Abstract
Background:
Along with high calorie and high protein diet, a new comprehensive dietary approach is needed to control cachexia caused by cancer and its related outcomes. This study was done to evaluate the effect of a Mediterranean diet on body composition, nutritional status, and inflammatory markers among cancer cachexia patients.
Methods:
In this randomized clinical trial, 46 patients with colorectal cancer-induced cachexia were included. After randomization, 23 patients were allocated to the intervention group (Mediterranean diet) and 23 to the control group (nutritional counseling for weight gain and prevention of weight loss in cancer patients). The primary outcome including muscle health, nutritional status, and inflammatory markers along with secondary outcomes such as quality of life, and serum proteins were evaluated at the start and the eighth week of the study. Statistical analysis was performed according to the intention-to-treat concept. To compare changes in dependent variables between the 2 groups, analysis of covariance (ANCOVA) was performed.
Results:
After adjustment for the baseline values, age, sex, and supplements use, in the Mediterranean diet group mean of weight (P < .001), lean body mass (P = .001), fat mass (P = .002), and muscle strength (P < .001) were significantly increased compared to the control group. Regarding inflammatory markers, the mean serum level of tumor necrosis factor-alpha (TNF-α) (P < .001), high sensitive-C-reactive protein (hs-CRP) (P = .01) and Interleukin 6 (IL-6) (P < .001) were significantly improved in the Mediterranean diet group. Moreover, in the Mediterranean diet group, the score for global health status (P = .02) and physical performance score (P < .001) were significantly increased.
Conclusion:
It appears that the implementation of the Mediterranean diet might be a strategy to improve nutritional status, quality of life, inflammatory markers, and body composition in patients with colorectal cancer cachexia.
Trial registration:
Iranian Registry of Clinical Trials (www.irct.ir); ID: IRCT20211027052884N1
Introduction
The prevalence and mortality rate of colorectal cancer has decreased due to the advancement of screening and treatment methods in recent years, but it is still the third most common cancer in the United States and the world. 1 About 41% of colorectal cancer is related to the proximal colon, 22% to the distal colon and 28% to the rectum. 2 Cachexia, which is identified as a continuous loss of skeletal muscle mass and fat, plays an important role in disease prognosis and the pathogenesis of weakness in cancer. 3 In the study by Poisson et al, 4 the prevalence of cancer cachexia among 157 patients with colorectal cancer was 53.9%. Cachexia is a multifactorial disease and systemic inflammation plays an important role in its pathophysiology. 5 The level of inflammatory factors such as IL-6, TNF-α, and CRP increases significantly in this disease compared to non-cachetic patients.5-7 An acute increase in inflammatory factors disrupts the role of neuropeptide Y, leptin, and the neuroendocrine axis, and ghrelin, thus causing continuous stimulation of anorexia pathways. 5 Also, these inflammatory cytokines lead to many of the metabolic changes observed in cachexia, such as a state of hypercatabolism, muscle destruction, increased lipolysis, and the acute phase response. 5
Various studies indicate the effective role of diet in controlling and reducing inflammation. 8 Also, several studies have shown that not only nutrients, but diets such as the Mediterranean diet can reduce inflammation in various diseases, including cancer.9,10 This diet contains high consumption of whole grains, olive oil, vegetables, legumes, fruits, nuts and seeds, and dairy products such as cheese and yogurt, while recommending low consumption of milk, red meat and sweets. It also recommends moderate consumption of eggs and fish. 9 Nutrients in the Mediterranean diet such as phytochemicals, polyphenols, oleuropein, hydroxytyrosol, antioxidants, monounsaturated fatty acid (MUFA) and omega-3 fatty acids (PUFA) by inhibiting oxidative stress and inflammatory cascade may reduce proteolysis and lipolysis in muscle tissue.11-14 Epidemiologic studies revealed that higher adherence to the Mediterranean diet was linked with higher muscle mass and strength.15,16 A randomized clinical trial (RCT) conducted by Gioxari et al to survey the effect of the Mediterranean diet intervention on inflammation in lung cancer showed that the advanced lung cancer inflammatory index (ALI) obtained from this formula (BMI × Alb/neutrophil lymphocyte ratio) was significantly increased in the control group more than the patients following the Mediterranean diet. 17 Additionally, various studies have shown that the quality of life decreases in patients with cachexia, and they also experience some degree of impairment in physical function.18,19 Patients with cancer cachexia may suffer from nausea, vomiting, loss of appetite and reduced food intake. 19 Therefore, nutritional support and following a healthy diet in order to reduce the aforementioned symptoms can be effective. 20 Baguley et al conducted an RCT on 23 men with prostate cancer and showed that following the Mediterranean diet significantly improved the quality of life after 3 months of intervention. 21 However, there is limited data available on the effect of this diet on cancer cachexia patients.
Considering the important role of the Mediterranean diet in controlling inflammation and reducing mortality from cancer and increasing muscle mass and strength, as well as considering that a few clinical trial studies has been done specifically in this field, the aim this clinical trial was investigating the effect of the Mediterranean diet on anthropometric indices, inflammatory factors, quality of life, and nutritional status in patients with cachexia caused by colorectal cancer.
Method
This randomized superiority controlled clinical trial on colorectal cancer cachexia patients was conducted in the Imam Khomeini hospital complex, Tehran, Iran, over 9 months (from January 2022 to September 2022). The local review board of the Tehran University of Medical Sciences accepted the current study protocol. Informed consent was obtained prior to randomization either from the patient or his/her legal representative. Stratified block randomization was applied based on BMI (≤23, >23) and the type of cancer (colon or rectum) via the www.randomization.com website. We published more details of the current protocol study previously. 22 The study was registered at www.irct.ir (ID: IRCT20211027052884N1). From January 2022 until September 2022, 46 patients with cachexia caused by colorectal cancer were recruited, 23 patients were in the intervention group (receiving a Mediterranean diet) and 23 patients were in the control group (receiving nutrition counseling).
Patients
All patients with ages ≥40 years with oral feeding and diagnosis of colorectal cancer in stages 2 to 4 based on TNM UICC 2010 system (on the oncologist detection) 23 and cachexia based on the Global Leadership Initiative on Malnutrition (GLIM) criteria 24 and patients’ functional status ≥70% according to Karnofsky scale 25 and individuals without serious underlying diseases like renal and/or hepatic disorders and without allergy history to the Mediterranean diet components, for example nuts or olive oil were included in the study. With regard to diagnosing cancer cachexia the GLIM criteria were applied. 24 The GLIM criteria included 3 phenotypic criteria (weight loss, low body mass index, and muscle mass loss) and 2 etiologic criteria (reduced food intake or reduced food absorption, and the presence of inflammation or disease). To diagnose cachexia, there must be at least one phenotypic criterion and one etiological criterion.
Intervention
Qualified patients were randomized into either an intervention or control group. We prescribed a Mediterranean diet regime with extra virgin olive oil (EVOO) for the intervention group, and nutritional instruction with dietary recommendations in cancer patients for the control group. All patients were visited by expert dietitians. The duration of intervention was 8 weeks. For the Mediterranean diet group, the required energy for the patients was estimated according to the ASPEN guideline 26 which started with 25 kcal/kg body weight per day, and then reached 35 kcal/kg body weight per day within 2 weeks. In this group energy requirement was determined as follows: 35% of daily calories from fats (less than 7% from saturated fats, 10% to 15% from polyunsaturated fats, and the rest from monounsaturated fats), 20% from protein, and 45% from carbohydrates. The weekly dietary menu was designed according to the Mediterranean regimen and EVOO was provided free of charge to the patients during the intervention period. All these points were explained to the patients in training sessions. Moreover, for the convenience of the patients and to increase their adherence to the protocol, the diet was personalized based on their tastes. To evaluate the compliance of patients with the prescribed dietary plan, a 1-day food record was taken from the patients every 2 weeks of the study. Higher than 60% adherence to the Mediterranean diet based on the prescribed energy and macronutrients was determined as the appropriate compliance of patients. The detailed information on designing the Mediterranean menu for the intervention group and nutritional advice for the control group were described in the published protocol. 22 In the control group, routine nutritional recommendations regarding weight gain and prevention of weight loss in cancer patients according to the clinical guidelines 27 were given as brochures. All the points in these brochures were clarified to the patients.
Treatment Protocol
New outpatients (newly diagnosed patients at the beginning of treatment) were treated based on the protocol of colorectal cancer treatment. 28 Accordingly, rectal cancer patients received first-line radiotherapy and colon cancer patients received first-line chemotherapy with 5-fluorouracil and oxaliplatin (FOLFOX), capecitabine and oxaliplatin (CAPOX), or capecitabine. The attending oncologist made decisions on the treatment protocol including chemotherapy and radiotherapy. All patients were received chemo and radiation treatment on an outpatient basis.
Outcomes
The primary outcomes were muscle strength, lean body mass, nutritional status, and inflammatory markers including high sensitive-C reactive protein (hs-CRP), tumor necrosis factor-alpha (TNF-a), interleukin-6 (IL-6), which assessed at the start and end (the eighth week) of the study. We assessed the patients nutritional status by the Patient Generated-Subjective Global Assessment Questionnaire (PG-SGA). 29 The questionnaire evaluates the nutritional status of patients according to information about a person’s medical history, diet history, and clinical symptoms. Based on the PG-SGA questionnaire guidelines, higher scores indicate the severity of malnutrition. Lean body mass as a part of body composition was calculated via the Bio-Electrical Impedance Analysis (BIA) (model: Portable Jawon medical). We assessed handgrip strength (muscle strength indicator) by the digital dynamometer (Seahan, model SH5003, Seahan Co, South Korea).
For measuring of inflammatory markers, a 5 mL venous blood sample in fasting state was taken from participants at the launch and end of study. Serum was separated by centrifugation and was reserved in a −80°C freezer. Then, the enzyme-linked immunosorbent assay (ELISA) was applied. The hs-CRP kit was from the LDN company, while IL-6 and TNF-α kits were from Invitrogen. Results for hs-CRP were expressed in ng/mL and for IL-6 and TNF-α were expressed in pg/mL.
We considered quality of life, serum albumin and total protein as secondary outcomes. Serum total protein and albumin were evaluated with an autoanalyzer (spectrophotometric method).
The quality of life of patients at the beginning and end of the research was evaluated by the researcher using the EORTC QLQ-C30 questionnaire introduced by the European Cancer Research and Treatment Organization. 30 We assessed 7 scales of this questionnaire such as functional status (physical, cognitive, emotional and social), appetite, constipation, diarrhea, nausea and vomiting, and global quality of life scale. A high score for the functional status and quality of life scale indicates a high or healthy level. But a high score for appetite, constipation, diarrhea, nausea and vomiting indicate a high level of symptoms and problems. 31 Other secondary outcomes including weight, body fat mass (BFM) and percent body fat (PBF) were also assessed through the Bio-Electrical Impedance Analysis (BIA) (model: Portable Jawon medical). It should be noted that there was a deviation from protocol for assessing CBC blood test from the patients. Because it was difficult for the patients to give us an additional 5 cc of blood and they did not cooperate in this matter, we did not perform the CBC blood test.
We evaluated other general information including economic status, sex, age, education, supplement use, medication, medical history, and COVID-19 history through standard questionnaires. The patient’s functional status was estimated by the Karnofsky scale. Height was also calculated using a tape measure.
Sample Size Calculation
Based on the formula that has been suggested for parallel clinical trials (n = 2 [(z1−α/2 + z1−β) 2 . s 2 ]/d 2 ), 32 and taking into account the nutritional status (score obtained from the PG-SGA questionnaire) as the main variable, 33 and considering the 15% probable dropout, 20 patients in each group were needed to include in the current study. More detailed on sample size calculation was clarified previously. 22
Statistical Analysis
Regarding dietary food records, the frequency of each food item was converted to grams per day. Then, the daily macro and micronutrients intake was calculated by Nutritionist IV software which is a modified food composition database based on the US Department of Agriculture. We performed the Kolmogorov Smirnov test and Q–Q plot to determine the normality of quantitative variables. If the data were not normal, logarithmic transformation was used to normalize them. Statistical analysis was performed according to the intention-to-treat concept. In the intention-to-treat analysis, all randomized patients who have the baseline measurements and followed the Mediterranean diet for at least 2 weeks were analysed. Thus, by using linear regression, imputed data were considered for patients who were excluded from the study during the intervention. 34 Mean (standard deviation) was used to describe quantitative variables and frequency (percentage) was used for qualitative variables. Independent sample t test was applied to determine the difference of quantitative variables between intervention and control groups. Chi-square test was done to determine the distribution of qualitative variables between intervention and control groups. Repeated measures ANOVA test was used to investigate the effect of Mediterranean diet on outcome variables. Then, in order to compare changes in dependent variables between 2 groups, analysis of covariance (ANCOVA) was performed. Data were analysed using SPSS version 26 software. P > .05 was considered statistically significant.
Results
In total, among the 46 participants who entered the randomization stage, 6 people were excluded from the study for the reasons mentioned in Figure 1, and finally, 40 people completed the study. The general characteristics of the participants in the study, separated by the group receiving the Mediterranean diet and the control group, are depicted in Table 1. There was no significant difference in terms of age, body mass index, scores obtained from the Karnovsky questionnaire, gender, marital status, education, job, underlying diseases, type of cancer, stage of cancer, type of cancer treatment, chemotherapy drugs, physical activity and history of Covid-19 between the 2 groups. However, there was a significant difference in the consumption of nutritional supplements between the intervention and control groups (P-value: .04).

Flow diagram of people participating in the study.
General Characteristics of the Participants in the Study by Intervention and Control Groups.
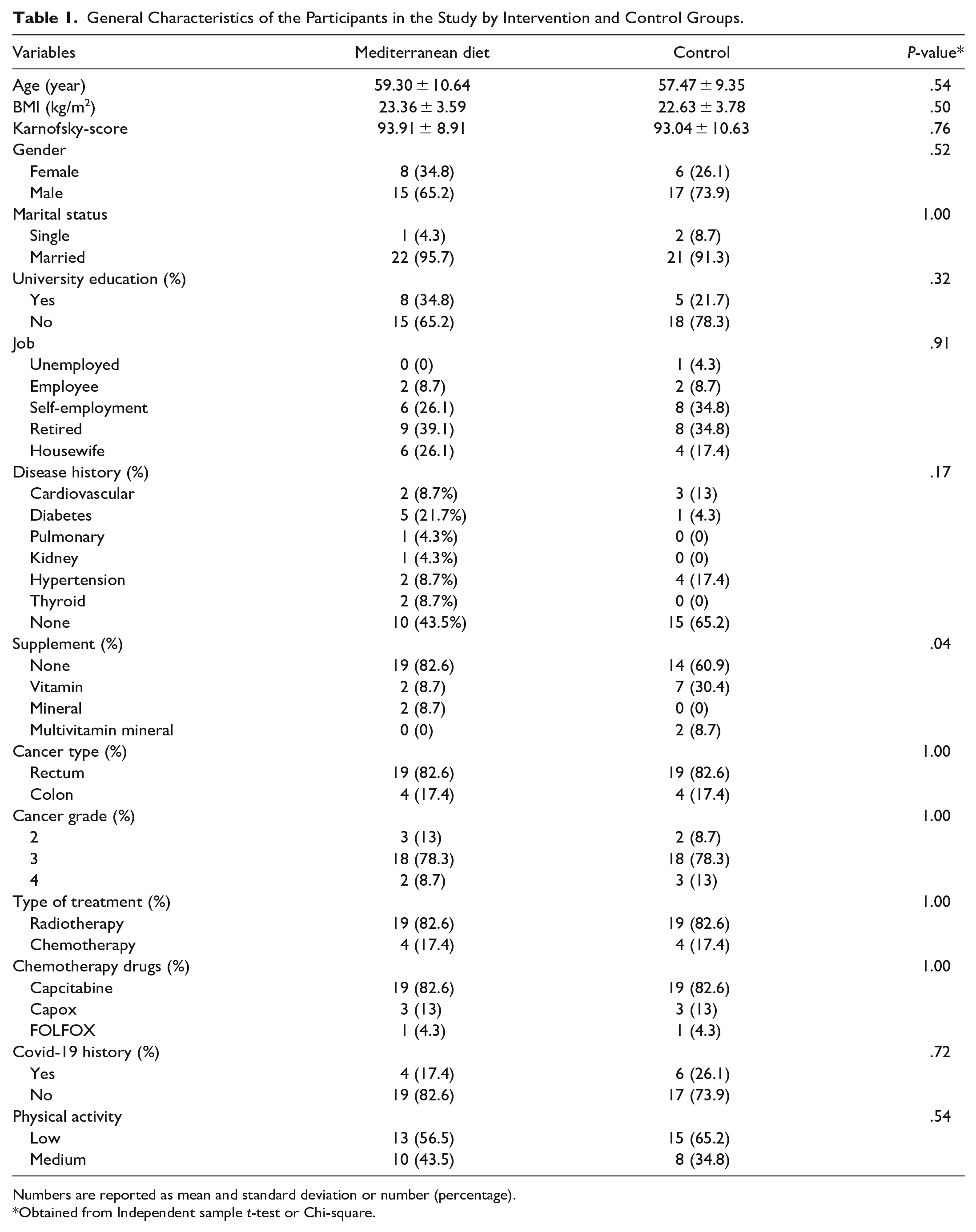
Numbers are reported as mean and standard deviation or number (percentage).
Obtained from Independent sample t-test or Chi-square.
The dietary nutrient intake of the participants during the study is shown in Table 2. The average energy, fat, and protein intakes in the Mediterranean diet group were significantly higher than the control group. Moreover, patients in the Mediterranean diet group had a higher intake of fiber, monounsaturated fats, oleic acid, omega-3, folic acid, vitamins E, A, B2, B3, B6, B12, magnesium, calcium, iron, and zinc than the control group. The dietary food groups intake during the study for intervention and control groups are shown in Supplemental Table 1. Patients in the Mediterranean diet group had a significantly higher intake of whole grains, white meat, vegetables, nuts and olive oil than the control group. Patients in the control group had a significantly higher intake of red meat, sweets, hydrogenated vegetable oil and liquid oil (liquid oils other than olive oil) than patients in the intervention group. However, there was no significant difference between the 2 groups for intake of refined grains, processed meat, low- and high-fat dairy products, fruits and legumes.
Mean and Standard Deviation of Daily Intake of Nutrients in Colorectal Cancer Patients in Intervention and Control Groups.

Numbers are reported as mean and standard deviation.
Obtained from independent sample t-test.
The mean and standard deviation of anthropometric indices, nutritional status, quality of life and inflammatory markers during the study for the intervention and control groups are shown in Table 3. Compared to the beginning of the study, body weight (kg) (mean change: 1.43, 95% CI: −5.75, 8.61; PTime × Group < .001), lean body mass(kg) (mean change: −0.50, 95% CI: −5.51, 4.40; PTime × Group < .001), and body fat (kg) (mean change: 2.00, 95% CI: −1.93, 5.94; PTime × Group = .004) was significantly increased in the Mediterranean diet group compared to the control group. In addition, there was a significant increase in muscle strength (mean change: 0.32, 95% CI: −5.22, 5.87; PTime × Group < .001) in the Mediterranean diet group after 8 weeks. A significant decrease in PG-SGA score (mean change: −3.04, 95% CI: −5.01, −1.08; PTime × Group < .001) was seen in the Mediterranean diet group, indicating that nutritional status was improved in this group. Concerning inflammatory markers, the serum levels of hs-CRP (mean change: 609.35, 95% CI: −547.76, 1766.46; PTime × Group = .03) and IL-6 (mean change: 0.76, 95% CI: 0.36, 1.16; PTime × Group < .001) increased significantly in the control group at the end of the study. Furthermore, the serum level of TNF-α in the intervention group was significantly decreased compared to the control group (mean change: −0.12, 95% CI: −0.47, 0.23; PTime × Group < .001). The total serum protein level in the control group decreased significantly (mean change: −0.34, 95% CI: −0.65, −0.03; PTime × Group = .03). The interaction effect of time × group on the scores obtained from the quality-of-life questionnaire revealed that physical performance (mean change: 7.37, 95% CI: −1.16, 15.91; PTime × Group < .001), appetite (mean change: −12.92, 95% CI: −27.98, 2.14; PTime × Group = .01), and constipation (mean change: 4.05, 95% CI: −7.75, 15.86; PTime × Group = .01) in the intervention group were improved significantly.
Mean and Deviation of Anthropometric Indices, Nutritional Status, Inflammatory Markers, and Quality of Life at the Beginning and End of the Study for Intervention and Control Groups.

Numbers are reported as mean and standard deviation.
Obtained from the repeated measure ANOVA test.
Table 4 shows the adjusted averages of changes in anthropometric indices, nutritional status, inflammatory markers and quality of life in both the Mediterranean diet and nutritional advice groups. After adjustment for the baseline values, age, sex, and supplements use, an average weight (changes from baseline in the Mediterranean diet vs nutritional advice groups: 0.94 vs −1.81, P < .001), lean body mass (intervention vs control: 0.35 vs −1.35, P = .01), fat mass (intervention versus control: 0.76vs −0.58, P = .002), fat percentage (intervention vs control: 0.98 vs −0.78, P = .02) and muscle strength (intervention vs control: 1.56 vs −2.29, P < .001) were significantly increased in the Mediterranean diet group compared to the control group. Moreover, the PG-SGA score was decreased significantly (intervention vs control groups: −4.04 vs 1.86, P < .001) in the intervention group after adjustment for confounding variables, showing the improved nutritional status in the Mediterranean diet group. Regarding inflammatory markers, after adjustment based on confounding variables, the mean serum level of TNF-α was significantly decreased in the Mediterranean diet group (intervention vs control groups: −0.47 vs 0.35, P < .001), and the mean serum levels of hs-CRP (intervention vs control groups: -63.23 vs 1406.62, P = .01) and IL-6 (intervention vs control groups: −0.02 vs 1.09, P < .001) were significantly increased in the control group compared to the intervention group. The average of total serum protein in the control group was significantly decreased compared to the intervention group (intervention vs control groups: 0.04 vs −0.46, P = .008) after controlling for covariates. Regarding the quality of life and its components, in the group receiving the Mediterranean diet the score for global health status (intervention vs control groups: 6.78 vs −3.95, P = .02), physical performance score (intervention vs control groups: 8.44 vs −6.32, P < .001), appetite (intervention vs control groups: −6.67 vs 13.29, P = .01), and diarrhea (intervention vs control groups: −2.14 vs 15.48, P = .02) were improved significantly after adjusting for confounding variables.
Adjusted Mean Change of Anthropometric Indices, Nutritional Status, Inflammatory Markers, and Quality of Life in Intervention and Control Groups.
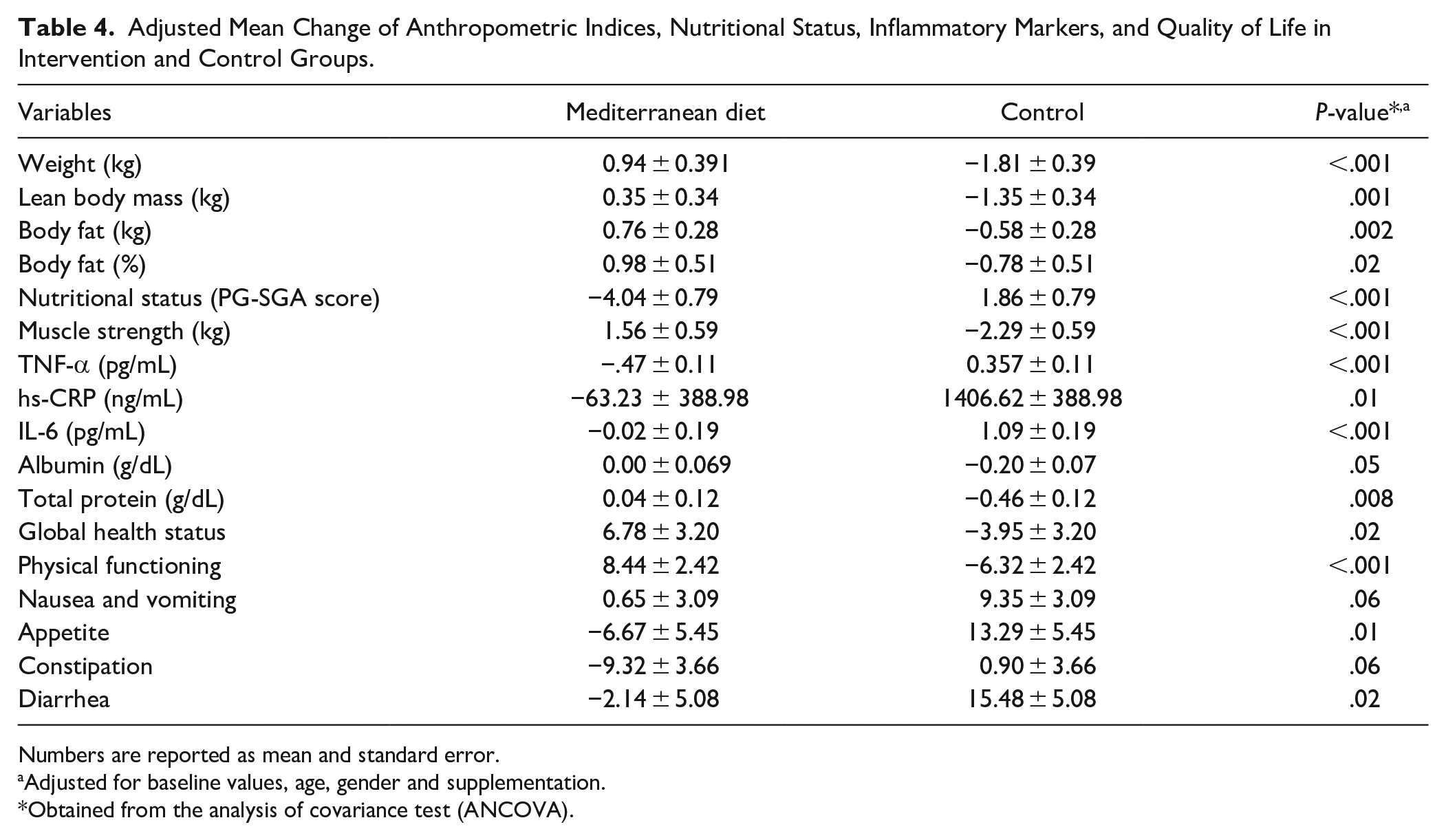
Numbers are reported as mean and standard error.
Adjusted for baseline values, age, gender and supplementation.
Obtained from the analysis of covariance test (ANCOVA).
Discussion
The findings of the present RCT showed that the Mediterranean diet rich in extra virgin olive oil led to an improvement in nutritional status, quality of life, inflammatory markers, and body composition in patients with cachexia induced by colorectal cancer compared to the control group.
Anthropometric indices are considered one of the most basic and important tools in the diagnosis of cachexia, as well as in predicting mortality in cancer patients.35,36 Various studies have shown that the reduction of muscle mass in cancer patients is associated with adverse outcomes, including more severe toxicity during chemotherapy and, as a result, a shorter life span.35,36 On the other hand, cancer is usually associated with a nutritional disorder, which is one of the effective factors in deteriorating the results of surgery, chemotherapy, or radiotherapy. 37 Therefore, one of the most important goals of clinical trials in cancer patients is to maintain or improve nutritional status. 38 Moreover, the increase in inflammatory factors provides the basis for progressive muscle breakdown, weight loss, high metabolism, and worsening the disease progress. 39 It should be noted that there is limited data available in this regard. Baguley et al conducted a 3-month clinical trial study in 2021 to determine the effect of the Mediterranean diet on 23 patients with prostate cancer. 21 The results of this study showed that the Mediterranean diet reduced body weight and muscle mass while improving quality of life and fatigue. However, they did not find a significant effect of following the Mediterranean diet on the levels of inflammatory markers such as IL-6 and IL-8. But we should mention that this study was conducted on overweight people (average BMI = 28.9). Another clinical trial study was conducted by Gioxari et al to investigate the effect of the Mediterranean diet on 30 patients with lung cancer for 3 months. 17 At the end of the intervention, there was no significant correlation between the 2 groups in BMI, body fat mass, and serum albumin levels. Nevertheless, the serum level of CRP decreased in the intervention group and increased in the control group. Of note, this difference in results could be due to the small sample size, different baseline BMI (average BMI = 27) and type of cancer, as well as the lack of adjustment for confounding factors in the analyses. It seems that due to the limited data available on the effect of the Mediterranean diet on cancer cachexia, a more relevant study is needed in this area.
The positive effects of the Mediterranean diet on nutritional status, quality of life, inflammatory markers, and body composition in patients with cancer cachexia can be explored from several points of view. As one of the healthiest diets in the world, the Mediterranean diet emphasizes the intake of healthier proteins obtained from whole grains, nuts, and white meats. 40 Getting the majority of dietary protein from these sources due to the presence of protein, antioxidant compounds, and polyphenols, in addition to reducing the amount of inflammation in the body, also helps to maintain and strengthen muscle tissue.41-43 Polyphenols, which are mainly found in fruits, vegetables, and whole grains, can reduce cancer-induced cachexia by possibly preserving muscle mass by inhibiting NF-kB signaling. 44 Another feature of the Mediterranean diet is the high content of dietary fats, especially PUFA and MUFA fatty acids. 40 Various studies have shown that adequate intake of PUFA and MUFA fats in the diet can be effective in maintaining weight through preserving muscle mass and fat mass, as well as reducing inflammatory factors.45,46 EVOO, as the main source of MUFA in the diet, can play a significant role in weight gain, reducing muscle mass and fat mass wasting, and improving nutritional status in cancer patients. 47 Studies have shown that hydroxytyrosol, as one of the main components of EVOO, prevents muscle wasting by inhibiting reactive oxygen species (ROS) and increasing antioxidant capacity. 47 The levels of oxidizing compounds such as ROS increase in cancer cachexia, and with the progress of cachexia, cell damage and inflammation cause disease deterioration.48,49 In addition to the anti-inflammatory and health-promoting effect of the Mediterranean diet, providing adequate energy and protein for the patients based on cancer guidelines may be another factor for weight gain and improving muscle condition of patients.26,50
To our knowledge, this is the first study investigate the effect of Mediterranean diet on body composition, quality of life, nutritional status, and inflammatory cytokines on colorectal cancer cachexia patients. The Mediterranean dietary regimes were personalized for each patient to increase their adherence. Moreover, each patient in the intervention group received EVOO free of charge. Another strength of the RCT was that we included patients with only colorectal cancer cachexia. Furthermore, to specify the intermediate mechanisms in cachexia, inflammatory markers were also assessed. One of the neglected aspects of cachexia, its psychological and social effects on patients. Thus, we evaluated the effect of Mediterranean diet on quality of life by EORTC QLQ-C30 questioner.
Serum and urine hydroxytyrosol concentrations is the key biomarker for specifying adherence to Mediterranean diet. 51 However, we did not evaluate this biomarker due to inadequate budget. Thus, the mean of four 24-hours dietary records during the study was used to evaluate adherence to the diets. Different protocol in treatment of cancer might have an effect on nutritional status; though patients were stratified based on type of cancer. Additionally, in the current RCT blinding study participants or staff was impossible because of nutritional interventions. However, the outcome assessor was blinded to the group assignment.
Conclusion
This RCT indicated that the Mediterranean diet rich in energy, protein and EVOO could improve nutritional status, quality of life, inflammatory markers, and body composition in patients with colorectal cancer cachexia compared to the control group. It appears that the implementation of this diet is a decent strategy to decrease the risk of cachexia. This trial conducted on patients with colorectal cancer cachexia, therefore, generalization our finding to other type of cancer or cachexia should be done with cautious.
Supplemental Material
sj-docx-1-ict-10.1177_15347354231195322 – Supplemental material for The Effect of Mediterranean Diet on Body Composition, Inflammatory Factors, and Nutritional Status in Patients with Cachexia Induced by Colorectal Cancer: A Randomized Clinical Trial
Supplemental material, sj-docx-1-ict-10.1177_15347354231195322 for The Effect of Mediterranean Diet on Body Composition, Inflammatory Factors, and Nutritional Status in Patients with Cachexia Induced by Colorectal Cancer: A Randomized Clinical Trial by Amir Bagheri, Farzaneh Asoudeh, Saied Rezaei, Mohammad Babaei and Ahmad Esmaillzadeh in Integrative Cancer Therapies
Footnotes
Author Contributions
AB, SR, MB, and AE contributed to the design of the work. AB and FA wrote the manuscript. AE and MB critically reviewed the manuscript and validated the procedure. The final manuscript was read and approved by all authors.
Availability of Data and Materials
The raw data can be made available by request to the corresponding author.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: More than half of the extra virgin olive oil was provided by the Jalalat Exir Olive Oil Company free of charge. For the remaining oil, the company received 50% of the oil price in the marketplace from Tehran University of Medical Sciences. However, this company did not contribute to the study project, intervention, procedures, evaluation, collection, data processing, or writing the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by Tehran University of Medical Sciences, Tehran, Iran (Grant number: 1400-2-212-54107). The Jalalat Exir Olive Oil Company provided extra virgin olive oils in a reduced price. The funders had no role in the trial procedures, intervention, and data processing.
Ethics Statement
This study was approved by the Ethic Committee of Tehran University of Medical Sciences (IR.TUMS.MEDICINE.REC.1400.601) on September 01 2021.
Informed Consent Statement
All patients were read the terms and conditions in the consent form before the study. Patients wishing to participate gave written informed consent.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
