Abstract
Introduction
Physical activity (PA) plays a significant role in cancer care, with inactivity contributing to increased risk of cancer development, 1 increased cancer-associated mortality, 2 decreased quality of life (QoL), and increase in symptom burden. 3 Patients with lung cancer are particularly burdened by severe symptoms4-6 and low activity tolerance, likely related to respiratory comorbidity and cigarette smoking.7,8 In lung cancer, lower exercise tolerance predicts worsened survival,5,6,9 and associations between inactivity and lower QoL have been discovered in survivors, 10 long-term survivors, 11 and patients with advanced disease. 12 PA interventions in patients with lung cancer have shown improvements in oxygen uptake, activity tolerance, QoL, and symptom burden. 13 However, most studies are small, few randomized trials are available, and studies enrolling patients with advanced-stage lung cancer are less common. 13
Adherence is a common barrier to PA interventions in lung cancer patients. Implementing a structured and physical therapy-based regimen, Temel and colleagues had 44% of subjects complete an 8-week intervention, 14 and a recent randomized trial implementing a walking regimen reported 44.5% of subjects completing the regimen. 15 Activity regimens that optimize adherence may be the most likely to improve outcomes. Factors reported to improve PA adherence in cancer patients include home-based regimens, 16 motivational techniques, 17 and lower intensity regimens. 18
In prior work with advanced-stage lung cancer patients, we have utilized weekly phone calls to deliver activity goals and provide motivation. We hypothesize that a patient-centered activity regimen (PCAR) utilizing a walking-based regimen, individualized patient goals, an educational session, and gain-framed text messaging (ie, those emphasizing the benefits of PA) would improve PA participation in patients with advanced-stage lung cancer. We chose to focus on gain-framed messages, as this type of messaging has been shown empirically to encourage PA 19 and, to our knowledge, has not been utilized in patients with lung cancer. Some of the results of this study have been previously presented in the form of a slide presentation at a national conference. 20
Methods
We compared patient participation and PA between a group receiving twice/daily, gain-framed text messages (institutional review board protocol number Pro00052856) and a historical group (institutional review board protocol number Pro00028353) that received weekly phone calls. Both studies implemented low-impact PA at the Medical University of South Carolina. The goal in both groups was to increase walking distance in patients with advanced-stage lung cancer. Advanced-stage lung cancer was defined as stage III or IV non–small cell lung cancer and limited or extensive stage small cell lung cancer. Patients were eligible for enrollment at diagnosis, during active treatment, or after treatment. In both motivational strategies, we recommended a home-based walking regimen. To monitor PA, we provided all patients with FitBit (San Francisco, CA) accelerometers that were returned at the end of the study.
The 2 groups differed by type of FitBit accelerometer, motivational strategy, and length of enrollment. The first study (n = 29 patients) was conducted between January 2015 and October 2015. Subjects wore a FitBit Zip that clips on to clothing, received weekly phone calls to deliver activity goals and remind them to exercise, and were enrolled for 4 weeks. The second study (n = 15 patients) was conducted between October 2016 and July 2017, and subjects participated in a multifaceted PCAR. The PCAR group used a wrist-bound FitBit Flex, received twice-daily text messages, and were enrolled for 12 weeks. The PCAR group also participated in an educational session at enrollment that lasts approximately 20 minutes and focuses on common symptoms in lung cancer, potential benefits of increasing PA, and what types of exercise have been studied in cancer. Text messages were personalized and included a weekly activity goal, current step count, gain-framed messages, and motivational statements. To assess patient interest and elicit potential changes in future regimens, patients in the PCAR group were given feedback questionnaires at the end of the intervention.
The same PA recommendations were made to both groups. After collecting daily step count for 7 days, an average daily step count was calculated. Step count goals were calculated by adding 400 steps/day to the average daily step count. The recommended increment of 400 steps/day is based on prior interventions in patients with chronic obstructive pulmonary disease. 21 At the end of each week, an average daily step count was calculated, and an updated walking goal was provided. If the patient reached their prior goal, the recommended step count goal was raised by another 400 steps/day. If the goal was not attained, then no change was made to their walking goal. If the patient averaged the recommended walking goal for healthy patients (ie, ⩾10 000 steps/day), continuation of current PA was recommended. For example, if the patient averaged 2000 steps/day during week 1, a walking goal of 2400 steps/day was recommended. If the patient averaged 2400 steps/day during week 2, a walking goal of 2800 steps/day was recommended. If, instead, the patient averaged 2300 steps/day during week 2, the walking goal of 2400 steps/day was maintained.
Based on prior walking studies in chronic obstructive pulmonary disease, data were considered not “usable” if <200 steps were collected/day or if <5 days were collected/week. 22 New weekly recommendations were not provided if “usable” data were unavailable. In this situation, the prior walking goal was maintained.
Analysis
To describe the study population and compare demographics between study phases, categorical and continuous variables are presented as N (%) and mean ± standard deviation, respectively. To estimate effect sizes (ESs) and to accommodate participants enrolled in both studies, repeated-measures, mixed-effects linear regression controlling for first week average, gender, and age was utilized. For accurate comparison, the 4 weeks of the phone-based group were compared with the first 4 weeks of the twice-daily text messaging group. Statistical significance was assessed at α = 0.05. All analyses were performed in SAS v9.4.
Results
We had a total of 35 unique patients with step count data. There were 22 subjects enrolled in the weekly phone calls group and 15 subjects enrolled in the twice-daily text messaging group. Two patients participated in both studies. Though 30 patients were originally enrolled in the weekly phone calls group, one patient was excluded due to inaccurate diagnosis, 6 patients never provided step count data (Table 1), and 1 patient provided a step count for only 1 day. Most patients in the study were male (70.3%) or stage IV (70.3%), and the average age of our participants was 66 years.
Demographics.
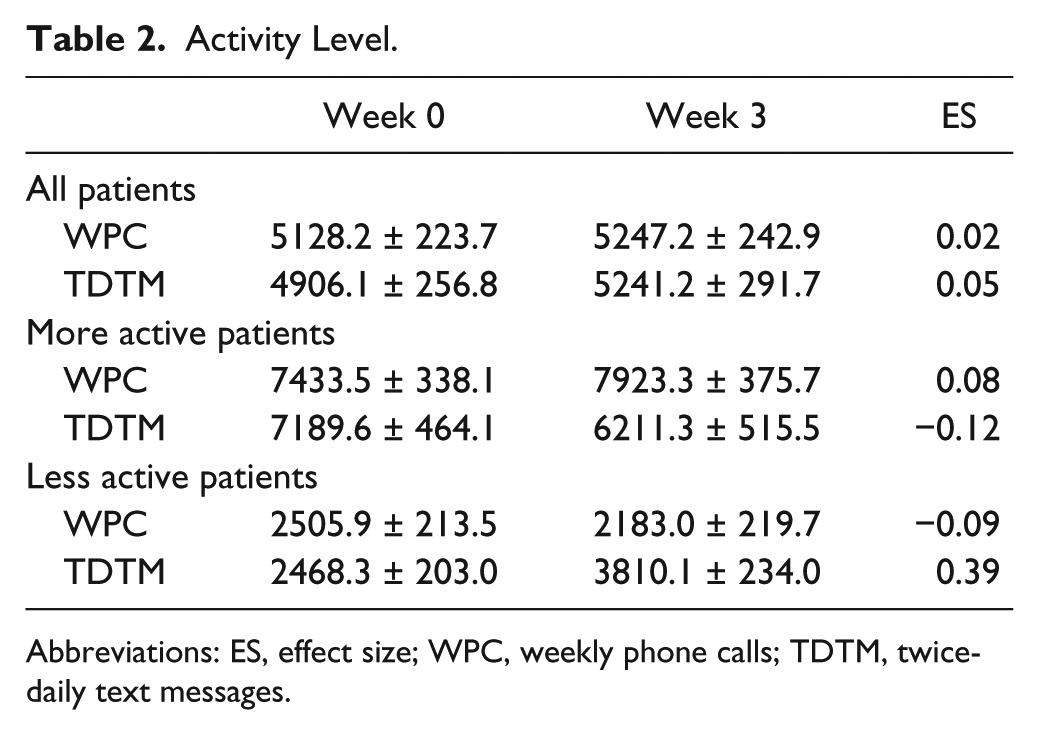
PA level was assessed by daily step count. Activity levels varied widely between patients and within an individual patient during the interventions. Figure 1 shows scatterplots of daily step counts for each group arranged by their average daily step count during the first week. The average, unadjusted step count for all 37 patients during the initial week was 4799 (Table 1). After adjusting for first week average, gender, and age, the average weekly change increased for both intervention groups. Overall, effect sizes were 0.02 for weekly phone calls and 0.05 for twice-daily text messages (Table 2).

(a) Individual daily step counts for group receiving weekly phone calls over time. (b) Individual daily step counts for PCAR (patient-centered activity regimen) group over time.
Activity Level.
Abbreviations: ES, effect size; WPC, weekly phone calls; TDTM, twice-daily text messages.
Participants were grouped into “more active patients” (MA patients; average baseline weekly step count > 4220) and “less active patients” (LA patients; average baseline weekly step count ⩽ 4220); the cutoff value was the median and divided each group into 2 equal parts. Changes were more pronounced and the directionality of change was opposite for the 2 interventions (Figure 2). For the weekly phone calls intervention, MA patients increased (ES = 0.08) and LA patients decreased (−0.12) their average weekly step counts between their baseline week (ie, week 0) and their third week. For the twice-daily text messages intervention, MA patients decreased (ES = −0.09) and LA patients increased (0.39) their average weekly step counts between their baseline week and their third week.
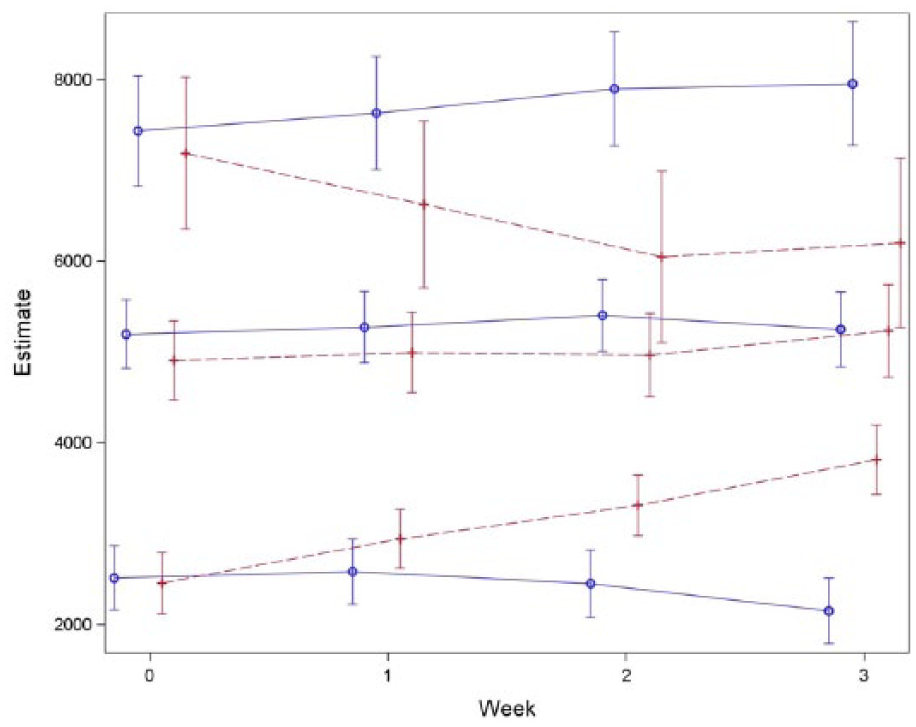
Repeated-measures model comparing weekly step counts between WPC and TDTM groups.
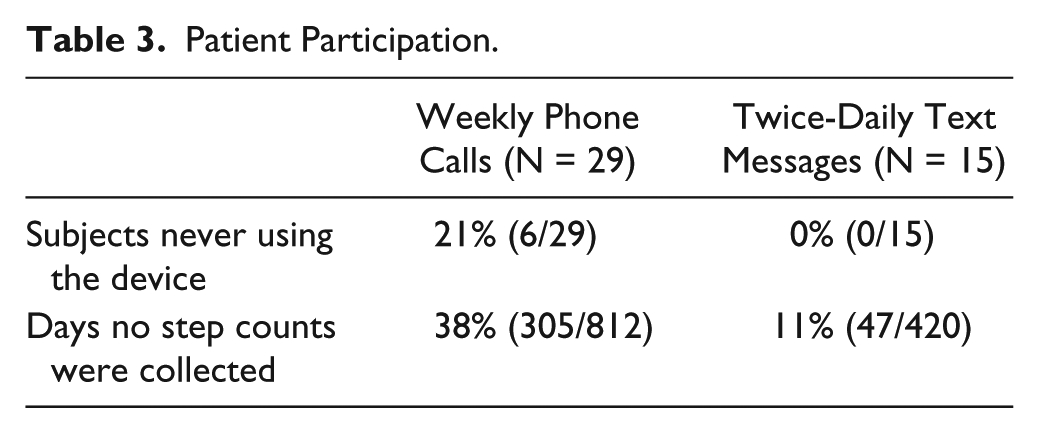
There was higher participation and less missing step count data in the twice-daily messaging group (Table 3). In the weekly phone call group, 6 patients (21%) did not provide any step count data. All patients provided step count data in the twice-daily text messaging group.
Patient Participation.
Evaluating responses to feedback questionnaires from patients in the twice-daily text messaging group (Table 4), most patients reported that they found the intervention helpful (92%), intend to continue tracking their activity (75%), and would participate in another PA study (85%). By contrast, most patients were not interested in participating in group activities (83%). Patients most frequently reported preferring text messaging once/day and at noon.
Feedback Survey for Patient-Centered Activity Regimen.

Discussion
This study has 3 important findings. First, a multifaceted activity regimen prioritizing patient education and twice-daily gain-framed text messaging improves patient participation compared with a weekly phone call-based regimen. Second, subjects participating in the PCAR were more likely to increase their PA compared with those receiving weekly phone calls, and patients who were less active at baseline showed the most improvement. Third, patients in the PCAR group reported high satisfaction with the intervention. These findings suggest that, compared with weekly phone calls, PCAR may be a more efficacious mechanism to implement PA in patients with advanced lung cancer.
This PCAR aims to address the limitations to PA interventions in lung cancer. Though PA interventions benefit patients with lung cancer, 13 PA implementation is limited by inconsistent adherence. Two possible explanations for low adherence are lack of a motivational component or the use of activity regimens that are inconvenient or impractical for patients. Recently, several trials have reported positive results with supervised PA interventions.23-25 To be sure, supervised PA interventions are able to implement more intense PA in a safe setting. However, the frequency of PA sessions is limited, travel burden is placed on the patient, and increases in activity outside the activity sessions are not assured. For example, a recent randomized trial implementing weekly activity sessions in advanced lung cancer showed reasonable adherence (69%), though no significant change in unadjusted daily moderate-vigorous activity at 6 months (as measured by accelerometers). 26 A patient-centered regimen that is home-based, low-intensity, and motivational is well suited to overcome these important practical barriers.
Since patients with advanced-stage lung cancer have a heavier symptom burden, 27 they may have greater improvements with PA interventions.28,29 However, few studies have implemented PA regimens in patients with advanced-stage lung cancer. 13 Recent literature reviews have reported both safety and feasibility of PA interventions in patients with advanced cancer and lung cancer.13,30 Though PA interventions are likely harder to implement in “sicker” patients, many studies suggest that patients with lower preintervention function may obtain the most benefit.28,31
Gain-framed text messaging is a unique way to motivate patients with advanced-stage lung cancer. Gain-framed messages refer to messages that emphasize the benefits of a certain behavior. 32 By contrast, loss-framed messages emphasize the consequences of not performing the same behavior and are sometimes used to make patient recommendations. Consistent with the tents (tenets) of prospect theory, gain-framed messaging has been shown to improve smoking cessation rates 32 and increase PA in older men, 33 healthy women, 34 patients in cardiac rehabilitation, 35 and overweight patients. 36 Since patients with advanced-stage lung cancer frequently have a life expectancy <2 years, they may not be motivated by potential negative outcomes. To our knowledge, the use of gain-framed messaging to motivate patients with advanced-stage lung cancer has not been previously reported.
In addition to showing that PCAR is an effective way to motivate patients with advanced-stage lung cancer toward PA, this study also provides several potential insights regarding PA implementation in this group. Figure 1 suggests that there are “more active” and “less active” groups in both the PCAR and weekly phone call cohorts. Though the overall (ungrouped) PA was unchanged, the “less active” group significantly increased their step count using PCAR, suggesting that the “more active” and “less active” groups may respond differently to motivational techniques. Another interesting finding is the potential positive correlation between baseline average daily step count and subsequent PA (see Figure 1). Clinically, it seems likely that subjects with lower baseline PA tolerance may not be able to achieve the same goals as patients with higher baseline PA. It is also possible that patients in the “more active” group may benefit from maintaining their current activity level (rather than increasing it). Overall, the findings of this study suggest that a “one-size-fits-all” approach to PA implementation in patients with advanced lung cancer may not be the most effective. Indeed, it would be worthwhile for future investigations to examine tailored interventions.
Finally, this study provides some guidance as to how patients would like PA recommendations provided via text messages. Almost all (92%) of patients reported finding PCAR helpful and were willing to participate in another activity study. Over half (55%) of patients preferred 1 text message/day that is delivered at noon, and most patients were not interested in group physical activities. Though these data provide some guidance for future messaging studies, the varied questionnaire response also suggest that motivational strategies (much like PA goals) may need to be individualized.
From a clinical perspective, knowing which patients are more likely to participate in low-impact, home-based exercise regimens would be very useful. Since this study suggests that patients respond differently to motivational strategies, future studies should evaluate the predictors of participation as well as potential clinical benefits of a PCAR. There are many patient and treatment factors that may influence participation in activity interventions in advanced-stage lung cancer (eg, baseline PA, smoking history, cancer treatment, depression, etc). Predictors to participation could then be used to design a clinical trial.
The strength of this study is the unique approach to motivating patients with advanced-stage lung cancer. Since adherence to PA studies in this group of patients is challenging, a technique that is patient-centered, improves participation, and has high patient satisfaction may be very clinically useful.
There are several weaknesses to this study. First, it is not clear what intervention component led to improvements in patient participation. In contrast to weekly phone calls, PCAR utilizes a different accelerometer, more frequent motivational strategies, and gain-framed text messaging. Second, the interventions were not run concurrently, and confounding variables could contribute to the study’s findings. Third, feedback questionnaires were not provided to the group of patients receiving telephone-based recommendations, so patient interest between the 2 strategies cannot be compared. Fourth, both patients receiving active treatment and survivors were included. It is possible that these 2 groups respond uniquely to PA interventions or motivational techniques.
Conclusions
A PCAR using patient education, wrist-bound accelerometers, and twice-daily gain-framed text messages improves patient participation and likelihood to improve PA compared with a weekly telephone-based system. Since PA interventions in patients with advanced-state lung cancer suffer from inconsistent adherence, a technique that improves participation may contribute to improved outcomes. Since both patient participation and feedback were favorable, PCAR should be used in a randomized controlled trial to evaluate for improvements in symptom burden, QoL, and mood.
Footnotes
Author Contributions
BCB contributed to study design, data acquisition, data analysis, and manuscript writing. JMH, AP, AMR, BAT, and GAS contributed to study design, data analysis, and manuscript revision. BTB contributed to study design, data acquisition, and manuscript revision. All authors have reviewed and approved this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded, in part, by the National Institutes of Health (NIH) Clinical and Translational Science Award (CTSA) program (NCATS Grant Number #UL1TR001450) and the Yale SPORE in Lung Cancer (P50 CA196530). This study was also supported by the South Carolina Clinical and Translational Research Institute with an academic home at the Medical University of South Carolina (NIH/NCATS Grant Number UL1TR000062). The contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIH.
