Abstract
Background:
The prescription of Chinese herbal medicine (CHM) consists of multiple herbs that exhibit synergistic effects due to the presence of multiple components targeting various pathways. In clinical practice, the combination of Erchen decoction and Huiyanzhuyu decoction (EHD) has shown promising outcomes in treating patients with laryngeal squamous cell carcinoma (LSCC). However, the underlying mechanism by which EHD exerts its therapeutic effects in LSCC remains unknown.
Methods:
Online databases were utilized for the analysis and prediction of the active constituents, targets, and key pathways associated with EHD in the treatment of LSCC. The protein-protein interaction (PPI) network of common targets was constructed and visualized using Cytoscape 3.8.1 software. Gene ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) analyses were performed to investigate the functional roles of core targets within the PPI network. Protein clustering was conducted utilizing the MCODE plug-in. The obtained results highlight the principal targets and pathways involved. Subsequently, clinical samples were collected to validate alterations in the levels of these main targets through Western blotting (WB) and immunohistochemistry (IHC). Furthermore, both in vivo and in vitro experiments were conducted to investigate the therapeutic effects of EHD on healing LSCC and elucidate its underlying mechanism. Additionally, to ensure experimental reliability and reproducibility, quality control measures utilizing HPLC were implemented for EHD herbal medicine.
Results:
The retrieval and analysis of databases in EHD medicine and LSCC disease yielded a total of 116 overlapping targets. The MCODE plug-in methods were utilized to acquire 8 distinct protein clusters through protein clustering. The findings indicated that both the first and second clusters exhibited a size greater than 6 scores, with key genes PI3K and ErbB occupying central positions, while the third and fourth clusters were associated with proteins in the PI3K, STAT3, and Foxo pathways. GO functional analysis reported that these targets had associations mainly with the pathway of p53 mediated DNA damage and negative regulation of cell cycle in terms of biological function; the death-induced signaling complex in terms of cell function; transcription factor binding and protein kinase activity in terms of molecular function. The KEGG enrichment analysis demonstrated that these targets were correlated with several signaling pathways, including PI3K-Akt, FoxO, and ErbB2 signaling pathway. On one hand, we observed higher levels of key genes such as P-STAT3, P-PDK1, P-Akt, PI3K, and ErbB2 in LSCC tumor tissues compared to adjacent tissues. Conversely, FOXO3a expression was lower in LSCC tumor tissues. On the other hand, the key genes mentioned above were also highly expressed in both LSCC xenograft nude mice tumors and LSCC cell lines, while FOXO3a was underexpressed. In LSCC xenograft nude mice models, EHD treatment resulted in downregulation of P-STAT3, P-PDK1, PI3K, P-AKT, and ErbB2 protein levels but upregulated FOXO3a protein level. EHD also affected the levels of P-STAT3, P-PDK1, PI3K, P-AKT, FOXO3a, and ErbB2 proteins in vitro: it inhibited P-STAT3, P-AKT, and ErbB2, while promoting FOXO3a; however, it had no effect on PDK1 protein. In addition, HPLC identified twelve compounds accounting for more than 30% within EHD. The findings from this study can serve as valuable guidance for future experimental investigations.
Conclusion:
The possible mechanism of EHD medicine action on LSCC disease is speculated to be closely associated with the ErbB2/PI3K/AKT/FOXO3a signaling pathway.
Keywords
Introduction
Laryngeal squamous cell carcinoma (LSCC) is a prevalent form of malignant cancer in the head and neck region, characterized by a poor prognosis. 1 Despite significant advancements in therapy, the incidence and mortality rate of LSCC remain alarmingly high, while the underlying pathogenic mechanism remains elusive.2,3
Erchen decoction plus Huiyanzhuyu decoction (EHD) is commonly employed in the treatment of patients with LSCC presenting phlegm syndrome and blood stasis syndrome, exhibiting promising clinical outcomes. Preliminary cellular experiments have demonstrated that EHD effectively suppresses the proliferation, invasion, and cell cycle progression while promoting apoptosis in LSCC cells. 4 Moreover, EHD can inhibit the STAT3/PD-L1 signaling pathway in LSCC and impede tumor growth in vivo experiments. 5 Comprising two classical prescriptions, namely Erchen decoction and Huiyanzhuyu decoction, EHD primarily functions to invigorate spleen function and resolve phlegm for treating damp phlegm syndrome. Additionally, Huiyanzhuyu decoction has exhibited remarkable benefits for patients with head and neck diseases associated with blood stasis syndrome. 6
The main clinical syndromes of LSCC are characterized by phlegm coagulation and blood stasis. The effectiveness of the herbal formula EHD in resolving phlegm and removing blood stasis highlights the value of Chinese medicine in syndrome differentiation and treatment. EHD is composed primarily of fifteen medicinal materials, including Citri Exocarpium Rubrum, Pinelliae Rhizoma, Poria, Glycyrrhizae Radix et Rhizoma, Zingiberis Rhizoma Recens, Mume Fructus, Persicae Semen, Carthami Flos, Platycodonis Radix, Rehmanniae Radix, Angelicae Sinensis Radix, Scrophulariae Radix Bupleuri Radix Aurantii Fructus,and Paeoniae Radix Rubra. Numerous studies have demonstrated the significant clinical effects of EHD. Our previous research indicated that modified EHD could alleviate symptoms and improve recovery, while reducing recurrence of precancerous lesions in LSCC diseases.7,8 Furthermore, basic experimental research has shown that EHD regulates the CyclinD1/STAT3 signaling pathway to promote apoptosis in LSCC cells and inhibits PD-L1 protein levels to influence the tumor immune microenvironment.9,10
The advantage of Chinese herbal medicine (CHM) in tumour treatment lies in its multicomponent and multitarget characteristics. 4 With the rapid progress of bioinformatics, the CHM network pharmacology approach has been established, shifting the focus from a “one target, one drug” paradigm to a new perspective of “multiple targets and multiple components,” which is considered promising for more cost-effective drug development.
Network pharmacology can be utilized to establish a network of medicinal products with specific mechanisms of action and predict targets based on public databases and existing published data. This approach can also be employed to analyze and construct the networks of overlapping targets between medicine and disease through bioinformatics analysis, as well as to investigate the pharmacological mechanisms of medicines within these networks. Therefore, in this study, we simulated the network relationships between the 15 components of EHD and the targets associated with EHD using network pharmacology in order to gain a better understanding of the synergistic effects and mechanisms underlying EHD. Additionally, we conducted clinical sample detection, cell experiments, and animal studies to validate the mechanism by which EHD mediates its therapeutic effect on LSCC.
Materials and Methods
Reagents
All herbs were purchased from KANGMEI Pharmaceutical Co., Ltd. (Guangzhou, China). RPMI-1640 medium and fetal bovine serum (FBS) were obtained from HyClone Laboratories Inc. (Logan, USA). Antibodies against P-PDKI, PI3K, P-AKT, P-STAT3, Foxo3a, and ErbB2 were purchased from Cell Signaling Technology Inc. (Danvers, MA, USA). The TU212 cell line was obtained from the Advanced Research Center of Central South University (Changsha, China). PBS, formaldehyde, paraffin, haematoxylin and DAB reagent kits were purchased from Weijia Technology Co., Ltd (Guangzhou, China). RIPA assay buffer was purchased from Cell Signaling Technology Inc. (Danvers, MA, USA). The bicinchoninic acid (BCA) protein assay kit was purchased from (Abcam, USA).
Preparation of EHD Lyophilized Powder
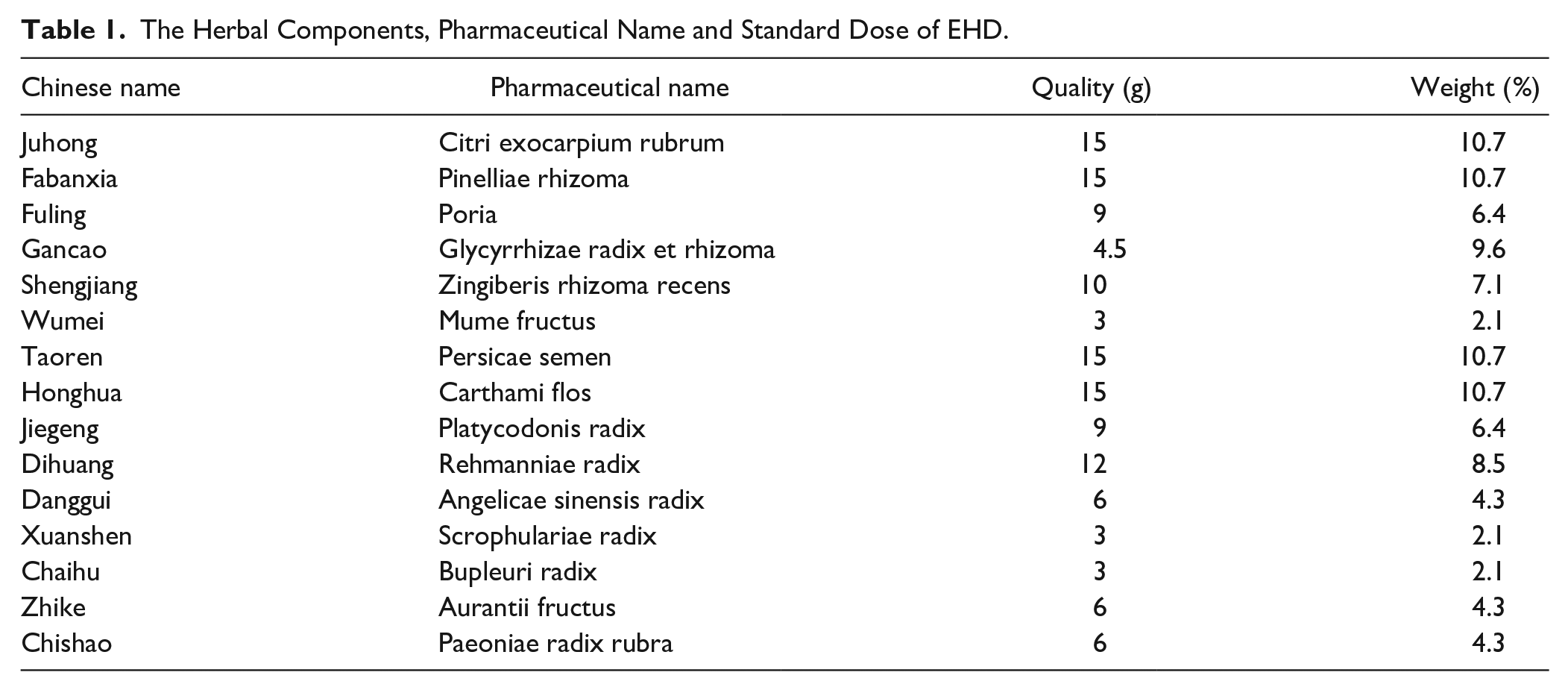
EHD is composed of 15 Chinese herbal medicines, the herbal components, pharmaceutical name and weight of one dose of EHD were consistent with those as shown in Table 1. The herbal components and standard dose of EHD have been published in a previous original publication. 10
The Herbal Components, Pharmaceutical Name and Standard Dose of EHD.
We mixed the medicines together according to the components and standard dose of EHD, and crushed the drugs with a swing high-speed universal machine. Five times deionized water of the total amount herbal medicines was added to the bottle. EHD was soaked in deionized water for 30 minutes before boiling twice at 100°C in deionized water in 10-fold volumes for 2 hours. The filtrates were concentrated with a rotary evaporator and dried by lyophilization to obtain dried powder. The extract was stored at 4°C. It was decocted with a water frying sleeve thermostat for 120 minutes, the drug residue was filtered with a 400 mesh filter screen, centrifuged at 3500 r/min (r = 40 cm) for 5 minutes. The medicine supernatant was obtained to concentrate the solution with a rotary evaporator. The final concentration is 1 g/mL. The concentrate is frozen at −80°C for 48 hours and then frozen with a vacuum freeze-drying machine for 72 hours.
Identification of EHD Active Ingredients and LSCC Targets
To determine the chemical compounds of the 15 plants, we performed a search of the Chinese Herbal Medicine Systems Pharmacology Database and Analysis Platform (TCMSP, http://tcmspw.com/index.php, updated on May 31, 2023). We used oral bioavailability (OB) ≥ 30% and drug-like (DL) ≥ 0.18 as criteria for the compound screening to identify the biologically active ingredients of EHD and their corresponding targets. PreOB predicts oral bioavailability. PreDL predicts drug likeness, and OB and DL are absorption, distribution, metabolism, and excretion (ADME)-related factors. LSCC-related targets were mined from DisGeNET (https://www.disgenet.org/). We set DPI (Disease Pleiotropy Index) > 0.615, and DSI (Disease Specificity Index) > 0.585 as the thresholds for the identification of candidate targets, and data from the GeneCards (Relevance score) ≥ 10, OMIM and NCBI databases were also retrieved simultaneously. The target genes of LSCC were obtained after removing duplicate genes. A protein-protein interaction (PPI) network was constructed with the online STRING database (http://string-db.org/) using “Homo sapiens” with score >0.99. The results were visualized by CytoScape 3.8.1. The clusterProfiler R package was utilized to perform GO functional enrichment and KEGG pathway analysis of common targets. Additionally, GO analysis encompassed 3 aspects: biological process (BP), molecular function (MF), and cellular component (CC). The obtained results were filtered with a significance threshold of P ≤ .01 and visualized using the “ggplot2” R package. Furthermore, the top 50 KEGG pathways were categorized and summarized based on the first 6 classifications in the KEGG PATHWAY Database (https://www.kegg.jp/kegg/pathway.html) with a significance threshold of P-value less than 1.0E-14. The proteins with overlapping expression patterns were subjected to bioinformatic annotation with the online BioinfoGF database (https://bioinfogp.cnb.csic.es/tools/venny/index.html). We took the intersection of the total disease targets and drug targets. After topological analysis of the protein interaction network nodes, including screening of key nodes to remove duplicates. The findings were visualized using CytoScape (version 3.8.1), and hub genes were further extracted using Molecular Complex Detection (MCODE, version 1.5.1) with the following parameters: MCODE scores > 3; node score cutoff = 0.2; k-core = 2; degree cutoff = 4; and max. depth = 100. To visually represent the relationship in the medicine-targets-signaling pathway, we constructed an interactive network of medicine-target genes-signaling pathway using CytoScape.
Clinical Specimen Collection and Testing
The clinical study of LSCC patients included is approved by the Ethics Committee of Guangdong Provincial Hospital of Chinese Medicine (grant no. BF2018-030-01; approval date: September 2018). Informed consent was provided by the patients clinically diagnosed with LSCC with phlegm coagulation and blood stasis syndrome. The tumor specimens and adjacent safe margins were procured from these patients. A total of 20 samples were obtained, representing 10 patients. The samples from 4 patients were stored at –80°C in a refrigerator specifically designed for Western blot (WB) experiments, while the immunohistochemistry (IHC) experiments utilized samples from 6 patients that had been preserved in formalin.
Animal Experiment Design
The animal experiment was carried out with the approval of the Experimental Animal Ethics Committee of Guangdong Provincial Hospital of Chinese Medicine (grant no. 2018013; approval date: July 2018). BALB/c nude mice (16.5 ± 1.0 g, females) were provided by Beijing Weitong Lihua Experimental Animal Technology Co., Ltd. (certificate no. SCXK; Beijing. China). The animals were reared in a specific pathogen-free (SPF) environment, housed individually in separate cages, and then randomly allocated into experimental groups: a model group (n = 5) and drug intervention groups (low, medium, and high dose groups; each consisting of 5 mice). Mice were subcutaneously injected in the right forelimb, below the armpit (2.0 × 106 LSCC cells/animal). When the tumors reached an average size of 10 mm 3 , the mice were administered varying concentrations of EHD daily via intragastric administration. The medium EHD dose administered to the mice was determined based on the daily equivalent dose administered to humans. In comparison to the medium-dose group, the high-dose group received a dosage twice as strong, while the low-dose group received half of the dosage. The recommended daily dosage of raw herbal material for EHD is typically 140.5 g. When this human dose was converted into an animal dose (based on a person weighing 60 kg and a conversion factor between humans and mice of 12.33), it was equivalent to the middle dose (28.87 g/kg, EHD-M) used in this study. Therefore, EHD was administered every day at doses of 14.44 g/kg (EHD-L), 28.87 and 57.74 g/kg (EHD-H) for different groups of mice. The intervention period was 4 weeks. The animal experiments ended with tumour diameter greater more than 2 cm, mouse death or 28 days after administration.
Cells Experiment Design
TU212 LSCC cells were divided into 2 groups: the control group and the EHD group. The cells were incubated in 6-well plates at a density of 2 × 105 cells in each well, and the adherent cells were treated with EHD (1.6 mg/mL lyophilized powder) for 48 hours. Then, the cells were washed twice with phosphate-buffered saline (PBS) and subsequently lysed using RIPA cell lysis buffer supplemented with phosphatase inhibitors and protease inhibitors. The protein concentration was determined using a BSA kit.
Construction of EHD Prints by HPLC
The EHD lyophilized powder was diluted to a concentration of 100 μg/mL and subsequently filtered through a 0.4 μm filter for analysis using an Agilent 1260 LC HPLC system equipped with an Agilent 5 TC-C18 column (260 × 4.6 mm, CA, USA). The mobile phase consisted of acetonitrile (B) and water (A) containing 0.05% phosphoric acid, while the column temperature was maintained at 30°C and the flow rate set at 1 mL/min with an injection volume of 10 μL. A gradient elution condition was applied as follows: 0-2 minutes, 5% A, 95% B; 2-4 minutes, 5%-8% A, 95%-92%B; 4-12 minutes, 8%-12% A, 92%-88% B; 12-14 minutes,12%-20% A, 88%-80% B; 14-18 minutes, 20%-25% A, 80%-75% B; 18-25 minutes, 25%-40% A, 75%-60% B; 25-30 minutes, 40%-40% A, 60%-60% B; 30-31 minutes, 40%-95% A, 60%-5% B; 31-35 minutes, 95%-5% A, 5%-95% B; 35-40 minutes, 5%-5% A, 95%-95%. The detection wavelengths were selected as 210, 230, 254, 270, and 280 nm.
Western Blot
The tumor tissues or cells from different groups were minced and lysed with RIPA assay buffer on ice for 30 minutes. The protein concentration was determined using a BCA protein assay kit. Equal amounts of protein (40 µg) per lysate were resolved on a Tris-glycine gel, transferred onto a nitrocellulose membrane, and blocked for 1 hour with 5% TBS containing 1% Tween-20. Membranes were incubated overnight at 4°C with the desired primary antibodies (anti-P-STAT3, anti-P-PDK1, anti-PI3K, anti-P-AKT, anti-Foxo3a, anti-ErbB2, or anti-GAPDH; diluted to 1:1000), followed by incubation with the appropriate secondary antibody (diluted to 1:3000) for 1 hour. Densitometric analysis was performed using a chemiluminescence imaging system. GAPDH was used as an internal control for each sample.
IHC Staining
Tumor specimens from patients with LSCC and mice were harvested and fixed in 4% paraformaldehyde for 20 hours. The embedded samples were cut into 5 μm sections, dewaxed, antigen retrieved, sealed, and incubated overnight at 4°C with antibodies against P-STAT3 (dilution, 1:800), P-PDK1 (dilution, 1:200), P-AKT (dilution, 1:200), PI3K (dilution, 1:500), Foxo3a (dilution, 1:300), and ErbB2 (dilution, 1:100). After washing the slides 3 times with TBS-Tween buffer solution to remove unbound primary antibody followed by secondary antibody incubation step using HRP-conjugated anti-rabbit IgG or anti-mouse IgG as appropriate. DAB reagent kit was used for staining followed by counterstaining with hematoxylin. Images were captured using a bright-field microscope.
Results
Network Analysis and Functional Enrichment Analysis of the EHD and LSCC Overlapping Targets
In total, 472 targets from the 15 herbs in EHD and 700 LSCC targets were obtained though topological analysis of the protein interaction network nodes. Duplicate targets were removed as shown in Figure 1A and B. The disease targets and drug targets were intersected by the online STRING database, with score > 0.99 as the criterion. Finally, using an online tool, a Venn diagram illustrating the overlap between targets of all medicines and disease-related targets was generated, a total of 116 key nodes were obtained as shown in Figure 1C. The 116 targets’ protein (Supplemental Table 1) clustering was conducted utilizing the MCODE plug-in, MCODE was used for a topological analysis of the PPI network, and 8 different protein clusters were acquired, as shown in Figure 2. Arranged in descending order based on the MCODE score, the first and second clusters exhibited a size greater than 6 scores, with key genes PI3K and ErbB occupying central positions (Figure 2A and B), while the third and fourth clusters were associated with proteins in the PI3K, STAT3, and Foxo pathways (Figure 2C and D).

Network analysis of targets. (A) PPI of EHD key targets, (B) PPI of key targets in LSCC, and (C) Venn diagram of medicine targets and disease targets.

The 116 targets’ protein clustering. Screened hub targets in Cytoscape with MCODE. (A and B) MCODE score ≥ 6; (C-E) MCODE score = 4. (F) MCODE score = 3.33. (G and H) MCODE score = 3.
GO annotation conducted through the DAVID website revealed that the overlapping targets between medicine and disease were predominantly associated with pathway of p53 mediated DNA damage and negative regulation of cell cycle in terms of biological function; the death-induced signalling complex in terms of cell function; transcription factor binding and protein kinase activity in terms of molecular function, as shown in Figure 3A. The KEGG enrichment analysis revealed a strong association between many target genes and the PI3K-Akt pathway, Foxo, and ErbB signalling pathway, as shown in Figure 3B. Cytoscape 3.8.1 was applied to build an interactive network of EHD medicines-target genes-signaling pathways shown in Figure 4. The map illustrates the relationship among 3 components: EHD herb medicines, overlapping targets, and signaling.
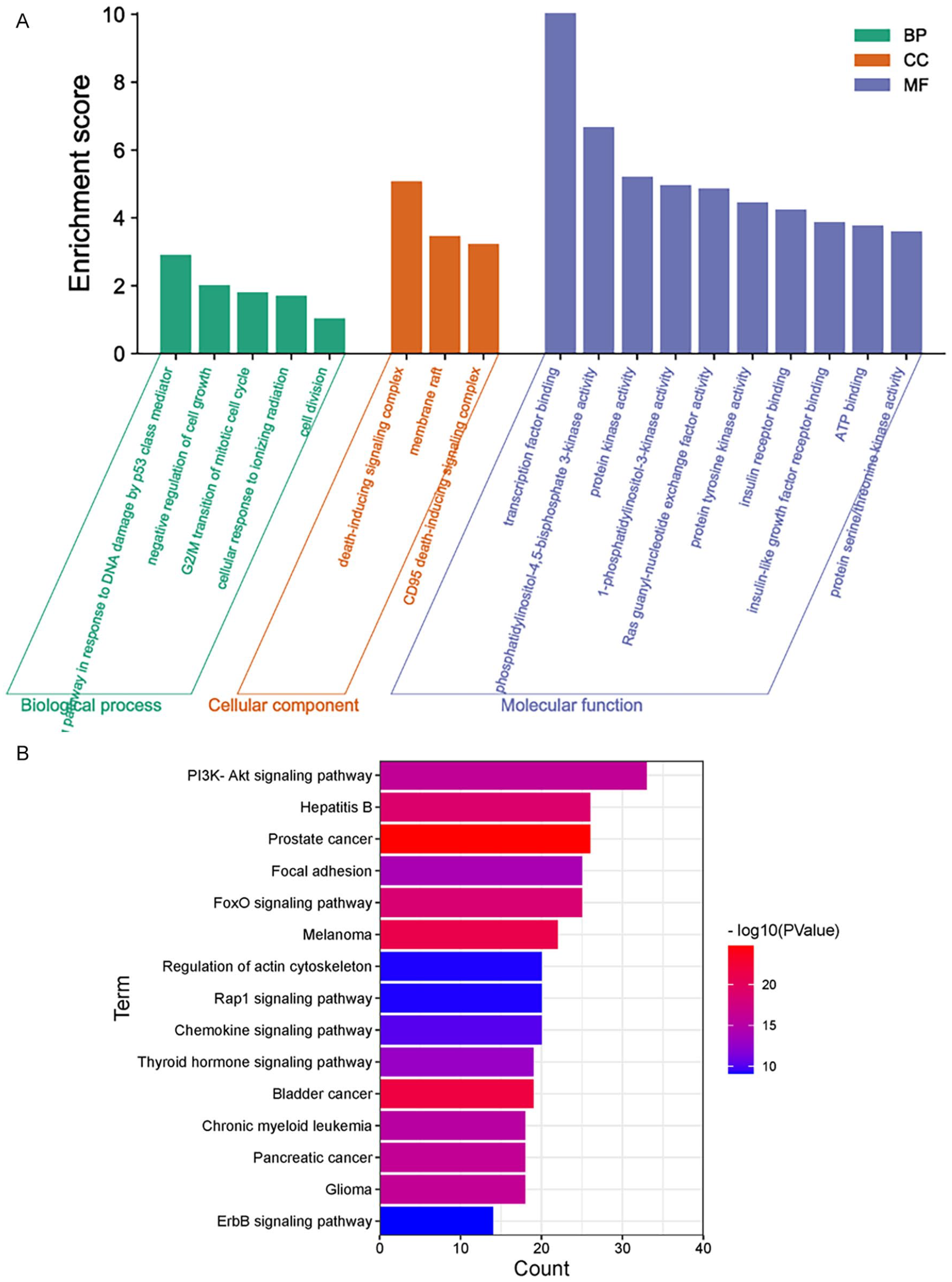
GO enrichment analysis and KEGG signaling pathways analysis of 116 common targets. (A) Cellar component analysis, biological process analysis, molecular functions analysis, (B) KEGG signaling pathways analysis. EHD medicines-common targets- signaling pathways interaction diagram.
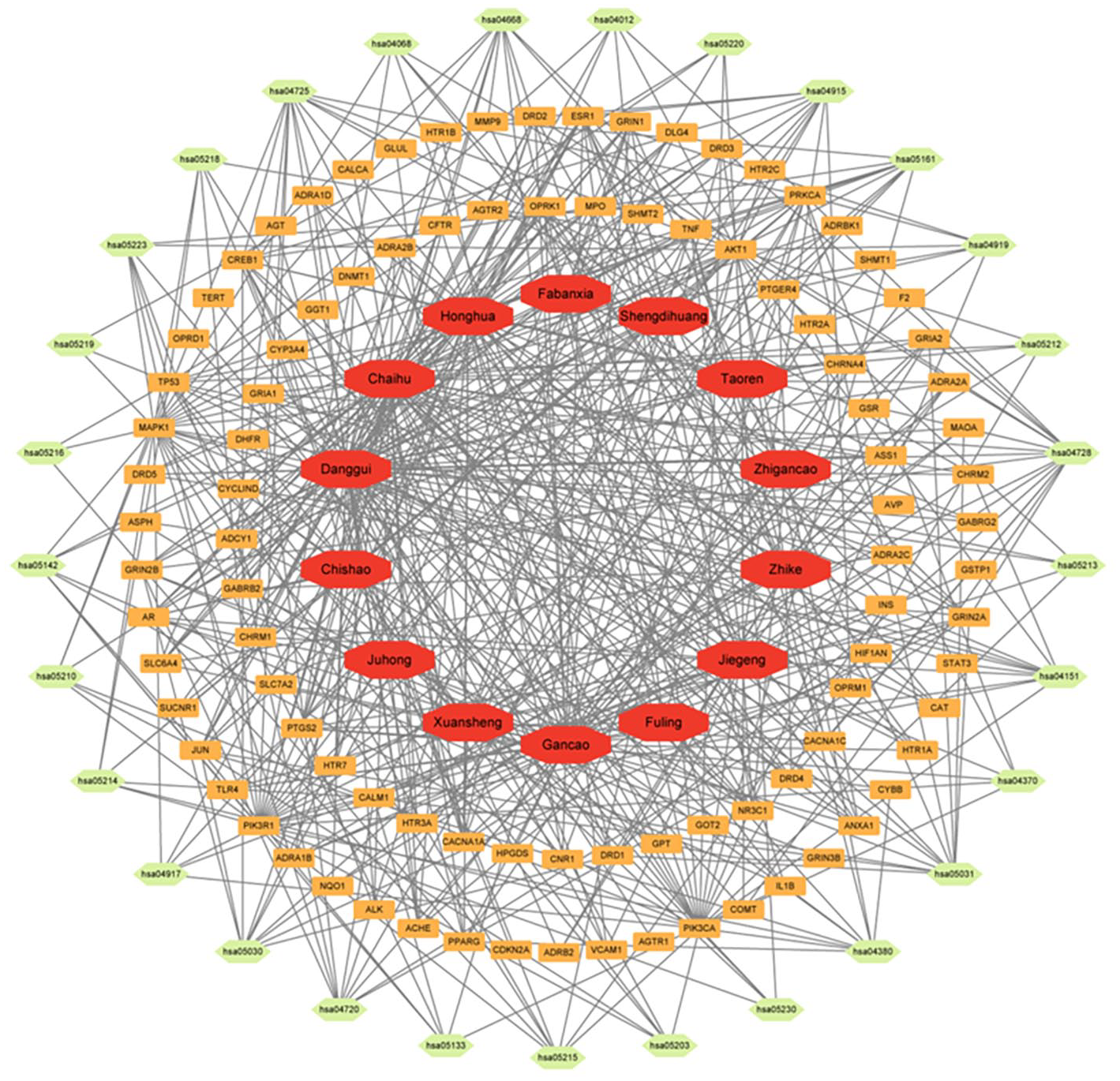
Network of EHD medicines- common targets-signaling pathways. Red, yellow, green oval represent EHD medicines, common targets, and signaling pathways, respectively, and black lines indicate mutual relationships.
The Protein Expression Levels in Tumor Specimens and Their Corresponding Normal Tumor Margin Tissues from Patients with LSCC
The KEGG enrichment analysis showed that PI3K-Akt, Foxo, ErbB signaling pathways were closely related to the occurrence of LSCC disease. The surgical specimens and corresponding normal tumor margin tissues from ten patients were collected. We conducted WB and IHC analysis to detect the levels of P-STAT3, P-PDK1, PI3K, P-AKT, Foxo3a, and ErbB2. The results of WB revealed differential expression levels of these genes between the 2 groups. Specifically, P-STAT3, P-PDK1, PI3K, P-AKT, and ErbB2 exhibited increased expression in the tumor sample compared to the normal tumor margin samples. Conversely, Foxo3a demonstrated decreased expression in the tumor tissue compared to the normal tumor margin tissue as shown in Figure 5A. The statistical significance of the difference between the 2 groups is illustrated in Figure 5B. The IHC experiment revealed the expression of P-STAT3, P-PDK1, PI3K, P-AKT, Foxo3a, and ErbB2 proteins in both groups. The results were consistent with those obtained from the WB experiments (Figure 5C).

Protein expression levels in tumor specimens and corresponding normal tumor margin tissues from LSCC patients. (A and B) Western blot analysis: P-STAT3, P-PDK1, P-AKT, PI3K, FOXO3a, and ErbB2 proteins expression level in clinical laryngeal cancer samples (Tu, Tumor) and safe margins (Adj, Adjacent), respectively. (P < .05 or P < .001 comparison of two groups). (C) Proteins in the tumor tissues samples from the different groups detected by immunohistochemistry.
The Administration of EHD Induces Alterations in the Protein Levels of P-STAT3, P-PDK1, PI3K, P-AKT, Foxo3a, and ErbB2 in LSCC Xenograft Nude Mice
To validate the mechanism by which EHD affects LSCC were identified by the KEGG enrichment analysis. The xenograft tumors of nude mice models were established. The model animals were randomly divided into 4 groups (0 g/kg EHD, 14.44 g/kg EHD, 28.87 g/kg EHD, 57.74 g/kg EHD, respectively). The previous study has demonstrated that EHD can effectively inhibit the growth of ectopic tumors and induce apoptosis in LSCC, as evidenced by in vivo experiments. 5 In this study we conducted a WB and IHC detection of P-STAT3, P-PDK1, PI3K, P-AKT, Foxo3a, and ErbB2 in the xenograft tumors of nude mice in different groups. The WB results showed that EHD downregulated the level of P-STAT3, P-PDK1, PI3K, P-AKT, and ErbB2 protein, while upregulating Foxo3a protein expression as demonstrated in Figure 6A. The statistical significance of the difference is illustrated in Figure 6B. The IHC experiment revealed the expression of P-STAT3, P-PDK1, PI3K, P-AKT, Foxo3a, and ErbB2 proteins in both groups. The results were consistent with those obtained from the WB experiment (Figure 6C). These findings suggest that EHD can modulate the PI3K-AKT signaling pathway as well as Foxo3a and ErbB2 pathways which is consistent with our functional enrichment analysis.

The expression of P-STAT3, P-PDK1, P-AKT, PI3K, FOXO3a, and ErbB2 proteins in LSCC xenografts nude mice was suppressed by EHD. (A and B) Western blot analysis: P-STAT3, P-PDK1, P-AKT, PI3K, FOXO3a, and ErbB2 proteins expression level in different groups. (**P < .05 compared to the control group; ***P < .01 compared to the control group; ****P < .001 compared to the control group). (C) Proteins in the tumor tissues samples from the different groups detected by immunohistochemistry.
The Expression Levels of P-STAT3, P-PDK1, PI3K, P-AKT, FOXO3a, and ErbB2 Proteins in the LSCC Cell Line are Modulated by EHD
To further elucidate the underlying mechanism by which EHD impacts LSCC in vitro, we conducted an analysis of the PI3K-AKT, FOXO3a, and ErbB2 signaling pathways. We have demonstrated that EHD inhibited invasion, migration and cycle of LSCC, and promoted the cell apoptosis in a preliminary experiment. 4 In this study WB analysis was performed to detect the levels of P-STAT3, P-PDK1, PI3K, P-AKT, FOXO3a, and ErbB2 in both the control group and EHD group as shown in Figure 7. The results demonstrated that EHD effectively inhibited P-STAT3, P-AKT, and ErbB2, while promoting FOXO3a as shown in Figure 7A. The statistical significance of the difference between the 2 groups is illustrated in Figure 7B. These findings suggest that EHD exerts significant effects on the PI3K-AKT, FOXO3a, and ErbB2 signaling pathways, thereby further confirming the results obtained from functional enrichment analysis.

(A) The expression of P-STAT3, PDK1, AKT, PI3K, FOXO3a, and ErbB2 proteins in LSCC cells was inhibited by EHD. (b) Western blot analysis: P-STAT3, P-PDK1, P-AKT, PI3K, FOXO3a, and ErbB2 proteins expression level in different groups.
The HPLC Diagrams for EHD Chinese Medicine are Established
To ensure the stability and repeatability of Chinese herb medicine in experimental research, we aimed to determine the EHD HPLC diagrams. A total of twelve components were identified in the fifteen herbs through HPLC analysis. Based on the retention time of the standard sample, 12 active compounds were identified in the whole EHD, namely paeoniflorin (Peak 1), amygdalin (Peak 2), harpagide (Peak 3), glycyrrhizic acid (Peak 4), liquiritin (Peak 5), saikosaponin A from Bupleurum falcatum (Peak 6), saikosaponin D from Bupleurum falcatum (Peak 7), ferulic acid (Peak 8), succinic acid (Peak 9), rehmannioside D (Peak 10), neohesperidin (Peak 11), 6-gingerol (Peak 12), as shown in Figure 8. Twelve components accounted for more than 30% of the whole EHD. The results can provide guidance for subsequent experimental studies.

Chinese herb quality control of EHD. The brown corresponds to wavelength 270 nm. The pink corresponds to wavelength 280 nm. The green corresponds to wavelength 230 nm. The red corresponds to wavelength 210 nm. The blue corresponds to wavelength 254 nm. The numbers refer to the main components identified by the control sample.
Discussion
Phlegm and blood stasis is one of the core pathologies of LSCC. Huatan Quyu decoction, which consists of Erchen decoction and Huiyanzhuyu decoction (EHD), has demonstrated efficacy in treating LSCC with phlegm coagulation and blood stasis syndrome. EHD has shown promising therapeutic effects in clinical practice due to its reliance on a holistic view, syndrome differentiation, and treatment administration theory. However, further exploration is needed to elucidate its mechanism of action. CHM possesses unique advantages in managing complex diseases. Moreover, Traditional Chinese medicine exhibits synergistic effects through multiple components targeting multiple pathways, thus benefiting from network pharmacology analysis.
Network pharmacology involves the analysis of biological systems through online databases based on systems biology theories. The systematic and holistic characteristics of this approach align with the principles of Chinese herbal medicine, including syndrome differentiation and treatment administration. In CHM research, network pharmacology analysis has been widely utilized. 11 The present study utilized online database analysis to identify corresponding overlapping targets for 15 Chinese herbs in EHD and LSCC disease, resulting in the construction of an interaction network between EHD medicines, overlapping targets, and signaling pathways using Cytoscape software. The changes in the levels of the main targets were verified using clinical samples, and both in vivo and in vitro experiments were conducted to investigate the effects of EHD on healing LSCC and elucidate its potential mechanism.
This approach can also be utilized for the analysis and construction of networks involving overlapping targets between medicine and disease through bioinformatics analysis. Ultimately, a total of 116 overlapping disease targets and drug targets were obtained via online retrieval and software analysis. The clustering analysis of these targets, as well as the GO functional enrichment analysis, revealed their predominant concentration in tumor-related pathways such as PI3K-Akt, Foxo, and ErbB signaling pathways. Clinical specimen testing has indicated a close association between the ErbB2/PI3K/AKT/Foxo3a signaling pathway and LSCC disease. Pharmacological studies conducted in vitro and vivo experiments have suggested that EHD inhibits P-STAT3, P-PDK1, PI3K, P-AKT, and ErbB2 while upregulating FOXO3a protein expression. All aforementioned data collectively demonstrate that EHD effectively impacts the PI3K-AKT, FOXO3a, and ErbB2 signaling pathways thereby further confirming the results obtained from functional enrichment analysis.
The phosphatidylinositol-4,5-bisphosphate 3-kinase (PI3K)/v-akt murine thymoma viral oncogene homologue (AKT) pathway is not only activated in LSCC but also frequently activated in various human cancers and has been considered a promising therapeutic target. 12 Various pan-PI3K inhibitors, isomer selective inhibitors, and PI3K/mTOR dual inhibitors are currently being tested in clinical trials. 13 However, the clinical efficacy of inhibitors targeting this pathway may not have met expectations thus far, 14 but this lack of success does not necessarily imply that the PI3K/AKT pathway is not a crucial player in carcinogenesis. In this study, we observed elevated protein levels of p-STAT3, PDK1, Akt, and PI3K in tumor core tissues compared to normal tumor margin tissues of LSCC patients. This finding suggests that the PI3K-Akt signaling pathway plays a significant role in LSCC development. Through both in vivo and in vitro experiments, it was demonstrated that EHD effectively resolves phlegm and removes blood stasis by downregulating p-STAT3, PDK1, Akt, and PI3K protein levels. These results provide evidence supporting the potential use of EHD as a regulator of the PI3K-Akt signaling pathway for treating LSCC.
The Forkhead box proteins of class O subgroup (FOXOs) are recognized as tumor suppressors due to their established roles in cell cycle arrest, apoptosis, aging, differentiation, DNA damage repair, and scavenging of reactive oxygen species. 15 FOXO3 negatively regulates a set of genes associated with angiogenesis and vascular remodeling, such as angiopoietin 2 (Ang2) and eNOS, thereby inhibiting blood vessel formation and maturation. 16 Elevated expression of FOXO3 has been linked to upregulation of matrix metalloproteinases (MMPs) and increased cancer metastasis. 17 FOXC1 can attenuate FOXO activity by enhancing the PI3K/Akt signaling pathway, 18 acting upstream of the FOXO transcription factor. In this study, it was observed that the level of FOXO3A protein was higher in tumors compared to normal margin tissues in patients with laryngeal squamous cell carcinoma (LSCC). Both in vivo and in vitro experiments demonstrated that EHD elevated the level of FOXO3A protein, potentially contributing to its mechanism for resolving phlegm and removing blood stasis.
Tyrosine kinase receptor 2 (ErbB), also known as NEU or CD340, is a membrane receptor encoded by proto-oncogene erbB-2 with a molecular weight of 185 kDa. It belongs to the epidermal growth factor receptor (EGFR) family and activates PI3K through various tyrosine kinase receptors (RTKs) on the cell membrane, including EFGR, ErbB2 (HER2), PDGFR, and VEGFR. 19 Additionally, inhibition of ERBB2, PI3K, or AKT has been shown to lead to compensatory FOXO-dependent RTK expression. 20 Our study found that the concentration of ErbB-2 protein was higher in tumors than in normal tumor margin tissues in LSCC patients. Furthermore, we discovered that EHD could inhibit ErbB-2 protein levels through both in vivo and in vitro experiments while resolving phlegm and removing blood stasis.
Here, we aimed to identify overlapping targets for medicines and diseases, as well as their associated pathways, through network pharmacological analysis. The findings of our study provide support for the notion that the ERBB2/PI3K/AKT/FOXO3 pathway plays a significant role in LSCC signaling. Based on the results from our biological experiments, it is plausible that EHD may be involved in the ERBB2/PI3K/AKT/FOXO3 signaling pathways for potential therapeutic applications in LSCC treatment. However, further investigation is required to fully elucidate the underlying mechanisms of this study. Given the complexity of CHM compound components, it is imperative to determine which targets are involved and extensive efforts are needed for their optimization.
Conclusion
The findings of the current investigation indicate that EHD may exert a role in the prevention and treatment of LSCC via modulation of the ErbB2/PI3K/AKT/FOXO3a signaling cascade.
Supplemental Material
sj-docx-1-ict-10.1177_15347354241259182 – Supplemental material for Experimental Verification of Erchen Decoction Plus Huiyanzhuyu Decoction in the Treatment of Laryngeal Squamous Cell Carcinoma Based on Network Pharmacology
Supplemental material, sj-docx-1-ict-10.1177_15347354241259182 for Experimental Verification of Erchen Decoction Plus Huiyanzhuyu Decoction in the Treatment of Laryngeal Squamous Cell Carcinoma Based on Network Pharmacology by Xi Tan, Qiulan Luo, Yiwei Hua, Shiqing Zhou, Guiyuan Peng, Renliang Zhu, Wenyong Chen and Yunying Li in Integrative Cancer Therapies
Footnotes
Authors’ Contributions
QLL, GYP, YYL, and XT designed the study. XT, YWH, and SQZ conducted the experiments. RLZ and WYC participated in the data collection and analysis. XT and QLL participated in the writing of the manuscript and data interpretation. All authors read and approved the final manuscript and agree to be accountable for all aspects of the research in ensuring that the accuracy or integrity of any part of the work is appropriately investigated and resolved. XT and QLL contributed equally to this work.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (No. 82305328, 82274591, 82205204) and the Traditional Chinese Medicine Bureau of Guangdong Province (No. 20233018, 20201147, 20211177, 20211200) and the Department of Education of Guangdong Province (No. 2021KQNCX014).
Ethical Approval
The experiments on animals and humans were approved by Ethics Committee of Guangdong Chinese Medicine Hospital (Guangzhou, Guangdong, China).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
