Abstract
Background:
Children and adolescents undergoing umbilical cord blood transplantation (UCBT) are faced with severe fatigue and a decline in quality of life (QoL) during the inpatient period.
Objective:
To investigate the effect of a structured exercise intervention on fatigue, QoL and clinical outcomes among children and adolescents during UCBT.
Methods:
In this randomized controlled trial, participants (n = 48) were randomized to a control group (CG: usual care) or an intervention group (IG: a structured exercise intervention). Fatigue and QoL were assessed at hospital admission, 14 days after UCBT, and at discharge using linear mixed model analysis. In addition, engraftment kinetics, supportive treatment, transplant-related complications, and hospital length of stay were derived from medical records.
Results:
4 patients completed the study, the IG participated in an average of 2.12 (1.36-2.8) sessions with a duration of 24 (16-34) min weekly, and the total rate of adherence to the training program was 70.59%. For fatigue and QoL, there was a significant effect of time in the control group, with the total score of fatigue decreased from T1 to T2 (73.9vs 60.9, P = .001) and T1 to T3 (73.9vs 65.6, P = .049), and the QoL scores decreased from T1 to T2 (73.9vs 66.1, P = .043). The hospital length of stay was less in the intervention group (P = .034).
Conclusion:
Our randomized study indicated that structured exercise interventions might exert a protective effect by attenuating the decline in fatigue and QoL, and shortening duration of hospitalization.
Keywords
Introduction
Umbilical cord blood (UCB) has been a valuable alternative stem cell source for treating malignant and nonmalignant hematopoietic disorders, especially for children.1,2 With improving survival rates, there has been an increase in the number of pediatric patients who have undergone UCB transplantation (UCBT).3,4 Compared to other types of hematopoietic stem cell transplantation (HSCT), UCBT patients often suffer from delayed engraftment, higher rates of infection and transplant-related mortality, and longer hospital length of stay (LOS) and higher hospital costs.1 -5 In addition, patients experience treatment-related negative symptoms during hospitalization, particularly severe fatigue and a decline in quality of life (QoL).6 -8
To date, there is sufficient evidence to confirm that exercise interventions can improve fatigue and QoL in adult patients undergoing HSCT.9 -11 However, few studies have described the effect of structured exercise interventions on pediatric HSCT recipients, and there has been high heterogeneity in the results. 12 Kabak et al 13 conducted a multimodal exercise program that including strengthening, endurance, stretching, and relaxation exercises for children undergoing autologous and allogenic HSCT. They reported that the moderate exercise program significantly promoted QoL levels. Smith et al 14 showed that a progressive structured exercise program performed 3 times weekly and once weekly on discharge for 6 weeks had a beneficial effect on fatigue but not on QoL scores. Senn-Malashonak et al 15 conducted a randomized study of 70 children and adolescents receiving autologous and allogenic HSCT. The study showed that patients’ QoL was not improved after their combined exercise interventions targeted a moderate intensity with 5 sessions weekly during the inpatient period. Despite the growing number of studies on exercise during pediatric HSCT, little is known about the effectiveness of structured exercise interventions in children and adolescents who undergo UCBT. In addition, the influence of structured exercise interventions on the pediatric patients’ clinical outcomes still needs to be clarified.16 -19
The primary aim of this study was to investigate the effect of a structured exercise intervention for improving fatigue and QoL in pediatric UCBT recipients. A secondary purpose was to examine the effectiveness of structured exercise interventions on clinical outcomes, including engraftment kinetics, supportive treatment, transplant-related complications, and hospital LOS.
Methods
Study Design
This prospective study was a randomized controlled clinical trial conducted from July 2017 to December 2018. The study was approved by the ethical commission of the First Affiliated Hospital of University of Science and Technology of China (USTC). Informed consent was obtained from all participants and/or their parents before the study. Patients were assigned to 2 groups at admission using a randomized table, starting from any number in a row, with odd numbers included in the control group and even numbers included in the intervention group.
Participants
A total of 116 children and adolescents with leukemia who received their first UCBT were included in this randomized study. The inclusion criteria were as follows: (1) age 8 to 17 years old, (2) Karnofsky performance score ≥70, and (3) able to read and speak Chinese. The exclusion criteria were evidence of cardiovascular, orthopedic, or neurological deficits.
Treatment Conditions
A health education brochure was provided from the nursing team on admission, which including breathing therapy, muscle relaxation and flexibility training. Children’s books and toys were provided by their parents for entertainment to eliminate the distress of isolation.
Intervention group (IG)
Patients received a structured exercise intervention comprising 3 sessions per week for a duration of 30 to 40 minutes from admission to discharge. The exercise intervention began with aerobic training, followed by resistance (3-6 exercises) and flexibility training (6-9 exercises). Aerobic training was defined as walking or rising on a tiptoe for 15 to 20 minutes in the isolation unit. The resistance routine included exercises that targeted major muscle groups of lower extremities and the core (back, lumbar, gluteal, thighs, and calves), and participants were asked to perform 2 to 3 series of 8 to 12 repetitions per exercise against gravity. The flexibility training consisted of dynamic and static stretching, dynamic stretching included low-back extension, abdominal crunch, waist and hip stretching, performing 10 to 12 repetitions for 1 to 2 sets, static stretching included maintaining hip extensors, knee flexors and ankle dorsiflexors in an extended position for 10 to 30s, performing 2 to 3 repetitions for each muscle. Considering patients’ upper limbs were inserted with peripherally inserted central catheters, we did not include exercises for the upper limbs in the program. Before exercise, the therapist checked the patients’ health state and potential contraindications, and each session was individualized and conducted under supervision of an exercise physiologist. Exercise intensity was maintained at a moderate intensity targeting to 12 to 14 using the Borg’s rating of perceived exertion (RPE). 20
Control group (CG)
Patients were advised to exercise following the health education brochure. Usual daily bed activities were not restricted during the study period. To avoid psychological bias, the controls were visited by a member of the research team with the same frequency and amount of time with playing.
Outcome Measures
Study outcomes comprised fatigue, QoL, and clinical parameters. Children over 6 years old in our center were not allowed to be accompanied by parents due to protective isolation during hospitalization. Therefore, the questionnaires of fatigue and QoL were conducted at the date of hospital admission (T1), 14 days after UCBT (T2) and at discharge (T3) by the patients independently. Clinical outcomes were derived from medical records.
Primary outcomes
The primary outcomes included fatigue and QoL.
Fatigue
Fatigue was documented via the Pediatric Quality of Life Inventory (PedsQL) Multidimensional Fatigue Scale (MFS) Acute Version, which is designed by Varni et al and colleagues. 21 The scale includes three dimensions (18 items): general fatigue, fatigue corrected with sleep/rest, and cognitive fatigue. The questionnaire is rated on a 5-point Likert scale, ranging from 0 (never) to 4 (always). Each item was reversed and rescaled to 0 to 100 (0 = 100, 1 = 75, 2 = 50, 3 = 25, 4 = 0), and the total score was obtained by averaging the 18 items. Higher scores indicate fewer symptoms of fatigue. The Cronbach’s alpha of the Chinese version of the PedsQL- MFS varied from .92 to .93 for the child self-report. 22
QoL
QoL was measured by PedsQL 3.0 Cancer Module, which is specifically tailored for pediatric patients with cancer. 21 The PedsQL 3.0 Cancer Module is a 27-item measure consisting of 8 subscales, including pain and hurt, nausea, and procedural anxiety, among others. Patients were asked to rate the scale the same as for the PedsQL-MFS, with higher scores indicating better QoL. The Cronbach’s alpha of the Chinese version of the PedsQL 3.0 Cancer Module varied from .84 to .99 for the child self-report. 23
Secondary outcomes
The secondary outcomes included engraftment kinetics (days to neutrophil engraftment and to platelet engraftment, and days of myelosuppression post-UCBT), supportive treatment (number of platelet/red blood cell transfusions, parenteral nutrition, and antibiotic treatment), transplant-related complications (number of infections after UCBT, grade of pre-engraftment syndrome (PES) and acute graft-versus-host-disease (aGVHD), and time from UCBT to PES and aGVHD), and hospital LOS. Neutrophil engraftment was defined as the first day when an absolute neutrophil count was higher than 0.5 × 109 /L for three consecutive days. Platelet engraftment was defined as the first day when the platelet count was higher than 20 × 109/L for seven consecutive days without transfusion support. 24
Statistical Analysis
Statistical analysis was carried out using SPSS version 22.0 for Windows (SPSS Inc., Chicago, IL, USA) and R software packages, version 4.0.5 (R Foundation for Statistical Computing, Vienna, Austria). All the data were tested for normal distribution before analysis. For demographics and medical parameters, the normal data are given as the means and standard deviations and were compared by an independent t test. The nonnormal data are presented as medians with ranges and were evaluated by the Mann–Whitney U test. Categorical data are presented as numbers and were examined by the chi-square test.
For fatigue and QoL, the linear mixed model (LMM) was used to estimate the effects of group (between-patient), time (within-patient), and interactions between the group and time. The statistical models were adjusted for age, gender, and disease type; post-hoc pairwise comparisons were conducted when the analysis demonstrated significance. The incidences of neutrophil and platelet engraftment were analyzed using the log-rank test. When P < .05 (2-sided), the results were considered statistically significant.
Results
Recruitment
The recruitment flow diagram is presented in Figure 1. A total of 116 children and adolescents who received their first UCBT were recruited between July 2017 and December 2018. The major exclusion reason was the age of potential participants (<8 years: n = 52), 9 patients declined to participate due to a lack of interest. Ultimately, 48 patients met the inclusion criteria and participated in the study, and 4 patients withdrew because of medical complications.

The recruitment flow diagram.
Baseline Characteristics
Patient and medical characteristics are shown in Table 1. Of the 44 patients (CG: n = 21; mean age (12.14 ± 2.90) years; IG: n = 23; mean age (11.52 ± 2.48) years), all the conditioning regimens were myeloablative, and there were no significant differences in demographic and medical characteristics at baseline, except for primary diagnosis, which was not comparable between groups (P = .002).
Patient and Medical Characteristics.

Abbreviations: BMI, body mass index; AML, acute myelogenous leukemia; ALL, acute lymphoblastic leukemia; CML, chronic myelogenous leukemia; CR, complete remission; PR, partial remission; PIF, primary induction failure; Flu, fludarabine; Bu, busulfan; Cyclo, cyclophosphamide; TBI, total body irradiation; KPS, karnofsky performance score; HLA, human leukocyte antigen; TNC, total nucleated cells.
Participation in Exercise Intervention
Forty-four patients (CG: n = 21; IG: n = 23) completed the study. During the hospitalization phase, the exercise intervention group participated in an average of 2.12 (1.36-2.8) training sessions with a duration of 24 (16-34) min per week, and the average RPE was 12.6 (12-13.7). The rate of participation in scheduled exercises was 70.59% (45.5%-93.3%), the primary barrier to participation including fever >38℃, severe dizziness, nausea and vomiting, diarrhea, severe anemia, hemorrhage, and extreme muscle pain. There were no exercise-induced adverse events throughout the study.
Effectiveness of Exercise for Fatigue
LMM analysis indicated no significant differences between the CG and IG on total and subscale scores of fatigue after adjusting for age, gender, and disease type in the model (Table S1, S2). However, time had significant impact on fatigue, and post-hoc pairwise comparisons of PedsQL MFS scores at the 3 time points showed a decline in the CG from T1 to T2 (73.9vs 60.9, P = .001) and T1 to T3 (73.9vs 65.6, P = .049) (Figure 2). Differences in the IG were not significant.
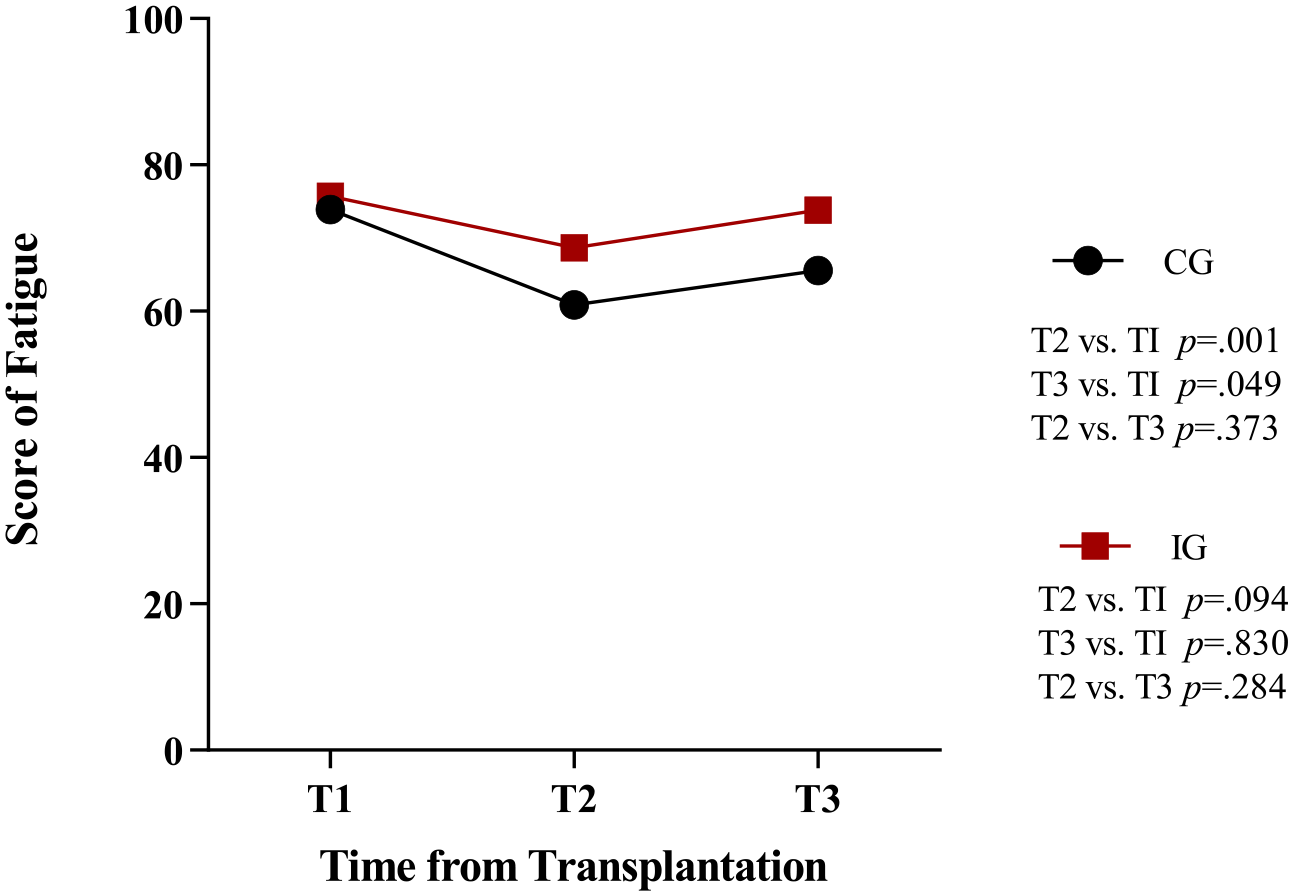
Total score of fatigue at the date of hospital admission (T1), 14 days after UCBT (T2), and at discharge (T3) in 2 groups. Higher values indicate less fatigue.
Effectiveness of Exercise for QoL
LMM analysis revealed no significant differences between the CG and IG on total and subscale scores of QoL after adjusting for age, gender, and disease type in the model (Table S3, S4). However, there was a significant time effect between scores at T1 and T2 in the CG (73.9vs 66.1, P = .043), while differences in the IG were not significant (Figure 3).

Total score of QoL at the date of hospital admission (T1), 14 days after UCBT (T2), and at discharge (T3) in the CG and IG. Higher values indicate higher QoL.
Effectiveness of Exercise for Secondary Outcomes
No significant differences were found for engraftment kinetics, supportive treatment, and transplant-related complications between 2 groups, however, the hospital LOS was less in the IG (Table 2; P = .034). Log-rank test showed no between-group differences in the incidences of neutrophil or platelet engraftment (Figure 4; p>.05).
Effects of Structured Exercise Interventions on Secondary Outcomes.

Abbreviations: PES, pre-engraftment syndrome; aGVHD, acute graft-versus-host-disease; LOS, length of stay.

The incidences of neutrophil and platelet engraftment in both groups.
Discussion
This study is the first to investigate the effect of a structured exercise intervention on fatigue, QoL, and clinical outcomes for pediatric UCBT recipients. The findings from this prospective, randomized study demonstrated that the structured exercise program has time effects on preventing the decrease in fatigue and QoL during hospitalization, and it is a successful strategy for shortening hospital length of stay.
Fatigue is one of the most troublesome symptoms during cancer treatment. 25 By diminishing the decrease in fatigue, the protective effect of a structured exercise intervention was demonstrated. This finding is in accordance with previous studies.9,14 Oberoi et al 9 selected 169 adult trials and 1 children trial in their systematic review, confirmed that physical activity was effective at reducing fatigue in patients with cancer and HSCT recipients. The effect of exercise interventions may work by improving physical fitness and promoting functional capacity.14,26,27 However, no significant differences were found between the 2 groups; this is in line with Rossi et al, 28 and may be related to the short-term intervention that was conducted only during hospitalization. In addition, fatigue is a symptom that might persist for years after treatment completion. 25 A speculation to guide future study is that fatigue may improve over time.
Our demonstration that patients’ QoL was positively maintained by the structured exercise intervention during hospitalization. This finding is consistent with reports that exercise contributes to promoting QoL when compared to usual care.12,13 Recent guidelines also demonstrated that movement and exercise training may provide benefits for QoL. 29 The QoL in the control group decreased significantly at 14d after transplant, which period is a short-term evaluation, and associated with more symptoms. However, it also returned to pre-UCBT at discharge, which was consistent with Liu et al 30 They conducted a prospective longitudinal study and found that health-related QoL decreased at 14-days post-HSCT, but increased at 1 week before hospital discharge. The fast recovery of QoL in the controls might be due to the encouragement to increase physical activity after engraftment. Therefore, it was suggested that a progressive exercise program be carried out to achieve greater benefits in the later phase of transplantation.
The structured exercise intervention was not effective at improving engraftment kinetics, supportive treatment, and transplant-related complications; this result is in line with prior studies.13,15,18 On the other hand, the result verified the safety of exercises, as it did not increase the risk and severity of UCBT. Contrary to our study, Morales et al 17 showed a reduced number of total and viral infections in the exercise group, where the exercise program was performed 5 times weekly for 60 minutes in children during HSCT hospitalization. Gleeson et al 31 reported that exercise may play an anti-inflammatory role by reducing the amount of visceral fat mass and inducing an anti-inflammatory environment post-exercise in chronic metabolic and cardiorespiratory disease patients. Considering that the patients were already receiving steroid, immunosuppressive, and antibiotic therapies, whether exercise can play an anti-inflammatory role in UCBT still needs further randomized controlled trials to verify. Although the number of infections was not decreased after UCBT, we found that the supervised exercise intervention was associated with a shorter length of hospital stay, which is in line with the previous finding. 19 To be noted, it is of major importance for the children to get home earlier and reduce the hospital costs, especially important in UCBT, where the duration of hospitalization is much longer compared to other kind of transplantation.5,32
This study has several limitations. Firstly, the exercise adherence was low (70.59%), which largely due to the delayed engraftment of UCBT recipients.3,7 In addition, children were not willing to exercise because of the severe symptom burden, and it was very hard to do the strict exercises in the isolation unit without peers. Secondly, the sample size was small, so that we could not conclude that the structured exercise intervention had effects on fatigue and QoL between 2 groups. Thirdly, physical capacities are important parameters, 33 but our research mainly focused on psychological characteristics, so that we did not include any physical fitness parameters, nor did we use a physical capacity test throughout this study.
Conclusion
This randomized study demonstrated that the structured exercise intervention was beneficial for counteracting the decline in fatigue and QoL for pediatric UCBT recipients. Future research should help to further determine exercise frequency, duration, and intensity, and verify the dose-dependent effects on physical fitness parameters for young patients.
Supplemental Material
sj-docx-1-ict-10.1177_15347354231210857 – Supplemental material for Psychological Effects of a Structured Exercise Intervention During Umbilical Cord Blood Transplantation in Children and Adolescents
Supplemental material, sj-docx-1-ict-10.1177_15347354231210857 for Psychological Effects of a Structured Exercise Intervention During Umbilical Cord Blood Transplantation in Children and Adolescents by Lu Huang, Yun Wu, Ming Xu, Yingying Wang, Chengzhen Jia, Guiqi Song, Kaidi Song, Yaohua Wu and Yongliang Zhang in Integrative Cancer Therapies
Footnotes
Acknowledgements
We are grateful to the patients and their families for their participation in this study.
Author Contributions
All authors contributed to the study design, material preparation, and data collection. Lu Huang performed statistical analyses and wrote the first draft of manuscript, Yongliang Zhang contributed to the design of review protocol, comments on subsequent versions of manuscript, and interpretation of results. All authors reviewed and agreed upon the manuscript content.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Research Ethics and Patient Consent
The study received ethical approval from the First Affiliated Hospital of University of Science and Technology of China, and it was conducted following the principles of the Declaration of Helsinki. Informed consent was obtained from all participants and/or their parents before the study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
