Abstract
Purpose:
This study was developed to evaluate the effects of moxibustion on tumor microenvironmental hypoxia in a murine model of Lewis lung carcinoma (LLC).
Methods:
Twenty-four tumor-bearing mice were randomized into tumor group (T), tumor + cisplatin group (TC), tumor + moxibustion group (TM), and tumor + cisplatin + moxibustion group (TMC) (n = 6/group). Six age-matched C57BL/6 mice were employed as control group (Ctrl). A tumor model was established by implanting LLC cells into the right flank of each mouse. Animals in the TM group received moxibustion treatment at the ST36 (bilateral) and GV4 acupoints on the day of visible tumor formation. Moxibustion treatment was performed every other day for a total of 7 sessions. Animals in the TC group were intraperitoneally injected with cisplatin (3 mg/kg) on day 3 after visible tumor formation, and this treatment was performed every 3 days for 4 times. Animals in the TMC group underwent combined moxibustion and chemotherapy treatment, following the same conditions as outlined above. Following treatment, the concentrations of hypoxia-inducible factor-1α (HIF-1α), vascular endothelial growth factor (VEGF), CD31, and Ki67 were measured using ELISA, Western blot, and immunohistochemical staining.
Results:
Compared to the tumor group, treatment in the TM, TC, and TCM groups resulted in varying reductions in tumor growth (P < .001 or P < .05), while tumor microenvironmental hypoxia was alleviated as evidenced by the downregulation of HIF-1α, VEGFA, and CD31(P < .001-P < .05).
Conclusion:
Our results suggest that a combined approach of moxibustion and cisplatin can alleviate intratumoral hypoxia, promote vascular normalization, and slow the growth of LLC tumors in mice.
Introduction
Hypoxia is defined by a lack of access to a sufficient oxygen supply, and it is a common finding in the tumor microenvironment (TME), as the rapid uncontrolled growth of most solid tumors ultimately restricts the availability of oxygen to many of these malignant cells. 1 A hypoxic TME is also closely linked to the aggressive growth and metastatic progression of many types of cancers. 2 As hypoxia is an extremely common characteristic of solid malignant tumors, activating hypoxia-inducible factors (HIFs) is nearly universal across various tumor types. HIF-1, functioning as the master regulator of cellular responses to hypoxic conditions, can promote the transcriptional activation of a series of target genes associated with proliferation, survival, glucose metabolism, angiogenesis, invasion, and metastasis.3-5 HIF-1α is a HIF-1 subunit, and its accumulation promotes VEGF upregulation, which is associated with angiogenic activity under hypoxic settings. 5 This HIF/VEGF signaling activity in response to hypoxia promotes angiogenic activity that can facilitate metastatic progression. 2
Moxibustion is a traditional Chinese medicine (TCM) practice common in many Asian nations. In this noninvasive procedure, the herb Artemisia vulgaris is burned on or above specific acupoints, which warms the area and helps to alleviate associated symptoms. 6 Moxibustion has been reported to inhibit tumor growth, 7 and to help reduce cancer-related fatigue 8 and pain, 9 in addition to reducing radiotherapy and chemotherapy side effects. 10 The therapeutic efficacy of moxibustion is attributed to warming stimulation, 11 which promotes blood circulation, enhances the local blood supply, and helps to mitigate ischemia and hypoxia. However, it remains to be seen whether moxibustion, when conducted at specific acupoints, can alleviate the ischemic hypoxic conditions within the TME and thereby reduce the invasiveness of associated tumor cells.
Moxibustion is commonly applied in cancer patients following surgery or concurrently with or following chemoradiotherapy. 12 Therefore, in this study, we aim to evaluate the combined effects of moxibustion and cisplatin chemotherapy on HIF-1α and VEGF expression within the TME using a murine model of Lewis lung carcinoma LLC. We hypothesize that moxibustion can alleviate intratumoral hypoxia, promote vascular normalization, and consequently reduce the aggressive growth of these malignancies.
Materials and Methods
Reagents
This study utilized Cisplatin (P4394) from Sigma-Aldrich (USA), high-glucose DMEM (10-013-CVRC), and Penicillin-streptomycin (ZY90307) from Corning, 0.25% EDTA (25200-056) and Fetal Bovine Serum (FBS) (10099141) from Gibco, and ELISA kits specific for HIF-1α (ab275281), VEGF (ab209882), and CD31 (ab204527) from Abcam. We also sourced antibodies for HIF-1α (ab228649), VEGFA (ab51745), CD31 (ab281583), Ki67 (ab16667), and beta Actin (ab8226) from Abcam.
Cell Culture
We cultured LLC cells, sourced from the Cell Bank of Shanghai Institutes for Biological Sciences (Chinese Academy of Sciences), in DMEM supplemented with 10% FBS and penicillin-streptomycin. We maintained these cells in a 37°C humidified 5% CO2 incubator at the Laboratory of Acupuncture and Tuina, Shanghai University of Traditional Chinese Medicine.
Animal
Female C57BL/6 mice (6-8 weeks old, 18-22 g) from Beijing Wei Tong LiHua Laboratory Animal Technology Co., Ltd., Beijing, China, were housed under controlled conditions (24°C, 40%-50% humidity, 12 hours light/dark cycle) with ad libitum food and water access. The Guidelines for the Care and Use of Laboratory Animals published by the Ministry of Science and Technology of China were used to conduct all animal studies, which received approval from the Animal Care and Use Committee of Shanghai University of Traditional Chinese Medicine (Permit Number: PZSHUTCM220711024). Twenty-four tumor-bearing mice were randomized into tumor group (T), tumor + cisplatin group (TC), tumor + moxibustion group (TM), and tumor + cisplatin + moxibustion group (TMC) (n = 6/group). Six age-matched C57BL/6 mice were employed as control group (Ctrl).
Tumor Model Establishment
After 1 week of acclimatization, mice were subcutaneously implanted in the right flank with 1×105 LLC cells in 100 μL of PBS. 13 Tumors were measured with digital calipers. Tumor volumes (mm3) were measured with the following equation: (length×width2)/2. 14 One week after tumor implantation, the mice were randomly assigned into different groups when the tumors became palpable. On day 21, mice bearing LLC tumors were weighed before euthanasia following deep anesthetization with 4% isoflurane in oxygen. Tumors were subsequently excised, weighed to calculate net body weight (body weight-tumor weight), and imaged alongside a ruler.7,15 We also assessed it by Tumor Growth Inhibition Rate (TGI). TGI was calculated as follows: TGI, % = (VT–VTM or VTC or VTMC) × 100/VT, where VT, VTM, VTC, and VTMC mean tumor volume (mm3) in T, TM, TC, and TMC groups, respectively. 15
Moxibustion and Cisplatin Treatment
From the day of visible tumor formation (ie, day 7), mice in the TM and TMC groups received moxibustion treatment at 2 points: the bilateral “Zusanli” acupoint (ST36, situated 2 mm lateral to the anterior tubercle of the tibia in the anterior tibial muscle and 4 mm distal to the bottom of the knee joint), and the “Mingmen” acupoint (GV4, located on the subspinous process of the second lumbar spine on the mid-dorsal line). Stimulation was performed 10 minutes per acupoint every other day for 14 days (7 sessions). 16 Briefly, mice were immobilized using a homemade moxibustion fixer, and the distance between the skin overlying the target acupoints and the lit end of the moxa sticks (length: 120 mm, diameter: 12 mm, Nanyang Hanyi Moxibustion Technology Development Co., Ltd., China) was controlled to within 2 to 3 cm. The distance was adjusted using an electronic temperature meter to maintain the temperature of acupoints in the 40°C ± 2°C range. Meantime, mice in the Ctrl, T, and TC groups were handled for 30 minutes.
Animals in the TC and TMC group were intraperitoneally injected with cisplatin (dose: 3 mg/kg, with 1 mg cisplatin powder diluted in 5 mL 0.9% sodium chloride 7 ) on day 3 after visible tumor formation (ie, day 10) and this treatment was performed every 3 days for 4 times. Meantime, mice in the Ctrl, T, and TM groups were intraperitoneally injected with an equal amount of 0.9% sodium chloride.
All treatment was performed at room temperature (25°C ± 1°C).
ELISAs
Following centrifugation, serum samples were stored at −80°C. Commercial ELISA kits were used to quantify serum HIF-1α, VEGF, and CD31 concentrations based on provided instructions.
Western Blot
The protein was extracted from tumor tissue samples by lysis buffer supplemented with a protease inhibitor cocktail (Pierce). These lysates were then separated via 10% SDS-PAGE, transferred to PVDF membranes (Millipore, MA, USA), and the blots were blocked for 60 minutes using 5% non-fat milk at room temperature. Blots were then probed overnight with antibodies specific for VEGFA, CD31, or HIF-1α at 4°C, followed by treatment with secondary antibodies conjugated to HRP. We subsequently employed a Western blotting detection system to identify protein bands, adhering to the instructions provided by the manufacturer.
Immunohistochemical (IHC) Staining
We performed IHC staining on paraffin-embedded tissue sections (4-5 μm). Briefly, these sections were deparaffinized, rehydrated, blocked, and probed overnight with antibodies specific for VEGFA, CD31, Ki67, or HIF-1α at 4°C prior to probing with streptavidin HRP. DAB (Santa Cruz Biotechnology, CA) was then used for color development, and slides were counterstained with hematoxylin prior to imaging with an Olympus IX81 microscope.
Statistical Analysis
GraphPad Prism 8 was used to analyze all data reported as the mean ±SEM. Results were compared with 2-sided Student’s t-tests or one-way ANOVAs, with P < .05 as the threshold for statistical significance.
Results
Moxibustion Combined With Cisplatin Inhibited Tumor Growth in LLC-Bearing Mice
We dynamically assessed the tumor growth curve in tumor-bearing mice treated with moxibustion, cisplatin, and moxibustion + cisplatin. As shown in Figure 1c, the mean tumor size was 55.30 ± 2.82 mm3, and there was no difference in the tumor volume between the groups before the intervention (ie, day 7). Starting from day 13, the three treatment groups showed a noticeable decrease in tumor growth rate and a clear reduction in tumor size compared to the T group (P < .001-P < .01), an effect that persisted until day 19 (P < .001-P < .05). By day 21, tumor weight values in the three treatment groups were significantly reduced compared to the T group (P < .01-P < .05) (Figure 1d). Moreover, compared to the T group, tumor volumes in the TM, TC, and TMC groups were significantly reduced (all P<0.01) such that the respective calculated degrees of tumor inhibition were 42.44%, 49.36%, and 58.72% (Table 1, Figure 1e).

The effect of combinatorial moxibustion and cisplatin treatment on tumor growth in LLC-bearing mice. (a) Flowchart of experimental interventions. (b) Tumor growth curve analysis of the LLC-bearing mice in each group at the corresponding time points. Compared to T group, *P < .05. **P < .01. ***P < .001 (n = 6). (c) Images of the dissected tumors from each group (n = 6). (d) Tumor weight after 14 days of treatment. Compared to T group, *P < .05. **P < .01 (n = 6). (e) Tumor volume after 14 days of treatment. Compared to T group, **P < .01 (n = 6). (f) and (g) Expression of Ki67 in tumor cells. Compared to T group, ***P < .001 (n = 6). Data were expressed as mean ± SEM.
Comparison of Tumor Volume and Inhibitory Rate Before and After Treatment (mean ±SEM).

Compared to T group, *P< .05. **P< .01.
Tumor cell proliferation, as reflected by Ki67 staining, was compared in tumor tissue samples of LLC bearing mice. Compared to the T group, Ki67 staining was significantly inhibited in the TM, TC, and TMC groups following the 14 days treatment period (all P < .001) (Figure 1f and g).
Moxibustion and Cisplatin Alter Relative Serum Cytokine Levels
Next, ELISAs were used to quantify serum HIF-1α, VEGF, and CD31 concentrations, revealing that all 3 were significantly upregulated in the T group compared to the Ctrl group (P < .001-P < .01). However, after a 14-day treatment period, these 3 proteins were significantly downregulated in the TM, TC, and TMC groups (P < .001-P < .01), and the combined treatment of moxibustion and cisplatin further inhibited HIF-1α and VEGF levels (both P < .01 vs TC or TM) (Figure 2a-c).
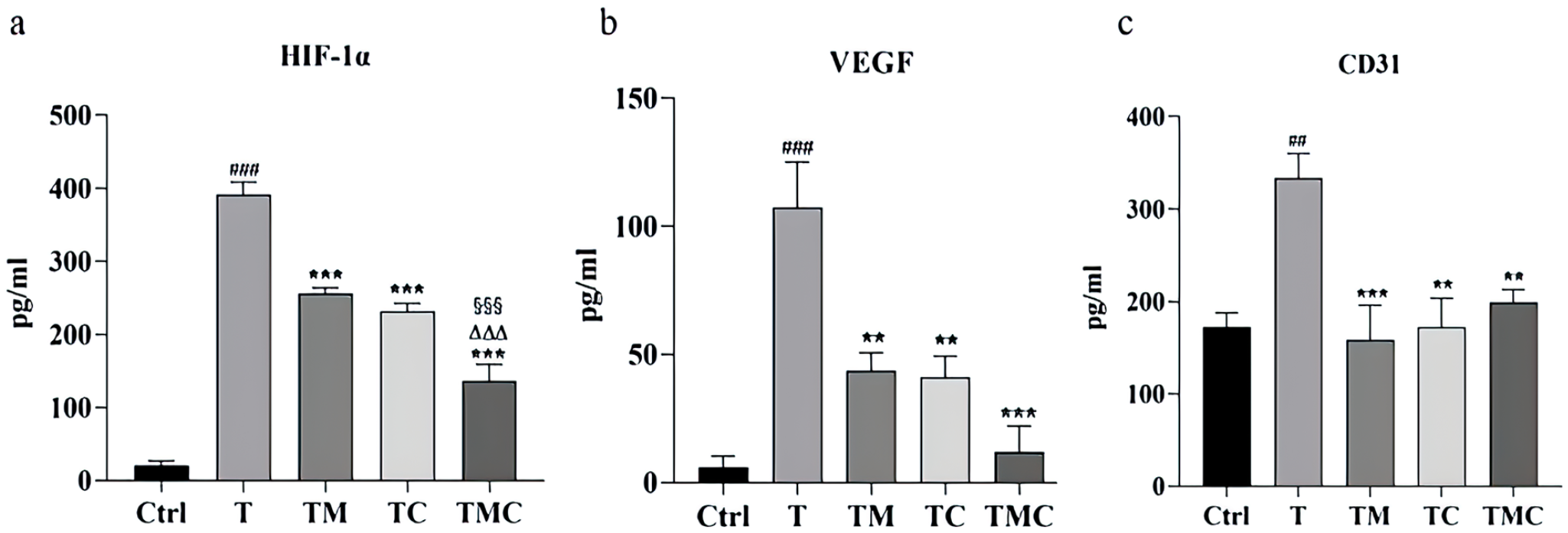
Expression levels of relative serum cytokine in peripheral blood. (a) For Peer Review HIF-1α expression level in serum. Compared to T group, ***P < .001; compared to Ctrl group. ###P < .001; compared to TM group. §§§P < .001; compared to TC group. △△△P < .001(n = 6). (b) VEGF expression level in serum. Compared to T group, **P < .01. ***P < .001; compared to Ctrl group, ###P < .001 (n = 6). (c) CD31 expression level in serum. Compared to T group, **P < .01. ***P < .001; compared to Ctrl group. ##P < .01 (n = 6). Data were expressed as mean ± SEM.
Moxibustion and Cisplatin Synergistically Suppress LLC Tumor Angiogenic Activity
Western blot analysis revealed that CD31, HIF-1α, and VEGFA were strongly expressed in the T group, while their expression was significantly weaker in the TM, TC, and TMC groups (P < .001-P < .05). The combined treatment of moxibustion and cisplatin further inhibited CD31, HIF-1α, and VEGFA expression (all P < .01 vs TC or TM) (Figure 3a-d). Consistently, IHC staining revealed significant inhibition in intratumoral HIF-1α, VEGFA, and CD31 positive expression in the TM, TC, and TMC groups compared to the T group (P < .001-P < .05) (Figure 4a-d).
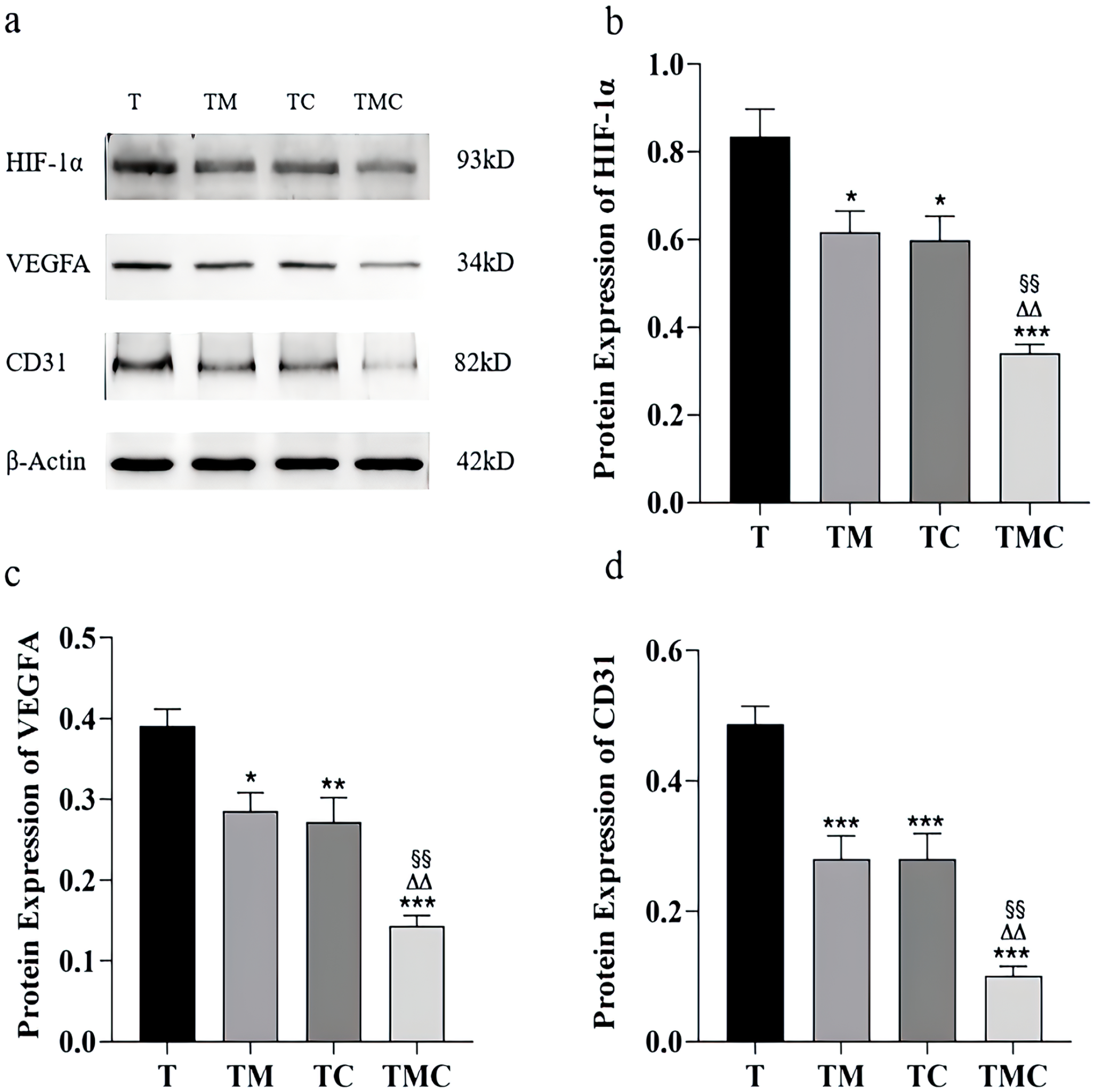
Moxibustion inhibited HIF-1α, VEGFA, and CD31 protein levels in tumor tissue. (a-d) Representative blots showing HIF-1, VEGFA, and CD31 protein levels. Compared to T group, *P < .05. **P < .01. ***P < .001; Compared to TM group, §§P < .01; Compared to TC group, △△P < .01 (n = 6). Data were expressed as mean ± SEM.
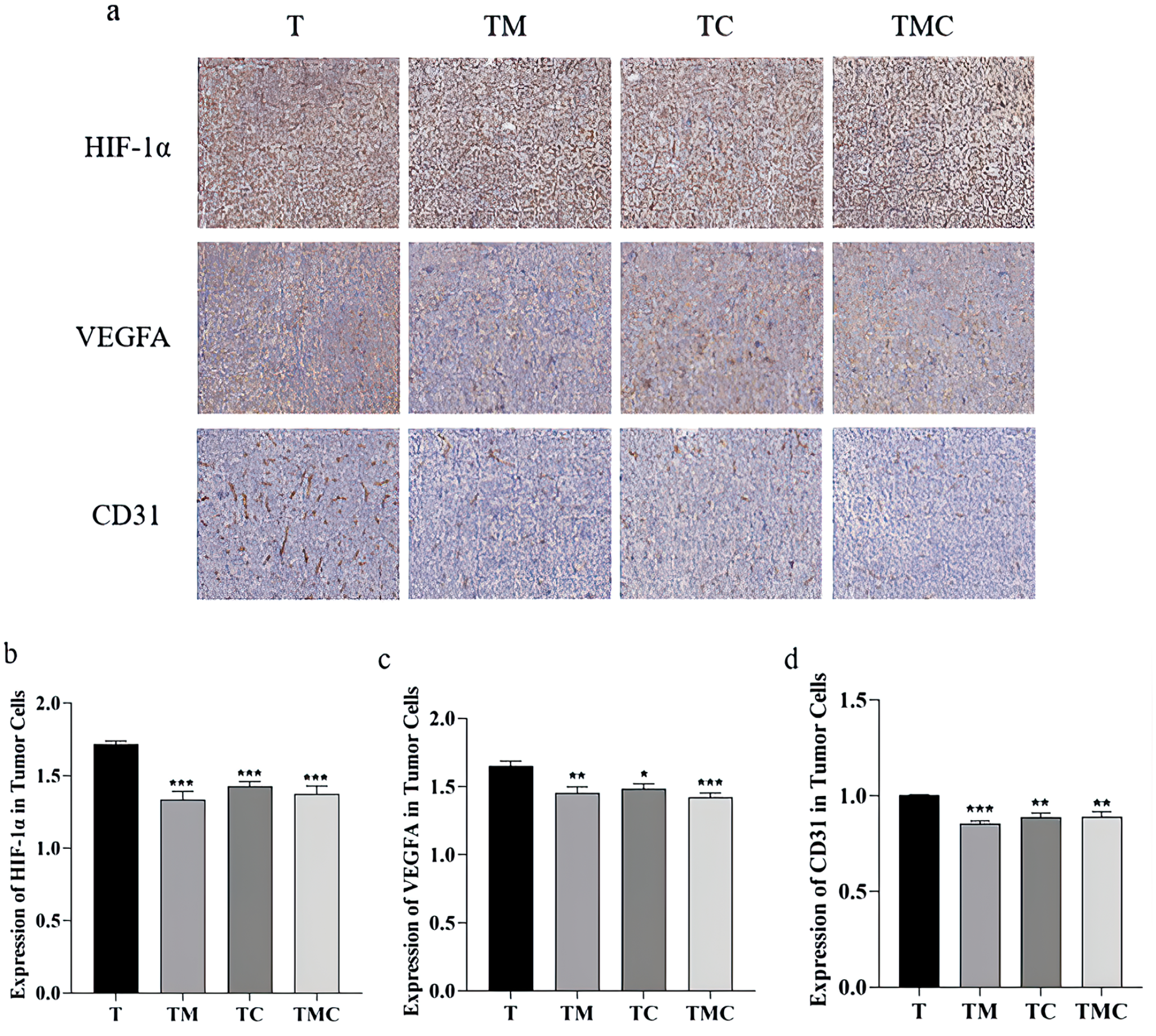
Moxibustion inhibited HIF-1α, VEGFA, and CD31 expression in tumor cells. (a) Representative tumor cross-sections from T, TM, TC, and TMC. The sections were stained with antibodies against HIF-1α, VEGFA, and CD31. 400×. (b-d) Representative Immunohistochemistry showing levels of HIF-1, VEGFA, and CD31 in tumor cells. Compared to T group, *P < .05. **P < .01. ***P < .001 (n = 6). Data were expressed as mean ± SEM.
Discussion
This study was focused on investigating the anticancer effect of moxibustion combined with cisplatin in Lewis lung cancer mice and exploring the possible mechanism of moxibustion on inhibiting tumor growth by alleviating hypoxia in TME.
The present results highlight the therapeutic value of moxibustion, cisplatin, and the combination of the 2, given the significant suppression of tumor growth in the TM, TC, and TMC groups compared to the T group. Consistently, the tumor weight and volume values in these 3 treatment groups were significantly lower, and there was a concomitant drop in the levels of Ki67, which serves as a biomarker of tumor cell proliferative activity. 17 Cisplatin is a broad-spectrum antineoplastic agent widely used in clinical settings to suppress the growth of lung cancer and many other types of malignancies. 18 While effective, the clinical application of cisplatin is limited by its side effects, inherent toxicity, 19 and issues of drug tolerance. 20 Moxibustion offers the combined benefits of suppressing tumor growth7,21,22 while alleviating chemotherapeutic drug side effects. 12 These data thus indicate that moxibustion can suppress lung tumor growth. Strikingly, moxibustion and cisplatin treatment can synergize to improve therapeutic outcomes.
Our study found significant increases in serum HIF-1α and VEGF levels in the LLC-bearing mice. This supports the hypoxic TME associated with these solid tumors, as reported previously.1,2,23 Such intratumoral hypoxia can increase HIF-1α expression, thereby promoting VEGF upregulation and ultimately promoting angiogenic activity conducive to further tumor growth.24-26 The malignant activity is also directly related to adverse effects including metastatic progression and therapeutic drug resistance.7,8,27 Angiogenesis is regulated by various factors, with VEGF being significantly important. 28 CD31 is highly expressed on the surface of endothelial cells and is well established as a marker for monitoring vessel density in malignant tissue. 29 VEGF and CD31 are well-defined markers of angiogenesis. VEGFA is involved in the growth of blood vessels and lymphatic vessels, and positive VEGFA expression usually indicates tumor metastasis or poor prognosis. 30 In this study, VEGFA was used as a marker of VEGF to detect neovascularization in tumor tissues specifically. Therefore, the key to moxibustion’s anticancer effect might be alleviating TME hypoxia and promoting vascular normalization.
Following moxibustion, cisplatin, or combined treatment, serum and tumor HIF-1α and VEGF levels fell significantly in these mice relative to those in the T model group. Noticeably, the TMC group showed a significant decrease in serum HIF-1α expression and intratumoral HIF-1α and VEGF levels compared to the TM and TC groups. Here, mice in the 3 treatment groups exhibited significantly lower serum and intratumoral CD31 expression than those in the T group. Notably, CD31 levels in these target tumors were significantly lower in the TMC group than in the TM and TC groups. Therefore, our results indicated that cisplatin and moxibustion could potentially disrupt tumor vascular development by mitigating the hypoxic conditions of the TME. Furthermore, these 2 treatment approaches synergistically enhance the anti-hypoxic effect, leading to improved outcomes.
Our study shows that either moxibustion alone or combined with cisplatin can constrain tumor growth by suppressing proliferation, consistent with prior evidence.7,21,22 Our investigation yielded different results in contrast to prior studies suggesting that moxibustion can stimulate the co-regulation of tumor vascular normalization and immune reprogramming 7 without inhibiting HIF-1α expression in the TME—likely due to their limited sample size (n = 3), which could introduce bias. Our study is the first to discern that either stand-alone or paired with cisplatin, moxibustion can inhibit HIF-1α expression in both TME and serum of LLC-bearing mice. These findings imply that moxibustion has the potential to enhance systemic circulation, subsequently improving local blood supply and reducing the likelihood of ischemia and hypoxia. Hence, alleviating hypoxia within the TME might be critical to moxibustion’s capability to impede tumor invasion.
In traditional Chinese medicine, hypoxia is considered Qi and blood deficiency, while ST36 and GV4 are commonly used in clinical practice to treat Qi and blood deficiency. Moxibustion at ST36 is widely used to improve the quality of life among cancer patients8,31,32 or inhibit tumor growth in tumor-bearing mice.7,21,22 In our study, moxibustion on ST36 and GV4 acupoints in these LLC-bearing mice significantly suppressed tumor growth. Alleviating the hypoxia in the TME might be the key mechanism by which moxibustion inhibits tumor invasion, associated with its effect on warming stimulation and promotion of blood circulation. 22 Overall, moxibustion thus exhibits excellent potential for its application as a treatment for cancer that specifically targets the hypoxic TME.
Conclusions
In summary, our study reveals that the co-administration of moxibustion and cisplatin effectively curtails tumor proliferation by mitigating intratumoral hypoxia. Reductions in HIF-1α, VEGF, and CD31 levels were observed across all treatment groups, indicative of angiogenesis suppression. The data thus provide evidence that the conjunction of cisplatin and moxibustion can suppress HIF-1α expression within the TME more potently, disrupting angiogenesis and thereby obstructing tumor growth. Incorporating this dual therapeutic approach in clinical practice could thus hold substantial potential for enhancing the prognosis of patients with lung cancer.
Footnotes
Authors’ Contributions
Conception and design by J.W. The method was developed by J.W. and H.M. Animal experiments were done by N.M., X.W., and C.W. Data analysis was performed and interpreted by J.W. and N.M. H.M. and N.M. wrote the manuscript. J.W. and H.M. wrote the reviewed and revised manuscript.
Availability of Data and Material
The data supporting this study’s findings are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is partly funded by grants from the National Natural Science Foundation of China (grant number 82274640) and the National Natural Science Foundation of Shanghai (grant number 21ZR1463300).
Ethics Approval
Approval from the Animal Care and Use Committee of Shanghai University of Traditional Chinese Medicine (Permit Number PZSHUTCM220711024).
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Code Availability
Not applicable.
