Abstract
Background:
The surgical intervention serves as the paramount and prevalent remedy for individuals afflicted with colorectal malignancies, with the significance of perioperative stewardship and convalescence being indisputable. Prehabilitation coupled with preoperative lifestyle modulation has demonstrated efficacy in patients subjected to certain classifications of abdominal procedures. However, the evidence pertaining to its impact on those battling colorectal cancer remains equivocal.
Methods:
A meta-analysis, grounded in pairwise contrast, of randomized controlled trials (RCTs) was orchestrated, coupled with a systematic review, to probe the efficacy of preoperative lifestyle modulation and prehabilitation on patients’ postoperative functionality and recuperation. An exhaustive exploration of 8 electronic databases and trial registries was undertaken to encompass all pertinent RCTs disseminated in English or Chinese from January 2012 through December 2022. Employing a random-effects model, we evaluated parameters such as the 6-minute walk test (6 MWT), complications, quality of life (QoL), aggregate and postoperative duration of hospitalization (tLHS and postLHS), and healthcare expenditure (HExp) for postoperative patients.
Results:
A total of 28 RCTs were incorporated into the systematic review and meta-analysis. Relative to conventional preoperative care, rehabilitation or preoperative lifestyle management was found to enhance postoperative 6MWT (SMD 1.30, 95% CI 0.30 to 2.29) and diminish the complication rate (OR 0.53, 95% CI 0.40 to 0.69). Nonetheless, no significant discrepancies were observed in QoL (SMD 1.81, 95% CI −0.26 to 3.87), tLHS (SMD −0.26, 95% CI −0.68 to 0.15), and postLHS (SMD −1.46, 95% CI −3.12 to 0.20) between the groups. HExp could not be evaluated due to a lack of sufficient data for synthesis. Most pooled outcomes exhibited significant heterogeneity, urging a cautious interpretation. Subgroup analysis revealed that nutritional interventions could mitigate the incidence of complications, and preoperative exercise could improve tLHS and postLHS. A combined approach of physical, nutritional, and psychological intervention or prehabilitation proved superior to any single intervention in enhancing postoperative capabilities.
Conclusion:
This meta-analysis delineated the efficacy of preoperative interventions on postoperative capabilities in patients with colorectal cancer, thereby offering evidence for clinical practice. It was concluded that preoperative interventions are unequivocally beneficial for postoperative functional recovery and the reduction of complication rates in patients with colorectal cancer. Nonetheless, the acquisition of more high-level evidence is still necessitated to further ascertain the effectiveness of this strategy for other patient groups and to establish its best practices. The heterogeneity in the pooled outcomes underlines the need for future studies to be more uniform in their design and reporting, which would facilitate more robust and reliable meta-analyses.
Keywords
Introduction
Colorectal cancer (CRC) is one of the most prevalent malignant neoplasms, responsible for approximately 11% of all cancer diagnoses worldwide and leading to 8.8 million deaths in 2018. 1 Over the past decade, an increase in CRC cases has been observed among individuals under 50, particularly in countries undergoing economic transitions, thereby escalating healthcare expenditures.1 -3 Surgical treatment, either via laparoscopic or open abdominal operations, is typically the preferred approach for most CRC cases, especially those in the early stages, when simple endoscopic treatment is not feasible. The choice between laparoscopic and open surgery largely depends on the surgeon’s expertise and the patient’s condition. 4 Therefore, the perioperative care of CRC patients is of paramount importance, with potential implications for surgical outcomes and prognoses.5,6 Beyond routine perioperative care, the Enhanced Recovery After Surgery (ERAS) protocol has emerged as one of the most employed strategies in the surgical management of CRC.7,8 A typical ERAS strategy often emphasizes the importance of preoperative management, but it primarily focuses on the postoperative phase. 9
In 2015, prehabilitation emerged as a complementary and potentially more efficacious strategy than ERAS, 10 aimed at augmenting postoperative functional capacities. This innovative concept in preoperative management serves as an extension of ERAS principles, wherein patients undergo interventions prior to the surgical stress. These interventions encompass cardiorespiratory conditioning primarily through aerobic and resistance exercises, nutritional support mainly through the supplementation of whey protein, and psychological assistance. 11 The effectiveness of prehabilitation has been demonstrated in diverse patient populations undergoing surgical treatments. Prehabilitation adopts a proactive approach to enhance the patient’s health and physical condition prior to surgery, with the ultimate goal of fortifying their ability to endure the stress of the procedure and expedite their recovery thereafter. 12
Typically, a prehabilitation approach encompasses 3 components: preoperative physical exercise, nutritional support, and cognitive intervention. 13 In essence, it is a preoperative lifestyle intervention that focuses on enhancing the functional capacity of patients. 11 Prehabilitation has proven effective in certain types of surgeries, particularly lung cancer and abdominal malignancy surgeries, with a multimodal prehabilitation approach often favored over a single preoperative lifestyle intervention.12,14 -16 However, there is a dearth of high-level evidence regarding the effectiveness of prehabilitation in CRC patients. The current evidence from randomized controlled trials (RCTs) is mixed, especially when considering studies from different regions. The practical application of prehabilitation in clinical settings also remains to be fully explored.
Perhaps due to the novelty of the prehabilitation concept, it has been referred to by various terms such as preoperative intervention and capacity improvement before surgery.13,17 This has resulted in a dispersion of related evidence, making it more challenging for clinical practice to reference. Furthermore, the specific role and impact of each component within prehabilitation in enhancing a patient’s capacity requires further investigation. Therefore, in this context, this study aims to provide evidence on the effectiveness of prehabilitation and other forms of preoperative lifestyle interventions on postoperative capability in CRC patients.
Methods
Study Design
This systematic review of randomized controlled trials used contrast-based frequentist pairwise meta-analysis to synthesize data. We reported the results according to the preferred reporting items for systematic reviews and meta-analyses (PRISMA) 2020 statement. 18 A protocol was prepared and registered a priori in PROSPERO (CRD42022359495).
Eligibility Criteria
We aimed to include all randomized controlled trials related to prehabilitation and preoperative lifestyle interventions in patients with CRC in the past 10 years. The eligibility criteria are displayed in Table 1.
Eligibility Criteria.

Search Strategy
We searched PubMed, Web of Science, Embase, CINAHL, Cochrane Central Register of Controlled Trials, CNKI, WanFang Data, and SinoMed for eligible articles published from 1 January 2012 to 15 December 2022 using terms “colorectal,” “colon,” “rectal,” “prehabilitation,” “lifestyle,” “nutrition*,” “exercise,” “psychologi*,” “congnition*,” and others. The full and detailed search strategy can be found in
Screening, Selection and Data Extraction
All references identified from the search were imported into EndNote 20 (Clarivate, PA, USA) to remove duplicates. After automatic exclusion by filtering title using excluding terms, the reviewers (J.-J.Z., Y.-R.H., H.-L.D., and Z.-M.H.) assessed the eligibility in the order of title, abstract, and full text. Each reference was decided independently by at least 2 reviewers, and arisen discrepancies were discussed and decided by the authors together.
Four reviewers (J.-J.Z., Y.-R.H., H.-L.D., and Z.-M.H.) independently extracted data using a standard data collection form. We resolved any disagreements by discussion and consensus.
Risk of Bias Assessment
Two reviewers independently assessed risk of bias of the included studies using Cochrane RoB 2 tools. 20 RCTs were categorized into the levels of “low,” “some concerns,” or “high” risk of bias based on the level of 5 different domains of risk of bias in RoB 2 tools.
Statistical Analysis
Primarily, we employed the “metafor” package within R for our data synthesis. A frequentist method rooted in pairwise contrast was utilized for the meta-analysis, with heterogeneity evaluated through I² and Q statistics. In instances where the I² statistic exceeded 50%, a random-effects model was applied in the meta-analysis, while a fixed-effect model was otherwise implemented. To further analyze heterogeneity, we conducted a subgroup analysis based on the characteristics of the participants and the types of interventions. Sensitivity analyses were carried out to gage the robustness of our results. The presence of publication bias was assessed using the Begg and Egger tests. For this study, the level of statistical significance was set at 0.05, two-tailed.
The collated data were presented as the standardized mean difference (SMD) for continuous data measurements, odds ratio (OR) for dichotomous data, and SMD for outcomes reported in both continuous and dichotomous formats. Additionally, 95% confidence intervals (95% CI) were disclosed.
Results
Study Characteristics
We identified 2620 publications and trial registrations from the initial search. After data selection, 28 studies were finally included in the review (Figure 1). The study characteristics were displayed in Table 2.
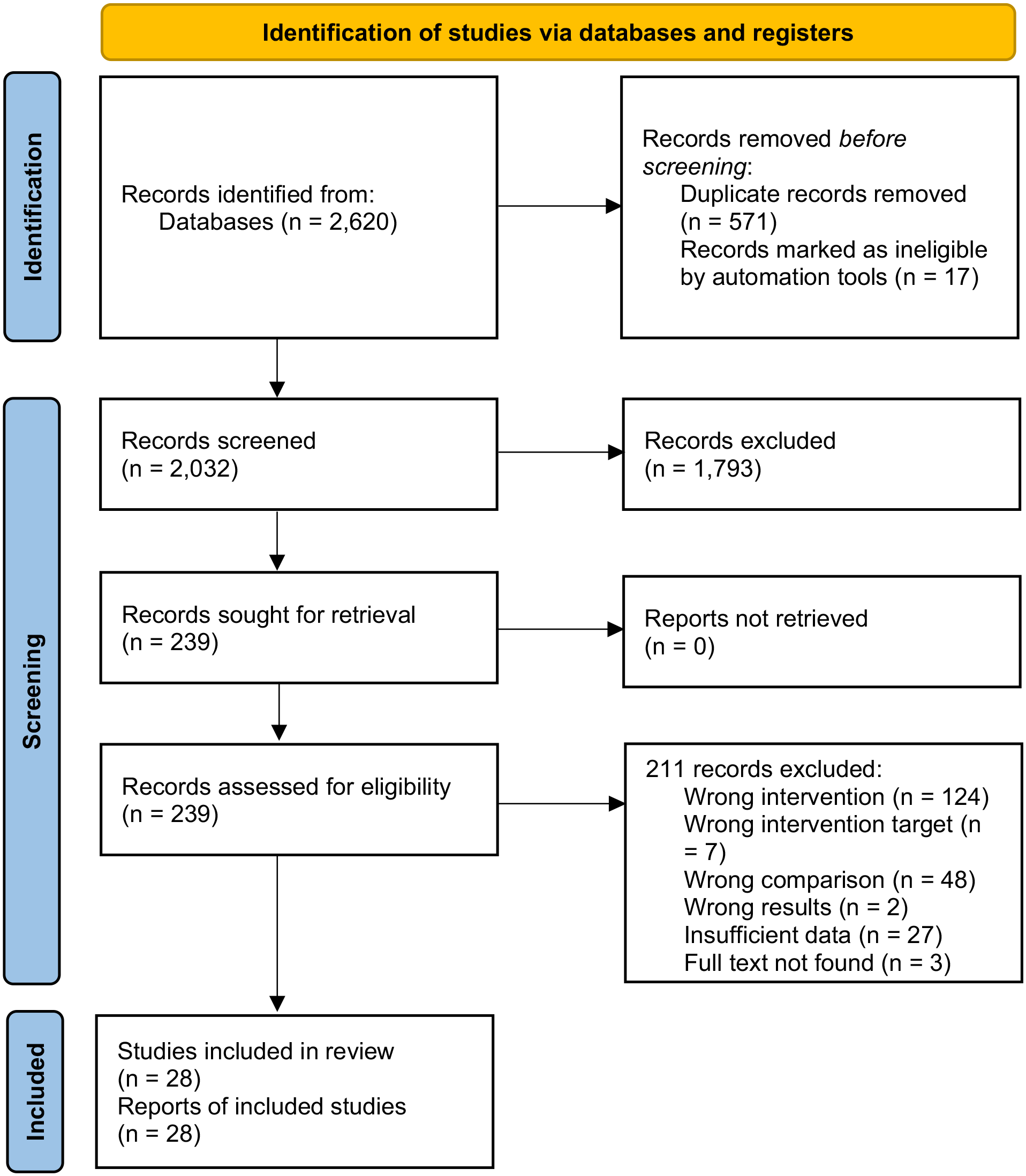
PRISMA flow diagram of study selection.
Study Characteristics.
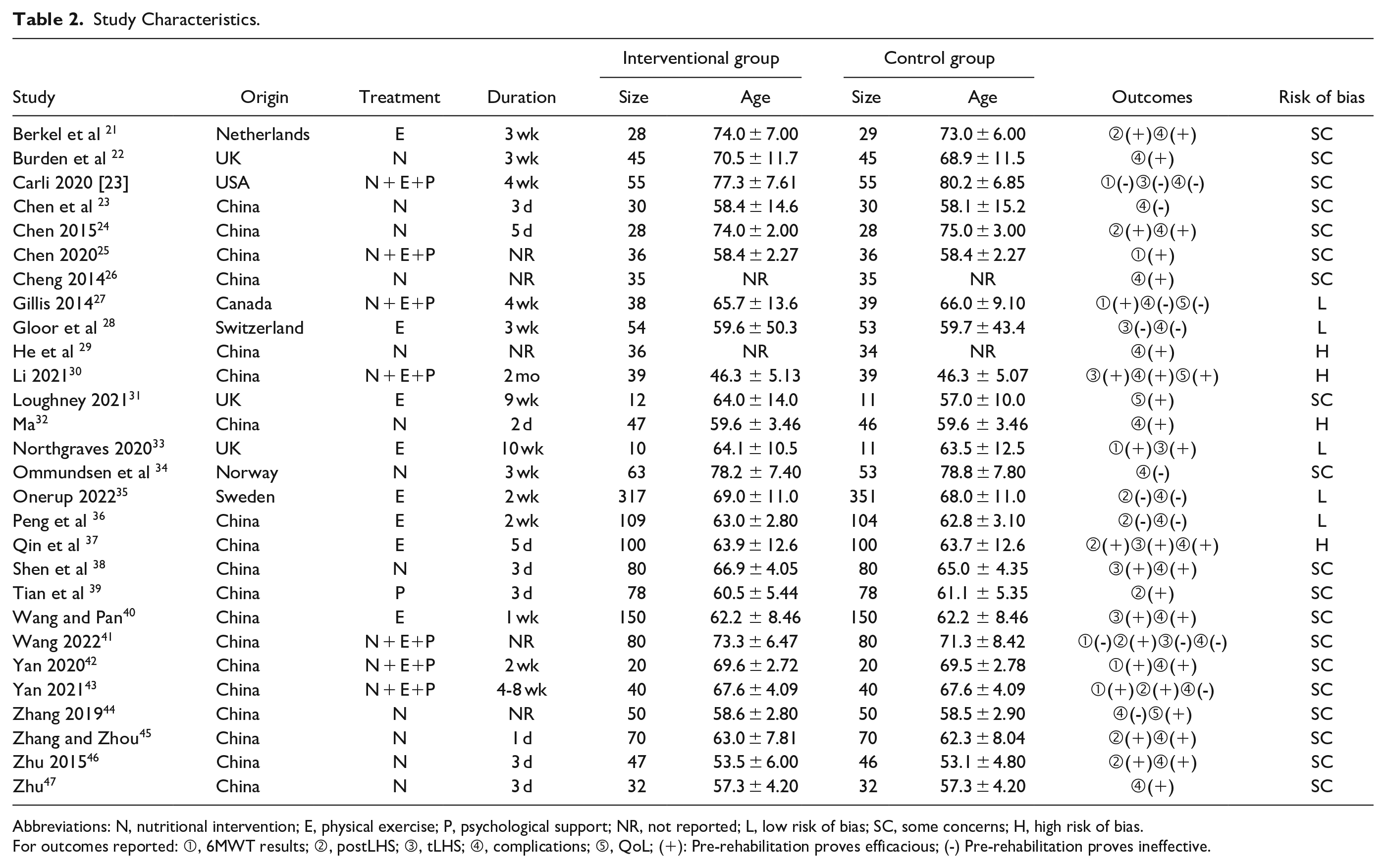
Abbreviations: N, nutritional intervention; E, physical exercise; P, psychological support; NR, not reported; L, low risk of bias; SC, some concerns; H, high risk of bias.
For outcomes reported: ①, 6MWT results; ②, postLHS; ③, tLHS; ④, complications; ⑤, QoL; (+): Pre-rehabilitation proves efficacious; (-) Pre-rehabilitation proves ineffective.
The bulk of the trials were disseminated within the past 5 years (2018-2023), with China serving as the primary source. The sample sizes spanned a spectrum from a mere 10 to a substantial 317, with the majority of participants being individuals of advanced age, exceeding 60 years. Given the absence of a standardized method for prehabilitation or lifestyle management, the selected interventions diverged across studies, and participants underwent varying durations of preoperative treatment, ranging from a single day to an extensive 9 weeks. Among the predetermined outcomes, health expenditure failed to be reported by the qualifying studies and thus was omitted from the final analysis.
Risk of Bias
Out of the 28 studies incorporated into our analysis, 4 studies (14%) were identified as carrying a high risk of bias, while 6 studies (21%) were deemed to bear a low risk of bias (as shown in Figure 2). The overall risk of bias in the eligible studies was deemed acceptable. The risk of bias assessment results for each included study are presented in Table 2.

Risk of bias assessment.
Primary Outcomes
Six-minute walk test
Seven studies,25,27,33,41 -43,48 encompassing 560 participants, reported results for the postoperative 6-minute walk test (6MWT). In these studies, a combination of nutritional, exercise, or psychological interventions was employed by 6 of them. The pooled analysis indicated that prehabilitation and/or preoperative lifestyle management significantly enhanced the patients’ postoperative activity capability compared to routine care (SMD = 1.30, 95% CI 0.30 to 2.29). However, substantial heterogeneity was observed (as depicted in Figure 3A).

Effect of prehabilitation on 6WMT. (A) pooled analysis. (B) sensitive analysis, omitting each single study, random-effect model. (C) sensitive analysis, adding each single study, fixed-effect model. (D) subgroup analysis of studies of different origin. (E) subgroup analysis of different interventional duration.
In this analysis there was no significant publication bias on Egger test (P = .065). A sensitivity analysis was performed, and the results were not significantly different from the total combined estimated value, which suggested that the meta-analysis results were relatively stable and reliable. Subgroup analysis revealed that the Chinese studies, which did not report their duration of intervention, contributed most to the observed heterogeneity (as shown in Figure 3D and E). Despite this, the result remained robust when any single study was omitted from or added to the pooled analysis, as well as when different models were utilized (as illustrated in Figure 3B and C).
We observed that when only non-Chinese research data were available, the heterogeneity was 0. The pooled analysis showed that prehabilitation and/or preoperative lifestyle management significantly improved the patients’ postoperative activity capability compared to routine care (SMD = 0.46, 95% CI 0.19 to -0.74), and the results were stable. (as shown in Figure 4).

Sensitivity analysis of 6-minute walk test (6WMT) results from western studies only.
Postoperative complications
Twenty-two investigations, encompassing 2989 participants, documented postoperative complications. The amalgamated analysis suggested that prehabilitation or preoperative lifestyle modifications could efficaciously diminish the occurrence of postoperative complications (OR = 0.43, 95% CI 0.40 to 0.69, Figure 5A), accompanied by a moderate, yet acceptable, heterogeneity (I2 = 51%). Subgroup Analysis by Country: heterogeneity was not reduced, indicating country is not the source of heterogeneity. Subgroup scrutiny inferred that both a multimodal intervention and standalone nutritional counseling could yield effective reductions in complications, with the latter demonstrating a larger effect size (Figure 5A). Sensitivity scrutiny pinpointed Onerup 2022 35 as a potential heterogeneity source, yet the amalgamated outcome remained fairly robust when excluding or incorporating a single study (Figure 5C and D). A sensitivity analysis was performed, and the results were not significantly different from the total combined estimated value, which suggested that the meta-analysis results were relatively stable and reliable.

Effect of prehabilitation on postoperative complications. (A) pooled analysis and subgroup analysis of different treatment types. (B) Funnel Plot, an evaluation of publication bias. (C) sensitive analysis, omitting each single study, random-effect model. (D) sensitive analysis, adding each single study, fixed-effect model. (E) subgroup analysis of different intervention duration.
Secondary Outcomes
Quality of life
A mere quartet of investigations27,30,31,44 proffered accessible Quality of Life (QoL) findings, utilizing disparate scales. No discernible distinction was observed between the intervention and control cohorts (SMD = 1.81, 95% CI −0.26 to 3.87), accompanied by pronounced heterogeneity (I 2 = 97%, P < .01, Figure 6). Sensitivity analysis, subgroup analyses and publication bias could not be performed due to the limited number of included studies.
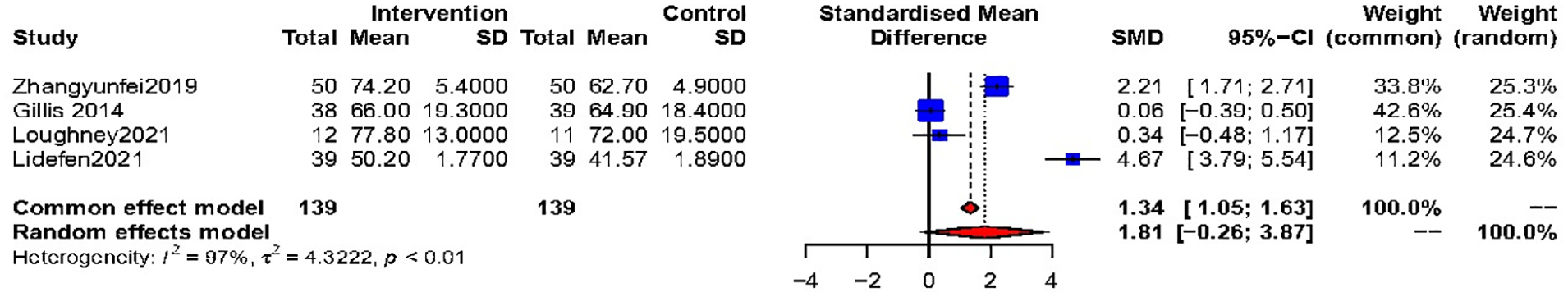
Effect of prehabilitation on QoL.
The impacts exhibited marked variation across the individual trials. Owing to the paucity of studies and participants incorporated, a heterogeneity analysis was not undertaken. Ultimately, the amalgamated outcome failed to illuminate the efficacy of prehabilitation on Quality of Life.
Postoperative length of hospital stay
A total of 10 studies, comprising 1823 participants, were amalgamated for the analysis of post-operative length of hospital stay (postLHS). The meta-analysis revealed no significant effect (SMD = −1.46, 95% CI −3.12 to 0.20, Figure 7A), and substantial heterogeneity was still apparent (I² = 96%, P < .01). A subgroup analysis of different treatment strategies suggested that the heterogeneity might primarily stem from trials that solely employed nutritional counseling as an intervention. When these RCTs were excluded, the effect attained significance (Figure 7B). However, the results were not consistently robust when a specific study 46 was omitted and also fluctuated between models (Figure 7C and D). Publication bias was assessed by Egger test and significant statistical publication bias was detected (P = .04). Sensitivity analysis was performed and demonstrated that one study might have been highly influential, suggesting that this result may be an outlier. 46
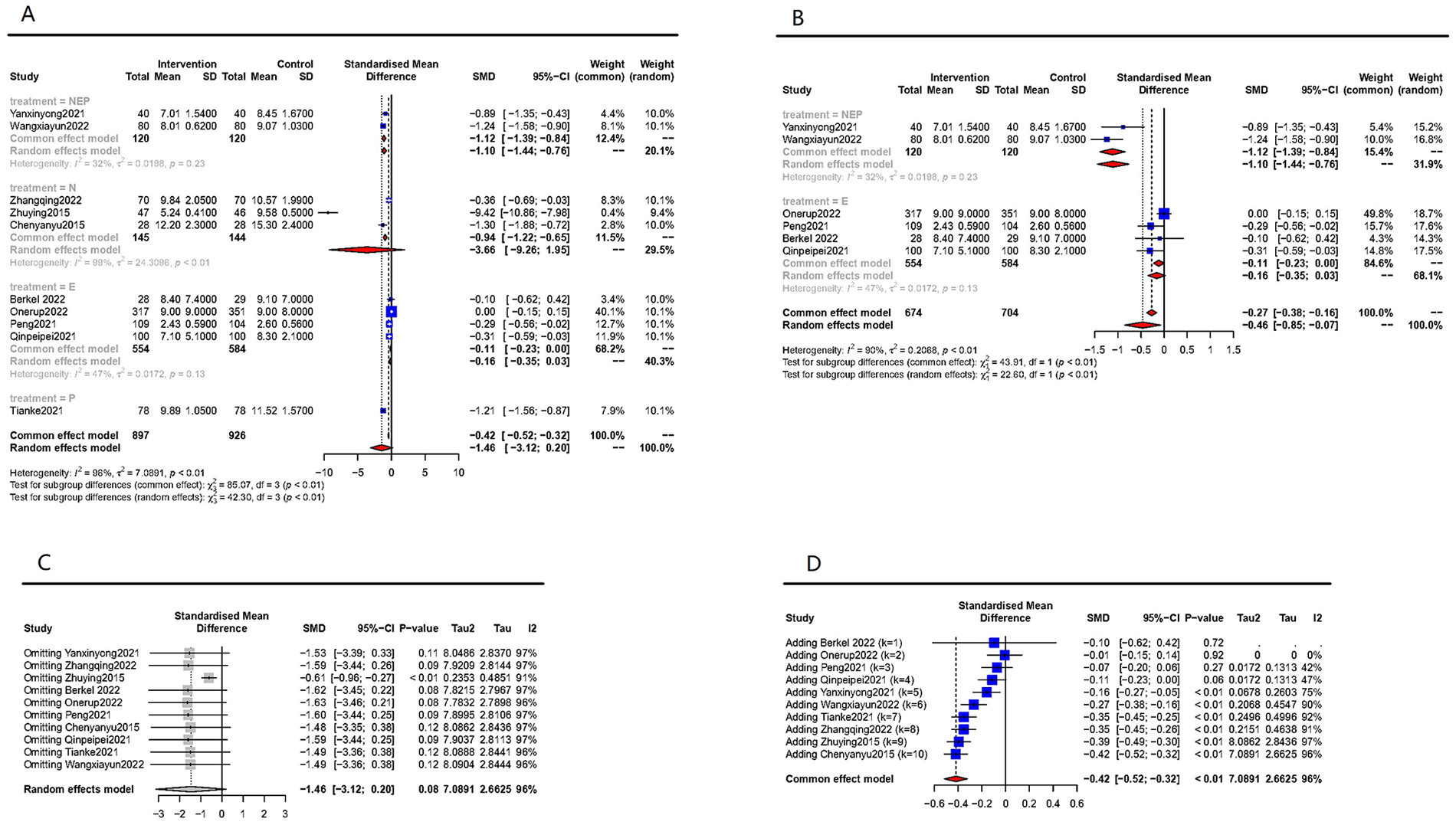
Effect of prehabilitation on postLHS. (A) pooled analysis and subgroup analysis for different intervention strategies. (B) pooled analysis excluding nutritional intervention-only trials. (C) sensitive analysis, omitting each single study, random-effect model. (D) sensitive analysis, adding each single study, fixed-effect model.
Total length of hospital stay
Total length of hospital stay (tLHS) was reported in 8 studies. Of these, 3 implemented multimodal interventions (nutritional, exercise, and psychological), while 4 only used exercise as an intervention. The pooled analysis did not reveal a significant difference between the groups (SMD = −0.26, 95% CI −0.68 to 0.15, Figure 8A), but there was relatively high heterogeneity (I² = 89%). Heterogeneity analysis suggested that trials which only used exercise as an intervention appeared to be more homogenous, and a positive effect was observed in the pooled analysis of these trials (SMD = −0.29, 95% CI −0.45 to −0.13, Figure 8A). Sensitivity analysis did not identify a single study that could account for the heterogeneity (Figure 8B and C). In this analysis there was no significant publication bias on Egger test (P = .9313). Sensitivity analysis was performed and demonstrated that one study might have been highly influential, suggesting that this result may be an outlier. 41

Effect of prehabilitation on total length of hospital stay. (A) pooled analysis and subgroup analysis of different treatment types. (B) sensitive analysis, omitting each single study, random-effect model. (C) sensitive analysis, adding each single study, fixed-effect model.
Discussion
This systematic review assembled and consolidated the current body of evidence regarding the influence of prehabilitation or preoperative lifestyle interventions on postoperative performance in colorectal cancer patients. On the whole, the postoperative physical capability, as measured by the 6WMT, as well as the incidence of complications, could be effectively lessened by these interventions. However, we did not observe a significant improvement in LHS and QoL, primarily due to the limited sample size and significant heterogeneity.
Prior to the introduction of the concept of prehabilitation, 10 there were already studies that implemented preoperative interventions and observed positive outcomes, such as the use of nutritional strategies to prevent postoperative malnutrition.24,26,46 However, these studies primarily adopted a robust nurse- or practitioner-led approach and predominantly utilized enteral nutrition as opposed to comprehensive nutritional counseling and food intake recording. The heterogeneity observed may be due to these early studies not adopting prehabilitation as their underlying rationale.
Another prominent source of heterogeneity was the type of treatment. In our analysis, we classified the studies into multimodal groups (comprising nutritional, exercise, and psychological interventions) and groups that only had nutritional, exercise, or psychological interventions. Most of the time, subgroup analysis indicated a significant difference between groups. However, the multimodal group did not appear to be more effective than the groups with a single intervention, especially when compared to the nutritional group. A likely reason inferred from the methods of intervention in the trials was that the nutritional interventions in the latter group were generally more rigorous than those in the multimodal ones. Most included prehabilitation trials employed nutritional counseling as well as exercise advice, while the trials with only one intervention used supervised exercise or enteral nutrition. However, the subgroup analysis did not suggest that the difference was clinically significant. We could not find data on patient compliance with different intervention strategies in the studies, but the appropriateness of the intensity and duration of the intervention should be considered in clinical practice.
In addition to the different combinations of lifestyle strategies, the duration of the intervention and the specific intervention plan (such as a menu or exercise plan) also contributed to the heterogeneity. However, we were unable to explain this via subgroup analysis using the currently available data, because the reporting quality of the included studies was relatively low: some participant characteristics as well as detailed intervention methods were not reported. Nevertheless, some individual studies appeared to be very different in their intervention methods. Also, the duration of the intervention varied greatly, ranging from 1 day to 9 weeks, and the timepoints at which postoperative measurements were recorded also differed. All of these factors could make the outcomes more difficult to interpret and therefore need to be applied with caution.
In our pooled analysis, both the 6-Minute Walk Test (6WMT) results and the incidence of complications were found to be significantly improved in prehabilitation groups, with relatively large effect sizes. The 6WMT is commonly used to assess the physical functional capacity of patients undergoing surgery or those with chronic diseases. 49 Our effect size was similar to that reported in an earlier Cochrane systematic review involving patients with colorectal cancer, which included 3 trials. 50 The 6WMT results of patients with other types of cancer who underwent surgery also improved with prehabilitation. 51 Patients who engaged in pre-operative exercises, such as muscle strength training, endurance training, etc., improved their physical fitness before surgery and therefore recovered faster after surgery.52 -54
The incidence of postoperative complications was also significantly reduced in the prehabilitation groups. However, this should be interpreted with caution, as the definitions and recording procedures for complications varied across different trials. Open-label strategies without blinding could also contribute to bias. Notably, previous studies have reported that exercise under preoperative supervision for abdominal surgery did not reduce the incidence of postoperative complications, 55 and triple prehabilitation did not significantly reduce postoperative mortality and complications in elderly patients undergoing surgery. 56
Postoperative and total Length of Hospital Stay (LHS) were found to be similar between the prehabilitation and control groups, which is consistent with some other publications. 50 However, this contradicts a systematic review published in 2018 that included older trials and cohorts. 57 Perioperative management strategies could be one of the important factors influencing LHS. Some hospitals have a fixed-period hospital stay plan, for example, patients are admitted 2 days before surgery and discharged 1 week postoperatively, regardless of the type of preoperative management. Researcher bias should also be considered in this context.
Although we performed a pooled analysis on Quality of Life (QoL), there were only 4 trials included and these demonstrated significant heterogeneity. Therefore, the effectiveness of prehabilitation on postoperative QoL remains to be explored. Evidence on QoL is also mixed in patients with other diseases undergoing surgeries.58 -60 Researchers continue to design new prehabilitation models for better QoL outcomes, 60 and a standardized prehabilitation intervention may contribute to this.
Limitations
This systematic review has several limitations. First, the heterogeneity of the included studies remains a significant barrier when interpreting the results. Subgroup and sensitivity analyses were performed to analyze probable sources of heterogeneity; however, more detailed participant and intervention information were not reported by the studies, making it impossible to identify all sources of variability. Furthermore, by including some articles published in Chinese journals, our results can more objectively reflect the outcome measures. However, the inclusion of certain Chinese articles with lower quality may also have an impact on our results. Second, reporting bias, including publication bias and selective outcome reporting, may impact the results. The Egger test and funnel plots indicated that publication bias existed for specific outcomes. Furthermore, a few trials were excluded because they did not report specific outcomes, and there was no response from the investigators when we attempted to contact them for this information. Third, meta-regression and statistical adjustment of the effect sizes were not conducted due to insufficient participant characteristic data reported in the studies. This means that potential confounding variables may not have been adequately controlled for in the analysis, which could influence the overall results and conclusions.
Conclusion
This investigation delineates the potency of prehabilitation, or the management of preoperative lifestyle, in patients diagnosed with colorectal cancer. Collectively, prehabilitation can enhance patients’ physical prowess post-surgery and mitigate complication rates. However, the impact on other metrics of functional capability warrants further exploration. Rigorously designed and methodologically robust Randomized Controlled Trials (RCTs) remain a necessity to quantify the influence of prehabilitation. A uniform prehabilitation protocol could be employed by healthcare professionals to bolster the prognosis of patients with colorectal cancer.
Footnotes
Acknowledgements
The authors would like to express deeply grateful to each of the researchers involved in the study, and anyone else who helped in this study.
Author Contributions
All authors have agreed on the final version and meet at least one of the following criteria (recommended by the ICMJE [*![]() ]): substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; drafting the article or revising it critically for important intellectual content.
]): substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; drafting the article or revising it critically for important intellectual content.
The author contribution box as follows:

| Criteria | Author Initials |
|---|---|
| Made substantial contributions to study design, acquisition of data, or analysis and interpretation of data; | JiaJun Zhang, YuRu Hu, HuiLi Deng |
| Involved in drafting the manuscript or revising it critically for important intellectual content; | JiaJun Zhang, YuRu Qu Shen |
| Given final approval of the version to be published. | JiaJun Zhang, YuRu Qu Shen |
| Agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. | JiaJun Zhang, YuRu Hu, HuiLi Deng, ZhongMou Huang, JianMei Huang, Qu Shen |
Data availability statement
All data used in this work are from published materials.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Humanities and Social Sciences Research Planning Fund of the Ministry of Education (21YJAZH071) and XMU Training Program of Innovation and Enterpreneurship for Undergraduates (2023 × 645).
Ethical Statement
This study was approved by the ethics committee of School of Medicine, Xiamen University on September 14 of 2022. (XDYX202209K22).
Informed consents
This systematic review does not need ethical approval or informed consents from participants.
