Abstract
Objective:
In China, grade 2 to 3 immune-related rash will probably lead to the interruption of immunotherapy. Corticosteroid (CS) is the main treatment, but not always effective. The external application of clearing heat and removing dampness, which is represented by Qing-Re-Li-Shi Formula (QRLSF), has been used in our hospital to treat immune-related cutaneous adverse events (ircAEs) for the last 5 years. The purpose of this study was to discuss its efficacy and safety in the treatment of grade 2 to 3 rash.
Methods:
A retrospective study of patients with grade 2 to 3 immune-related rash in our hospital from December 2019 to December 2022 was conducted. These patients received QRLSF treatment. Clinical characteristics, treatment outcome, and health-related quality of life (HrQoL) were analyzed.
Results:
Thirty patients with grade 2 to 3 rash (median onset time: 64.5 days) were included. The skin lesions of 24 cases (80%) returned to grade 1 with a median time of 8 days. The accompanying symptoms were also improved with median time of 3 to 4 days. The addition of antihistamine (AH) drug didn’t increase the efficacy of QRLSF (AH + QRLSF: 75.00% vs QRLSF: 83.33%, P = .66). No significant difference was observed in the efficacy of QRLSF treatment regardless of whether patients had previously received CS therapy (untreated population: 88.24% vs treated population: 69.23%, P = .36). During 1-month follow-up, 2 cases (8.33%) underwent relapses. In terms of HrQoL, QRLSF treatment could significantly reduce the median scores of all domains of Skindex-16, including symptoms (39.58 vs 8.33, P < .0001), emotions (58.33 vs 15.48, P < .0001), functioning (46.67 vs 13.33, P < .0001) and composite (52.60 vs 14.06, P < .0001).
Conclusion:
External application of clearing heat and removing dampness was proven to be an effective and safe treatment for such patients. In the future, high-quality trials are required to determine its clinical application in the field of ircAEs.
Keywords
Introduction
Entering the revolutionary era of cancer immunotherapy, targeting the programed cell death protein 1 (PD-1)/programed cell death ligand 1 (PD-L1) axis has achieved great success in the clinical treatment of different types of malignant tumors. 1 PD-1/PD-L1 inhibitors can enhance the activities of effective T cells and restore immune surveillance in order to exert the antitumor effect. 2 However, the mechanism of PD-1/PD-L1 blockers is closely associated with the occurrence of dermatologic immune-related adverse events (irAEs) due to the shared T cell antigen between tumor tissue and skin, and the increased level of pro-inflammatory cytokines and antibodies released by activated T and B cells.3,4 Besides, human leukocyte antigen variants are considered as potential biomarkers for risk assessment of specific irAEs (including skin toxicity). 4
Cutaneous toxicity is the most common irAE induced by PD-1/PD-L1 blockers, with the incidence of approximately 30% to 40%.5,6 Rash occurs frequently and a wide range of manifestations can present, including maculopapular rash, lichenoid dermatitis, acneiform rash, etc. 3 According to the guideline of the management of immune checkpoint inhibitor-related toxicity formulated by Chinese Society Of Clinical Oncology (CSCO), 7 management involves the use of topical or/and systemic corticosteroid (CS) for grade 2 or 3 rash. Concurrently, patients with grade 3 rash are required to withhold immunotherapy and this measure should be considered for grade 2. However, most of these recommendations are based on low level evidence. Furthermore, the efficacy is far from satisfactory. Up to 25% of rash is CS-refractory. 8 The presence of rash may be a sign of clinical benefit and interruption of the treatment may lead to disease progression. 8 Therefore, how to relieve grade 2 to 3 immune-related rash and ensure patients to continue anti-PD-1/PD-L1 therapy is extremely important.
Chinese herbal medicine (CHM) is commonly prescribed in treating and preventing antitumor therapy related skin toxicity in clinical practice, particularly in Asian countries such as China. 9 In the past 5 years, our center has developed a new method for treating immune-related cutaneous adverse events (ircAEs), which is mainly based on the theory of external treatment and the principle of clearing heat and removing dampness, as summarized in a proven recipe, (Qing-Re-Li-Shi Formula, hereinafter referred to as “QRLSF”). QRLSF is empirically formed by adding Coicis Semen (Yi yi ren) and Angelicae Dahuricae Radix (Bai zhi) to Si-Miao-Yong-An decoction (SMYAD), which has been applied externally to treat sores, carbuncles and ulcers that resemble the symptoms of ircAEs. Modern pharmacological research has shown that SMYAD can regulate the differentiation and recruitment of monocytes, macrophages and T cells, reduce the secretion of inflammatory factors and inhibit platelet aggregation and activation.10 -12 All these pharmacological activities are mainly attributed to the inhibitory effect of SMYAD on inflammatory reaction, which may be related to the pathological factor of ircAEs. Thereby, QRLSF is speculated to be a potential formula to resolve dermatologic toxicity induced by PD-1/PD-L1 inhibitors and has been proven to be highly effective in our daily clinical practice. Here, we conducted an initial retrospective study to report 30 patients with grade 2 to 3 immune-related rash, and discussed their changes in clinical features and health-related quality of life (HrQoL) to assess the efficacy and safety of external application of clearing heat and removing dampness, which was represented by QRLSF, for moderate to severe rash.
Methods
Study Design and Patients
We retrospectively analyzed cancer patients who developed grade 2 or 3 immune-related rash after receiving the therapy of PD-1/PD-L1 inhibitors and were referred to the First Affiliated Hospital of Zhejiang Chinese Medical University (Zhejiang Provincial Hospital of Chinese Medicine) for the treatment of skin lesions from December 2019 to December 2022. The diagnosis and severity of the lesions was determined according to the clinicopathologic data or the consultations of dermatologists, and the guideline of the management of immune checkpoint inhibitor-related toxicity formulated by CSCO. 7 The body surface area (BSA) of the rash was calculated in accordance with the Chinese Rule of 9. The inclusion criteria were as follows: (1) Age ≥ 18 years old; (2) Diagnosed with grade 2 or 3 PD-1/PD-L1 inhibitors-related rash; (3) Suffered from the failure of the therapeutic regimes based on guidelines or expert consensus such as CS or refused to accept such treatments; (4) Treated with QRLSF for external use. The patients who had history of immune-related skin disorders or with insufficient clinical data were excluded.
This study was prepared in line with STROBE guideline and approved by the Ethics Committee of the First Affiliated Hospital of Zhejiang Chinese Medical University (Zhejiang Provincial Hospital of Chinese Medicine) (No. 2022-KL-040-01). We obtained the written informed consent for publication of all the patients.
External Application of QRLSF
Through reference to the case history, the clinical symptoms of these cases with rash were manifested as erythema, papules, pustules, or lichen planus, red tongue with greasy fur, accompanied by pruritus, pain, edema, tightness, burning, purpura, or desquamation, etc. Guided by the theory of traditional Chinese medicine (TCM), it belonged to “drug toxicity rash,” which was characterized by “damp-heat” syndrome. Due to its effect of clearing heat and removing dampness, QRLSF was prescribed to these cases for the external treatment of rash. QRLSF was composed of 30 g Lonicerae Japonicae Flos (Jin yin hua), 30 g Scrophulariae Radix (Xuan shen), 15 g Angelicae Sinensis Radix (Dang gui), 30 g Coicis Semen (Yi yi ren), 15 g Angelicae Dahuricae Radix (Bai zhi), and 30 g Glycyrrhizae Radix et Rhizoma (Gan cao). These herbs were identified and processed by TCM preparation room of the First Affiliated Hospital of Zhejiang Chinese Medical University. In a recent study, this formula was analyzed by high-performance liquid chromatography (HPLC) for quality control. 13 The brief process was as follows: a constituent was soaked for 30 minutes and decocted twice with eightfold water for 1 h each time. The extracts were combined, filtered, and finally concentrated to 200 mL at 100°C. The final concentration of the prescription was 0.75 g/mL (raw herbs). After cooling, sterile medical non-woven fabrics were immersed in the prepared Chinese herbal decoction and gently covered the skin lesions without drips. During the treatment, the medical non-woven fabrics were always kept moist and the local skin reactions were observed. Each hydropathic compress was applied for 20 minutes, twice per day.
Data Collection and Measurements
The relevant clinical data were recorded in each patient’s medical record. The following data were collected: demographic variables, medical history, clinical manifestations, physical signs, treatment, outcome, HrQoL, and concomitant medication. An improvement of rash to grade 1 was defined as valid because PD-1/PD-L1 inhibitors could be re-challenged.
The Skindex-16 questionnaire was used to evaluate the HrQoL of the patients with rash induced by PD-1/PD-L1 inhibitors before and after QRLSF treatment. Skindex-16 contains 16 items and the score of each item is on a scale of 0 to 6, with 0 implying “never bothered” and 6 implying “always bothered.” It covers 3 domains: symptoms (4 items), emotions (7 items), and functioning scales (5 items). The score is expressed in a linear scale varying from 0 to 100. The Chinese version of the Skindex-16 questionnaire has been proven to be of great reliability and validity. 14 Higher score represents poorer quality of life (QoL).
Statistical Analysis
GraphPad Prism version 5.0 and IBM SPSS version 25.0 were used to draw graphs and analyze data. Descriptive statistics were used to summarize the data. The Wilcoxon signed rank test was used for comparison of measurement data that didn’t conform to a normal distribution. The χ2 test or Fisher’s exact test was used for comparison of enumeration data. The level of statistical significance was set at P < .05 (two-tailed).
Results
Clinical Characteristics
The clinical characteristics of the patients are shown in Table 1. Thirty eligible patients with grade 2 to 3 rash (12 at grade 2 and 18 at grade 3) were included in our analysis. The ratio of male to female was 13:2. The average age was 65.03 ± 10.06 (range: 40-83). The tumor types included lung cancer (n = 14), gastric cancer (n = 3), nasopharyngeal cancer (n = 1), hepatic cancer (n = 4), chondrosarcoma (n = 1), prostatic cancer (n = 1), renal cancer (n = 1), esophageal cancer (n = 2) and pancreatic cancer (n = 3). The types of PD-1/PD-L1 inhibitors contained atezolizumab (n = 1), camrelizumab (n = 9), pembrolizumab (n = 5), toripalimab (n = 2), tislelizumab (n = 6), and sintilimab (n = 7). Based on previous treatment, our study included the treated population who had suffered from the failure of CS regime (n = 13) and the untreated population who had refused to receive CS treatment (n = 17).
The Clinical Characteristics of the Patients (n = 30).
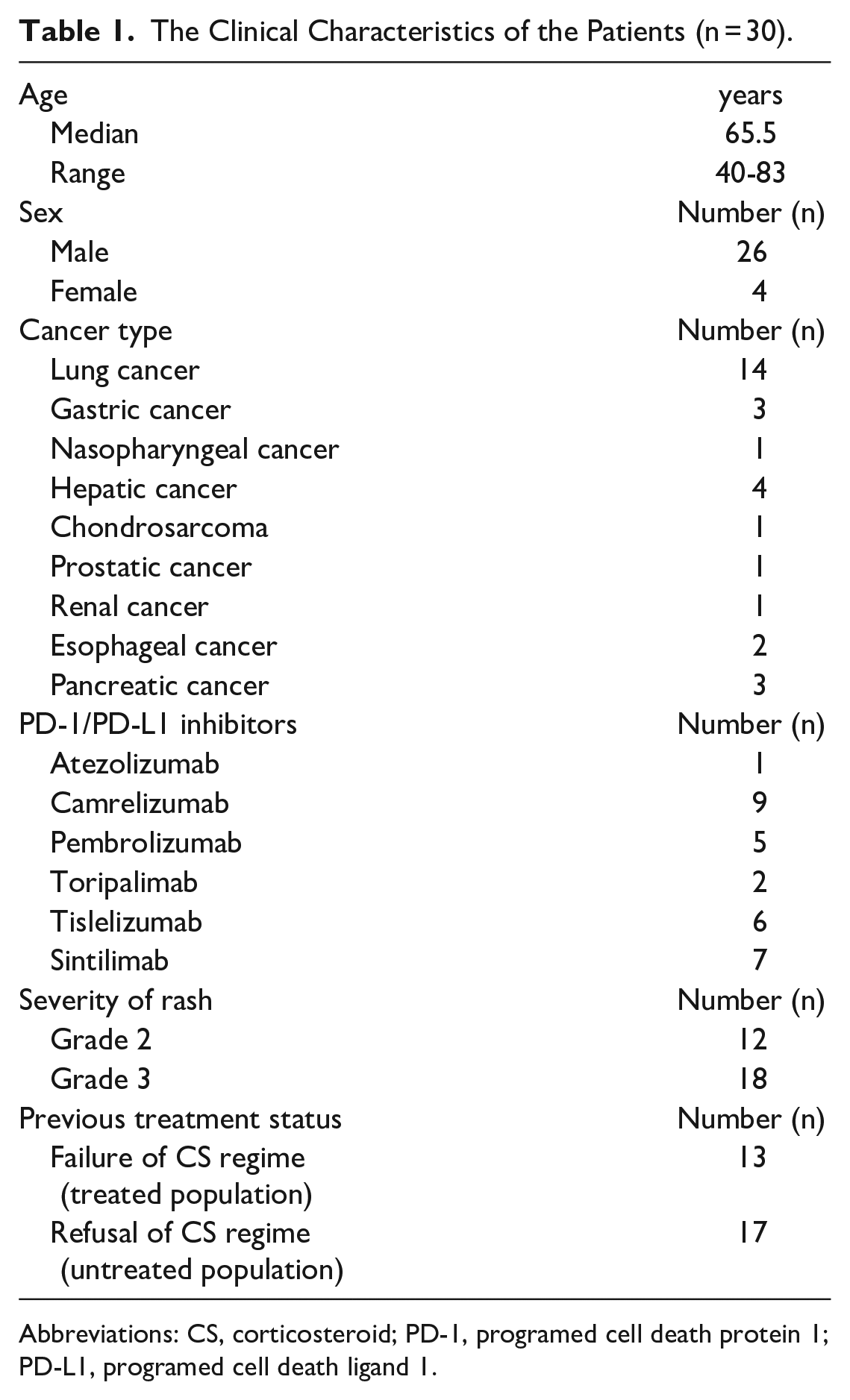
Abbreviations: CS, corticosteroid; PD-1, programed cell death protein 1; PD-L1, programed cell death ligand 1.
Efficacy and Safety of QRLSF
These patients received external application of QRLSF; the longest use time was 1 month. After treatment with PD-1/PD-L1 inhibitors, the median onset time of grade 2 and 3 rash was 182.5 and 57.5 days, respectively. The median time for rash occurrence among all subjects included in this study was 64.5 days. The representative photos before and after QRLSF treatment are shown in Figure 1A. Ten cases with grade 2 rash (median time: 5 days; range: 3-11 days; effective rate: 83.33%) and 14 with grade 3 (median time: 13 days; range: 1-19 days; effective rate: 77.78%) achieved the standard for re-treatment of PD-1/PD-L1 blockers and their skin lesions were eventually reduced to grade 1. In the whole population, the median time for rash to recover to grade 1 was 8 days and the total effective rate was 80% (Figure 1B and C). The accompanying symptoms of rash (eg, itching, pain, burning and tightness) were also greatly improved. Twenty-three out of 26 patients (effective rate: 88.46%) with itching experienced symptom relief, with a median time of 4 days (range: 1-15 days). The remission of symptoms occurred in all 13 patients with initial pain, with a median time of 3 days (range: 1-28 days). Similarly, the alleviation of burning and tightness was also observed in 12 patients, with a median time of 3 days (range: 1-15 days) (Figure 1D). During the treatment of QRLSF, 12 patients also used antihistamine (AH) at the same time. However, the application of such drugs did not increase the remission rate of rash (AH + QRLSF: 75.00% vs QRLSF: 83.33%, P = .66) (Figure 1E). Before QRLSF treatment, 13 patients were defined as the treated population because they had received CS therapy on the basis of guide recommendation, but none obtained clinical benefit. Seventeen were defined as the untreated population because they were unwilling to receive CS treatment and had never taken other methods. The result showed that there was no significant difference in the efficacy of QRLSF between these 2 types of patients (untreated population: 88.24% vs treated population: 69.23%, P = .36) (Figure 1F). During 1-month follow-up, 2 cases out of the 24 valid patients underwent relapses (recurrence rate: 8.33%). Simultaneously, no adverse reactions related to QRLSF were documented in any medical record.
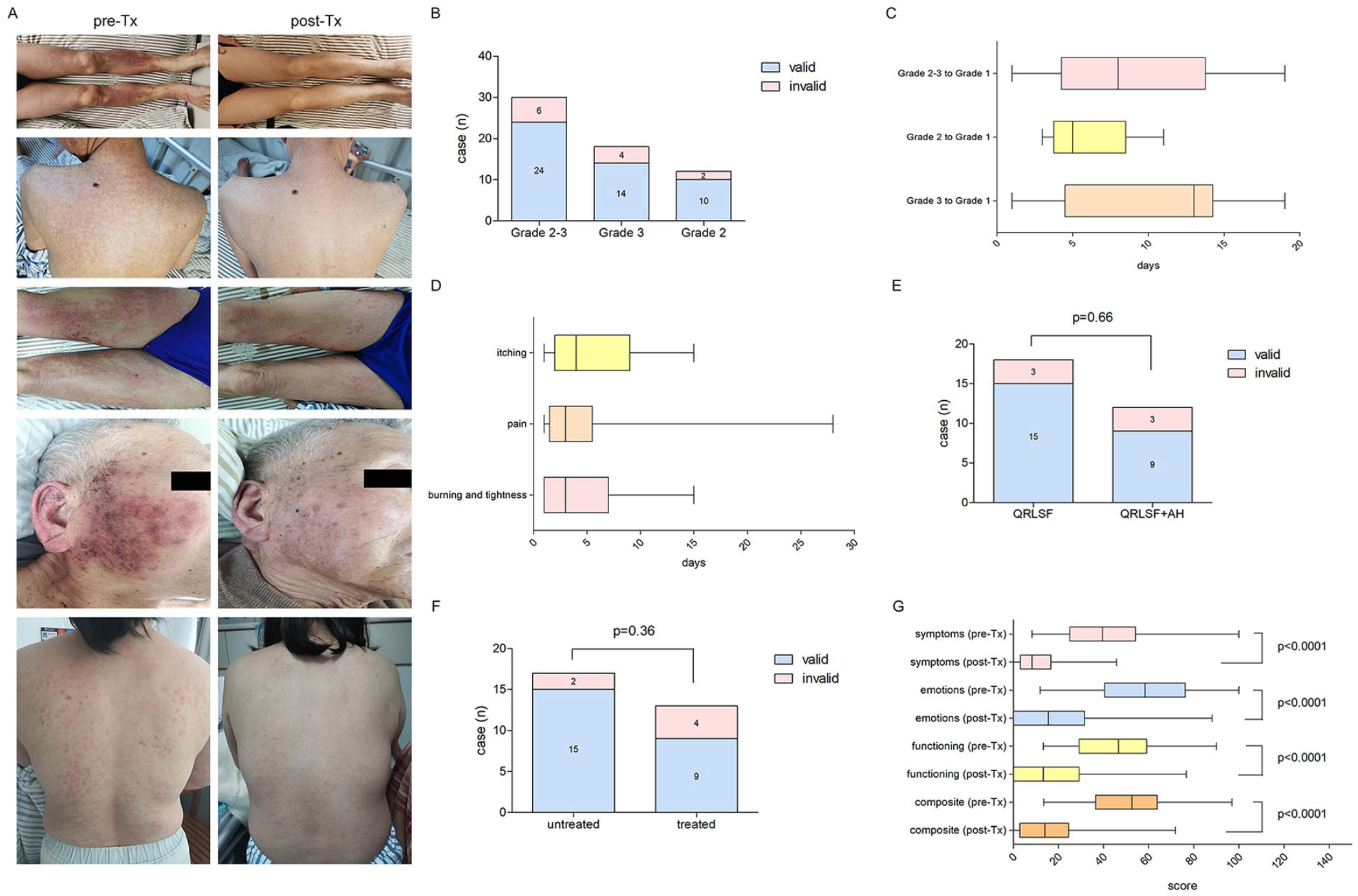
The efficacy of QRLSF on grade 2 to 3 immune-related rash induced by PD-1/PD-L1 inhibitors. (A) The representative photographs of clinical manifestation of immune-related rash before (left) and after (right) QRLSF treatment. (B) Number of patients who benefited from QRLSF treatment. An improvement of rash to grade 1 was defined as valid. (C) The median time for rash to recover to grade 1. (D) Median time for relief of accompanying symptoms of rash (itching, pain, burning and tightness). (E) Comparison of the effective rate between the patients who received AH therapy or not during QRLSF treatment. (F) Comparison of the effective rate of QRLSF treatment between the treated and untreated patients. (G) Changes of the scores in each subscale of Skindex-16 questionnaire (symptoms, emotions, functioning and composite) after QRLSF treatment. Boxes represented interquartile range (25% percentile, 75% percentile). Central horizontal line indicated median. Outer horizontal lines indicated the range from minimum to maximum. Abbreviations: AH, antihistamine; QRLSF, Qing-Re-Li-Shi Formula; pre-Tx, pre-treatment; post-Tx, post-treatment.
Analysis of HrQoL
As shown in Table 2, the total median score of Skindex-16 questionnaire was decreased after QRLSF treatment (52.60 vs 14.06, P < .0001), indicating that QRLSF could improve HrQoL of patients with PD-1/PD-L1 inhibitors-associated rash. We further analyzed the scores of each subscale in the Skindex-16 questionnaire. When comparing with the baseline, QRLSF treatment could significantly reduce the median scores of all 3 domains, including symptoms (39.58 vs 8.33, P < .0001), emotions (58.33 vs 15.48, P < .0001), and functioning (46.67 vs 13.33, P < .0001) (Figure 1G).
Changes in Skindex-16 Scores Before and After QRLSF Treatment.

Abbreviations: QRLSF, Qing-Re-Li-Shi Formula; pre-Tx, pre-treatment; post-Tx, post-treatment; p25, 25% percentile; p75, 75% percentile; CI, confidence interval.
Discussion
Rash induced by immune checkpoint inhibitors (ICIs) is very common in our clinical practice. PD-1/PD-L1 inhibitors are the main types of ICIs. Some patients also develop rash even after stopping PD-1/PD-L1 immunotherapy owing to the continuous activation of the immune system and inflammatory response. 15 Worryingly, once grade 2 to 3 rash occurs, withholding ICIs therapy and treating with high potency topical and systemic CS are the limited options for such patients, 16 which may hinder the anti-tumor treatment. A high-quality meta-analysis has indicated that there is a positive association between the presentation of irAEs (including ircAEs) and objective response rate, progress free survival, and overall survival. 17 Concurrently, the Multinational Association of Supportive Care in Cancer has also mentioned that the occurrence of ircAE is always accompanied by a better prognosis. 8 Therefore, interruption of anti-PD-1/PD-L1 treatment will undoubtedly reduce the antitumor efficacy, and even lead to disease progression. In addition, the efficacy of CS therapy is not satisfactory and remains controversial. Some scholars reported that the median time to resolution of all-grade lichenoid and maculopapular rash after drug discontinued or systemic CS treatment was up to 36.5 and 37.5 days, respectively, while the severe and other subtype required even longer. 18 About a quarter of immune-related rash patients are steroid-refractory. 8 The application of CS may result in multiple complications (eg, emotional change, infection, diabetes, etc.), not only disrupting future treatment, but also influencing the QoL. 19 Though a previous study showed that use of CS to treat irAE other than other indications would not impact the tumor response 20 ; the other studies concluded that the administration of CS before the initiation or the early period of immunotherapy would hinder the efficacy of ICIs.21,22 Up to now, the correlation between CS used for ircAE and inferior outcome of ICIs therapy still remains indeterminate.
TCM has unique advantages in reducing adverse reactions related to anti-tumor treatment and improving the QoL of patients.23 -25 However, the application of CHM in the treatment of immune-related rash is still in the exploratory phase and the relevant studies are rarely reported. With the guidance of external treatment theory of TCM, immune-related rash belongs to “drug toxic rash.” According to its common clinical manifestations of rash with or without symptoms (eg, pruritus, pain, purpura, edema, burning or tightness etc.), immune-related rash is mainly diagnosed as heat-damp syndrome. Hence, clearing heat and removing dampness is identified as the fundamental treatment principle. External application of TCM is considered as a more effective way to treat rash. Hydropathic compress with CHM, as one of the external treatment in TCM, has been widely adopted in the therapy of skin disorders. It exerts pharmacological effects by directly penetrating the liquid medicine into the skin lesions and increasing the intensity of local drug action to accelerate wound healing. Throughout history, this method has been proven to be safe and effective. Complying with the above evidence, a clinical proved recipe QRLSF has been empirically summarized on the basis of SMYAD. It primarily consists of 6 Chinese herbal medicines. Among them, Lonicerae Japonicae Flos has been shown to play an anti-inflammatory role by inhibiting the generation and release of inflammatory factors (eg, tumor necrosis factor alpha (TNF-α), interleukin-6 (IL-6), inducible nitric oxide synthase (iNOS), etc.) and regulating the related signaling pathways (janus kinase (JAK)/signal transducer and activator of transcription (STAT), Toll-like receptor 4 (TLR4)/nuclear transcription factor-κB (NF-κB), etc). 26 Scrophulariae Radix exhibits its anti-inflammatory activity by suppressing infiltration of eosinophils, macrophages, neutrophils and lymphocytes. It can also reduce the level of inflammatory cytokines, including TNF-α, IL-6, interleukin-17 (IL-17), etc. 27 It has been reported that the anti-inflammatory effect of Angelicae Sinensis Radix is associated with the activity of reducing the level of TNF-α and transforming growth factor-beta1 (TGF-ß1). It also exerts the function of anti-platelet aggregation and promoting blood circulation. 28 Pharmacology has confirmed that inhibiting inflammation is one of the most important properties of Coicis Semen. Its extract can down-regulate the expression of cyclooxygenase-2 (COX-2), monocyte chemoattractant protein-1 (MCP-1), NF-κB, etc. 29 Angelicae Dahuricae Radix is known as a common herb used in skin diseases. Lowering infiltration of inflammatory cells and level of cytokines (eg, nitric oxide (NO), TNF-α, prostaglandin E2 (PGE2), IL-6, interleukin-10 (IL-10), etc.) are its pharmacological functions. 30 Glycyrrhizae Radix et Rhizoma has been verified to have an anti-inflammatory mechanism related to a decrease in the secretion of inflammatory factors in macrophages and containment of the critical enzymes in the arachidonic acid (AA) metabolism process of PGE2 synthesis. 31 Based on the modern pharmacology and theory of TCM, the method of clearing heat and removing dampness, which is represented by QRLSF, has been applied to treat multiple types of cutaneous toxicity induced by a variety of antitumor drugs in our medical center for a long period of time. Here our study reviewed and summarized our clinical experience in relieving grade 2 to 3 immune-related rash.
In this retrospective study, we identified 30 patients (including 26 males and 4 females) with grade 2 to 3 immune-related rash. The reason for gender imbalance may be related to regional factors. In daily clinical work, there are more males than females seeking medical advice in our center. The median time of the presentation of ≥grade 2 rash reported in our cohort was 64.5 days. Previous research reported the cutaneous toxicity ≥grade 2 usually appeared after 3 months of ICIs therapy.18,32 We also found that there was a significant difference in the median onset time of grade 2 and 3 rash. In our study, grade 2 rash appeared later than grade 3. The reason may be related to individual differences. Rash can occur at any time after the application of ICIs, ranging from 1 to 1000s of days. 18 Additionally, the type of skin adverse events, geographical region and types of ICIs are all influencing factors for the above results. For the moment, the treatment of topical and systemic CS is the first choice for such patients according to the national and international recommendations. Unfortunately, it was not applicable for these 30 cases because they belonged to the refractory types or refused to receive CS treatment due to concern about its side effects. But it was encouraging that 80% of such patients could reach remission through the management of QRLSF treatment with a median time of 8 days. We conducted further subgroup analysis on patients with grade 2 and 3 rashes. Ten cases with initial grade 2 and 14 cases with grade 3 immune-related rash who finally achieved grade 1 with the median time of 5 and 13 days respectively could have the opportunities to receive anti-PD-1/PD-L1 treatment again. Comparing with the previous literature, the median resolution time of the strategies of topical steroids, antipruritics, and systemic immunomodulators (including CS) for the treatment of ≥grade 2 cutaneous toxicity was 41 days, 18 which was much longer than that of QRLSF in our study. The repair of skin lesions requires a certain period of time and the affected area may not have obvious changes, but the accompanying symptoms often change first. In this study, we analyzed common accompanying symptoms such as itching, pain, burning and tightness, which could disrupt patients’ normal life, affect their sleep, and lead to decreased physical strength, lack of concentration, and mental exhaustion. Nevertheless, half of patients experienced symptom alleviation within 3 to 4 days after QRLSF treatment, further confirming its efficacy in treating PD-1/PD-L1 related rash. In the course of treatment, although some patients received concurrent AH therapy, this did not increase the effective rate, revealing that the monotherapy of QRLSF showed a great effect. That is to say, QRLSF may become a novel choice to resolve problem of ircAEs. The CS therapy is not always effective and may hinder the efficacy of antitumor immunotherapy. Immunomodulators are the primary recommendations for the management of such patients with CS-refractory rash, yet there is little data on their curative effect or safety.33,34 Our study reported that patients could benefit from QRLSF treatment regardless of whether they had previously received CS therapy. The effective rate of 13 patients who had previously received CS treatment was nearly 70%, indicating that QRLSF was probably a candidate method for CS-refractory cases. The guideline 7 has suggested that PD-1/PD-L1 blockers can be reconsidered when rash gets back to grade 1, but did not mention the recurrence rate after rechallenge. As suggested by Simonaggio et al, 35 more severe initial irAE including skin toxicity was associated with the higher risk of recurrence rate. In this paper, only 2 patients (8.33%) underwent relapses after rechallenge. The recurrent rash was not more severe than the initial event and no other different types of irAEs were observed, indicating that QRLSF had latent capacity to prevent the recurrence of immune-related rash. In addition, the safety of CHM is especially attended by the public. There was no evidence in our study that QRLSF-related adverse events existed. Taken together, these factors demonstrated that external application of QRLSF was safe and reliable.
Increasing evidence has indicated that the symptoms of immune-related rash have a profound negative influence on the sufferers’ HrQoL, including patients’ social participation, emotional state, and daily activities, etc.15,18 IrcAEs are associated with the better treatment response, 36 which highlights the urgent need for effective treatment to maintain the QoL and therapeutic intensity of ICIs. Skindex-16, which is not limited to the type of dermatosis, is an appropriate instrument to assess the QoL of patients with ircAEs. 37 Changes in scores exceeding 10 points are considered as clinically significant. 38 After QRLSF treatment, the changes of the median scores on the subscales of symptoms, emotions, functioning and composite were more than 10 points, with statistical significance. As we know, the improvement of symptoms is always accompanied by the recovery of patients’ ability of daily life and alleviation of their anxiety. We believe that QRLSF treatment can improve the HrQoL of the patients with grade 2 to 3 immune-related rash. It is a candidate of therapeutic drugs for the treatment of immune-related rash, especially for patients with high-grade.
In spite of the positive findings, there were still some limitations of this research. Primarily, the sample size was small. Then, this was a single-center retrospective study and the control of variables was not ideal compared to prospective study. Also, lack of a control group would bring some selective bias to analyze the efficacy of QRLSF. Besides, the specific mechanism of QRLSF in the treatment of immune-related rash remained to be explored due to the lack of molecular characteristics. Therefore, some high-quality, prospective, large sample size and randomized controlled trials, and well-designed basic research are essential to be conducted to further explore the clinical efficacy and molecular mechanism of QRLSF in treating ircAEs, as well as its more accurate range of application. Even so, our single-center retrospective study has first discussed the potential of external application of clearing heat and removing dampness in the treatment of immune-related rash. The clinical data has profound guidance for clinical practice.
Conclusion
In conclusion, external application of clearing heat and removing dampness was an effective and safe method for patients with grade 2 to 3 immune-related rash. It could effectively relieve symptoms and improve the QoL of such patients. With the wide application of anti-PD-1/PD-L1 therapies in multiple types of cancer, it is necessary to strengthen the comprehensive understanding and management of cutaneous toxicity. The findings in our research will be important for the optimization of the administration of ircAEs. The identification of effectiveness of this new treatment for ircAEs, especially in CS-inapplicability, described in our single-center retrospective study provides a prerequisite for the design of high-quality trials that will further explore its potential biomarker targets, efficacy and safety in the future.
Footnotes
Acknowledgements
We show our appreciation to Department of Statistics of Zhejiang Chinese Medical University for their helpful statistical consultation.
Author Contributions
Shu-yi Chen, Fang-min Zhao, and Rui Yu contributed equally to this work. Shu-yi Chen and Qi-jin Shu contributed to the research conception and design. Shu-yi Chen, Fang-min Zhao, Rui Yu, Lin Sun, Yu-dan Yin, Gao-chen-xi Zhang, and Jia-yao Yang participated in data collection. Shu-yi Chen, Fang-min Zhao and Rui Yu were responsible for data analysis and interpretation. Shu-yi Chen and Fang-min Zhao finished the writing-original draft. All authors were accountable for the final approval of the manuscript.
Data Availability
The data of this study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Zhejiang Traditional Chinese Medicine of Science and Technology Program [grant number: 2022ZA075], “San Ying” Talent Training Project 3.0 and Key Disciplines of Zhejiang Provincial Hospital of Chinese Medicine.
Ethical Approval
This study was conducted according to the principles of Helsinki Declaration and approved by the Ethics Committee of the First Affiliated Hospital of Zhejiang Chinese Medical University (Zhejiang Provincial Hospital of Chinese Medicine) (No. 2022-KL-040-01).
Informed Consent
Written informed consent for the publication of their information was obtained from all patients.
