is a debilitating and burden symptom experienced by a significant proportion of cancer patients, significantly impacting their quality of life. 17 Moreover, the exact mechanisms underlying cancer-related fatigue are complex and multifaceted, involving an interplay of physiological, psychological, and environmental factors. 41 Thus, in recent years, virtual reality has emerged as a promising intervention for managing fatigue, with several studies demonstrating its efficacy in reducing fatigue levels in cancer patients.32 -37 However, despite these promising findings of included studies, the exact mechanisms underlying the fatigue-relieving effects of immersive virtual reality remain not fully understood. However, based on the existing literature, it is possible to elucidate two primary perspectives of how immersive virtual reality may alleviate fatigue, including physical and psychological effects. To illustrate, from a psychological perspective, immersive virtual reality can help decrease fatigue by providing a healthy distraction from the physical and emotional burden symptoms of cancer, as well as offering a sense of control and empowerment.43,44 Thus, this distraction hypothesis suggests that virtual reality can redirect the individual’s attention away from their fatigue and toward a more engaging and immersive experience, thereby reducing perceived fatigue. 45 Furthermore, the sense of control and empowerment afforded by immersive virtual reality may enhance an individual’s self-efficacy, leading to improved motivation, increased sense of relaxation, and then reduced fatigue. 46 Notably, these hypotheses are supported by the findings of recent clinical trials.32,33,36,37 The results of these trials indicated that the virtual reality intervention led to noticeable improvements in various aspects of quality of life, including fatigue, pain, depression, anxiety, and stress. Moreover, the participants reported experiencing feelings of relaxation and enjoyment and expressed a strong likelihood of continuing to use the interventions. These findings provide further evidence for the potential of immersive virtual reality as a therapeutic tool for managing fatigue and improving overall well-being in individuals with cancer.
In the context of the effectiveness of immersive virtual reality in managing cancer-related fatigue, a limited number of randomized control trials have been conducted among patients with cancer to assess the effectiveness of virtual reality in cancer-related fatigue, in this study a total of 6 clinical trials were found and included. Based on the result of these studies, the integration of virtual reality technology with meditation techniques has a significant impact on reducing fatigue among patients diagnosed with different types of cancer. Furthermore, the inclusion of virtual reality as an adjuvant to conventional therapies has yielded positive outcomes for patients, resulting in a sense of calm, heightened relaxation, refreshment, and a strengthened sense of self-empowerment among the individuals undergoing cancer treatment. 33 Furthermore, there was a notable enhancement in overall well-being in addition to cancer-related fatigue. 34 Still, there is a gap in the literature related to the effectiveness of virtual reality in managing cancer-related fatigue among patients with ovarian cancer; only one study was found and included in this review that examined the efficacy of using virtual reality as an intervention to alleviate cancer-related fatigue and other symptoms among individuals diagnosed with ovarian cancer in China. Thus, it was observed that the use of mindfulness training based on virtual reality technology resulted in a significant reduction in cancer-related fatigue among ovarian cancer patients undergoing chemotherapy. 35 Therefore, still, there is a need to conduct more studies among patients with ovarian cancer worldwide.
Additionally, when considering the effectiveness of virtual reality in addressing cancer-related fatigue in individuals diagnosed with breast cancer, it is important to note that 2 separate clinical trials were conducted to explore the efficacy of this novel technology.32,36 These trials provided valuable insights into the impact of virtual reality interventions, revealing a significant reduction in fatigue levels immediately following the intervention, which persisted even 48 hours later. Furthermore, participants who underwent virtual reality experiences reported experiencing positive emotions such as relaxation, comfort, and enjoyment, indicating a strong desire to continue utilizing these interventions in the future. In accordance, these findings are in line with the result of previous systematic reviews that support the effectiveness of virtual reality in managing fatigue among patients with advanced cancer, as well as the result showed that immersive virtual reality could provide many opportunities for patients with breast cancer, including reducing fatigue levels. 40 Notably, the results of one study also suggested that virtual reality may prove to be more effective than traditional music therapy in alleviating symptoms of anxiety, depression, and fatigue among patients with advanced breast cancer. 32 However, it is evident that there exists a substantial gap in the existing body of literature concerning the utilization of innovative virtual reality technologies for the management of fatigue. Consequently, we recommend that further randomized trials be undertaken to thoroughly investigate the efficacy of various virtual reality interventions in reducing cancer-related fatigue on a global scale.
Within the realm of symptoms related to cybersickness and other undesirable side effects that are associated with the usage of virtual reality among cancer patients, there exists a considerable discrepancy in the methods and tools employed to evaluate these side effects across various studies. Nevertheless, the most frequently reported negative effect among participants was feelings of nausea, with other complaints including feeling overheated, experiencing visual strain, and finding the headset to be cumbersome or uncomfortable.34,36 Additionally, certain individuals who utilized virtual reality-based interventions for managing cancer-related symptoms encountered adverse effects during their interaction with virtual reality headsets. These unfavorable effects encompassed feelings of being confined or “claustrophobic” and instances of dizziness following engagement with various interactive elements within the virtual environment during their initial exposure to immersive virtual reality. 36 Nevertheless, virtual reality is generally considered to be a safe intervention with minimal or temporary side effects that typically resolve within 24 hours without the need for management.32,35 However, to ensure the safety of implementing virtual reality technology for patients with cancer, it is crucial to conduct extensive clinical trials that thoroughly evaluate any potential side effects. As well as prior to utilizing virtual reality, it is essential to discuss these potential side effects with participants.
In the context of virtual reality-based intervention protocols, there exists a significant variation in the number and duration of sessions. The range of sessions included in clinical trials varies greatly, with some studies utilizing a single session lasting between 10 and 20 minutes,32,33 while others implement a more extensive regimen of fourteen sessions spread over a 2-week period, with each session lasting 10 minutes per day. 36 Additionally, a study conducted by Mao et al, 35 employed a virtual reality-based intervention for a duration of 4 weeks. However, the implementation of immersive virtual reality protocols in clinical trials exhibits significant variations, which are largely driven by the specific study objectives and the range of symptoms that researchers aim to manage among participants, including but not limited to cancer-related fatigue, depression, anxiety, and distress. 35 Thus, the adjustment of session frequency and duration depends on the burden symptoms that the researchers aim to manage in addition to cancer-related fatigue, and based on that, the protocol of virtual reality implementation will be adjusted to achieve the study objective.44,47
In conclusion, adopting an immersive virtual reality protocol for managing cancer-related fatigue requires a tailored approach that considers the specific study objectives, symptom burden, and individual patient needs. By understanding the complex relationships between immersive virtual reality, cancer-related fatigue, and various physiological, psychological, and environmental factors, researchers can develop more effective immersive virtual reality-based interventions that improve patient outcomes. Thus, there is a great need to establish a consistent and comprehensive protocol for the application of virtual reality among patients with cancer and suffering from cancer-related fatigue.
This study has a number of strengths and limitations. One of the notable strengths was the comprehensive search conducted across multiple reputable databases, gray literature sources, and thorough examination of reference lists from all the studies included in this review. This rigorous approach ensured the inclusion of all relevant studies conducted within the last decade, maximizing the comprehensiveness of this review. It is noteworthy that this systematic review stands out as the first of its kind which focused on investigating the effectiveness of immersive virtual reality in alleviating cancer-related fatigue. However, it is important to acknowledge the limitations of this study. Firstly, the number of clinical trials included in this review was relatively small. This can be attributed to the limited number of trials conducted in the specific context of utilizing virtual reality for managing cancer-related fatigue. Although efforts were made to include all available studies. Secondly, the included studies were conducted with small sample sizes. This highlights the necessity for future research to design trials with larger populations to enhance the robustness and generalizability of findings in this emerging field.
Implication for Practice and Future Research
The findings of this systematic review indicate that the use of virtual reality interventions among patients with cancer patients can significantly reduce cancer-related fatigue. Additionally, virtual reality is generally considered a safe intervention with minimal and temporary side effects that typically resolve within 24 hours without the need for intervention. As a result of these conclusions, it is advised that healthcare professionals incorporate virtual reality interventions into the care of cancer patients, including during chemotherapy, radiation therapy, post-surgery, and even during end-of-life care, in order to create protocols that empower patients and enhance their comfort. Future research should focus on larger sample sizes, longer intervention durations, and improved methodological quality, as well as investigating the potential negative side effects and cost-effectiveness of virtual reality interventions. Furthermore, all of the included clinical trials have been conducted in inpatient settings, it is recommended to also examine the effectiveness of virtual reality interventions in outpatient settings, such as in patients’ homes.
Conclusion
This systematic review has demonstrated that the use of virtual reality-based interventions holds great promise in effectively managing cancer-related fatigue. Virtual reality, being a new and innovative technology, can be easily incorporated into both inpatient and outpatient settings. Notably, a significant number of participants who underwent virtual reality therapy expressed feelings of relaxation, comfort, and excitement, and expressed a desire to continue using this intervention. Moreover, virtual reality therapy was found to be associated with minimal or no adverse effects. Furthermore, based on the findings from these studies, it is strongly recommended that healthcare professionals integrate virtual reality interventions into the treatment and support of patients with cancer. Future research in this area should prioritize larger participant groups, longer durations of intervention, and higher-quality methodological approaches. Additionally, it is crucial to explore any potential adverse effects and assess the cost-effectiveness of virtual reality interventions.
Footnotes
Author Contributions
The three authors of this systematic review contributed equally to the development of the research question and design, literature search, data extraction, and quality assessment of the studies. Specifically, Author 1 led the development of the research question and design, while Authors 2 and 3 contributed to the literature search, data extraction, and quality assessment. As well as, all three authors participated in the data synthesis and interpretation of the results, as well as the writing of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
