Abstract
Background:
Soothing the liver (called Shu Gan Jie Yu in Chinese, SGJY) is a significant therapeutic method for breast cancer in TCM. In this study, 3 liver-soothing herbs, including Cyperus rotundus L., Citrus medica L. var. sarcodactylis Swingle and Rosa rugosa Thunb. were selected and combined to form a SGJY herbal combinatory.
The Aim of the Study:
To investigate the inhibiting effect of SGJY on breast cancer in vivo and vitro, and to explore the potential mechanisms.
Materials and Methods:
SGJY herbal combination was extracted using water. A breast cancer rat model was developed by chemical DMBA by gavage, then treated with SGJY for 11 weeks. The tumor tissue was preserved for RNA sequencing and analyzed by IPA software. The inhibition effects of SGJY on MCF-7 and T47D breast cancer cells were investigated by SRB assay and cell apoptosis analysis, and the protein expression levels of SNCG, ER-α, p-AKT and p-ERK were measured by western blotting.
Results:
SGJY significantly reduced the tumor weight and volume, and the level of estradiol in serum. The results of IPA analysis reveal SGJY upregulated 7 canonical pathways and downregulated 16 canonical pathways. Estrogen receptor signaling was the key canonical pathway with 9 genes downregulated. The results of upstream regulator analysis reveal beta-estradiol was the central target; the upstream regulator network scheme showed that 86 genes could affect the expression of the beta-estradiol, including SNCG, CCL21 and MB. Additionally, SGJY was verified to significantly alter the expression of SNCG mRNA, CCL21 mRNA and MB mRNA which was consistent with the data of RNA-Seq. The inhibition effects of SGJY exhibited a dose-dependent response. The apoptosis rates of MCF7 and T47D cells were upregulated. The protein expression of SNCG, ER-α, p-AKT and p-ERK were all significantly decreased by SGJY on MCF-7 and T47D cells.
Conclusion:
The results demonstrate that SGJY may inhibit the growth of breast cancer. The mechanism might involve downregulating the level of serum estradiol, and suppressing the protein expression in the SNCG/ER-α/AKT-ERK pathway.
Background
Breast cancer is the most common type of cancer among women, accounting for about 31% of all female cancer cases.1,2 Among all cancer types, the mortality rate of breast cancer is the fifth highest, but ranks first in several undeveloped or underdeveloped countries. As such, breast cancer is regarded as one of the most critical social health problems around the world. 3
As a traditional therapy for depression, meta-analysis evidence has suggested that soothing liver-qi stagnation is effective when clinically applied for cancer-related depression. 4 In TCM, the general belief is that liver-qi stagnation is closely related to the occurrence and development of breast cancer. 5 The results of a previous study by the present authors revealed that women who have been experiencing low life satisfaction or had experienced low life satisfaction are more prone to a Qi-depressed constitution than those with a high life satisfaction, indicating that negative emotion is indeed a precipitating factor for Qi-depressed constitution. 6 Thus, soothing the liver is a significant therapeutic method for breast cancer, and is commonly adopted in clinical practice. 7 Previous investigations by the present authors showed that almost all TCM practitioners in China had taken soothing the liver as the main therapeutic method for breast cancer, 5 and the frequency of Chinese medicines with the function of soothing the liver was the highest in all Chinese medicines used for the treatment of breast cancer. 8 Jia-Wei-Xiao-Yao-San (JWXYS), which is a famous recipe with soothing liver-qi stagnation, was found to decrease LC3-II protein expression when taken in a low dose in combination with tamoxifen; and patients with estrogen receptor-α(+) or progesterone receptor(+) breast cancers had a good outcome. 9 In the present study, 3 liver-soothing herbs (SGJY), including Cyperus rotundus L., Citrus medica L. var. sarcodactylis Swingle and Rosa rugosa Thunb. were selected and combined as a working model representing the liver-soothing therapeutic method to treat breast cancer. For instance, Cyperus rotundus exhibits the anticancer activity on MCF-7 and MDA-MB-231 breast cancer cells,10,11 α-Cyperone was an active natural terpenoid in Cyperus rotundus water extraction that had inhibitory effects on tumor-derived DNA trigger microglia by STING pathway.12,13 Citrus medica var. sarcodactylis has antimicrobial, antioxidant and immunosuppressive activities and main effective components are flavonoids,14 -17 hesperidin was one of the most bioactive ingredient from flavonoids which showed apoptosis induction in breast cancer cell lines, 18 and a mechanism via the Estrogen Receptor/Mitochondrial Pathway. 19 Gallic acid and quercetin were main active ingredients extracted from Rosa rugosa Thunb. and had been reported antioxidant and anti-cancer effect.20 -22 So we used hesperidin,α-cyperone, gallic acid and quercetin as the 4 main effective components in SGJY herbal combination. The aim of the present study was to evaluate the antitumor effect of the SGJY on DMBA(7,12-Dimethylbenz[a]anthracene)-induced breast cancer in female rats, and the inhibition effect on breast cancer cells. Further, the mechanism was identified by means of RNA-Seq technology and Qiagen Ingenuity Pathway Analysis (IPA), and the related SNCG/ER-a/AKT-ERK signal pathway was evaluated.
Methods
Chemicals and Reagents
Female Sprague Dawley rats (6 weeks old) were provided by Hunan Slack Jingda Experimental Animal Technology Co., Ltd. (Certificate No. is SCXK (Xiang) 2013-0004, Changsha, China). All animal studies were carried out in SPF laboratory of the Department of Laboratory Animal, Jiangxi University of Chinese Medicine. Tamoxifen (Yangzijiang Pharmaceutical Group Co., Ltd., lot number:16041211). Citrus medica L. var. sarcodactylis Swingle (originating Guangxi province, China, batch number 160301), Rose rugosa Thunb. (originating Shandong province, China, batch number 160301) and Cyperus rotundus L. (originating Henan province, China, batch number 160301) were purchased from Hubei Daodi Medicinal Materials Technology Co., Ltd. Gallic acid, hesperidin, quercetin, and α-cyperone were purchased from Si Chuan Wei Ke Qi Biotech Co., Ltd. (Chengdu, China). Human breast cancer cell lines MCF-7 and T47D were purchased from Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences (Shanghai, China). MCF-7 cell culture medium (#CM-0149) was purchased from Procell Life Science & Technology Co., Ltd. (Wuhan, China). DMBA (#D3254) was purchased from Merck Life Science Co.,Ltd. (Shanghai, China) ). Rat estradiol Elisa kit (#) was purchased from Cusabio Bioengineering Co., Ltd (Wuhan, China). Total RNA Extraction Kit (#LS1040) was purchased from Promega(Beijing) Biotech Co., Ltd (Beijing, China). PrimeScript™ RT reagent Kit with gDNA Eraser (#RR470A) and SYBR® Premix Ex Taq™ II kit (#RR820A) were purchased from Takara Biomedical Technology(Beijing) Co., Ltd (Beijing, China); BCA protein assay kit (#23225) was purchased from Thermo Fisher Scientific Inc. (Santa Clara, United States). Annexin V-FITC/PI apoptosis kit (#AT101) was purchased from Hangzhou Lianke Biotechnology Co., Ltd (Hangzhou, China). SNCG antibody (#ab52633) and Estrogen Receptor alpha antibody (#ab32063) were purchased from Abcam (United States). Ak t(#4685S), p-Akt (S473) (#4058S), Erk1/2 (#4695T). and p44/42Erk1/2 (#4370T) antibody were purchased from Cell Signaling Technology, Inc. (Beverly, MA, USA). β-actin antibody (#20536-1-AP) was purchased from Proteintech Group, Inc (Wuhan,China).
SGJY Water Extraction Preparation
SGJY is composed of 3 traditional Chinese herbs: Cyperus rotundus, Citrus medica var. sarcodactylis, and Rosa rugosa. The dosage ratio of 3 herbs is 1:1:1. About 1.8 kg of resultant mixture was extracted (refluxed) with 10 times (18 000 mL) water solution at 95℃ for 2 h; the mixture was filtered. Subsequently, the residual was extracted with 8 times volume water solution for a second time at 95℃ for 1 h. The 2 filtrations were concentrated under reduced pressure at approximately 60℃. Finally, the concentrate was lyophilized at −80℃ to obtain a powder. The yield was approximately 35%. Moreover, the content of SGJY in this extract was approximately 0.35 g per 1 g herbal mixture. HPLC analysis of 4 kinds of active ingredients and the validation of the HPLC Method is shown in Supplemental Material 1 and 2.
Animals and Experimental Design
Female Sprague Dawley rats were housed in standard plastic cages and maintained under a 12-hour light-dark cycle at 22 ± 2°C, with free access to water and standard rodent diet. The experimental protocol was approved by the Animal Experimental Ethics Committee of the Jiangxi University of Chinese Medicine (JZLLSC2018023).
Seventy female rats were randomly divided into five groups: Group I (control, n = 12), orally received 1 mL sesame oil; Group II (DMBA, n = 20), administered intragastrically DMBA solution (20 mg/mL in sesame oil, 80 mg/kg for rats with one time,) to induce breast cancer; Group III (DMBA+TAM, 1.9mg·kg-1, n = 16); Group IV (DMBA+H-SGJY, 12.96 g/kg/d, n = 16); Group V (DMBA+M-SGJY, 6.48 g/kg/d, n = 16) ; Group VI (DMBA+L-SGJY, 3.24 g/kg/d, n = 16). According to the China Pharmacopeia 2020 Edition, the human dosage of each herb is 6 g for human use. The body surface area conversion algorithm was used to calculate the dosage for the rat. The dosage of 3.24, 6.48, and 12.96 g/kg/d were equivalent to 2, 4, 8 times of human consumption. one gram DMBA was dissolved in 50 mL sesame oil (concentration is 20 mg/mL), and a single dose of 80 mg/kg body weight was administered orally. After administering DMBA, all rats were administered SGJY or normal saline. Rats were checked for the tumor appearance weekly starting from the fourth week, and measured for body weight weekly. The tumor volume was calculated using the standard formula: width2 × length × 0.5. All the treatment was given each day and lasted for 11 weeks. The tumor tissue was cut and kept in RNA preservation solution. In this experiment, the tissue was from Group II and Group IV and was analyzed by programed RNA-SEQ, with the result validated by RT-PCR.
Analysis of Estradiol in Serum by Enzyme-Linked Immunosorbent Assay (ELISA)
The concentrations of estradiol in serum was measured by rat estradiol ELISA kits according to the manufacturer’s instruction. Briefly, biotinylated antibody reagent was added to 96-well plates, to which was added serum 50 μL, HRP-conjugate mixed solution 50 μL to each well of the plates, incubated at 37°C for 1 h. Plates were washed by filling each well with Wash Buffer (200 μL) 3 times. Then 50 μL of Substrate A and 50 μL of Substrate B was added to each well, mixed well and incubated for 15 minutes at 37°C. After adding Stop Solution 50 μL to each well, we used a microplate reader set to 450 nm to determine the optical density of each well within 10 minutes.
RNA-Seq Technology
Two hundred milligram cancer tissue was added to 10 times lysis solution and extracted by EastepTM Super Total RNA Extraction Kit. The integrity of the RNA sample was measured by the Agilent Bioanalyzer 2100 system (Agilent Technologies, CA, USA), and was between 1.8 and 2.1. The BGISEQ-500 platform (BGI, Shenzhen, China) was programed for the RNA-sequence analysis. Gene expression levels were quantified by Expectation Maximization (RSEM). 23 Differentially expressed genes (DEGs) are defined with FDR ≤ 0.001 and fold change ≥1. The cutoffs of probability ≥ .6 and |log2 ratio| >=0.58(ratio = 1.5) were applied to select DEGs.
Ingenuity Pathway Analysis (IPA) for Core Analysis, Upstream Analysis and Interaction Network Analysis
Ingenuity Pathway Analysis (IPA, http://www.ingenuity.com; Qiagen) is a useful web-based bioinformatics tool. The DEGs excel file is uploaded to IPA and then the core analysis is run. IPA uses a right-tailed Fisher’s Exact test to define significance. Upstream analysis used the number of differentially expressed genes to predict upstream regulators of the biomarker genes. The Z-score was used to evaluate changes among treatment groups.
Validation SNCG, CCL21, and MB by Real-Time PCR
Genes were selected based on the DEGs results. One hundred milligram cancer tissue was added 10 times lysis solution and extracted by the EastepTM Super Total RNA Extraction Kit. The integrity of the RNA sample was measured by the Agilent Bioanalyzer 2100 system. One microgram total RNA was used to synthesize First-stand cDNA by the PrimeScriptRT reagent Kit with gDNA Eraser following the manufacturer’s instructions. In the first step, genomic DNA contamination was eliminated with gDNA Eraser for 2 minutes at 42°C. The subsequent reverse transcription reaction was: PrimeScript RT Enzyme Mix I:1.0 μL; RT Primer Mix*4 : 1.0 μL; 5×PrimeScript Buffer 2: 4.0 μL; RNase Free dH2O: 4.0 μL. The reaction was set on 37℃ for 15 minutes and then changed into 85℃ for 5 seconds. Primers and templates were mixed with TB Green Premix Ex Taq II,using the following primers (all primers listed in the 5′to 3′orientation): Synuclein-gamma (SNCG) : GGAGGCCAAAGAGCAAGAGGA; AGCGTCTGGAAGGTGATCCGAA; Chemokine ligand 21 (CCL21): GAGCCTAGGTTGCCCCATCC; GCCGCTCCGCCATAGAAGA. The muscle-associated respiratory protein myoglobin (MB): GACAACATGCTGCTGAGATCCA; TTGACCGGGATCTTGTGCTT. β-actin: GAAACTACCTCAACTCCATC; CGAGGCCAGGAGGAGCCGCC. The data analyzed by 2 –ΔΔCt method and β-actin was utilized as the reference gene.
Cell Culture
Human breast cancer cell lines MCF-7 and T47D are both estrogen-dependent human breast cancer cell lines. T47D cells were cultured in RPMI 1640 medium supplemented with 10% fetal bovine serum (FBS), penicillin (100 U/mL), and bovine insulin (0.2 U/mL) at 37°C with 5% CO2. MCF7 cells were cultured in MEM medium supplemented with bovine serum (FBS), penicillin (100 U/mL), and bovine insulin (0.01 mg/mL) at 37°C with 5% CO2.
The Inhibitory Effect of SGJY on the Proliferation of Breast Cancer Cells by the Sulphorhodamine (SRB) Assay
The 2 cell lines were trypsinized, and the density was provided at 5×103cells/well for testing in 96 plates. After 24 h, 100 μL various concentrations of extracts (1.25, 2.5, 5, 10, and 20 mg/mL) was added and incubated for 24 h, then 10% trichloroacetic acid (50 μL) was added to the wells gradually. The plates were incubated at 4°C (1 h) and then washed 3 times with tap water, and air-dried. The dried plates were stained with SRB (100 μL) and kept for 30 minutes at room temperature in the dark. The unbound dye was washed 3 times with 10% acetic acid. One hundred microliter of 10 mM Tris base buffer was added to the wells for solubilizing the dye. The absorbance was measured by a microplate reader (Biorad Instruments, Germany) at 570 nm and the percentage of inhibition was calculated.24,25
Apoptosis Analysis by the Flow Cytometer
Approximately 8 × 105 /plates T47D and MCF7 cells were seeded in 60 mm plates, after 24 h, cells were adherent to the wall. The culture medium was removed and 2 mL SGJY (1.25, 2.5, 5 mg/mL)-containing culture medium was added for another 24 h. Subsequently the cells were harvested with 0.25% Trypsin-EDTA, collected (including cells from culture supernatant), centrifuged and washed with pre-cooled PBS, then diluted with 5 times binding buffer and 1 times working fluid, taking 500 μL resuspended cells. The resuspended cells were added to 5 μL Annexin V-FITC and 10 μ PI, and then waited for 15 minutes to program by flow cytometer.
The Effect on Protein Level Was Determined With Western Blot
Approximately 2 × 106 /plates T47D and MCF7 cells were seeded in 100 mm plates and incubated for 24 h. We then removed the original culture medium and added 4 mL SGJY (1.25, 2.5, 5 mg/mL)-containing culture medium for another 24 h. Then the cells were harvested with 300 μL RIPA Lysis Buffer (Beyotime, Shanghai, China), samples (50 μg protein/lane) were fractionated by SDS-PAGE and then transferred to polyvinylidene fluoride (PVDF) membranes by the wet blotting method. The membranes were blocked with 5% milk in Tris-buffered saline Tween20 (TBST) for 1 h at room temperature, then incubated with primary antibodies (SNCG 1:1000, ER-α 1:1000, AKT 1:1000, p-AKT 1:1000, ERK 1:1000, p-ERK 1:1000, β-actin 1:1000) at 4°C overnight, subsequently incubated with the horse-radish peroxidase-conjugated goat anti-rabbit secondary antibody (dilution 1:5000) for 1 h at room temperature. Protein bands were then detected using enhanced chemiluminescence Western blot detection reagents (Thermo Fisher Scientific, MA, USA).
Statistical Analysis
Statistical analyses were performed using SPSS 22.0. The differences between values were evaluated using one-way analysis of variance (ANOVA) followed by pairwise comparison with the Student–Newman–Keuls test. For the occurrence rate, the Chi-squared test was used. Statistical significance was accepted at a value of P < .05. Data were presented as mean ± standard deviation (SD). Graph pad Prism 8.0 software was used to make the corresponding figures.
Results
Effect of the SGJY on DMBA-Induced Breast Carcinogenesis in Rats
Table 1 showed the results of treatment with SGJY for DMBA-induced breast cancer in rats. An observation can be made that 60% to 75% of the rats in the group that received DMBA developed carcinogenesis. Regarding treatment with TAM (tamoxifen), tumors were found in only 2 rats lasting for 11 weeks, 10 rats didn’t appear to bear the tumor when they died at week 6 to 7, another 2 rats had tumor but also died at week 10, so the mortality rate was higher than the other group. It is noted that compared with the group of DMBA alone, the mortality in the group with TAM increased. Compared with the group with TAM, the mortality of the group with SGJY is significantly lower, indicating that SGJY at least did not increase the toxicity of DMBA. From this point of view, SGJY has higher safety than TAM.
The effect of SGJY on DMBA-induced breast carcinogenesis in rats.

The data represent the mean ± SD (n = 6 or 7 per group). Chi-squared test analysis.
P < .01. **P < .01 ***P < .001, compared with DMBA-induced breast cancer model group.
The body weights of TAM treatment decreased the body weights in the rats from Week 2 to Week 11, and was statistically significant when compared with the control group (P < .001) (Figure 1A). There was no significant difference in final body weight between the SGJY group and the Control group at the end of the experiment, indicating that SGJY could strengthen the body and increase the body weight of rats with DMBA-induced breast cancer.
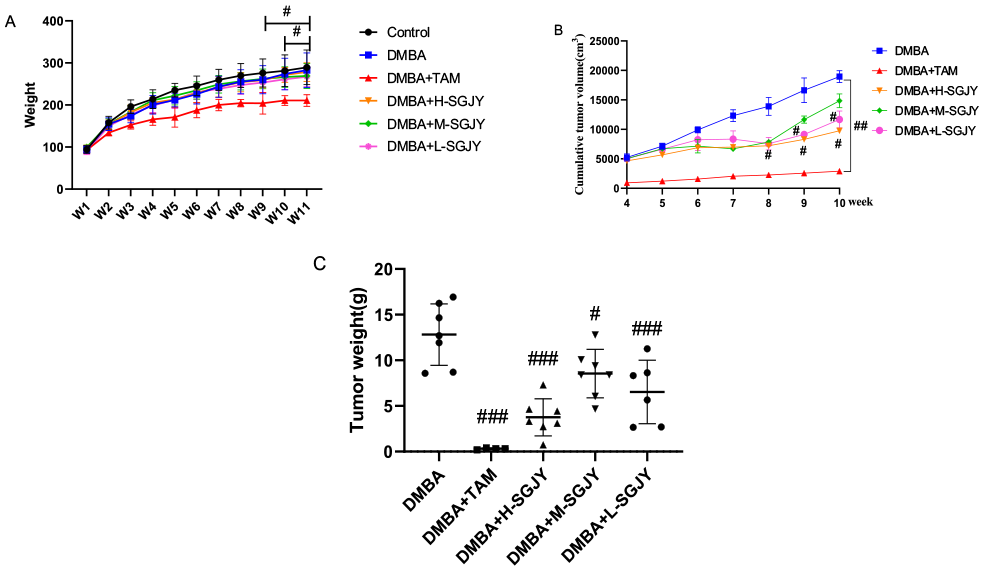
Tumor weight and cumulative tumor volume after treatment with SGJY on DMBA-induced breast carcinogenesis in rats. The data represent the mean ± SD (n = 7 per group). One-way ANOVA analysis. (A) The changes of weight in all groups. (B) Cumulative tumor volume in all groups. (C) Tumor weight of all groups.
To investigate whether SGJY had an effect in inhibiting the development of DMBA-induced breast cancer model, Figure 1B shows the cumulative tumor volumes during 4 to 10 weeks. Compared with the DMBA group, the H-SGJY group had statistical significance during W8-W10 (P < .05); the L-SGJY group had statistical significance during W9-W10 (P < .05). The tumor weights all exhibited significant decreases, and were 4.252 ± 1.527 g (F = 3.373, P < .001), 8.956 ± 2.42 g (F = 0.826, P < .05) and 6.54 ± 3.175 g (F = 0.224, p<.01) respectively (Figure 1C). The data show that the H-SGJY group had the highest inhibition effect on DMBA-induced breast cancer.
The Effect of SGJY on Estradiol Level by ELISA
Because DMBA-induced breast cancer is an estrogen-dependent mammary adenocarcinoma, the level of estradiol was the important parameters in the model.26,27 Figure 2. reveals the level of estradiol was 455.05 ± 81.58 pg/mL in the control group; it was sharply increased in the DMBA-induced model group (1179.11 ± 251.14 pg/mL) which had a significant difference (P < .001). After treatment of SGJY at doses of 12.96, 6.48 and 3.24 g/kg/d for 11 weeks, as Figure 2. shows the levels of estradiol were decreased in all 3 groups, at 572.93 ± 102.46, 620.47 ± 98.79 and 541.45 ± 174.13 pg/mL respectively and had significant changes when compared with the DMBA model group (P < .001). These data indicated SGJY decreased the level of estradiol in DMBA-induced breast cancer model.

The levels of estradiol in serum by ELISA. The data represent the mean ± SD (n = 7 per group). One-way ANOVA analysis. ***P < .001, compared with the control group; ###p < 0.001, compared with DMBA-induced breast cancer model group.
The Effect of SGJY on Genes and Transcripts
Based on the RNA-seq data, a total of 14 499 genes were identified in the DMBA versus the SGJY groups. The cutoffs of probability ≥.6 and |log2 ratio| ≥ 0.58(ratio = 1.5) were applied to select DEGs. The number of differentially expressed genes were 225; 62 genes were upregulated and 163 genes were downregulated.
Ingenuity Pathway Analysis (IPA) for Core Analysis
Ingenuity Pathway Analysis, a web-based bioinformatics tool, was used for further pathway analysis. Z-score ≤ 2 & Z-score ≥ 2 was considered meaningful. 28 Figure 3 illustrates the top canonical pathways including 7 increased by SGJY (Systemic Lupus Erythematosus In T Cell Signaling Pathway, T Cell Receptor Signaling, Role of NFAT in Regulation of the Immune Response, ICOS-ICOSL Signaling in T Helper Cells, PKCθ Signaling in T Lymphocytes, Dilated Cardiomyopathy Signaling Pathway, Neuroinflammation Signaling Pathway), and 16 decreased by SGJY(Apelin Liver Signaling Pathway, Intrinsic Prothrombin Activation Pathway, Apelin Cardiomyocyte Signaling Pathway, Factors Promoting Cardiogenesis in Vertebrates, Colorectal Cancer Metastasis Signaling, Gustation Pathway, IL-6 Signaling, MSP-RON Signaling In Macrophages Pathway, p38 MAPK Signaling, GP6 Signaling Pathway, Cardiac Hypertrophy Signaling, Actin Cytoskeleton Signaling, HOTAIR Regulatory Pathway, Estrogen Receptor Signaling, Tumor Microenvironment Pathway, Hepatic Fibrosis Signaling Pathway) (Supplemental Material 3).

Canonical pathways analysis of SGJY in DMBA-induced breast cancer by IPA (Z-score ≤ 2 & Z-score ≥ 2 was considered meaningful).
Upstream Regulator Analysis by IPA
The IPA Upstream Regulator Analysis was assessed to identify the upstream regulators that may be responsible for gene expression changes in the study. IPA predicts activated or inhibited upstream regulators to explain which is the upregulated or downregulated genes. 29 There were more altered regulators decreased by SGJY (z score < −2), they were 4 transmembrane receptors (IGF1R, IL6R, SEMA7A, CD36); with 22 transcription regulators (MEF2C, MYOD1, SMARCA4, CEBPB, JUN, TEAD3, MEF2D, ZEB1, GLI1, RB1, SIRT1, EPAS1, SOX9, MYB, HMGA1, STAT6, CREBBP, CALR, TARDBP, SNAI1, TAL1, TRIM24); or with 8 chemical - endogenous mammalian (beta-estradiol, cholecalciferol, corticosterone, L-triiodothyronine, D-glucose, aldosterone, 2-methoxyestradiol, epinephrine). The data pointed out SGJY would suppress the level of beta-estradiol, and that will help for treatment of breast cancer. In addition, the upstream regulator network scheme showed 86 genes could affect the expression of beta-estradiol, including SNCG, CCL21 and MB. SNCG was further selected for functional validation in a breast cancer cell experiment. There were 3 genes connected with the Estrogen Receptor Signaling (Figure 4). There are 10 increased by SGJY with transcription regulator (CIITA, KDM5A, CBX5, EZH2, CDKN2A, CTCF, RAD21, RBPJ, EOMES and NCOA2) or with transmembrane receptor (FAS) (z score > 2).

Upstream Regulator beta-estradiol influenced by 86 genes and connected with Estrogen Receptor Signaling by IPA. SNCG, CCL21, and MB were labeled by black rectangles.
Validation of SNCG, CCL21and MB Gene Expression by Rt- PCR
As shown in Figure 5, the relative expression of genes in the RT-PCR data aligned with the RNA-seq data, suggesting that the DEGs detected from our RNA-seq analysis are valid. SGJY (6.48 and 3.24 g/kg/d) decreased the expression of SNCGmRNA (P < .01), SGJY (12.96, 6.48, and 3.24 g/kg/d) decreased the expression of MBmRNA (P < .001, P < .01, and P < .01) and increased the expression of CCL21 (P < .001, P < .01 and P < .01).
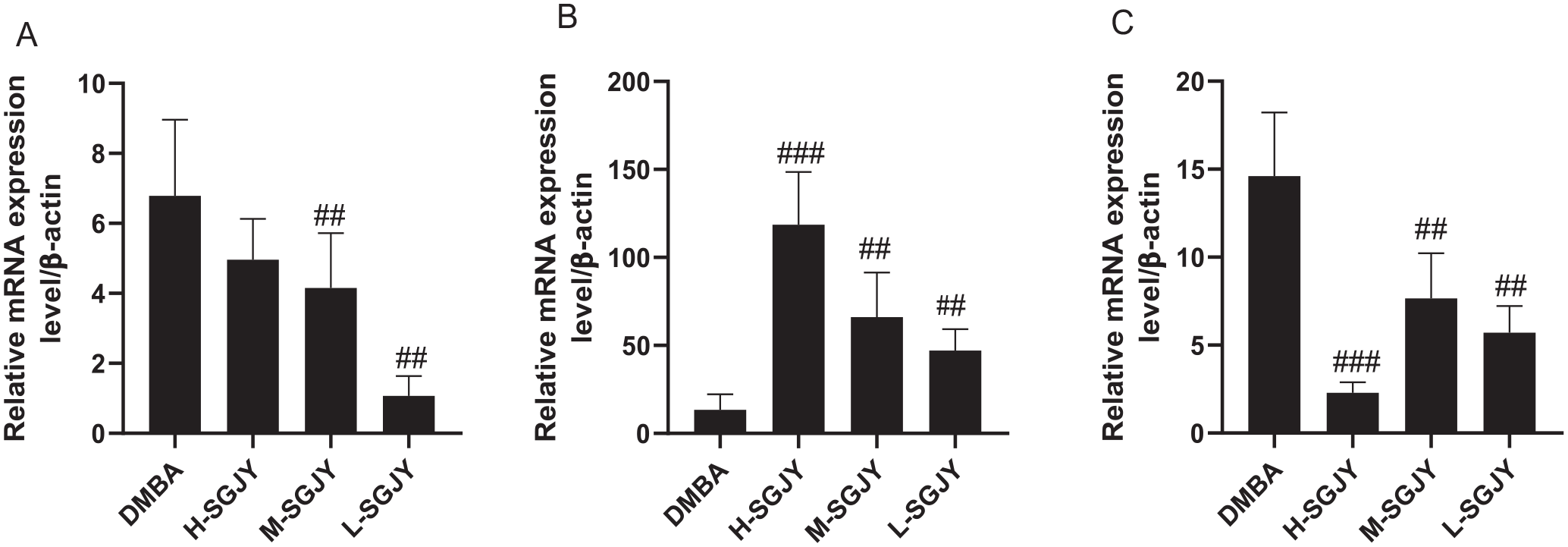
Real-time qPCR analysis of 3 selected DEGs. (A) SNCGmRNA. (B) CCL21mRNA. (C) MBmRNA; (n = 3 per group). One-way ANOVA analysis. ##P < 0.01 and ###P < 0.001 compared with DMBA-induced breast cancer model group.
The Anti-Proliferative Effect of SGJY on Breast Cancer Cell by the Sulphorhodamine (SRB) Assay
Compared to the untreated group, Figure 6 shows that SGJY at a concentration of 2.5 to 20 mg/mL inhibited the growth of 2 types of breast cancer cells. The inhibition effects of SGJY exhibited a dose-dependent response when the concentrations of SGJY were from 2.5 to 5 mg/mL on MCF7 cells and from 2.5 to 10 mg/mL on T47D cells. The proliferation rates of SGJY were between 49.7 ± 2.45% to 29.2 ± 2.44% on MCF7 cells at a concentration of 2.5 mg/mL at 24, 48, and 72 h (P < .05) after treatment; and 71.7 ± 3.4% to 48.9 ± 2.3% (P < .05) on T47D cells at a concentration of 5 mg/ mL at 24, 48, and 72 h (P < .05) after treatment. The other concentration of SGJY were the same. These data show SGJY exhibited a time-dependent manner in terms of the inhibitory effects on cell proliferation and had more effect on MCF7 cells.

Inhibitory effect of SGJY on the proliferation of breast cancer MCF7 and T47D cells. The proliferation of cells was detected by SRB assay. Inhibitory rate of breast cancer MCF7 and T47D cells treated with different concentrations of SGJY (1.25, 2.5, 5, 10, and 20 mg/mL) for 24, 48, 72 h. (n = 3 per group). One-way ANOVA analysis. *P < .05, **P < .01 and ***P < .001, compared with the control group.
Apoptosis Analysis by the Flow Cytometer
To explore the mechanism of SGJY in the inhibition of breast cancer cells, a flow cytometer was used to measure the apoptosis of MCF7 and T47D cells treated with SGJY at different concentrations (0, 1.25, 2.5, and 5 mg/mL) for 24 h. The late apoptotic rates were increased in MCF7 and T47D cells and especially in the SGJY 5 mg/mL group(P < .01). There was no significant difference in early apoptosis. That indicated SGJY had the ability to induce apoptosis (Figure 7).

SGJY induced apoptosis of MCF7 and T47D cells by flow cytometry. (A) Flow cytometry of cell apoptosis. (B) Apoptotic rate of MCF7 and T47D cells at different concentrations of SGJY. Cells treated with SGJY (0, 5, 2.5, and 1.25 mg/mL) for 24 h. Following treatment, cells were stained with Annexin V-FITC/PI and analyzed by flow cytometry. (n = 3 per group). *P < .05, **P < .01 and ***P < .001, compared with the control group.
SGJY Inhibited SNCG/ER-α/AKT-ERK Pathway Related Protein Expression
Due to the close relationship between SNCG and the activities of ER-alpha, AKT and ERK, 30 the levels of the proteins involved in the SNCG, ER-alpha and ERK /AKT pathways were assessed by means of western blot. Figure 8 shows that SNCG and ER-α were significantly decreased by SGJY at all doses in MCF-7 cells, and the phosphorylated levels of AKT and ERK were decreased by high and medium doses of SGJY. In parallel, the same trend was observed with SGJY in T47D cells. The experimental results confirm the prediction that SGJY can inhibit breast cancer cell by inhibiting key targets in the SNCG/ER-α/AKT-ERK pathway.

Changes in the expression of SNCG-related protein in SGJY-treated MCF7 and T47D cells. (A) SNCG -associated proteins, as detected by western blot analysis. β-actin was used as a reference protein. (B) Relative optical density of ER-α, SNCG, AKT, p-AKT, ERK and p-ERK proteins in MCF7 cells following treatment. (C) Relative optical density of ER-α, SNCG, AKT, p-AKT, ERK, and p-ERK proteins in T47D cells following treatment. n = 3 per group. One-way ANOVA analysis. *P < .05, **P < .01 and ***P < .001, compared with the control group.
Discussion
Since soothing the liver is a common therapeutic method for breast cancer, evaluation of the effect of such treatment on inhibiting breast cancer and exploration of the potential mechanisms are of considerable significance, which will provide a basis for the precise application of the liver-soothing method in the treatment of breast cancer, as well as promote the development of TCM. In cancer, different combinations are being tested in clinical trials, such as chemotherapeutic agents, hormonal therapies, and radiation, 31 with particular focus on the combinations of cytotoxic agents and biotherapies (such as monoclonal or polyclonal antibodies, gene therapy, vaccines, cytokine therapy). 32 Notably, the combination of treatments, as a frequent medical form, has been utilized for over 2500 years in TCM. Combined medication, rather than the use of single herbs, can appropriately reflect one therapeutic method to a large extent. 33 As an example, Aneustat (OMN54) is a mixture of extracts of Scutellaria barbata, Ganoderma lucidum and Salvia miltiorrhiza, which was developed by Omnitura Therapeutics Inc., USA, and used as a first-in-class multivalent immuno-oncology therapeutic. 34 Such treatment was demonstrated to have anticancer activity via inhibition of aerobic glycolysis. 35
In the present research, 3 common liver-soothing herbs for breast cancer treatment were selected and combined as a representative working model of the liver-soothing method. Firstly, DMBA-induced mammary tumors were selected to investigate the suppressive effect on breast cancer in vivo. With the treatment of a SGJY herbal combination at doses of 12.96, 6.48, and 3.24 g/kg/d for 11 weeks, the tumor weight and volume decreased significantly. Further, the inhibitory effect was also observed on MCF7 and T47D breast cancer cells in vitro, including inhibiting the proliferation and promoting apoptosis. The results demonstrate that the liver-soothing therapeutic method has a direct therapeutic effect on breast cancer, in addition to relieving physical discomfort caused by the illness or various treatments.
Endocrine disorders, especially hyperestrogen secretion, are closely related to the occurrence and development of breast cancer via causing the production of breast epithelial cells and fibrous tissue hyperplasia. The tumors in the present DMBA-induced breast cancer model were estrogen-dependent mammary adenocarcinomas, and the increase in estradiol level was a significant parameter. Notably, the serum estrogen levels of female patients with liver stagnation syndrome are also significantly increased, and the clinical symptoms caused by increased estrogen are generally similar to that of liver stagnation syndrome. 36 Moreover, a recent clinical study showed that Chaihu Shugan Powder with the function of soothing the liver could reduce serum estrogen levels of patients with stage III breast cancer. 37 The results of the present study are consistent with such studies. The serum estradiol levels of the rats induced by DMBA were significantly increased, whereas, all 3 different doses of SGJY could reduce the estradiol level caused by DMBA. The results suggest that decreasing the serum estradiol level could be a mechanism of SGJY inhibiting breast cancer.
RNA-Seq has been a preferred method of global gene expression analysis, and given the large scale and complexity of transcriptomes, 38 such sequencing is widely used to reveal the etiology of diseases or the therapeutic mechanism of drugs. Particularly for cancers, RNA-Seq is beneficial for identifying lineage-specific biomarker signatures for cancer types. 39 According to the present results of RNA-Seq data in vitro, SGJY could suppress the expression levels of SNCG, CCL21, and MB genes and decrease estradiol expression. The pair of C-C chemokine ligand 21/chemokine receptor 7 (CCL21/CCR7) accelerated growth and metastasis in breast cancer,40 -42, while the CCL21/CCR7 chemokine axis regulates the expression and secretion of lymphangiogenic factor VEGF-C, thereby advancing proliferation, migration and tube formation of the primary human lymphatic endothelial cell markers (LECs). 43 The muscle-associated respiratory protein myoglobin (MB) is expressed in breast tumors, 44 and the positivity thereof could be detected in up to 40% of primary breast tumors, especially in estrogen receptor (ER)-positive cases. 45 MB expression allows breast cancer cells to utilize oxygen more efficiently during hypoxia, leading to the inhibition of HIF-1α stabilization and the prevention of the hypoxic response.46 -48 At the same time, the canonical pathways were identified by means of Qiagen Ingenuity Pathway Analysis (IPA). The top canonical pathways showed SGJY downregulated estrogen receptor signaling and modulated 9 genes, which confirmed the close relationship between SGJY and estrogen receptor signaling. Potential upstream regulators detected by IPA included 4 transmembrane receptors, 22 transcription regulators and 8 endogenous mammalian chemicals, and the beta-estradiol expression was especially inhibited. The canonical pathways and upstream regulators could serve as potential avenues for elucidating the mechanisms by which the SGJY could inhibit the DMBA-induced breast cancer model. As such, an assumption was made that inhibiting SNCG and estradiol expression may be a treatment mechanism of SGJY combinations.
SNCG (γ-Synuclein gene), which is identified as breast cancer specific gene 1 (BCSG1), is upregulated in advanced breast cancers. SNCG leads to a sharp increase in cell motility and invasiveness in vitro, as well as a profound augmentation of metastasis in vivo in breast cancer, 49 SNCG siRNA could downregulate the expression of phosphorylation of AKT and ERK and then decrease the proliferation and migration in breast cancer cells,30,50 and siSNCG treatment affected MCF7 and T47D breast cancer cells, which are 2 estrogen receptor-positive breast cancer cells. 51 A recent study reported that miRNA-15a mediated the cell cycle and induced apoptosis in breast cancer cells by targeting SNCG. 52 Such results indicate that SNCG is a tumor marker in breast cancer, and treatment that downregulating SNCG may be an effective strategy in breast cancer.53,54 According to the present results of RNA-Seq data using rat tissue and the experiment in vitro, SGJY could suppress the expression of SNCG, whether at the gene level or the protein level. The present report is the first in which liver-soothing herbs have been shown to have the effect of inhibiting SNCG expression to reduce the occurrence and development of breast cancer.
The molecular targets of SNCG aberrant expression in breast cancer have been identified, indicating that SNCG is as an ER-α-associated chaperone to interact with the Hsp-ER-α complex, enhance the high-affinity ligand-binding capacity of ER-α, and stimulate ligand-dependent activation of ER-α.54,55 Further studies have indicated that SNCG protects Hsp90 client proteins such as ER alpha 36 and stimulates rapid membrane-initiated estrogen signaling in breast cancer cells. At present, ER-α is regarded as a transcription factor promoting the initiation and progression of breast cancer. 56 ER-alpha 36 is recognized as a molecular abnormality which solidly correlates to the aggressiveness, carcinogenesis, and therapeutic response of breast cancer. 57 The data of the present study show that SGJY not only induced the apoptosis, but also suppressed the protein expression levels of SNCG, ER-α, p-AKT, and p-ERK in MCF-7 and T47D cells. To the present knowledge, the present report is the first in which such herbs have been shown to exhibit inhibition effects on the aforementioned indexes. Accordingly, an assumption could be made that the inhibition of SGJY on breast cancer is also closely related with the inhibition of SNCG, ER-α, p-AKT, and p-ERK.
Notably, Chinese medicine combinations can have multiple components and multiple targets, and thus, there is uncertainty whether the suppressing effects on ER-α, p-AKT, and p-ERK is caused by the inhibition of SNCG. Thus, further research is needed to confirm whether there is a causal relationship between the inhibition of SNCG and the activities of ER-α, p-AKT, and p-ERK. Regardless, the present study is a significant reference for applying SGJY to treat breast cancer, exploring the mechanism of action, and identifying the possible material basis for the future. In addition, there is still a lack of fundamental research on SGJY in terms of pharmacodynamics, pharmacokinetics, toxicology and drug combination research.
Conclusion
SGJY exhibits chemo-preventive effects on DMBA-induced breast cancer in vivo and inhibition on the growth and the promotion of apoptosis of breast cancer cells in vitro. The mechanism might involve downregulating the level of serum estradiol, and suppressing the protein expression on the SNCG/ER-α/AKT/ERK pathway. The results indicate that SGJY might be an effective therapy for breast cancer.
Supplemental Material
sj-docx-1-ict-10.1177_15347354241233258 – Supplemental material for The Herbal Combination Shu Gan Jie Yu Regulates the SNCG/ER-a/AKT-ERK Pathway in DMBA-Induced Breast Cancer and Breast Cancer Cell Lines Based on RNA-Seq and IPA Analysis
Supplemental material, sj-docx-1-ict-10.1177_15347354241233258 for The Herbal Combination Shu Gan Jie Yu Regulates the SNCG/ER-a/AKT-ERK Pathway in DMBA-Induced Breast Cancer and Breast Cancer Cell Lines Based on RNA-Seq and IPA Analysis by Yi Zhao, Linan Zhao, Tao Wang, Zhenghao Liu, Suyuan Tang, Hongxia Huang, Li Wu and Youzhi Sun in Integrative Cancer Therapies
Supplemental Material
sj-docx-2-ict-10.1177_15347354241233258 – Supplemental material for The Herbal Combination Shu Gan Jie Yu Regulates the SNCG/ER-a/AKT-ERK Pathway in DMBA-Induced Breast Cancer and Breast Cancer Cell Lines Based on RNA-Seq and IPA Analysis
Supplemental material, sj-docx-2-ict-10.1177_15347354241233258 for The Herbal Combination Shu Gan Jie Yu Regulates the SNCG/ER-a/AKT-ERK Pathway in DMBA-Induced Breast Cancer and Breast Cancer Cell Lines Based on RNA-Seq and IPA Analysis by Yi Zhao, Linan Zhao, Tao Wang, Zhenghao Liu, Suyuan Tang, Hongxia Huang, Li Wu and Youzhi Sun in Integrative Cancer Therapies
Supplemental Material
sj-xls-3-ict-10.1177_15347354241233258 – Supplemental material for The Herbal Combination Shu Gan Jie Yu Regulates the SNCG/ER-a/AKT-ERK Pathway in DMBA-Induced Breast Cancer and Breast Cancer Cell Lines Based on RNA-Seq and IPA Analysis
Supplemental material, sj-xls-3-ict-10.1177_15347354241233258 for The Herbal Combination Shu Gan Jie Yu Regulates the SNCG/ER-a/AKT-ERK Pathway in DMBA-Induced Breast Cancer and Breast Cancer Cell Lines Based on RNA-Seq and IPA Analysis by Yi Zhao, Linan Zhao, Tao Wang, Zhenghao Liu, Suyuan Tang, Hongxia Huang, Li Wu and Youzhi Sun in Integrative Cancer Therapies
Footnotes
Acknowledgements
Not applicable.
Abbreviations
HPLC: High Performance Liquid Chromatography
DMBA: 7,12-Dimethylbenz[a]anthracene
RNA-SEQ: High-throughput mRNA sequencing
IPA: Ingenuity Pathway Analysis
DEGs: Differentially expressed genes
SNCG: γ-Synuclein gene
ER-α: Estrogen Receptor-α
p-AKT: Phosphorylated protein kinase B
p-ERK: Phosphorylated extracellular regulated protein kinases
CCL21: Chemokine ligand 21
MB: Muscle-associated respiratory protein myoglobin
SRB: Sulphorhodamine assay
Authors’ Contributions
Yi Zhao: performed the experimental study, analyzed the data, and drafted the manuscript. Youzhi Sun: designed the research and drafted the manuscript. Linan Zhao: completed the animal experiments. Tao Wang: completed the cell experiments. Zhenhao Liu: completed the HPLC experiments. Suyuan Tang: completed the cell experiments. Hongxia Huang: analyzed the data. Li Wu: analyzed the data and formal analysis. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Availability of Data and Materials
The data used to support the findings of our study are included within the article, or within the Supplementary data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Natural Science Foundation of China (82360961, 82060868), Natural Science Foundation of Jiangxi Province (20203ABC28W019), Science and Technology Research Project of Jiangxi Provincial Department of Education (GJJ2200907), Young Jing Gang Scholar of Jiangxi Province, Jiangxi University of Chinese Medicine Science and Technology Innovation Team Development Program(CXTD22007, CXTD22008).
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
