Abstract
Introduction
Cancer cachexia is characterized by significant weight loss, sarcopenia, and an underlying inflammatory process that often leads to higher morbidity and mortality. 1 It is a metabolic disorder affecting up to 80% of patients with advanced cancer. 2 Although sarcopenia can occur independent of cachexia, it is a hallmark of the progressive disease and an indicator of decreased physical function, loss of quality of life, and shorter survival in cancer patients. 3 Corresponding to loss of lean muscle mass are other parameters indicative of increased tissue breakdown such as increased lactate dehydrogenase (LDH) activity especially in cancer cachexia, increased C-reactive protein (CRP) due to localized and systemic inflammation in patients with gastrointestinal (GI) cancer, and lower prealbumin levels associated with poor nutrition and loss of necessary protein mass. 4 Body composition can further indicate a response to inflammation and loss of lean body mass. Bioelectrical impedance analysis (BIA) allows the indirect analysis of cellular health as intracellular water (ICW) serves as an indicator of inflammation.5,6 Furthermore, lean body mass measured through BIA relates to skeletal muscle mass as a major protein source and storage for the body. Using BIA to detect loss of lean body mass together with a shift in body water from intracellular to extracellular may therefore serve as an indicator of the progressive nature of inflammation in cancer patient.
Current interventions for the treatment of cancer cachexia are limited to symptomatic pharmacotherapy such as megestrol acetate and other corticosteroids that temporarily increase protein synthesis and halt muscle degradation. 7 Complementary approaches include cannabis and appetite stimulants as well as nutritional supplements high in protein content that can counteract muscle loss and stabilize catabolic activity in some patients. However, responses to each of these are typically short in duration, with side effects common or access limited.
Given that lean muscle loss is likely occurring as a consequence of underlying inflammation related to both progressive malignancy and chemotherapy, 8 symptomatic relief alone may not provide a lasting benefit to patients and often delays administration of chemotherapy and worsens quality of life. Acupuncture has a long history of use as part of traditional Chinese medicine as well as a well-researched and established treatment option for a range of disorders including acute and chronic pain 9 and irritable bowel syndrome. 10 Clinical studies indicate that acupuncture provides benefits to cancer patients by reducing symptoms of nausea and vomiting, 11 increasing gastric emptying, 12 and altering physiological functions of hormones (ghrelin and leptin) that play important roles in GI functions.13,14 Acupuncture stimulation is able to differentially affect inflammatory reactions by reducing CRP and LDH 15 and demonstrates a decrease in inflammatory response (eg, tumor necrosis factor [TNF]-α, interleukin-6, CRP) in animal studies16-18 and TNF-α in patients with congestive heart failure. 19 These biomarkers are particularly relevant for patients with cancer cachexia given the important component of cachexia development in systemic inflammation and muscle wasting, which is accelerated by the malignancy.20,21
Although many studies have employed acupuncture to mitigate symptoms of cachexia and other cancer-related symptoms,22,23 it is not consistent among studies of how acupuncture points are selected for sham acupuncture and true acupuncture. We defined that targeted acupuncture (TA) is a verum/true acupuncture by the definition of the National Cancer Institute (NCI) at the National Institutes of Health (https://www.cancer.gov/publications/dictionaries/cancer-terms/def/verum-acupuncture), and the acupuncture points were predetermined based on the specific characteristics and to the pathways of the condition to study. For our study, predetermined points were selected based on the characteristics of cancer cachexia to modulate systemic inflammation, anorexia, and muscle wasting. In contrast, sham acupuncture is defined as “needles do not go as deep and are not used at the same points on the body.” (NCI: https://www.cancer.gov/publications/dictionaries/cancer-terms/def/verum-acupuncture). Nontargeted acupuncture (NTA) is a modified sham acupuncture. In our study, the control group (CG) received NTA, which was focused on predetermined points that are nonrelevant to cachexia points on the body such as headache, sore throat, or nasal congestion in the same sense of sham acupuncture. The NTA points employed, however, indeed penetrated as deep as the TA group, which was not under the NCI definition of sham acupuncture. These predetermined points were employed consistently for all patients without deviations or additional points based on individual symptoms. This allows for strict standardization of the protocol removing potential bias.
The results presented in this article evaluate 2 central hypotheses of the preliminary study: TA in complement to standard of care chemotherapy (1) maintains muscle mass and weight and (2) improves quality of life through increased appetite stimulation in patients with GI cancer–associated cachexia.
Methods
Study Design
This pilot study was conducted using a 2-group, parallel, single blind, randomized controlled design to examine the effect of TA on improving appetite and weight in GI cancer patients with cachexia. The study was approved by the Institutional Review Board (IRB-01) of the institution where the study was conducted and registered to the ClinicalTrials.gov database (NCT#02148159). Informed consent was obtained from all individual participants included in the study.
Sample and Setting
Patients were recruited from 2 oncology outpatient (academic and community) clinics. Inclusion criteria included adult subjects (>21 years old) with (1) a medical diagnosis of primary GI cancer (eg, gastric, biliary, small intestine, or colorectal), (2) weight loss of at least 5% over the last 6 months, (3) ability to communicate in English, and (4) ability to follow the research protocol. Exclusion criteria included those who (1) planned to have surgical procedures at the time of recruitment or during the month prior to the study, (2) would receive radiation therapy during the study period, (3) underwent surgery during the study or would not have chemotherapy after the surgery, (4) had any comorbidities that may affect the interpretation of study findings (eg, HIV, AIDS, Alzheimer’s disease, movement disorder, acute myocardial infarction within last 3 months, hepatitis), (5) had open burn sites or infected wounds, (6) were diagnosed with esophageal cancer with a mechanical swallowing difficulty, (7) had an uncorrected, mechanical digestive obstruction, or inability to tolerate enteral nutrition, (8) had a diagnosis of pancreatic adenocarcinoma, and (9) had a life expectancy of less than 6 months.
Randomization and Allocation Concealment
Randomization was preassigned by the statistician who was not part of patient care or communicated with the patient care team during the study. To prevent selection bias of assigning patients to a certain group, allocation sequence was strictly based on the date and time of the informed consent signed without exception. Each patient received a consecutive personal identification number in a first consented, first serve order, and was assigned to 1 of the 2 groups by personal identification number using random length permutated blocks to keep the randomization balanced between the treatments. There was no stratification provision. Due to the nature of the intervention, the study was designed as a single-blind randomized controlled trial.
Study Intervention
Acupuncture Protocol
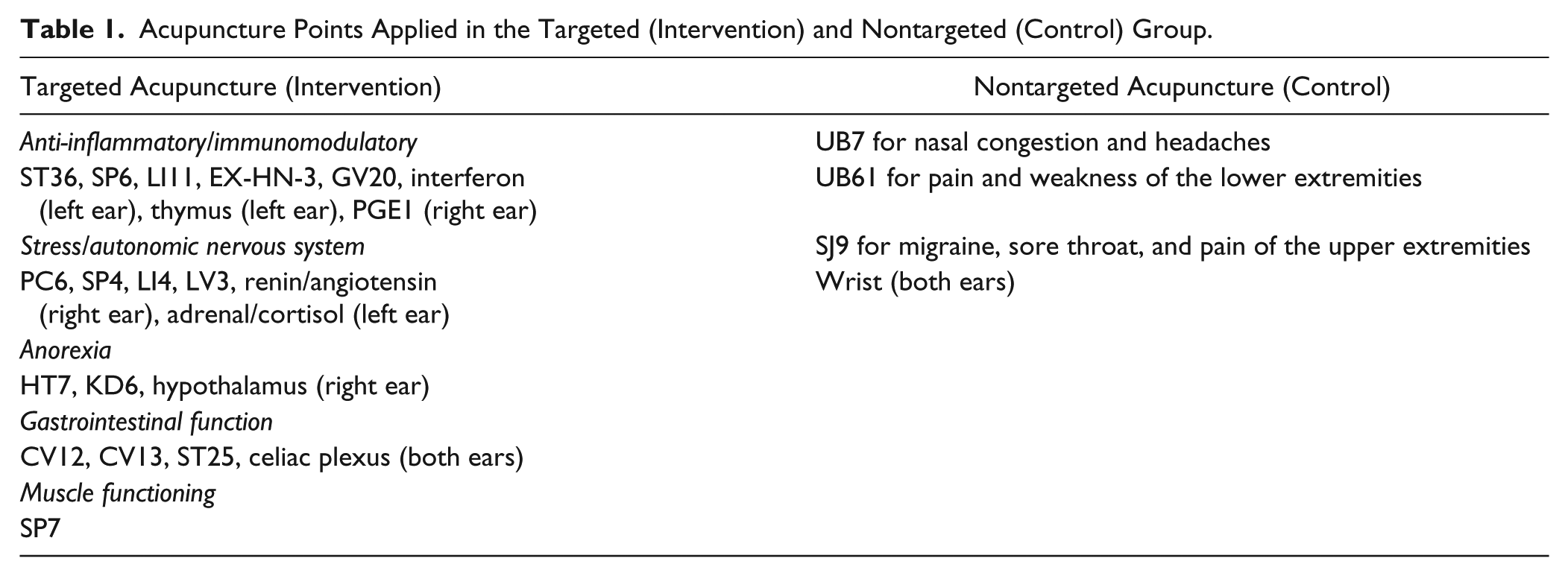
The selection of TA points was based on the potential mechanism of action for cachexia.24,25 The selected acupuncture points were linked to specific biological factors that affect the processes involved in the initiation, progression, or maintenance of cachexia (anti-inflammatory/immunomodulation, stress/autonomic nervous system, anorexia, and muscle wasting) using auricular 26 and traditional Chinese medicine (Table 1). 27 All the targeted points were needled with the patients fully dressed and in a supine position. Acupuncture needles were single-use, sterile stainless steel, and disposable measuring 0.20 × 15 mm or 0.20 × 30 mm. The auricular points identified by anatomical locations of the ear, and needles were inserted at the appropriate locations. Next, traditional body acupuncture points were located using their standard anatomical location and needled at proper needling depth (0.5-1.5 cm) bilaterally. Needles were retained for 15 to 20 minutes followed by application of manual stimulation with even rotating method until the needling (de qi) sensation or needle grasp sensation was obtained. Total time for an acupuncture session was approximately 45 to 50 minutes per treatment. All patients in the TA group (TAG) received the same 23 auricular and body acupuncture points while all patients in the control group (CG) received NTA, which consisted of 5 acupuncture points that were not specific to the mechanisms of cachexia. Both groups received 8 weekly acupuncture interventions in consecutive weeks. For replicability and generalizability of this protocol, the Standards for Reporting Interventions in Clinical Trials of Acupuncture recommendations were implemented.28,29 Both CG and TAG received the same frequency and duration of treatment and same interaction with the acupuncture physician in the same environment during each visit. Acupuncture was provided to all patients by an acupuncturist who is trained and licensed in the United States, has long-term experience with oncology patients, and is certified by the Memorial Sloan-Kettering Cancer Center.
Acupuncture Points Applied in the Targeted (Intervention) and Nontargeted (Control) Group.
Measures
All study assessments (unless otherwise noted) were measured at baseline and then weekly until interval completion. Demographic information including cancer type and treatment, age, gender, education, marital status, ethnicity, and insurance type/status was collected at baseline.
Body Composition
Body composition was measured using the ImpediMed Imp SFB7 (ImpediMed Ltd, Eight Mile Plains, Queensland, Australia). This BIA device is known to be reproducible, noninvasive, and validated in cancer patients. 30 It is an objective tool to measure fat-free mass (FFM), ICW, and extracellular water (ECW). Weight was measured weekly and height at baseline. This information was entered into the BIA device to obtain the body composition values.
Biomakers
Blood samples were collected at the clinics for appetite hormones (leptin, ghrelin), systemic inflammation (CRP, TNF-α), nutrition status (prealbumin), and LDH. Collected blood samples were immediately stored on ice and transported to the laboratory. These samples were processed within an hour and stored at −80°C. Samples for CRP and LDH were sent to the institution’s hospital laboratory for immediate analysis while the rest of the samples remained stored until all samples were analyzed in batch.
Statistical Analysis
Independent Student’s t test and simple linear regression with P < .05 were used for statistical comparisons. All data were analyzed using Microsoft Excel 2013 (version 15.0, Microsoft, Seattle, WA) and GNU PSPP (http://www.gnu.org/software/pspp/).
Results
Cohort demographics at study enrollment are shown in Table 2. A total of 38 patients enrolled in the study, of which 30 completed the 8-week intervention (attrition rate of 21%). A majority of patients who completed the study were treated for colorectal cancer in both the TAG and CG (Table 2). Otherwise, the 2 groups were comparable in baseline demographics. Of 8 patients who withdrew from the study, 2 completed 5 treatments before withdrawal and 6 completed only 1 treatment before withdrawing. Of the 2 patients completing 5 treatments, 1 was dropped by the research team due to a protocol violation, while the other withdrew for personal reasons. The reasons for withdrawal from the study after the first treatment were primarily related to extra transportation arrangements and decreased interest in study participation. Overall, this pilot study demonstrated that the institutional setting was feasible for recruitment given the completion rate as previously confirmed by a smaller feasibility study. 31
Demographics of all Enrolled Patients and Those Who Completed the Study.

Body Composition and Weight
The FFM as an indicator of skeletal muscle mass was significantly higher among the TAG at baseline and during the whole intervention period (Figure 1A). However, no loss in FFM was noticed in either the TAG or CG with a significant increase in FFM in the CG at week 8 compared with baseline (increase by 1.671%, P = .016). The relative change in body weight over all eight weeks trended upward in the TAG during weeks 7 and 8 relative to the CG (Figure 1B). In contrast, the absolute body weight was lower in the TAG throughout the study by an average of 12 to15 lbs. and did not change significantly (Figure 1B).

(A) Absolute (in % of total body water) and relative change (in %) in fat-free mass (FFM) at baseline (week 1), after 4 weeks, and at the end (8 weeks) of the study for intervention and control groups. #P < .05 compared with control at same week, *P < .05 compared with baseline. (B) Absolute (in lbs) and relative change (in %) in body weight at baseline (week 1) and each week of the study until the end (week 8) for intervention and control groups.
Appetite Markers
The hormones leptin and ghrelin presented with variability in the TAG and CG. The absolute leptin blood levels were significantly lower in the TAG both at the baseline (3019 ± 3982 vs 7494 ± 7941 pg/mL) and end (2439 ± 3517 vs 9060 ± 8813 pg/mL) of the intervention (Figure 2). However, the relative change in concentration resulted in an average nonsignificant increase in leptin levels by 34% and 37% in the CG and TAG, respectively. The appetite-stimulating hormone ghrelin was more variable and did decrease on average by 17% in the TAG by the end of the study, which was not significant compared with the CG (Figure 2).

Absolute (in ng/mL) and relative change (in %) in leptin and ghrelin blood levels at baseline (week 1) and at the end (8 weeks) of the study for intervention and control groups. *P < .05 compared with control at same week.
Inflammatory Markers
CRP as a general inflammatory marker did not differ between the CG and TAG during the study (Figure 3A). For both groups, it remained within the normal range of 0 to 10 mg/dL. In the CG, the levels were more variable and initially higher with a decrease at week 4 and return to baseline levels at week 8. The prealbumin levels were also not different between CG and TAG although for the CG the level increased during weeks 4 and 8 compared with baseline (Figure 3A).
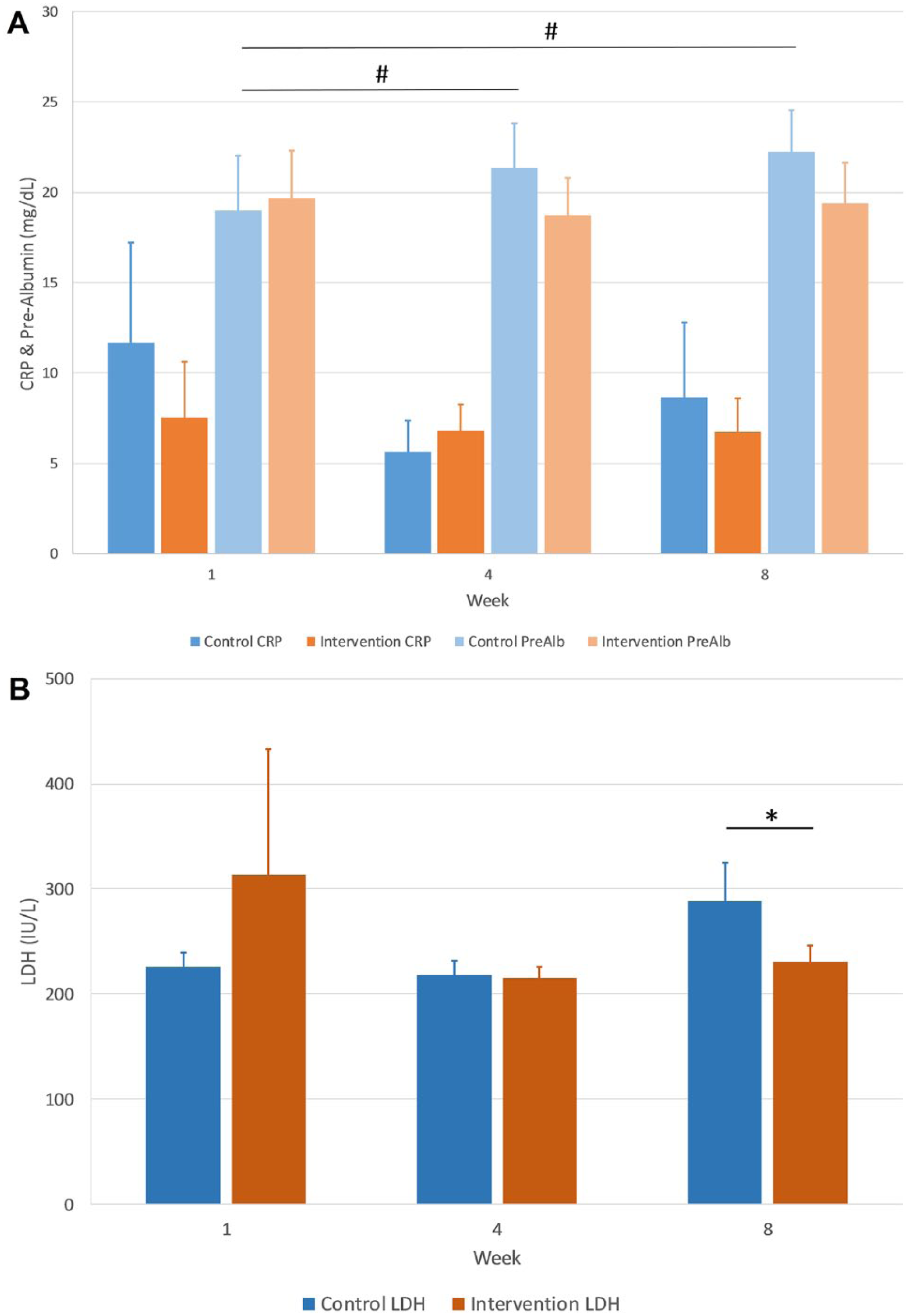
(A) C-reactive protein (CRP) and prealbumin (PreAlb) blood levels in mg/dL at baseline (week 1), after 4 weeks, and at the end (8 weeks) of the study for intervention and control groups. #P < .05 compared with baseline. (B) Lactate dehydrogenase (LDH) activity in IU/L at baseline (week 1), after 4 weeks, and at the end (8 weeks) of the study for intervention and control groups. *P < .05 compared with control at same week.
The LDH activity as a measure of muscle tissue damage was elevated for both the TAG and CG above normal range (100-190 IU/L) but increased significantly for the CG in week 8 compared with the TAG (288 IU/L vs 230 IU/L, P = .04) and to baseline (Figure 3B).
The ratio of ECW to ICW as an unspecific indicator of inflammation and cellular integrity was significantly higher for the TAG during weeks 4 and 8 compared with the CG (week 4: 0.93 vs 0.84, P = .05, week 8: 0.94 vs 0.84, P = .05) (Figure 4A). A further indicator of inflammation was the increased level of TNF-α in the TAG at study end compared with the CG (13.15 vs 9.24 pg/mL, P = .04) (Figure 4B).

(A) Extracellular-to-intracellular ratio at baseline (week 1), after 4 weeks, and at the end (8 weeks) of the study for intervention and control groups. *P < .05 compared with control at same week. (B) Tumor necrosis factor (TNF)-α at baseline (week 1) and at the end (8 weeks) of the study for intervention and control groups. *P < .05 compared with control at same week.
The outcomes evaluated in this part of the study demonstrate feasibility and utility of the objective measurements for a future larger scale trial although some of the parameters did not reach statistical significance given the small sample size.
Patients in either the TAG or CG did not report adverse events associated with the acupuncture treatment as evaluated by both the acupuncturist and the research team members who collected data. The pilot study confirmed safety of acupuncture intervention in GI cancer patients with cachexia.
Discussion
Cancer patients with cachexia are at increased risk to suffer from inferior quality of life and increased morbidity and mortality. 32 Interventions that aim to interfere with this biologic process have the potential to significantly affect the clinical benefit of a large number of patients engaged in palliative care. Weight stabilization is a critical goal for cachectic patients and serves as an indicator for maintenance of chemotherapy scheduling, quality of life, and nutritional status. 33 While the absolute weight gain in this pilot study was minimal in the TAG, no relative weight loss occurred in either group. Since neither body weight nor FFM decreased in the TAG, the pro-inflammatory mediator TNF-α may serve a different purpose as a signaling molecule. If this is specific to lipolysis or gluconeogenesis requires further research.34,35
The hormones leptin and ghrelin influence appetite oppositely with leptin suppressing food intake, while ghrelin stimulates appetite. 36 Both leptin and ghrelin blood concentrations were significantly higher in patients compared with reference values, which has been observed in prior studies for ghrelin but not for leptin. 37 While absolute leptin levels were significantly lower in the TAG, the relative change between baseline and the end of the study indicated a nonsignificant increase in leptin levels for both groups. The decrease in absolute leptin blood concentration (−0.58 ng/mL) in the TAG indicates a beneficial outcome of TA since food intake is less suppressed compared with the CG with increased leptin concentrations (+1.566 ng/mL). The significant elevation of ghrelin levels in both the CG and TAG has been reported previously in patients with GI cancer and cachexia. 37 It has been hypothesized to be associated with high insulin resistance in cachectic patients. 38 Therefore, a lowering of elevated ghrelin levels in the TAG (−5.59 ng/mL) may actually indicate an improvement of insulin resistance. However, in this study, the reduction was not significant compared with the CG potentially requiring a longer acupuncture intervention or follow-up observation.
Increased CRP serves as an unspecific indicator of systemic inflammation commonly observed in patients undergoing chemotherapy, which worsens if necessary protein cannot be provided to support cellular functions. 39 In this pilot study, both the TAG and CG had normal CRP levels and prealbumin as a marker of readily available protein remained stable in the TAG while it increased in the CG at the end of the study. It is not clear if this is related to the acupuncture points chosen for the CG, which may have increased absorption of amino acids from the GI tract. Despite this uncertainty, both groups did not indicate clinical significant levels of unspecific inflammation as indicated by CRP. Another body composition measure that relates to lean muscle mass and protein status is FFM, which was maintained throughout the study in the TAG. The absolute percentage FFM was significantly higher in the TAG from the CG, but the relative change in FFM was unaffected in although it increased in the CG, which may correlate to the increase in prealbumin, gain in protein, and increased lean muscle mass related to the choice of acupuncture point. The selection of acupuncture points for the CG may also inadvertently have increased appetite or decreased stress; outcomes that were not intended for the CG.
A more specific indicator of muscle degradation is LDH, which correlates well to lean muscle mass loss and localized inflammation. While both the TAG and the CG had clinically elevated LDH activity, by the end of the study only the CG had increased LDH activity while the TAG had lowered activity at week 4 and remained stable at week 8. This correlates to the nonsignificantly elevated CRP levels in the CG by the end of the trial. While LDH activity is specifically related to muscle tissue breakdown, it also contributes to general inflammatory processes that are reflected in elevated CRP levels. 40
Another indicator of morbidity and nutritional status is the ratio between ECW and ICW (ECW:ICW), which is less than 0.9 in healthy populations indicating a higher ICW distribution. 41 With loss of cell integrity and cell mass, a shift toward a higher amount of ECW occurs, which was the case for the TAG at weeks 4 and 8 in this study. While this shift was minimal, it indicates that cellular integrity got worse that may be linked to an ongoing inflammatory process. Confirming the elevated inflammatory process is the increased measure of TNF-α in the TAG at the end of the study. It has been shown that increased TNF-α levels are associated with longer survival times in cachectic cancer patients that also gained weight. 42
There are several limitations of this study. First, given our findings, future studies should increase the sample size and duration of acupuncture intervention to allow for the potential treatment effect to be fully observed. Second, due to inclusion of patients with various types of GI cancer and stages in this pilot study, the heterogeneity of the patient population and chemotherapy regimens limit interpretation of weight changes and appetite as well as generalizability of findings. As a pilot study, we aimed at feasibility and included subject enrollment from both academic and community practices to improve the diversity of subject inclusion. Finally, the study included sham acupuncture, which requires careful selection of acupuncture points for both the TA and the CG to exclude potential covariate effects that could lead to unexpected influences on observed parameters. The predetermined acupuncture points used in the CG are unlikely to have therapeutic effect on cachetic symptoms or underlying conditions; however, the discrepancy in total needles used between TAG and CG may have introduced bias. The 23 needles used in the TAG group is similar to what is being used for other complex conditions such as depression 43 or irritable bowel syndrome. 44
Conclusions
In this 8-week pilot study we demonstrated feasibility in design, conduct, and completion of a TA protocol in GI cancer cachexia suggesting scalability in the future. While the TAG presented with less muscle degradation (as indicated by the LDH) at the end of the study, a more active inflammatory process may have been present as suggested by a higher ECW/ICW ratio and elevated TNF-α levels. Specifically, some contradictory findings such as lower LDH levels but increased prealbumin likely due to the small sample size and heterogeneity of the patient population will require further investigation. In addition, the leptin levels in the TAG were significantly lower, which is in agreement with increased appetite and a potential weight gain observed toward the end of the study. While both groups maintained their weight, the TAG demonstrated a trend in weight gain in weeks 7 and 8, suggesting a delayed benefit to the intervention or need for prolonged administration of the TA. Further studies with a larger sample size, more homogenous patient population, and prolonged follow-up or intervention are warranted.
Footnotes
Acknowledgements
The authors would like to express deep appreciation to the patients who participated in the study and the research staff who assisted during all stages of the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the University of Florida Opportunity Research Fund, partly by the Clinical Translational Science Institute (CTSI), the National Center for Advancing Translational Sciences of the National Institute of Health (UL1TR001427), and the UF PRICE-UF Health Cancer Center Seed Grant Program to Dr. Yoon. Publication of this article was funded in part by the University of Florida Open Access Publishing Fund to Dr. Grundmann.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Institutional and/or National Research Committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
