Abstract
Extracts from Euglena gracilis have been shown to prevent cancer growth in mouse models. However, the molecular mechanism of this anti-cancer activity has not been determined nor has the effect of Euglena extracts on tobacco smoke carcinogen-induced carcinogenesis. Here, we investigate the hypothesis that this anti-cancer activity is a result of changes in the intestinal microbiota induced by oral administration of the extract. We found that a Euglena gracilis water extract prevents lung tumorigenesis induced by a tobacco smoke-specific carcinogen (NNK) in mice treated either 2 weeks before or 10 weeks after NNK injection. Both of these treatment regimens are associated with significant increases in 27 microbiota metabolites found in the mouse feces, including large increases in triethanolamine, salicylate, desaminotyrosine, N-acetylserine, glycolate, and aspartate. Increases in the short-chain fatty acids (SCFAs) including acetate, propionate and butyrate are also observed. We also detected a significant attenuation of lung carcinoma cell growth through the induction of cell cycle arrest and apoptosis caused by low levels of SCFAs. This study provides strong evidence of anti-cancer activity in Euglena gracilis extracts against tobacco smoke carcinogen-induced tumorigenesis and demonstrates that this activity is linked to increased production of specific gut microbiota metabolites and the resultant induction of cell cycle arrest and apoptosis of lung carcinoma cells.
Introduction
Lung cancer is one of the most commonly diagnosed cancers in both men and women and the leading cause of cancer-related deaths worldwide. 1 An estimated 130 180 Americans are expected to die from lung cancer in 2022, accounting for over 21% of all cancer-related deaths. 2 Tobacco smoke is the leading cause of lung cancer and associated deaths due to long-term exposure to carcinogens. Compared with non-smokers, smokers have as much as a 14-fold increased risk of developing lung cancer.3,4 However, lung cancer also remains a leading cause of cancer-related death among non-smokers. 5 Although many preventive and therapeutic strategies are being implemented to limit the casualties, lung cancer patients still have a lower rate of survival than any other cancer. 6 Therefore, the development of novel and efficacious preventative, diagnostic, and therapeutic strategies against lung cancer is urgently required.
Traditional therapeutic options for cancer can be limited by the severity of the side effects, and cancer prevention has focused on lifestyle changes, which encounter significant barriers. Some traditional therapeutics, such as paclitaxel, have been derived from natural products. Many more natural products have been popularly touted for cancer prevention or treatment, but few have demonstrated any significant efficacy in scientific studies. Despite this, natural products remain a hopeful source for the development of cancer therapeutics and preventatives. Euglena gracilis (E. gracilis), a unicellular micro-alga, is found in both fresh and saltwater and has features of both animals and plants. This alga is used as a nutritional and functional dietary supplement and has also been shown to alter the intestinal microbiome and modulate host immunity. 7 It has been reported that whole E. gracilis and its main component, paramylon (a water-insoluble granule or linear form of β-1,3-glucan), have medicinal properties, such as antimicrobial, 8 anti-viral,9-11 and antitumor12-14 activities. In addition, our previous study showed that the partially purified extract derived from E. gracilis (EWE), devoid of water insoluble mature paramylons, prevents lung tumor growth in mice by stimulating host antitumor immunity through the attenuation of myeloid-derived suppressor cell (MDSC) populations. 15 These results suggest that E. gracilis contains multiple bioactive components, not only paramylon. E. gracilis, therefore, has potential as a source of natural products that might improve the efficacy of therapeutic strategies against diseases such as lung cancer and aid in their prevention.
There is an increasing interest in modulation of the gut microbiota using prebiotics and probiotics to develop preventative and therapeutic strategies against cancer. The gut microbiota plays a role in the modulation of the host immune system by producing bioactive microbiota-derived metabolites. Short-chain fatty acids (SCFAs) are one of the typical metabolites produced by microbiota. It has been reported that SCFAs can regulate host physiological functions and influence the development of immune and inflammatory responses, both locally and at distant sites, such as in the lungs, and by playing a role in lung diseases such as asthma, chronic obstructive pulmonary disease (COPD), and lung cancer.16-19 A recent study reported that the intake of dry powder of E. gracilis alters microbiota composition in the human gut and increases the production of butyrate, a SCFA, resulting in improved digestive health and increased rate of defecation. 7 Therefore, the use of natural products that target the alteration of gut microbiota and metabolites could open a new avenue for cancer prevention and/or therapy.
Our recent study demonstrated that oral administration of either a partially purified water extracts from E. gracilis (EWE, the term EWE is used to indicate the water extract from Euglena gracilis) or boiled EWE (bEWE, the term bEWE is used to specifically indicate that the EWE was obtained after an additional boiling step in the extraction process) alters intestinal microbiota compositions, thereby attenuating the growth of lung tumors in mice. 20 However, the detailed mechanisms by which Euglena water extract induced this alteration have remained unknown. Additionally, whether EWE is also effective in inhibiting tobacco smoke carcinogen-induced lung cancer, which more closely mimics human clinical lung cancer,21-24 has not previously been studied.
Here, we report that bEWE prevents tumor growth in a mouse model of lung cancer induced by a tobacco-specific carcinogen, 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK). Since this NNK-induced lung cancer mouse model mimics human lung adenocarcinoma in morphology and mutation characteristics,21-24 this study is likely to have relevance for cancer in humans. In this study, high-throughput metabolic profiling revealed that oral administration of bEWE significantly increased intestinal microbial metabolites: short-chain fatty acids (SCFAs) and their precursors. In addition, treatment with the SCFAs significantly suppressed proliferation of both human and murine lung cancer cell lines in cell culture via induction of apoptosis, suggesting the anti-cancer effects of bEWE are attributable to changes in the intestinal metabolites.
Methods
Animals
All mice were housed humanely according to university, state, and federal guidelines (AAALAC) in the AAALAC-accredited animal resource facilities of the Kansas State University College of Veterinary Medicine. All the procedures for the handling of animals were approved by the Kansas State University Institutional Animal Care and Use Committee (Protocol # 4346) and Institutional Biosafety Committee (Protocol # 1433). Wild-type male and female A/J mice were purchased from Charles River Laboratories International, Inc. and were housed in a clean facility with controlled humidity and temperature on 12-hour light-dark cycles. The temperature of the room was set at 20°C to 26°C and the relative humidity was 30% to 70%. Before treatment, all the mice were acclimatized in the facility for a week. The mouse condition was observed every day and their body weights were monitored every 3 days. The study is being reported in accordance with ARRIVE guidelines.
Reagents
4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) was purchased from Thermo Fisher Scientific (Waltham, MA, USA). Short-chain fatty acids (sodium acetate, sodium butyrate, and sodium propionate) were from Sigma-Aldrich (St. Louis, MO, USA). Fluorescent conjugated antibodies targeting Annexin V and 7-aminoactinomycin D (7-AAD) were purchased from BioLegend (San Diego, CA, USA).
Cell Culture
The mouse Lewis lung carcinoma (LLC) cell line (CRL-1642) and human H1299 lung carcinoma cell line (CCL-5803) were purchased from American Type Culture Collection (ATCC, Manassas, VA, USA). Dulbecco’s Modified Eagle’s Medium (DMEM) and RPMI 1640 were obtained from Mediatech, Inc. (Manassas, VA, USA). Fetal bovine serum (FBS) was from Biowest (Riverside, MO, USA). The penicillin-streptomycin stock was obtained from Lonza Rockland, Inc. (Allendale, NJ, USA). The cells were cultured at 37°C in a humidified atmosphere containing 5% CO2. The cell lines were authenticated by short tandem repeat (STR) DNA profiling. Both the cells were maintained in low passages (<15) for this study.
Preparation of Euglena Water Extract
The boiled Euglena water extract (bEWE) was prepared using a protocol described previously. 20 Dried powder of Euglena gracilis was obtained from Euglena Co., Ltd. (Tokyo, Japan). It is composed of 29.4% carbohydrates, 42.3% protein, and 19.0% lipid. Approximately 70%–80% of the carbohydrate content is paramylon. 10 Briefly, dried powder of E. gracilis (5 g) was suspended in 40 mL sterile phosphate buffered saline (PBS) and incubated at 37°C for 30 minutes with periodic sonication for 30 seconds (water extraction). The suspension was centrifuged at 11 405g for 20 minutes, removing Euglena cell debris and mature paramylon (insoluble β-1,3 glucan aggregates, 2-8 µm diameter). 15 The supernatant was filtered through one layer of water-soaked Whatman #1 filter paper to remove floating materials (removal of majority of lipids). The filtrate was boiled for 10 minutes (denature the large size structured proteins), following cooling down to the room temperature (accelerating aggregation of the denatured protein). After the centrifugation at 11 405g for 10 minutes (removal of all heat-induced denatured proteins), the supernatant was filtered through a 0.22 μm sterile disk filter (Midwest Scientific, Valley Park, MO, USA) to remove microscopic paramylon particles and potential contamination by microorganisms larger than 0.22 µm diameter. The dry weight was calculated after drying a 400 μL aliquot at 55°C for 48 hours from which the dry weight of PBS was subtracted. The protein and carbohydrate levels from 3 different preparations of the final extract were determined by BCA assay (Pierc BCA Protein Assay Kit, Thermo Fisher Scientific, Waltham, MA, USA) and cysteine-sulfuric acid method, 25 respectively. Assay results indicated that the 1 mg/mL bEWE preparation contained 0.17 ± 0.01 mg/mL protein and 0.0519 ± 0.0003 µg/mL carbohydrate (n = 3). Notably, the protein level of bEWE (0.17 ± 0.01 mg/mL) was much smaller than that of the parent EWE, which is prepared without boiling (0.49 ± 0.12 mg/mL, n = 3). The bEWE was stored at −20°C until further use.
Effect of Orally Administrated bEWE on the Growth of NNK-Induced Lung Tumor
Wild-type male and female A/J mice were administered bEWE as described in Figure 1A. For the induction of lung tumors, the A/J mice (n = 10; 5 males and 5 females per group) were intraperitoneally (i.p.) injected with 100 mg/kg of NNK as described previously. 26 The mice were visually inspected for any signs and symptoms on a routine basis after the NNK injection. After 19 weeks of NNK injection, the mice were euthanized by exposure to saturated CO2 followed by cervical dislocation, and then the lungs were collected. The whole lung was stained by injecting India Black ink solution through the trachea and the tumor nodules in the lungs were counted under the stereo microscope. All of the measurements throughout this study were carried out by a single observer in a blinded fashion. The lungs were fixed in 10% formalin for histological analysis.
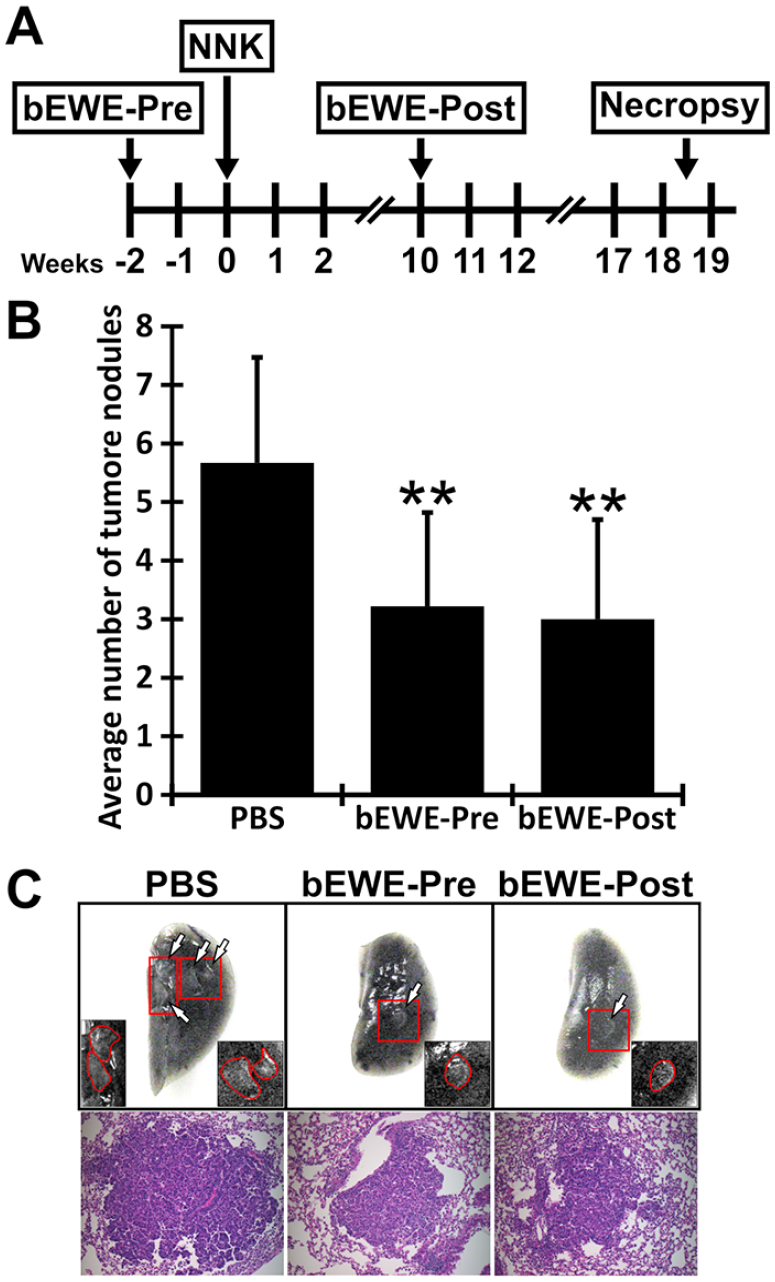
Pre- and post-treatment with bEWE significantly attenuated the growth of NNK-induced lung tumorigenesis in mice. (A) Schematic illustration of the study design. To evaluate the effect of orally administrated bEWE on the growth of NNK-induced lung carcinoma, 3 treatment regimens were performed: (1) PBS control, (2) 90 to 210 mg/kg of bEWE pre-treatment for 2 weeks prior to NNK injection until the end of the experiment (designated bEWE-Pre), (3) 90 to 210 mg/kg of bEWE post-treatment after NNK injection (designated bEWE-Post). PBS or bEWE were administered orally via drinking water. The weight of water pouches was measured every 3 days to monitor the daily intake of the drinking water containing bEWE. Based on the average water consumption (4 mL/mouse/day), the doses of bEWE were adjusted. (B) Average lung tumor nodule numbers in each treatment group. PBS served as a control. Results are presented as mean ±SD (n = 10). **P < .01 as compared to the PBS group. (C) Upper images: Representative lung images from each group. Arrows indicate tumor nodules in the India Black ink-injected lungs and inserts shown are the enlarged views of the tumor nodules. Lower images: Representative HE-stained tumor nodules from each group.
Metabolic Profiling and Absolute Quantification of Short-Chain Fatty Acids
To analyze the profile of primary metabolites in the fecal samples collected from NNK-induced tumor-bearing mice treated with the PBS control or bEWE, fecal samples were collected at the end of the study. These samples were analyzed by automatic liner exchange/cold injection gas chromatography-time of flight mass spectrometry (ALEX-CIS GC-TOF-MS) at the West Coast Metabolomics Center (University of California, Davis, Davis, CA, USA) using their proprietary methods. Similarly, the absolute quantification of SCFAs was determined by a targeted metabolite analysis at the West Coast Metabolomics Center using their proprietary methods. The absolute value of SCFAs in the fecal sample was quantified using authentic SCFA standards.
Following the normalization and filtering using proprietary algorithms, the values obtained were log-transformed and only the metabolites with known annotations were included in the analysis for this study. Multivariate analysis was used to clarify the clustering pattern based on the covariance of the data. The difference in the overall metabolite profile between the groups was visualized by a partial least squares-discriminant analysis (PLS-DA) score plot. The analysis of variance (ANOVA) contrasts and PLS-DA were conducted using the MetaboAnalyst online tool. 27 Based on the variable importance in projection (VIP) score of each metabolite, which was generated from the PLS-DA model, the variable/feature metabolites that were different between the groups were further identified. A VIP score equal to or greater than 1.5 was considered as a different variable/feature between the groups and the difference was visualized by heatmap based on the abundance ratio of metabolites. Further, univariate statistical analysis was conducted to analyze the features that vary significantly between the treatment groups. A Student’s t-test was performed to identify the metabolites that were significantly different between the treatments and the data was plotted in GraphPad Prism (version 9.3.1).
Growth Inhibition of Lung Carcinoma Cells by SCFA in Cell Culture
The murine LLC (2 × 103 cells/well) and human H1299 (2 × 104 cells/well) lung carcinoma cells were seeded into a 24-well plate with a 1 mL growth medium. After 48 hours, the cells were treated with SCFA (sodium acetate, sodium butyrate, or sodium propionate) with concentrations of 1, 5, 10, or 30 mM. PBS was used as the negative control. The effect of the SCFA (1, 5, and 10 mM) on the growth of human normal lung bronchial epithelial cell line, BEAS-2B (CRL-9609, ATCC), was evaluated. Trypan blue staining was performed according to the manufacturer’s instructions and the number of viable cells (trypan blue negative cells) were counted using a hemocytometer under an inverted microscope.
Flow Cytometric Analysis of the Apoptotic Status of Lung Cancer Cells Treated With SCFAs In Vitro
Anti-Annexin V antibodies/7-AAD was used to quantify the percentage of apoptosis in LLC cells treated with SCFA (sodium acetate, sodium butyrate, and sodium propionate). The LLC cells were seeded into a 12-well plate (5 × 104 cell/mL) and incubated with different SCFAs with three different concentrations at 0.1, 1, and 5 mM for 48 hours. The cells were collected and washed with PBS and co-stained with anti-Annexin V antibodies and 7-AAD. The sample was then analyzed by flow cytometry (BD LSRFortessa X-20; BD Biosciences, San Jose, CA, USA) and analyzed by BD FACSDiva software (BD Bioscience). Early apoptotic cells and late apoptotic cells were identified as populations that showed phenotypes of Annexin V+7-AAD− and Annexin V+7-AAD+, respectively.
Flow Cytometric Analysis of Cell-Cycle Arrest of Lung Cancer Cells Treated With SCFAs In Vitro
Cell-cycle (G1, S and G2/M phases) and apoptosis (Sub G1 phase) in LLC cells treated with SCFA (sodium acetate, sodium butyrate, and sodium propionate) was analyzed using flow cytometry. The LLC cells were seeded into a 6-well plate (5 × 104 cell/well) and incubated with different SCFAs with 3 different concentrations at 0.1, 1, and 5 mM for 48 hours. The cells were collected, fixed by 70% ethanol and stained with staining solution containing 0.1% (v/v) Triton X-100, 0.2 mg/mL RNase A and 0.02 mg/mL propidium iodide (PI) in PBS.
Statistical Analysis
The data are presented as mean ± standard deviation (SD) of the mean. The experiment was conducted with multiple sample determinations with several samples. For the global profiling of metabolites in mouse feces, 10 biological repeats were used for each condition. For the targeted quantification of SCFAs, 8 biological repeats were used per treatment condition. The data were analyzed and visualized using MetaboAnalyst software, 27 GraphPad Prism (v9.3.1), and Microsoft Excel (v16.16.27). Any statistical significance was assessed by a 2-tailed unpaired t-test. The following convention for symbols to indicate statistical significance was set. ns: P > .05, *: P < .05, **: P < .01, ***: P < .001, ****: P < .0001.
Results
Oral Administration of Euglena Water Extract Attenuated the Growth of NNK-Induced Lung Tumorigenesis
The antitumor effects of bEWE were investigated using tobacco smoke carcinogen (NNK) -induced lung tumorigenesis in mice. No differences were observed in mouse body weight (Supplemental Figure S1) or water consumption (Supplemental Figure S2) among the 3 mouse groups described in Figure 1A: a PBS control group, a pretreatment group receiving bEWE for 19 weeks (bEWE-Pre), and a post-treatment group receiving bEWE for 9 weeks (bEWE-Post). As shown in Figure 1B, both pre- (3.3 ± 1.6, P < .01) and post- (3.0 ± 1.7, P < .01) treatment with bEWE significantly reduced the numbers of NNK-induced lung tumor nodules in the lung as compared with that of the control PBS (5.7 ± 1.8). It has been reported that a single dose of NNK (100 mg/kg, i.p.) induces tumor formation 6 to 14 weeks after the treatment.28-30 The nodules in the present study were similar to adenomas at early stages of adenocarcinoma (Figure 1C). Typical glandular cells observed in adenocarcinomas were not yet observed. Because bEWE treatments were started 2 weeks before (bEWE-Pre) or 10 weeks after (bEWE-Post) NNK injection, these results indicate that the daily administration of bEWE prevents tobacco smoke carcinogen-induced lung tumorigenesis, even after microtumor formation.
Multivariate Analysis Identified Several Important Fecal Metabolites
The metabolic profiling of fecal samples was conducted in bEWE-treated NNK-induced tumor-bearing mice. Two-dimensional (2-D) partial least squares-discriminant analysis (PLS-DA) scores plot revealed a clear separation of the overall fecal metabolite profile between the bEWE (both pre- and post-treatments) and PBS treatment groups (Figure 2A), indicating that marked metabolomic changes occurred due to bEWE administration. Based on the variable importance in projection (VIP) scores, a total of 25 metabolites appeared to be the most important fecal metabolites for distinguishing the differences between the PBS, bEWE-Pre and -Post treatment groups as shown in the Figure 2B.

The partial least squares discriminant analysis (PLS-DA) score plot and VIP score revealed the alteration of fecal metabolites in NNK-induced tumor-bearing mice treated with bEWE-Pre treatment and bEWE-Post treatment. (A) The 2-D PLS-DA scores plot exhibited differences in metabolite profiles for each group. (B) A variable importance in projection (VIP) plot depicted the 25 most important fecal metabolites for distinguishing the differences between the treatment groups. The significance of the contributors was tested with the VIP score, and a value equal to or greater than 1.5 was used as a threshold of significance. A heat map with an abundance ratio is shown to the right. n = 10 per group.
Oral Administration of Euglena Water Extract Altered the Fecal Metabolite Levels in NNK-Induced Lung Tumor-Bearing Mice
Further comparison of metabolite levels among the individual groups was performed and 27 metabolites were found to be significantly altered in either bEWE-Pre or -Post treated mice when compared with the PBS control group (Figure 3 and Supplemental Figure S3). Of those, 17 metabolites were significantly higher (Figure 3), and 10 metabolites were significantly lower (Supplemental Figure S3) in either bEWE-Pre or -Post treated mice. Triethanolamine, salicylic acid, desaminotyrosine, N-acetyl serine, glycolic acid, aspartic acid, succinate, galactinol, and malate were significantly higher in both bEWE-Pre and -Post treated mice. Similarly, glyceric acid, glutamic acid, deoxycholic acid, adenine, and 4-aminobutyric acid were significantly higher in only bEWE-Post treated mice. Guanine, lactose, and threonine were significantly higher in only bEWE-Pre treated mice (Figure 3). Overall, the metabolic profiles of bEWE-Pre and -Post treated fecal samples were observed to be similar to one another.

Oral administration of bEWE altered the fecal metabolite levels in NNK-induced tumor-bearing mice. The difference in each metabolite level between the 3 groups: PBS, bEWE-Pre (Pre), and bEWE-Post (Post) treatment groups, was compared using a t-test. Results are presented as means ±SD (n = 10).
Euglena Water Extract-Induced Metabolite Changes Strongly Contribute to SCFA Metabolism
As shown in Figure 3 and Supplemental Figure S3, oral administration of bEWE altered microbiota metabolites. It was reported that SCFAs from gut microbiota attenuate lung carcinogenesis. 31 In the present study, therefore, the change in SCFAs and the metabolic pathways involved in production of SCFAs by gut microbes were further investigated. As depicted in Figure 4, malate and succinate are key metabolites for biosynthesis of SCFAs such as propionic acid and butyric acid. 32 Furthermore, amino acids such as aspartic acid, glutamic acid, and threonine can be metabolized to acetate, butyrate, and propionate.33,34 Other metabolites such as glyceric acid, acetylserine, and triethanolamine can also contribute to the synthesis of SCFAs like butyrate and acetate. 35 As shown in Figure 4, the metabolites that are highlighted in blue were increased in either bEWE-Pre or -Post treated mice suggesting the possibility of alteration of SCFA metabolism occurring in the bEWE treated mice. Therefore, the blue-highlighted metabolites could be potentially important intermediate metabolites relevant to EWE-induced prevention of lung tumorigenesis.
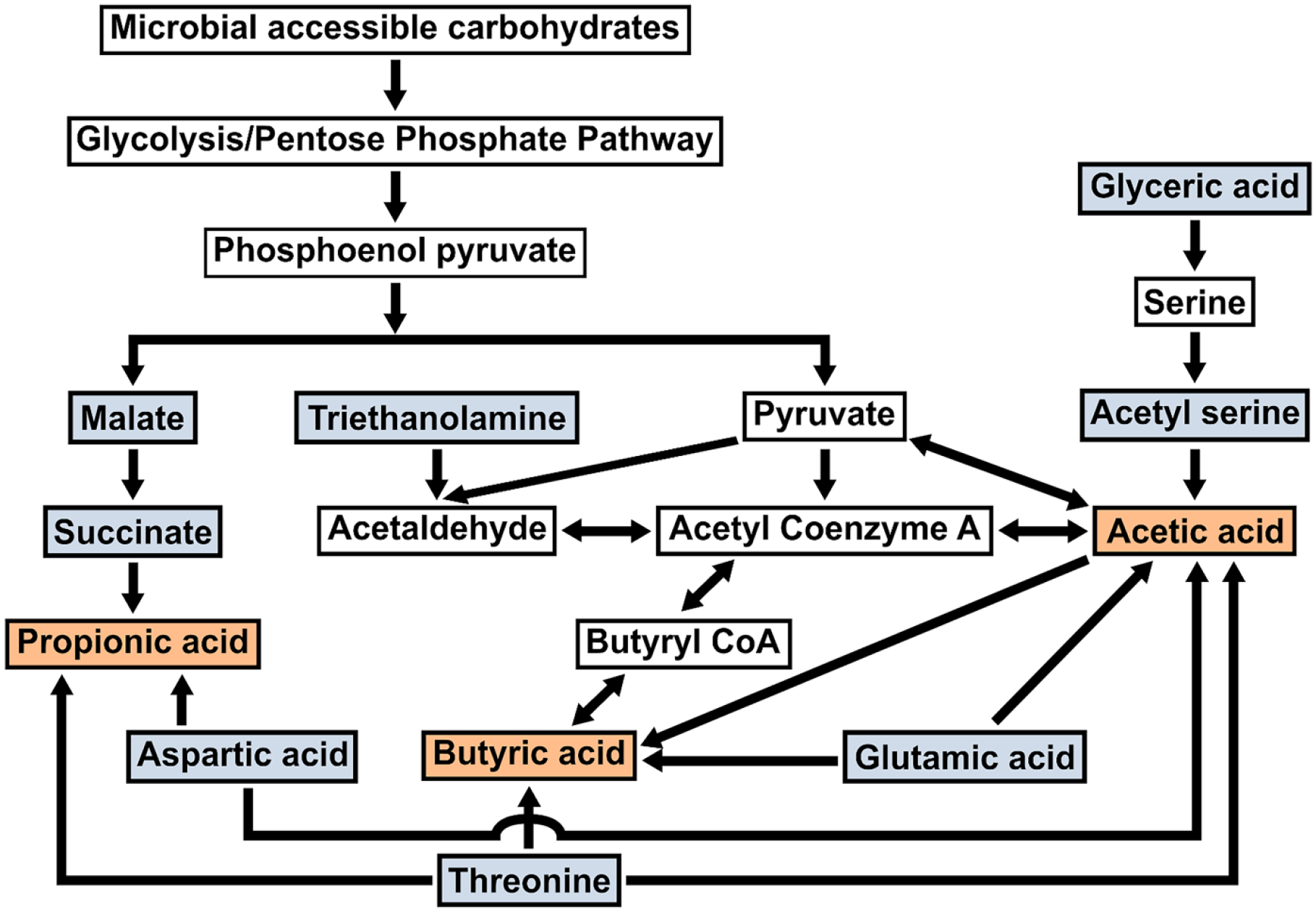
An outline of the metabolic pathways involved in the gut microbial production of SCFAs. The 3 primary SCFAs, acetic acid (acetate), propionic acid (propionate), and butyric acid (butyrate) (highlighted in orange) are produced by various bacterial fermentation of microbial accessible carbohydrates and metabolism of different amino acids. The metabolites highlighted in blue are significantly increased with both bEWE-Pre and -Post treatment groups, except glyceric acid (P < .05 in bEWE-Post: n.s. in bEWE-Pre), glutamic acid (P < .05 in bEWE-Post: n.s. in bEWE-Pre), and threonine (P < .05 in bEWE-Pre: n.s. in bEWE-Post).
Euglena Water Extract Treatment Increases the Production of SCFAs by Gut Microbiota in NNK-Induced Lung Tumor-Bearing Mice
The results from the metabolic profiling (Figures 3 and 4) suggest the possibility of involvement of SCFA metabolism in the antitumor effects of bEWE. To test this hypothesis, a separate targeted quantitative analysis of SCFA levels in the feces from tumor-bearing mice treated with either bEWE or PBS was conducted. As shown in Figure 5, acetic acid was significantly higher in both bEWE-Pre and -Post treated groups compared to the PBS control group (Figure 5A). On the other hand, propionic acid and butyric acid were significantly higher in only bEWE-Post treated mice (Figure 5B and C). However, there was no significant difference in valeric acid, formic acid, or isovaleric acid levels among the 3 treatment groups (Figure 5D-F). These results suggest that there were associations between fecal primary SCFA metabolism and the antitumor effects of Euglena water extract in NNK-induced tumorigenesis in the mouse lung.

bEWE treatment increased SCFA metabolism in the gut of NNK-induced tumor-bearing mice. The concentration of the following SCFAs (ng/mL): (A) acetic acid, (B) propionic acid, (C) butyric acid, (D) valeric acid, (E) formic acid, and (F) isovaleric acid, in the feces of mice treated with PBS, pre-treated with bEWE (Pre), or post-treated with bEWE (Post) are shown in the graphs. PBS served as the control. Results are presented as means ±SD (n = 8/group).
SCFA Treatment Inhibits the Proliferation of Murine and Human Lung Carcinoma Cells by Inducing Apoptosis In Vitro
To clarify the involvement of SCFAs in the antitumor effects of bEWE, the effect of the SCFAs on the proliferation of lung cancer cells was investigated by a trypan blue staining assay in cell culture. The SCFA treatment dose-dependently attenuated the growth of both types of lung carcinoma cells (Figure 6), but not in normal human lung epithelial cells, BEAS-2B (Supplemental Figure S4). Butyrate treatment inhibited the proliferation of both lung cancer cell lines more efficiently in comparison to acetate and propionate. These results strongly support our hypothesis that the antitumor effects of bEWE are induced by the stimulation of SCFA production in the gut of mice.
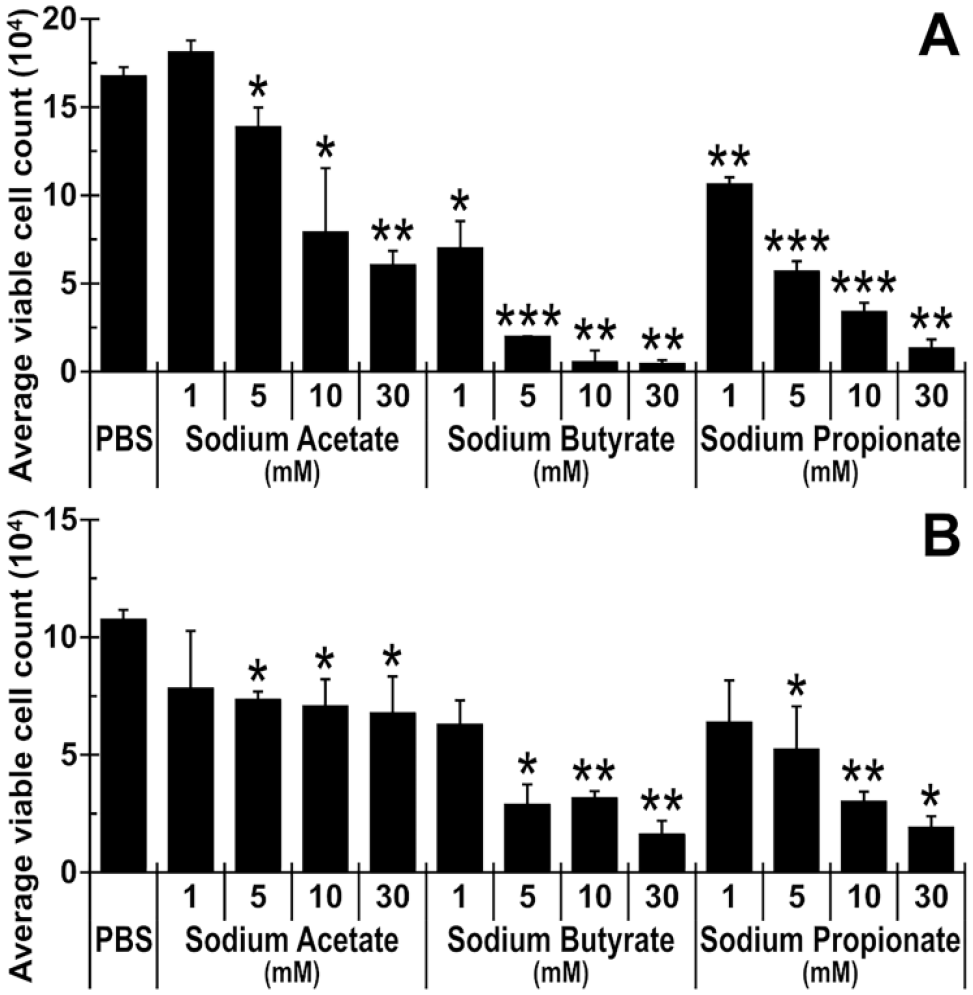
SCFA treatment dose-dependently attenuated the growth of murine and human lung carcinoma cells. The average viable cell number of (A) LLC murine lung carcinoma cells and (B) H1299 human lung carcinoma cells are presented. PBS served as the control. Results are presented as means ± SD (n = 3).
Furthermore, an apoptosis analysis carried out using anti-Annexin V antibodies and 7-AAD demonstrated that the SCFA-dependent inhibition of lung cancer cells is due to the induction of apoptosis (Figure 7). The results obtained from the LLC cells treated with SCFAs for 48 hours showed an increase in the apoptotic cells. The time course at 24 hours indicated that SCFAs induced negligible apoptosis for LLC cells treated with any of the 3 SCFAs (data not shown). When compared to the PBS control, the total apoptosis rate was significantly higher in cells treated with butyrate in comparison to acetate and propionate for 48 hours time point. Treatment with SCFA also affected the cell cycle of the LLC cells. As shown in Figure 8 and Supplemental Figure S5, 5 mM butyrate significantly decreased cell population in S and G2/M phases as well as Sub G1 phase compared with PBS control. The results suggest that butyrate treatment causes cell cycle arrest at the G1/S checkpoint, resulting in increased apoptotic (Sub G1) cells. These results also suggest that the apoptosis induced by SCFAs contributes to attenuating the growth of lung cancer cells.

SCFA treatment increased apoptosis in LLC murine lung carcinoma cells. LLC cells were treated with SCFAs (sodium acetate, sodium butyrate, sodium propionate) at 0.1 to 5 mM for 48 hours and the percentage of apoptotic cells was determined using flow cytometry. PBS served as a control. Results are presented as means ± SD (n = 2).

SCFA treatment increased the population of Sub G1 population representative of apoptosis and G1/S cell cycle arrest in LLC murine lung carcinoma cells. LLC cells were treated with SCFAs (sodium acetate, sodium butyrate, sodium propionate) at 0.1 to 5 mM for 48 hours and the percentage of G1, S, G2/M and Sub G1 (apoptotic cells) phases were determined using flow cytometry. PBS served as a control. Results are presented as means ± SD (n = 3).
Discussion
Because of its wide variety of nutritional components, Euglena gracilis is often used as a nutritional dietary supplement. It has been shown that Euglena extract and its main components, paramylon (water-insoluble aggregates of β-1,3-glucan), contain biological properties including immunomodulatory, anti-bacterial, anti-viral, and therapeutic properties against various diseases, including cancer.7,10,11,13,36 While there is an increasing interest in exploration of the therapeutic potential of E. gracilis and paramylon, the detailed mechanisms of their anti-cancer properties have not previously been clarified. Our previous study showed the anti-cancer effects of orally administrated Euglena water extract, devoid of paramylon, on lung cancer growth in an orthotopic LLC cell allograft model by altering immune suppressor cell populations: myeloid-derived suppressor cells (MDSC), 15 as well as by altering gut microbiota compositions. 20 However, the effect of Euglena water extracts without mature paramylon on the fecal metabolites in lung tumor-bearing mice remains unknown.
Among the various carcinogens involved in the induction of lung cancer, tobacco-specific nitrosamine (4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone, also known as NNK) has been documented as a potent lung carcinogen in mice. 22 NNK induces the formation of DNA adducts and, therefore, results in multiple mutations in critical cancer control genes.23,37 NNK-induced tumorigenesis in mice mimics human lung cancer development and it has been reported that the inhalation of toxic chemicals in smoke induces dysbiosis of the human gut microbiota.38-40 In addition, there is a strong correlation between NNK-induced lung adenocarcinoma and the disruption of gut microbiota and metabolite profiles in mice, signifying that this avenue should be researched further to develop better interventional targets against lung cancers. 41 Therefore, the anti-cancer properties and underlying mechanisms of Euglena water extract against lung tumorigenesis by NNK were investigated.
Microorganisms and their metabolites in the gut play crucial roles in human and animal health. 42 The prospects of developing a therapeutic approach for lung cancer via improving the gut microenvironment, including gut microbiota compositions and/or metabolites, are promising. The present study was conducted to clarify 2 subjects; (1) to evaluate the antitumor effects of orally administrated Euglena water extract in NNK-induced lung tumor models and (2) to identify the mechanism by which Euglena water extract inhibits lung cancer development based on changes in gut metabolites.
In the present study, both pre- and post-treatments with bEWE significantly reduced NNK-induced tumor development in comparison to the PBS treatment, and the attenuation was similar in both bEWE-Pre and -Post treatment groups (Figure 1B). Interestingly, bEWE reduced tumor burden when administered 10 weeks after NNK injection (bEWE-Post treatment); at this point, the tumors had already been developed. 29 However, in our previous study with the murine LLC allograft model, tumor growth was attenuated only with pre-treatment (3 weeks prior to the LLC cell inoculation), but not post-treatment (immediately after LLC cell inoculation for 3 weeks). 15 This discrepancy between the 2 studies may be explained by the durations of treatment with Euglena water extract; it appears that the growth inhibition of tumors may require at least 3 weeks or longer. This speculation is supported by our preliminary study in which 3 weeks of oral administration of Euglena water extract altered intestinal microbiota populations. 20 It is also supported by our previous study in which fecal transplantation using feces collected from mice treated with bEWE orally for 3 weeks attenuated orthotopic lung tumor growth in an LLC syngeneic lung cancer mouse model. These experiments indicate that daily oral administration of bEWE for 3 weeks altered microbiota populations, and bEWE-induced alteration of microbiota and their metabolites mediates lung cancer prevention in mice. Transplantation of the EWE-altered intestinal microbiota by fecal transplantation also prevented lung carcinoma growth in mice. 20 Nevertheless, these results suggest that bEWE could potentially be applied to lung cancer treatments associated with tobacco smoke carcinogens if administered in the early stages of tumor development. This result may increase interest in usage of E. gracilis extracts, not only for the prevention of lung cancer but also in the treatment of lung cancer, even after clinical diagnosis.
Since the mouse lung cancer model with LLC cancer cell graft employed in our previous studies15,20 does not mimic the development/progression of human lung cancer well, an NNK carcinogen-induced mouse model, which is clinically relevant to human lung cancer, was employed in the present study. The effect of bEWE was evaluated with the same experimental settings (pre- and post-treatment) using this model. The treatment with bEWE significantly attenuated NNK-induced tumor growth in both pre- and post-treatment settings (Figure 1), suggesting that bEWE-based lung cancer prevention/therapy may be a viable option for human lung cancer prevention.
Multivariate statistical analysis showed a clear separation of the fecal metabolite profiles between the bEWE treatment groups and the PBS control group. On the other hand, there was an overlap of fecal metabolites between bEWE-Pre and -Post treatment groups (Figure 2A). This overlap between bEWE-Pre and -Post treatment shows that bEWE treatment induces similar alterations in the gut microenvironment, regardless of the timing of treatment in NNK-induced tumor-bearing mice. The metabolomic profiling identified a significant increase in SCFA precursor metabolites in bEWE treated groups compared to PBS treated groups (Figures 3 and 4). To further understand the role of SCFAs in bEWE-induced anti-tumorigenesis, a quantitative analysis targeting SCFA levels in feces from bEWE-treated NNK-induced tumor-bearing mice was conducted. The results showed significant increases in acetic acid, propionic acid, and butyric acid, while no significant change in formic acid, isovaleric acid, or valeric acid in the bEWE-Pre or -Post treatments compared with the PBS treatment in the NNK-induced tumor-bearing mice (Figure 5). SCFAs are gut microbial metabolites that play a role in various physiological processes and impact the immune cell functions of the host. 43 Acetate, propionate, and butyrate are the major SCFAs that are produced in the intestinal compartment and can permeate the bloodstream and affect functions at distal sites, such as the lungs. 44 Moreover, SCFAs have many functions, which, either directly or indirectly, influence various cell types, tissues, and immune cells, 17 and can induce lung cancer cell apoptosis and cell cycle arrest, propionate especially. 45 Therefore, the present results suggest that the SCFAs acetate, propionate, and butyrate, derived from the bEWE-stimulated microbiota, may play an important role in the attenuation of NNK-induced lung carcinoma growth.
Since our results indicate that the increase of biosynthesis of SCFAs (acetate, propionate, and butyrate) by bEWE-stimulated gut microbiota may correlate with the antitumor activities of bEWE, the effects of SCFAs on the growth of lung carcinoma cell lines was evaluated in cell culture. The treatment with SCFAs dose-dependently attenuated the growth of both murine and human lung carcinoma cells (Figure 6), but not in normal human lung epithelial cell line: BEAS-2B, except high concentration (10 mM) butyrate (Supplemental Figure S4) which may suggest that SCFA-dependent cytotoxicity is selective for cancer cells over normal lung bronchial epithelial cells. Furthermore, the flow cytometric analysis of apoptosis in LLC cells treated with SCFAs showed an increase in the total (early and late apoptosis) apoptosis in the cells when compared with the PBS treated group (Figure 7). The apoptotic cells were dose-dependently increased by acetate, butyrate, and propionate treatments at 48 hours post-treatment. Strong induction of apoptosis was observed by the butyrate treatment, especially, as compared to the other 2 SCFAs (Figure 7). This induction of apoptosis by butyrate treatment was also observed in the Sub G1 population in cell cycle analysis (Figure 8). These results suggest that induction of apoptosis is a potential mechanism for the inhibition of lung cancer cell growth. These findings align with previous studies that report propionate and butyrate attenuating lung cancer cell growth by inducing apoptosis.45-47 Taken together, these in vitro studies support our hypothesis that attenuation of NNK-induced lung tumorigenesis by bEWE treatment is attributable to an increase in SCFAs produced from gut microbiota.
As shown in Materials and Methods, it is suggested that major components of Euglena, such as paramylon, chloroplasts, lipids, and large proteins (molecular weight higher than 10-20 kDa) have been removed in the process of the preparation of Euglena water extract. Therefore, bEWE contains various small peptides, water soluble β-glucans and various small molecules including amino acids and polyamines, etc. In our previous study which compared the anticancer activity of water extract from Euglena (EWE) and boiled EWE (bEWE), it was shown that the anticancer activity per unit mass of EWE was significantly increased by the heat treatment and subsequent removal of the denatured proteins. 20 It has been reported that dietary fiber enhances the number of beneficial microorganisms in the gut, and dietary fiber can be converted to the SCFAs by the microorganisms.48,49 Since the anticancer activity of bEWE can be analyzed only by mouse studies, we were unable to pinpoint what molecule or group of molecules is responsible for this bioactivity. However, since we are currently conducting further purification to figure out the active component(s), we hope to precisely answer this question in future studies.
Conclusions
The present study revealed that Euglena water extracts can be orally administered for the prevention and treatment of lung tumorigenesis in mice induced by the tobacco smoke carcinogen, NNK. Daily oral administration of bEWE caused a significant alteration in several fecal metabolites, either with pre- or post-treatment, including an increase in malate, succinate, triethanolamine, glycerate, acetyl serine, aspartate, glutamate, and threonine, which can be metabolized to SCFAs. Furthermore, the increase in fecal SCFAs (acetate, propionate, and butyrate) with bEWE treatment, and the inhibition of lung cancer cells with SCFAs, confirmed that the attenuation of NNK-induced lung tumorigenesis is associated with the alteration of gut microbiota metabolites. The present study strongly suggests that daily oral administration of bEWE alters gut metabolites, thereby attenuating NNK-induced lung tumorigenesis.
Supplemental Material
sj-docx-1-ict-10.1177_15347354231195323 – Supplemental material for Euglena gracilis Extract Protects From Tobacco Smoke Carcinogen-Induced Lung Cancer by Altering Gut Microbiota Metabolome
Supplemental material, sj-docx-1-ict-10.1177_15347354231195323 for Euglena gracilis Extract Protects From Tobacco Smoke Carcinogen-Induced Lung Cancer by Altering Gut Microbiota Metabolome by Deepa Upreti, Susumu Ishiguro, Morgan Phillips, Ayaka Nakashima, Kengo Suzuki, Jeffrey Comer and Masaaki Tamura in Integrative Cancer Therapies
Footnotes
Acknowledgements
We thank Sarah Devader (Kansas State University) for providing critical comments and discussing the manuscript.
Author Contributions
Conceptualization, D.U. and M.T.; Methodology, D.U., S.I. and M.T.; Resources, A.N. and K.S.; Investigation, D.U., S.I. M.P. and M.T.; Validation and Formal Analysis, D.U. and S.I.; Data Curation, D.U., S.I. and M.T.; Writing—Original Draft Preparation, Writing—Review & Editing and Visualization, D.U., S.I. M.P. J.C. and M.T.; Supervision, Project Administration and Funding Acquisition, J.C. and M.T.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs. Nakashima and Suzuki report being employed by Euglena Co., Ltd, which manufactures and markets products containing Euglena gracilis. Drs. Tamura and Comer have received a research grant from Euglena Co., Ltd. This does not alter the authors’ adherence to the journal policies on sharing data and materials. There are no patents or products in development to declare. The remaining authors (D.U. and S.I.) have no conflicts of interest to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by Kansas State University (KSU) College of Veterinary Medicine Dean’s funds [2018 CVM-SMILE] (M.T.), Euglena Research Fund [2018EU-3] (J.C. and M.T.), KSU Johnson Cancer Research Center [2020 JCRC-CRA] (M.T.), K-INBRE Bridging Award [P20 GM103418] (M.T.).
Data Availability Statement
All data is available upon request to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
