Abstract
Objective:
Powerful adjuvant strategies are required to improve the survival of patients with completely resected stage ΙΙΙA non-small cell lung cancer (NSCLC). We aimed to compare the efficacy of traditional Chinese medicine (TCM) treatment versus observation after adjuvant chemotherapy in these patients.
Methods:
Eligible patients were randomized 1:1 to receive either oral decoctions based on Qi-Yin syndrome differentiation (TCM group) or observation (observation group). The intervention lasted for 12 months. The primary endpoint was 1-year disease-free survival (DFS). Secondary endpoints were DFS, quality of life, regulatory T cells (Tregs), and cytotoxic T lymphocyte-associated antigen-4 (CTLA-4) on the surface of Tregs in peripheral blood. We used EORTC QLQ-LC43 to evaluate quality of life.
Results:
Between Apr 29, 2019, and Nov 11, 2021, 75 patients were randomly assigned to oral decoctions based on Qi-Yin syndrome differentiation (n = 38) or observation (n = 37). The full analysis set included 35 patients in the TCM group and 35 in the observation group. After a median follow-up of 24.2 months, oral decoctions based on Qi-Yin syndrome differentiation improved DFS compared with observation (HR 0.378, 95% CI: 0.157-0.912; P = .03). One-year DFS was 82.1% in the TCM group and 61.9% in the observation group (P = .06). Three months after randomization, scores of total health, role function, emotional function, and social function in the TCM group were higher than those in the observation group (P < .01 for all), scores of fatigue, pain, insomnia, appetite loss, constipation, cough, and chest pain were lower than those in the observation group (P < .05 for all); there was no significant difference in the proportion of Tregs between the TCM group and the observation group (P = .58); the proportion of CTLA-4+Tregs in the TCM group was lower than that in the observation group (P = .046). There were no adverse events that occurred in both groups.
Conclusions:
Oral decoctions based on Qi-Yin syndrome differentiation after adjuvant chemotherapy prolonged DFS, reduced the risk of disease recurrence and metastasis, improved quality of life, and down-regulated the proportion of CTLA-4+Tregs in completely resected stage ΙΙΙA NSCLC patients.
Trial Registration:
Chinese Clinical Trial Register, No. ChiCTR1800019396. Date of registration: 9 November 2018.
Keywords
Introduction
Lung cancer ranks as the first cause of cancer death globally. 1 Approximately 80% to 85% of all lung cancers are non-small cell lung cancer (NSCLC). Resected stage Ⅲ NSCLC is mainly stage ⅢA disease, which accounts for about half of stage Ⅲ NSCLC. 2 5-year survival rates are 92% in stage IA1 NSCLC and only 36% in stage ⅢA NSCLC. 2 Over a median follow-up of about 5 years, 76% of patients with stage Ⅲ disease have disease recurrence or die after surgery, regardless of the use of adjuvant chemotherapy. 3 Prevention of recurrence and metastasis for resected stage ⅢA NSCLC patients is essential, which is meaningful to prolong survival.
The goal of adjuvant treatment for postoperative patients with lung cancer is to prevent disease recurrence and metastasis. Adjuvant cisplatin-based chemotherapy yielded only a 5-year survival benefit of 5% in resected NSCLC.3,4 Adjuvant epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors (TKIs), such as gefitinib, 5 erlotinib, 6 icotinib, 7 and osimertinib8,9 have a greater improvement in DFS compared with adjuvant chemotherapy in resected EGFR mutation-positive NSCLC patients, and adjuvant osimertinib therapy translates into overall survival (OS) benefit. 10 The EVEN study only enrolled EGFR mutation-positive stage ⅢA NSCLC patients, adjuvant erlotinib for 2 years compared with adjuvant chemotherapy for 4 cycles could reduce the risk of disease recurrence or death by 73.2%. 6 The Impower010 trial is the first adjuvant immune checkpoint inhibitor (ICI) study to publish results, which gained a DFS benefit with adjuvant atezolizumab compared with best supportive care (BSC) in resected stage II–IIIA NSCLC after adjuvant chemotherapy (HR 0.79, 95% CI 0.64-0.96; P = .02), with superior benefit in patients whose tumors expressed programmed death 1 ligand (PD-L1) on 1% or more of tumor cells (HR 0.66, 95% CI 0.50-0.88; P = .0039). 11
The survival benefit of adjuvant chemotherapy is limited, the benefit population of adjuvant EGFR-TKIs is restricted, and adjuvant ICIs await more clinical research results. The advantages of TCM treatment include enhancing quality of life, improving immune function, and prolonging the survival of advanced NSCLC patients.12-15 The efficacy characteristics of TCM treatment are expected to improve the prognosis of resected stage ⅢA NSCLC patients by improving their disease resistance. Based on these, we designed a randomized controlled trial to evaluate whether oral decoctions based on Qi-Yin syndrome differentiation after adjuvant chemotherapy gained survival benefits in patients with resected stage ⅢA NSCLC.
Methods
Study Design
The trial was a randomized controlled study of oral decoctions based on Qi-Yin syndrome differentiation versus observation after adjuvant chemotherapy (adjuvant radiotherapy for stage IIIA-N2 was allowed) in completely resected stage ⅢA NSCLC patients, who were enrolled at Longhua Hospital affiliated with Shanghai University of Traditional Chinese Medicine. In this trial, outcome assessors and statisticians were blinded to the allocated treatment.
Qi-Yin Syndrome Differentiation Criteria 16
Syndrome differentiation criteria were based on “The Guiding Principles of Clinical Research of New Chinese Medicine (Trial)” 17 2 associate chief physicians conducted the diagnosis. The diagnosis could be made if at least 2 of the main symptoms and 1 of the secondary symptoms were present.
Qi deficiency syndrome: The main symptoms were coughing, a large amount of sputum, poor appetite, apathy and weakness, and pale and bulgy tongue. Secondary symptoms included spontaneous sweating, loose stool, and thin superficial and smooth pulse.
Yin deficiency syndrome: The main symptoms were coughing, a small amount of sputum, dry mouth, and red tongue. Secondary symptoms included night sweating, insomnia, low fever, and thready and rapid pulse.
Qi and Yin deficiency syndrome: The main symptoms were coughing, a small amount of sputum, shortness of breath, apathy and weakness, and thirst without wanting to drink. Secondary symptoms included spontaneous sweating, night sweating, reddish tongue, or tongue with teeth marks, and thready and weak pulse.
Participants
Eligible patients were diagnosed with primary NSCLC by pathological biopsy, 18 were completely resected (R0), had stage ⅢA disease per the seventh edition of the TNM classification, 19 did not receive neoadjuvant therapies, whose tumors were negative or unclear for EGFR or ALK alterations, were within 1 month after completing 4 cycles of adjuvant chemotherapy (stage ⅢA-N2 patients were allowed to receive adjuvant radiotherapy), were without recurrence or metastasis after adjuvant chemotherapy, were 18 to 75 years old, had ECOG PS score of 0 to 3, had normal liver and kidney functions, exhibited Qi and Yin deficiency, Qi deficiency, or Yin deficiency, and signed informed consent.
Exclusion criteria comprised: history of other malignant tumors in the past 5 years; previous exposure to TCM treatment after being diagnosed with NSCLC; existence of serious heart, liver, kidney, or any medical diseases which would interfere with the study; allergy to a drug used in the study; mental or cognitive disorders.
Randomization
Random numbers were automatically generated using the computer with no stratification factors. Eligible patients were randomly assigned 1:1 to either the TCM group or the observation group by opening an opaque envelope numbered in sequence after informed consent was obtained.
Treatment and Follow-Up
The intervention started within 1 week of randomization and lasted for 12 months. Patients received oral decoctions based on Qi-Yin syndrome differentiation as the sole adjuvant therapy in the TCM group. Professor Liu Jiaxiang at Longhua Hospital affiliated with Shanghai University of Traditional Chinese Medicine prescribed these 3 herbal formulas (Table 1) based on their functions (“Benefiting Qi and Detoxication Formula,” “Benefiting Yin and Detoxication Formula,” and “Benefiting Qi and Yin and Detoxication Formula”). All herbs were harvested according to the Good Agricultural Practice (GAP) and sourced from the same production area. Herbal compound formulas for all patients were uniformly prepared and decocted by Longhua Hospital affiliated with Shanghai University of Traditional Chinese Medicine and underwent quality control by complying with the Good Manufacturing Practice (GMP) specific to Chinese herbal medicine. Chinese herbals were administered with 150 mL of decoction daily for drinking, 30 minutes after breakfast and dinner. Patients did not receive any anti-cancer treatment but only were observed in the observation group.
The Ingredients in 3 Chinese Herbal Medicine Formulas.

Herbal compound formulas for all patients were uniformly prepared and decocted by Longhua Hospital affiliated with Shanghai University of Traditional Chinese Medicine and underwent quality control.
All patients in both groups received regular scans for disease recurrence and metastasis. Chest CT was performed every 3 months, upper abdomen and brain CT or MRI every 6 months, and bone emission CT every 12 months. Scans were also conducted if clinically indicated. Participants were followed up every 3 months until the cutoff date of analysis or death.
Endpoints and Assessment
The primary endpoint was 1-year DFS, which was defined as the proportion of patients without recurrence or death within 1 year after randomization. Progressive disease was assessed per RECIST 1.1.
Secondary endpoints were DFS, quality of life, regulatory T cells (Tregs), and immunosuppressive phenotype cytotoxic T lymphocyte-associated antigen-4 (CTLA-4) on the surface of Tregs in peripheral blood. DFS was defined as the time from the date of randomization to death from any cause or first disease recurrence. Quality of life was assessed using the EORTC QLQ-LC43, which included EORTC QLQ-C30 and QLQ-LC13. All patients self-administered the questionnaire at and 3 months after randomization. Flow cytometry assay was performed on the proportion of Tregs and CTLA-4+Tregs in peripheral blood at and 3 months after randomization. Lysing Buffer (Cat#555899), Stain Buffer (Cat#554656), APC/CY7 anti-human CD3 (Cat#300318), FITC anti-human CD4 (Cat#300506), PE anti-human CD25 (Cat#302606), APC anti-human CD127 (Cat#351316), and PE/Cy7 anti-human CD152 (Cat#349914) were purchased from BioLegend. Patients were monitored for adverse events by using the National Cancer Institute’s Common Terminology Criteria for Adverse Events v4.0 every 3 months during the intervention period.
Sample size
Completely resected stage ⅢA NSCLC patients who had finished adjuvant chemotherapy had a 1-year DFS of approximately 65%. 6 Based on the validity of assumptions and clinical experience in the past, we assumed that the 1-year DFS in our study would reach 86%. The sample size calculation was established using PASS software version 15, considering an inspection level of α = .05, grasp 1-beta of .80, and a 10% dropout rate. The sample was estimated to be 75 patients, 38 for the TCM group and 37 for the observation group.
Statistical Analyses
We assessed the efficacy in the full analysis set population. Statistical analyses were completed with SPSS 26. Median DFS and 1-year DFS were estimated by Kaplan-Meier methodology, and treatment comparisons were based on the log-rank test. HR for DFS was calculated by a Cox model, including a 2-sided 95% CI. HR for the effects of prognostic factors was calculated and shown in a forest plot. We used a paired t-test or paired rank sum test for intra-group comparisons and 2 independent samples t-test or rank sum test for inter-group comparisons. The Wilcoxon rank sum test and the Wilcoxon Mann-Whitney test assessed changes in EORTC QLQ-C30 and LC13 scores and the proportions of CTLA-4+Tregs. Changes in the proportions of Tregs were assessed by t-test. Statistical tests were conducted based on a 2-sided α = .05 and 95% CI.
Results
Patients
Between Apr 29, 2019, and Nov 11, 2021, 81 patients were screened. Seventy five patients were randomly assigned, 38 to oral decoctions based on Qi-Yin syndrome differentiation and 37 to observation; 2 patients in the TCM group didn’t receive the study medication, and 1 patient withdrew for personal reasons before completing the study medication and did not undergo any assessment; 1 patient in the observation group didn’t receive assigned observation, and 1 patient withdrew for personal reason before completing the study protocol and did not undergo any assessment; 70 patients were in the full analysis set population (Figure 1). Baseline characteristics were balanced between the 2 groups (Table 2).

Flow diagram of the study.
Baseline Characteristics.

Data are median (Q) or n (%).
DFS and 1-Year DFS
Median follow-up in the full analysis set was 24.2 months at the data cutoff. Seven (20%) of 35 patients in the TCM group and 17 (48.6%) of 35 patients in the observation group had DFS events; median DFS wasn’t reached in the TCM group and 25.5 months (95% CI: 10.9-40.1) in the observation group; HR for DFS was 0.378 (95% CI: 0.157-0.912, P = .03; Figure 2). One-year DFS was 82.1% in the TCM group and 61.9% in the observation group (P = .06). A DFS benefit of oral decoctions based on Qi-Yin syndrome differentiation over observation was evident in most subgroups (Figure 3), although these exploratory analyses should be interpreted with caution.

Disease-free survival.

Forest plot of the treatment effect on DFS in subgroup analyses in the full analysis set.
Quality of Life
Seventy patients had completed the EORTC QLQ-C30 and LC13 questionnaire at and 3 months after randomization. Three months after randomization, scores of total health status, social function, cognitive function, emotional function, role function, and physical function in the TCM group were higher than those at the time of randomization (P < .05 for all); scores of fatigue, pain, shortness of breath, insomnia, loss of appetite, constipation, and diarrhea were lower than those at the time of randomization (P < .05 for all); scores of shortness of breath, cough, chest pain, and arm and shoulder pain in the lung cancer subscale were lower than those at the time of randomization (P < .05 for all); the score of role function in the observation group was higher than that at the time of randomization (P < .05); scores of appetite loss and diarrhea were lower than those at the time of randomization (P < .05 for both); scores of cough and arm and shoulder pain in lung cancer subscales were lower than those at the time of randomization (P < .05 for both) (Table 3).
Standardized Scores of QOL and Self-Comparison of the 2 Groups.

Data are median (Q).
Abbreviations: AP, appetite loss; CF, cognitive functioning; CO, constipation; DI, diarrhea; DY, dyspnoea; EF, emotional functioning; FA, fatigue; FI, financial difficulties; HA, haemoptysis; LC-CO, coughing; LC-DS, dysphagia; LC-DY, dyspnea; LC-HR, alopecia; LC-PA, pain in arm/shoulder; LC-PC, pain in chest; LC-PN, peripheral neuropathy; LC-PO, pain in other parts; LC-SM, sore mouth; NV, nausea and vomiting; PA, pain; PF, physical functioning; QL, general health status; RF, role functioning; SF, social functioning; SL, insomnia.
All domains in EORTC QLQ-C30 and LC13 were balanced between the 2 groups at randomization. Three months after randomization, scores of total health, role function, emotional function, and social function in the TCM group were higher than those in the observation group (P < .01 for all); scores of fatigue, pain, insomnia, loss of appetite, and constipation in the TCM group were lower than those in the observation group (P < .05 for all); scores of cough and chest pain in the lung cancer subscale in the TCM group were lower than those in the observation group (P < .05 for both) (Table 4).
Comparison of quality of life between the 2 groups.

Tregs and CTLA-4+Tregs
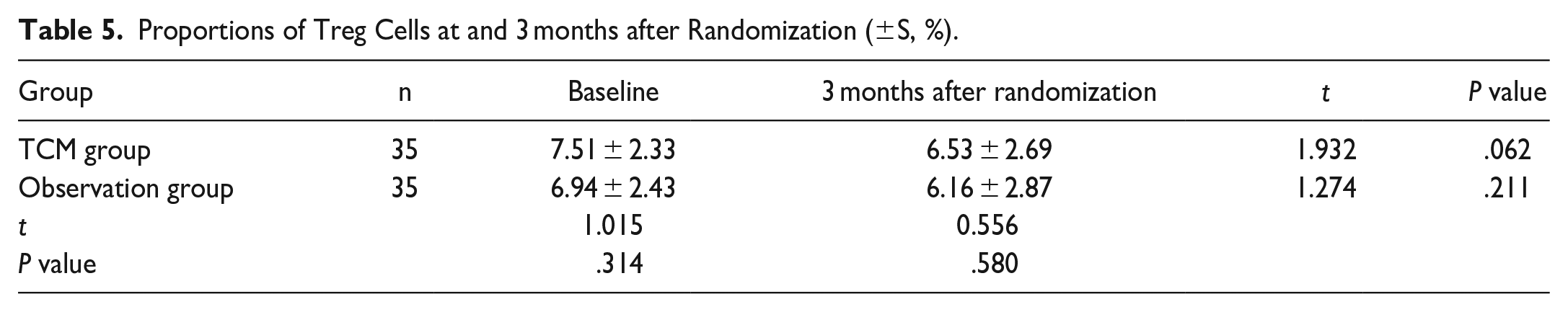
Three months after randomization, the proportions of Tregs and CTLA-4+Tregs in the TCM group were both lower than those at the time of randomization, and the difference was not statistically significant (P = .062, P = .544); the proportion of Tregs in the observation group was lower than that at the time of randomization, and the difference was not statistically significant (P = .211); the proportion of CTLA-4+Tregs in the observation group was higher than that at the time of randomization, and the difference was statistically significant (P = .041).
The proportions of Tregs and CTLA-4+Tregs were both balanced between the 2 groups at randomization (P = .314, P = .581). Three months after randomization, there was no significant difference in the proportion of Tregs between the TCM group and the observation group (P = .580); the proportion of CTLA-4+Tregs in the TCM group was lower than that in the observation group, and the difference was statistically significant (P = .046) (Table 5, 6 and Figure 4).
Proportions of Treg Cells at and 3 months after Randomization (±S, %).
Proportions of CTLA-4+Tregs at and 3 months after Randomization (M[Q], %).


The proportions of Tregs and CTLA-4+Tregs (Flow cytometry assay). (A) TCM group (B) Observation group.
Adverse Events
We conducted routine blood tests, and liver and kidney function tests on patients every 3 months during the intervention period. No adverse events occurred in both groups.
Discussion
Adjuvant chemotherapy yields only about 5% improvement in 5-year survival for postoperative NSCLC patients.3,4 With such poor gain in survival, powerful adjuvant treatment regimens remain to be studied. Adjuvant osimertinib therapy for 3 years, with or without previous adjuvant chemotherapy, reduced the risk of death by 51% versus placebo in resected stage Ⅱ-ⅢA EGFR mutation-positive NSCLC patients. 10 Adjuvant erlotinib for 2 years improved 2-year DFS compared with adjuvant chemotherapy in resected stage ⅢA EGFR mutation-positive NSCLC patients, and reduced the risk of recurrence or death by 73.2%. 6 Impower010 proved a DFS benefit with adjuvant atezolizumab versus BSC after adjuvant chemotherapy in resected stages Ⅱ-ⅢA NSCLC patients and reduced the risk of recurrence or death by 21% in all patients and 34% in patients whose tumors expressed PD-L1 on 1% or more of tumor cells. 11 Adjuvant EGFR-TKIs and ICIs are becoming powerful options for patients with stage ⅢA NSCLC in the adjuvant setting.
TCM treatment is becoming more and more accepted for its effects in preventing and treating malignant tumors. 20 TCM treatment after adjuvant chemotherapy for resected stage ⅢA NSCLC is widely used in China. Still, the optimal administration timing and duration of TCM treatment in these patients remain at the empirical level. Professor Liu Jiaxiang analyzed the syndromes of 310 patients with lung cancer, 21 248 patients exhibited Yin deficiency or Qi and Yin deficiency, which accounted for 80%. Therefore, we conducted this randomized controlled study to compare the efficacy of oral decoctions based on Qi-Yin syndrome differentiation versus observation after adjuvant chemotherapy in patients with resected stage ⅢA NSCLC. By administering oral decoctions based on Qi-Yin syndrome differentiation after adjuvant chemotherapy, the intervention in the adjuvant setting was strengthened, to prevent recurrence and metastasis more effectively.
A previous cohort study included 140 postoperative patients with stage ⅢA NSCLC. Chinese medicine combined with chemotherapy could prolong DFS by 7.94 months compared with adjuvant chemotherapy alone. 22 A retrospective study showed that TCM therapy was an independent protective factor for DFS of resected NSCLC patients: long-term TCM treatment could prevent the recurrence and metastasis in resected NSCLC. 23 In this study, oral decoctions based on Qi-Yin syndrome differentiation after adjuvant chemotherapy prolonged DFS compared with observation in resected stage ⅢA NSCLC patients, and reduced the risk of recurrence and metastasis by 62.2%. The increased median DFS was observed in most subgroup analyses, indicating the stability of the study findings. This study was consistent with the Impower010 trial (adjuvant ICIs) for the intervention phase (after adjuvant chemotherapy) and the duration of the intervention (12 months), and both trials yielded consistent encouraging results. Modern pharmacological research confirmed that core herbals of strengthening body resistance in these 3 formulas such as Radix Glehniae, Radix Ophiopogonis, Radix Astragali, Rhizoma Atractylodis Macrocephalae, Fructus Ligustri lucidi, and Gynostemma pentaphyllum Makino not only served as conventional tonics but also as tumor cell inhibitors. 24
Clinically, a DFS benefit may not be associated with improvement in quality of life. We used the EORTC QLQ-C30 and LC13, which are widely used and internationally recognized quality of life questionnaires for lung cancer patients and also suitable for Chinese patients, to evaluate the quality of life. 25 We demonstrated that oral decoctions based on Qi-Yin syndrome differentiation after adjuvant chemotherapy improved quality of life compared with observation in these patients. These 3 compound formulas enabled patients to have better living conditions.
CTLA-4 is an important inhibitory molecule that mediates tumor immune escape. Yiqi Yangyin Jiedu Formula down-regulated CTLA-4 expression when tumor cells and lymphocytes were co-cultured in vitro. 26 A randomized controlled trial showed that regulating the serum concentration of soluble CTLA-4 (sCTLA-4), which shares the same biological effects with CTLA-4, might be 1 of the mechanisms of TCM maintenance treatment of NSCLC. 27 In this study, we also demonstrated that oral decoctions based on Qi-Yin syndrome differentiation after adjuvant chemotherapy down-regulated the immunosuppressive phenotype CTLA-4 on the surface of Tregs. These 3 herbal formulas improve immune function by preventing peripheral immune escape.
The trial was carried out per the principles of the Declaration of Helsinki and guidelines for Good Clinical Practice. However, it also had some limitations. Firstly, the study was lack of placebo control, and participants and investigators were not blinded to the interventions. To control the bias, outcome assessors and statisticians were blinded to the allocated treatments. Secondly, we observed quality of life and blood indicators only at the third month rather than regularly observation within 1 year, partly out of consideration for patients mostly recovered from surgery and adjuvant chemotherapy after 6 months. Further limitations of the study were the small size and with limited ethnic/racial diversity. Moreover, mature data for DFS and OS in this study are needed.
Conclusion
Oral decoctions based on Qi-Yin syndrome differentiation after adjuvant chemotherapy, which prolonged DFS, reduced the risk of disease recurrence and metastasis, improved quality of life, and down-regulated the proportion of CTLA-4+Tregs, is a powerful strategy for completely resected stage ⅢA NSCLC patients.
Footnotes
Acknowledgements
We thank the clinical staff from Shanghai Chest Hospital and Shanghai Pulmonary Hospital for providing eligible patients.
Author contributions
Yi Jiang and Ling-shuang Liu designed the study. All authors contributed to the acquisition and interpretation of the data and preparation of the manuscript. All authors approved the final manuscript.
Availability of data and materials
The data and materials are available from the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the National Natural Science Foundation of China (No. 82374533), Shanghai Municipal Health Commission (No. 201840158, 202140370), Natural Science Foundation of Shanghai (No. 21ZR1463700), and Science and Technology Commission of Shanghai Municipality (No. 21MC1930500).
Research ethics and patient consent
The study and consent process were both approved by the Ethics Committee of Longhua Hospital affiliated with Shanghai University of Traditional Chinese Medicine, Shanghai, PRC (No. 2018LCSY057).
