Abstract
Vikil 20 is an herbal formula produced in Ghana and is widely marketed as a product to boost immunity as well as for general well-being. However, the pharmacological effect of this herbal preparation has not been proven scientifically. Therefore, this study was aimed at investigating the antioxidative as well as the anti-prostate cancer effects of the product. To assess the antioxidative effect of Vikil 20, the DPPH and ABTS activities were investigated. The total phenolic content was investigated using the Folin-Ciocalteu method. The cytotoxic effect of Vikil 20 against prostate cancer (PC-3) cells as well as normal (RAW 264.7) cells was investigated using the MTT assay whereas its anti-metastatic effect was analyzed using the cell migration assay. The effect of Vikil 20 on cell adhesion was analyzed via the cell adhesion assay whereas its effect on TNF-α secretion was investigated using a TNF-α detection kit. Vikil 20 demonstrated significant antioxidant effects by suppressing 57.61% and 92.88% respectively of DPPH and ABTS radicals at 1000 µg/mL with total phenolic contents of 140.45 mg GAE/g. Vikil 20 suppressed the proliferation of PC-3 cells by reducing the number of viable cells to 49.5% while sparing the RAW, 264.7 cells. Further, Vikil 20 significantly suppressed both cellular migration and adhesion of prostate cancer cells. Finally, suppression of cellular migration and adhesion is associated with a reduction in TNF-α secretion by PC-3 cells. Taken together, Vikil 20 was found to possess significant antioxidant and anti-prostate cancer effects in vitro.
Introduction
Among the several non-communicable diseases, cancer (of any origin) follows cardiovascular diseases as the leading cause of death. 1 Cancers are the principal cause of adult mortality worldwide, though some children are not spared, hence increasingly considered a major public health problem as the incidence and mortality increase continuously. 2 Prostate cancer is the second most common cancer in men after lung cancer with 1.4 million global cases in 2020 and approximately 375 000 deaths globally. 3 Even though the actual cause of prostate carcinogenesis is not fully explained, non-modifiable factors like age and ethnicity and modifiable risk factors like obesity, unhealthy diet, alcohol consumption, cigarette smoking, and oxidative stress are implicated. 4 Elevated oxidative stress is a key contributory factor in mutagenesis and progression of the stages of prostate cancer. 4 Current treatment options include chemotherapy, hormonal therapy, radiation therapy, and surgical interventions. Though there are significant limitations to all these methods, drugs offer the only approach in treating cases where the disease has spread through the body. Although chemotherapy has resulted in improvement in patients, the use of orthodox drugs is generally implicated in adverse effects including cardio-, neuro-, and nephrotoxicity. 5 Therefore, the need for more effective and less toxic therapies is the focus of modern research.
Bioactive chemicals abounding in herbal medicinal preparations make them the preferred alternatives in cancer treatment. In previous research, it was reported that herbal preparations exhibit favorable effects compared to conventional chemotherapeutic agents. 6 Because carcinogenesis is a multistage process, a single agent could not effectively alter the carcinogenesis processes. Therefore, the synergism between nutrients in plant-based extracts may offer protection against cancer.
Vikil 20 is an herbal formula with Khaya senegalensis, Azadirachta indica, and Vernonia amygdalina as active ingredients. It is perceived to support the immune system and to improve general wellbeing. Khaya senegalensis is known for its antioxidant as well as antiproliferative effects against colorectal cancers. 7 Azadirachta indica elicits antioxidant properties via activating antioxidant enzymes such as cytochrome p450-dependent monooxygenases as well as glutathione transferase and has also demonstrated anticancer activities by suppressing the growth of cell lines such as MCF-7 and HeLa. 8 Crude extracts of Vernonia amygdalina also suppress the proliferation of MCF-7 in vitro. 9 Despite its rich therapeutic composition, Vikil 20 has never been tested for its antioxidant and anticancer effects. Therefore, this experimental study was aimed at investigating the in vitro antioxidant as well as the anti-prostate cancer potentials of Vikil 20 .
Materials and Methods
Chemicals and Reagents
Reagents used in this study were MTT (Roche Applied Science, Mannheim, Germany) and nitric oxide (NO) detection kits (iNtRON Biotechnology, Seongnam-si, Korea). TNF-α ELISA kit was bought from Research and Development Systems (Minneapolis, MN) whereas Dulbecco′s Modified Eagle Medium, 2,2-diphenyl-1-picrylhydrazyl (DPPH) and 2,2-azino-bis-(3-ethylbenzthiazoline-6-sulfonic) acid (ABTS) were bought from Sigma Aldrich in South Korea.
Methods
Pre-experimental processing of Vikil 20 extract
Vikil 20 (batch number JV20/090) was obtained from pharmacies within the Ho municipality (Figure 1). Five bottles of the herbal formula (250 mL each) were concentrated by rotary evaporation. To attain a stock dose of 10 mg/mL (w/v), the Vikil 20 was reconstituted in double distilled water.

A picture showing the commercial herbal formula Vikil 20 and its package.
1,1-diphenyl–2 picrylhydrazyl (DPPH) radical scavenging assay
The DPPH scavenging activity was determined using the modified method by Gyamfi et al. 10 Briefly, 40 µL of (100, 500, and 1000) µg/mL of Vikil 20 was added to 160 µL of 0.1 mmol/L DPPH. The mixture was kept at ~25°C for 30 minutes. Further, the absorbance of the mixture was measured using 517 nm wavelength on DNM-9602 microplate reader. Ascorbic acid (200 µg/mL) was used as a standard antioxidant. The formula below was used to calculate the radical scavenging activity: (%) inhibition = {[(A0 − Ac)/A0] × 100}. Where A0 = the absorbance of the blank (in which the same volume of distilled water was used in place of the test sample) and Ac = the absorbance in the presence of the test samples (Vikil 20 ) or standard (Ascorbic acid).
2,2′-azino-bis (3-ethylbenzthiazoline-6-sulfonic) acid (ABTS) scavenging assay
The ABTS scavenging activity was determined using the modified method by. 11 Briefly, 40 µL of (100, 500, and 1000) µg/mL Vikil 20 was mixed evenly with 160 µL ABTS• + solution. The ABTS• +-Vikil 20 mixture was incubated at 30°C for 10 minutes. The absorbance of the ABTS• +-Vikil 20 mixture was read at 734 nm. Ascorbic acid (200 µg/mL) was used as a standard antioxidant. The ABTS• + scavenging activity was calculated using the formula: (%) inhibition = {[(A0 − Ac)/A0] × 100}. Where A0 = the absorbance of the blank (in which the same volume of distilled water was used in place of test sample) and Ac = the absorbance in the presence of the test samples (Vikil 20 ) or standard (Ascorbic acid).
Estimating the total phenolic content of Vikil 20
This was determined using the modified method by. 12 Initially, 5 mg of Vikil 20 was completely dissolved in 1 mL of methanol. Of the resultant solution, 200 µL was mixed with 800 µL of double distilled water. Subsequently, 1 mL of the above mixture was mixed with distilled water (5 mL) and Folin–Ciocalteu reagent (0.5 mL), with intermittent vortexing. The mixture was incubated for 5 minutes at room temperature, after which 20% (w/v) sodium carbonate (1.5 mL) and distilled water (10 mL) were added. The mixture was incubated at 25°C for 10 minutes after which the optical density was read at 750 nm, photometrically (UV–vis Jasco V-630). The total phenolic content was expressed as Gallic Acid Equivalent (GAE) in µg/mg basis on sample dry weight.
Cell viability and morphology assay
MTT cell viability assay was used to determine the cell viability of the RAW 264.7 and PC-3 cells. Briefly, each cell line was seeded in 96 wells at 2 × 103 cells/well and allowed to attach at 37⁰C in a humidified atmosphere with 5% CO2 and 95% air for 24 hour. Later, different concentrations of Vikil 20 (100, 250, 500, and 1000 µg/mL) were added to the RAW 264.7 and PC-3 cells; and incubated for 48 hour. Afterward, 20 µL of MTT (5 mg/mL) solution was added to each well and after 4 hour incubation, formazan crystals were dissolved in 100 µL DMSO. The optical density of the final product was read at 570 nm using the DNM-9602 microplate reader. Cell viability was calculated as a percentage of untreated controls. To monitor morphological changes in cell death in malignant (PC-3) cell lines, morphological studies were performed by visualizing both treated and untreated cells under a standard inverted microscope with 40x magnification, 48 hour after treatment.
Cell adhesion assay
Briefly, 2 × 105 cells/well were treated with Vikil 20 (1000 µg/mL) for 24 hour. The treatment was done on a 6-well plate. The cells were then detached and seeded again on a fresh plate. After 24 hour of incubation, the attachment as well as morphology of the cells were observed and photographed with a Cossim industrial digital camera (E3ISPM12000KPA). As a control, cells were cultured on the same plates without Vikil 20 .
Cell migration assay
Briefly, 3 × 106 cells were cultured in each well plate at 37°C for 24 hours. The confluent cell monolayer formed was scratched with a pipette tip. Floating cells were removed with a fresh medium. The cells were then photographed (at 0 hour) using an inverted microscope with 40x magnification, the media was replaced and Vikil 20 (1000 µg/mL) added. Control cells had only fresh media without Vikil 20 . After incubation at 37°C for 48 hour, photographs were taken and cell migration ability was observed.
Analysis of TNF-α secretion
Levels of TNF-α in supernatant were measured using a TNF-α kit according to the manufacturer’s protocols. PC-3 cells (2 × 105 cells/well) in 6 wells were treated with Vikil 20 (1000 µg/mL) for 48 hour after which supernatants were collected. Approximately 50 µL of supernatant from treated cells was added to the same volume of assay diluent RD1F for 2 hour at room temperature after which 200 µL of Human TNF-α conjugate was added for 2 hour. This was followed by the addition of 200 µL of substrate solution for 30 minutes after which 50 µL of stop solution was added for 15 minutes. The optical density was determined at 450 nm. The control group consisted of supernatants from untreated cells.
Data Analysis
All data were expressed as mean ± standard deviation (SD) of independent experiments using Microsoft Excel 2016. All tests were carried out in triplicates. P < .05 was considered to indicate a significant difference.
Results
Antioxidant Activity and Total Phenolic Content of Vikil 20
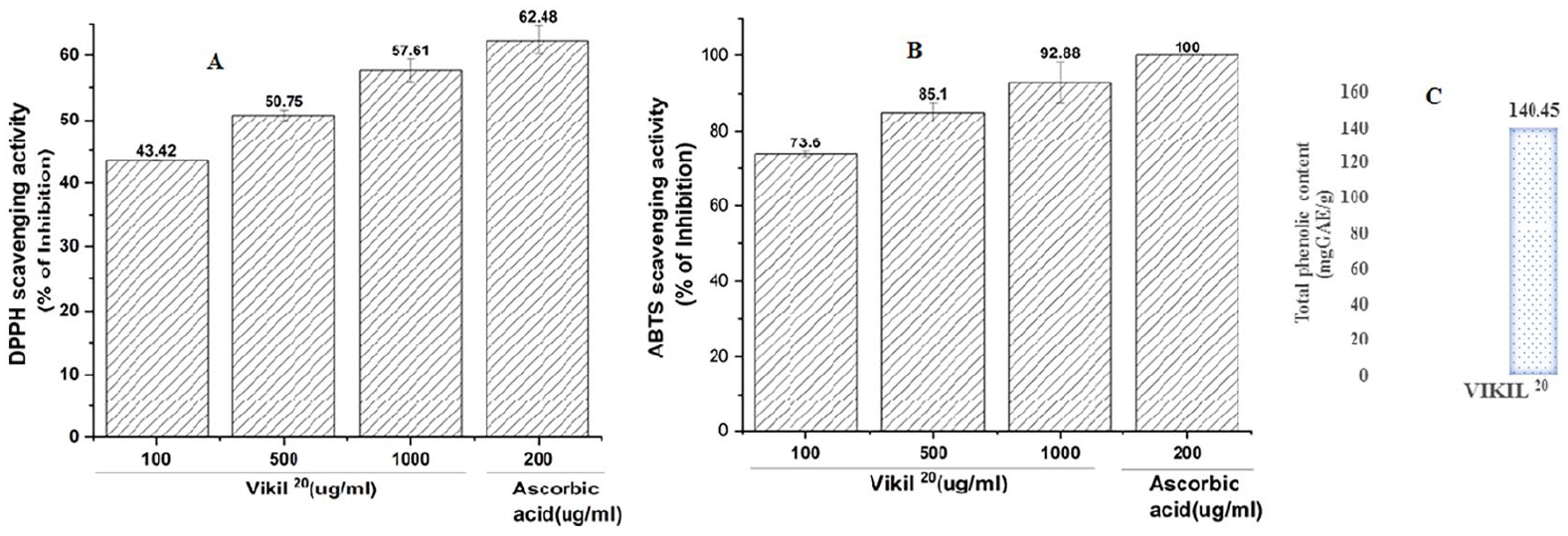
Vikil 20 demonstrated a significant DPPH free radical scavenging effect which was dose-dependent, quenching 43.42%, 50.75%, and 57.61% respectively at 100, 500, and 1000 µg/mL whereas ascorbic acid suppressed 62.48% at 200 µg/ml (Figure 2A). Furthermore, Vikil 20 also demonstrated an impressive ABTS radical scavenging activity which was also dose-dependent. It suppressed 73.60%, 85.10%, and 92.88% ABTS radicals at 100, 500, and 1000 µg/mL respectively (Figure 2B). Moreover, the total phenolic content was significantly high and was recorded at 140.45 mg gallic acid equivalent in 1 g dry weight of Vikil 20 extract (Figure 2C).
Cytotoxic Potential of Vikil 20
To investigate the selective cytotoxic effect of Vikil 20 on PC-3 prostate cancer cell line, PC-3 or the non-cancer RAW 264.7 cells were each treated with various doses of Vikil 20 for 48 hour after which viability was measured using the MTT assay. Vikil 20 suppressed the growth of the prostate cancer (PC-3) cells whereas sparing the normal (RAW 264.7) cells (Figure 3A and B). The viabilities of PC-3 cells were 66.7%, 58.9%, 52.5%, and 50.5% respectively at 100, 250, 500, and 1000 µg/mL of Vikil 20 . The normal (RAW 264.7) cells were however spared, with viabilities of 119.4%, 118%, 217.43%, and 257.68% after 48 hour of treatment with 100, 250, 500, and 1000 µg/mL of Vikil 20 (Figure 3B). PC-3 cells treated with Vikil 20 also demonstrated significant morphological changes especially, at 1000 µg/mL (Figure 3C). Whereas the untreated cells exhibited a homogenous monolayer with typical morphology, there was a remarkable inhibition in monolayer formation in treated cells, especially at 1000 µg/mL where the morphology of the cells was altered, making them round and separated from each other (Figure 3C).
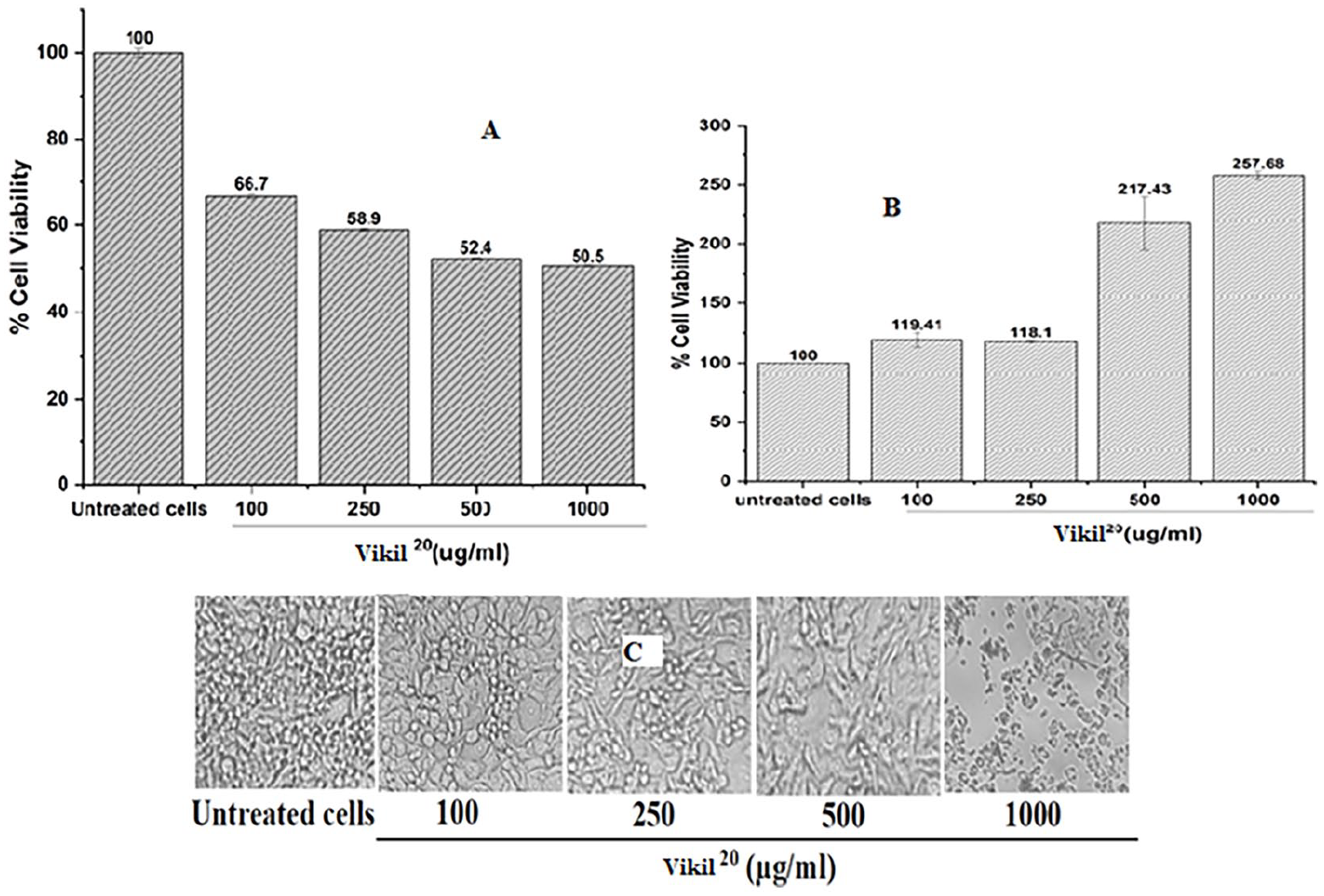
Effect of Vikil 20 on cell viability and morphology. PC-3 and RAW 264.7 cells were treated with various doses of Vikil 20 for 48 hour. The viabilities of (A) PC-3 and (B) RAW 264.7 cells were assessed using the MTT assay. (C) Cell morphology was visualized under an inverted microscope with 40x magnification.
Effect of Vikil 20 on Cell Migration and Adhesion
The effect of Vikil 20 on the migration of PC-3 prostate cancer cells was analyzed using the scratch wound healing assay. Confluent PC-3 cells were scratched with a pipette tip before treatment with or without Vikil 20 for 48 hour. In the untreated cells, there was significant migration into the denuded zone, However, the migration of PC-3 cells was suppressed prominently within 48 hour of incubation with Vikil 20 (Figure 4A). In the cell adhesion assay, PC-3 cells that had grown to 80% confluence were treated with Vikil 20 for 24 hour after which cells were trypsinized and transferred to a new 6-well plate for 24 hour. Vikil 20 significantly suppressed the adhesion of PC-3 cells (Figure 4B).

Effect of Vikil 20 on cell migration and adhesion. (A): Cell migration was investigated by the scratch wound assay. A straight scratch was made using a pipette tip on the confluent cell monolayer and remaining attached cells imaged (at 0 hour) and treated with Vikil 20 , except for the control. Following 48 hour incubation, images were captured and cell migration ability was observed. (B): Cells were treated with Vikil 20 for 24 hour and then detached and plated back on a fresh plate for 24 hour after which the attachment status was observed and photographs captured under an inverted microscope with 40x magnification.
Cytokines such as TNF-α, IL-6, and IL-1β are known to influence tumor growth as well as the onset of metastasis. 13 TNF-α is associated with promoting metastasis and invasion in prostate cancers. 14 Serum levels of TNF-α have been found to correlate positively with the progression of prostate cancer disease. 15 The basal level of TNF-α in supernatants of control cells was measured at 1.6 pmol/L. However, treatment with Vikil 20 reduced the expression of TNF-α moderately to 1.3 pmol/L (Figure 5).

Discussion
Prostate cancer is a leading cause of morbidity and mortality in men worldwide. 16 After lung, breast and colon cancers, prostate cancer is the fourth most diagnosed cancer. In 2018, more than 0.35 million men died from prostate cancer and if the current trend continue, there will be more than two million cases in 2040. 17 Although significant progress has been attained regarding prostate cancer treatment, the adverse nature of existing chemotherapeutics is still a major challenge. Herbal medicines have become popular due to their effectiveness against a variety of chronic diseases largely as a result of the presence of a plethora of phytocompounds with different mechanisms of action working in synergy. 18 Vikil 20 is an herbal formula made from the combination of Khaya senegalensis, Azadirachta indica, and Vernonia amygdalina and is perceived to be efficacious as an immune booster, to combat exhaustion, to ease hangover, to enhance athletic performance and to improve digestion.
The onset and progression of cancers are thought to be mediated by oxidative stress. Reactive oxygen species (ROS) levels may increase the risk of cancer. This is because oxidative stress has been implicated in tumor development and contributes to the progression of the tumor. 19 Reports indicate that prostate cancer is commonly associated with increased oxidative stress, 20 hence, antioxidants are therefore important in the prevention as well as the treatment of prostate cancers. In the current study, Vikil 20 suppressed DPPH free radicals in a dose-dependent manner. Furthermore, it also inhibited ABTS free radicals. Phenolic compounds are well known for their health benefits which include antioxidant, anti-inflammatory, and anticancer activities. 21 Substances rich in polyphenols have shown significant association with decreased risk of cancer mortality as well as oxidative stress. 22 Vikil 20 was found to have a significantly high total phenolic content which was recorded at 140.45 mg gallic acid equivalent per gram dry weight (mg GAE/g) of the herbal formula (Figure 2C). Our result is similar to that of Chanthasri et al 22 who measured the total phenolic contents of 20 polyherbal remedies in Thailand. Total phenolic contents were within the range of 384.6 mg GAE/g to 39.3 mg GAE/g with an average of 146.3 mg GAE/g. Languon et al 23 carried out a similar study in which the total phenolic contents of some herbal products on the Ghanaian market were 45.16, 18.55, 12.3 and 10.24 mg GAE/g respectively for MUT, STO, K-HER, and K-BA.
Despite the advances made in cancer biology, cancer remains one of the major causes of mortality worldwide and diagnoses are sharply increasing globally. 6 As the second leading cancer in men worldwide, prostate cancer is a major public health challenge. Reports indicate that it can be even more aggressive and may advance to a castration-resistant disease in older men of African descent if left untreated. 24 Available treatment regimens are quite expensive, have a plethora of harmful effects, are challenged with resistance and are not easily accessible. Hence, the search for affordable, less harmful, effective, and readily available alternatives is imperative. The most desired necessities of cancer therapeutics are efficacy and selective toxicity. 25 The ability of a drug to suppress proliferation in cancer cells, while paring normal cells is a key requirement for an effective cancer treatment. 26 From the results of our study, Vikil 20 significantly suppressed the proliferation of PC-3 cells whereas it had no adverse effect on the normal (RAW 264.7) cells (Figure 3A and B). At 1000 µg/mL, the viability of PC-3 had significantly reduced to 50.5% whereas that of RAW.264.7 was at 257.68%. Plants have a good number of secondary metabolites that act via different mechanisms in suppressing cancers and yet are neutral to normal cells. Vikil 20 is made up of at least 3 plant species, each of which has various phytochemicals that are known to have numerous health benefits, including the inhibition of certain cancers. Although this study focused on the anti-prostate cancer effect of Vikil 20 , future studies into its efficacy against other cancer types are recommended.
Cancer hallmarks include 8 biological capabilities that early cancer cells acquire during the multistep progression of human tumors. These include maintaining growth signaling, evading growth inhibitors, evading immune destruction, achieving replicative immortality, activating invasion and metastasis, inducing angiogenesis, and protecting against cell death and deregulation of cellular energetics. 27 Chemotherapeutics which can substantially suppress one or more of these acquired characteristics would be of enormous importance in the battle against cancer. Metastasis is a complex process in which malignant cells leave a major organ, migrate to distant sites, and eventually occupy a clinically detectable location in a distant organ. Patients with advanced breast and prostate cancer usually develop bone metastases, causing severe bone pain. 27 Unfortunately, research into the development of drugs that inhibit metastasis has not been very successful. In the present study, the migration of PC-3 was significantly suppressed within 48 hour of treatment with Vikil 20 (Figure 4A). Additionally, tumor invasion requires the adhesion of cancer cells to the extracellular matrix, 27 hence drugs that inhibit adhesion are important in the fight against cancer migration. From our results, treatment with Vikil 20 for 48 hour significantly suppressed the adhesion of PC-3 cells compared to the untreated controls.
TNF-α mediated pro-survival signaling is a major signaling cascade implicated in cell survival and the subsequent resistance to therapy. 28 Increased quantities of TNF-α in cancer patients and its enhanced expression in pre-cancerous and tumor tissues have been widely reported. This suggests that TNF-α plays an important role in tumor progression. 28 There is, therefore, the need to down-regulate the expression of TNF-α in prostate cancer. This is evident from our results in Figure 5 in which Vikil 20 suppressed the expression of TNF-α compared to the untreated cancer cells. Several inhibitors of TNF-α have been approved for management of several human diseases. However, the risk of toxicity and other untoward health problems 29 have made the use of TNF-α an unpopular therapeutic target. Hence, Vikil 20 serves as a novel and promising candidate in the inhibitors of TNF-α in prostate cancer.
Conclusion
Taken together, our results indicate that Vikil 20 possesses significant antioxidant activity and selectively suppresses the proliferation of PC-3 prostate cancer cells while sparing normal cells and may therefore stand as a promising candidate for the management of oxidative stress related diseases like prostate cancer. In vivo anti-prostate cancer assays involving mechanisms of action of specific molecular targets as well as toxicological studies are also recommended.
Footnotes
Author Contributions
C. O. T. and D. B. conceived and planned the experiments. C. O. T., E. A. and D. B. carried out the experiments. H. K-B. and C. O. T. contributed to sample preparation. E. K. E. and C. O. T. contributed to the interpretation of the results. C. O. T. and E. A. took the lead in writing the manuscript. E. K. E., A. A. B. and N. K. K. did a critical revision of the manuscript. All authors provided critical feedback and helped shape the research, analysis and manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
