Abstract
Background:
Aromatase inhibitors (AIs) are recommended as the preferred therapy for postmenopausal women with hormone receptor-positive (HR+) breast cancer. As a result, aromatase inhibitor-associated musculoskeletal symptom (AIMSS) have become a major problem leading to therapy discontinuation and decreased quality of life in patients receiving adjuvant AIs treatment. Multiple therapies have been attempted, but have yielded limited clinical results. This study will be performed to determine whether acupoint thread embedding (ATE) combined with Wenshen Bugu Decoction can effectively treat AIMSS, so as to improve the AIs medication compliance of postmenopausal breast cancer patients.
Methods:
This study will utilize a randomized, 2 parallel groups controlled trial design. A total of 128 eligible postmenopausal breast cancer women with AIMSS will be randomized to receive a 12-week treatment with Wenshen Bugu Decoction alone (control group) or in combination with ATE (treatment group) in a 1:1 ratio. The primary outcome will be the 12 week Brief Pain Inventory Worst Pain (BPI-WP) score. The secondary outcome measures will include response rate, Brief Pain Inventory-Short Form (BFI-SF), Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC), Functional Assessment of Cancer Therapy-Endocrine Symptom (FACT-ES), Functional Assessment of Cancer Therapy-Breast (FACT-B), bone marrow density (BMD), blood markers of bone metabolite, Morisky medication adherence scale-8 (MMAS-8), credibility and expectancy, and survival outcomes.
Discussion:
This trial may provide clinical evidence that ATE combined with Wenshen Bugu Decoction can be beneficial for treating AIMSS among postmenopausal breast cancer survivors. Our findings will be helpful to enhance the quality of life and reduce the occurrence of AIs withdrawal.
Keywords
Introduction
Breast cancer becomes the most frequently diagnosed malignancy among women globally and in China. As outlined in the latest cancer statistics, the number of new breast cancer cases rapidly reached 2.26 million worldwide by 2020, a figure that is forecast to rise to 4.4 million by 2070.1,2 Early-stage breast cancer accounts for more than 80% of all breast cancer, and 68% of breast cancer diagnoses are hormone receptor-positive (HR+), with approximately two-thirds being postmenopausal women.3,4 For decades, 5 year tamoxifen has been regarded as the gold standard for adjuvant endocrine therapy for early breast cancer. 5 Compared with tamoxifen, the third-generation aromatase inhibitors (AIs) have been shown to significantly improve disease-free survival (DFS) and reduce the risk of recurrence of contralateral breast cancer among postmenopausal women with HR+ breast cancer. 6 As a result, the third-generation AIs are recommended as the mainstay treatment of HR+ breast cancer in postmenopausal women. 7
Five-year treatment with AIs is associated with approximately 40% reduction in 10 year breast cancer mortality among early-stage HR+ breast cancer. 8 In breast cancer survivors, AIs suppress the aromatase enzyme, resulting in reduced estrogen concentrations and improved survival. 9 However, many patients experience adverse effects, the most common of which is aromatase inhibitor-associated musculoskeletal symptom (AIMSS), which occurs in approximately 47% of breast cancer survivors 10 and contribute to frequent discontinuation of AIs therapy and increased mortality.11,12 The primary clinical presentations of AIMSS patients include symmetrical pain or stiffness in the wrist, knee, and finger joints, as well as myalgia. 13 Symptoms can self-resolve within 2 weeks after AIs discontinuation but re-emerge upon resumption of use of AIs.10,14 It has been shown that extended use of AIs for 5 to 10 years reduces the risk of breast cancer recurrence and breast cancer in the contralateral region, 15 while prolonging the treatment period will inevitably be accompanied by a longer duration of pain and other uncomfortable symptoms. Hence, it has become an urgent issue to alleviate AIMSS and improve AIs medication adherence, to maintain or prolong survival and improve the quality of life of breast cancer patients.
Despite the fact that a considerable number of studies have evaluated systemic therapies for AIMSS in recent years, including vitamin D, duloxetine, omega-3 fatty acids, bisphosphonates, and physical exercise, 10 there are few evidence-based solutions for AIMSS management due to the large heterogeneity of intervention and study methodologies. 16 Due to current therapies having a poor curative effect on AIMSS, many clinicians and patients are trying to achieve benefits through Traditional Chinese Medicine (TCM) therapy.
The typical TCM therapies are composed of Chinese Herbal Medicine (CHM) and acupuncture-related techniques (ART). Our previous study found that Wenshen Bugu Decoction could decrease bone loss and reduce the risk of fracture caused by AIs of postmenopausal women with breast cancer. 17 Still, there are a considerable number of patients with AIMSS who cannot be relieved by Wenshen Bugu Decoction alone. ART has been widely used to control cancer pain, 18 joint pain, 19 and osteoporosis. 20 ART is recommended for breast cancer patients who suffer from AIMSS. The results of a high-quality study that enrolled 226 postmenopausal women with early-stage breast cancer and AIMSS demonstrated that acupuncture twice a week for 6 weeks followed by 1 session per week for another 6 weeks could statistically reduce joint pain and stiffness, although the clinical significance of improvement was uncertain and the cost of acupuncture intervention was out of insurance coverage. 21 Another ART is developed based on extended needle retention and is called acupoint thread embedding (ATE). By implanting absorbable catgut into acupoints, the acupuncture sensation would be magnified and prolonged, thus theoretically enhancing the efficacy. 22 Its simulation induction holding time can reach 7 to 14 days, which compensates for the disadvantages of acupuncture, such as short periods, more visiting times, difficulty in consolidating efficacy, and ease of relapse.23,24 As a result, ATE is expected to achieve better efficacy than traditional acupuncture by inserting and manipulating needles in treating AIMSS. No single mitigation strategy has been found to be effective for AIMSS. The present study aims to evaluate the efficacy and safety of ATE combined with Wenshen Bugu Decoction for the treatment of AIMSS in postmenopausal early-stage breast cancer survivors.
Methods and Design
Study Design
We will conduct a prospective, randomized, parallel-group controlled trial. An estimated 128 eligible subjects will be randomized to receive either Wenshen Bugu Decoction alone (control group) or in combination with ATE (treatment group). The study protocol follows the Consolidated Standards of Reporting Trials (CONSORT) statements 25 and the Standards for Reporting Interventions in Clinical Trials of Acupuncture (STRICTA). 26 The study flow diagram is shown in Figure 1, while the schedule of enrollment, interventions, and assessments is illustrated in Figure 2.

Study flow diagram.

The schedule of trial enrollment, interventions, and assessments.
Participant Recruitment
Postmenopausal breast cancer women, experiencing AIMSS after taking AIs, admitted to the breast clinic will be enrolled at Yueyang Hospital of Integrative Chinese and Western Medicine, Shanghai University of Traditional Chinese Medicine (referred to as “Yueyang Hospital”) between October 2022 and December 2023, with follow up to December 2026. After being informed about the study, and potential risks/benefits, patients are required to give written informed consent prior to randomization, followed by eligibility screening.
Inclusion criteria
Patients will be included if they fulfill all of the following criteria:
(1) Postmenopausal females aged 45 to 75 years. If one of the following criteria is met, these women are defined as postmenopausal: (i) after bilateral ovariectomy; (ii) aged ≥60 years; (iii) aged <60 years, natural menopause for at least 12 months, without chemotherapy, tamoxifen, toremiphene, or ovarian removal within 1 year, and follicle-stimulating hormone (FSH) and estradiol reached postmenopausal levels; (iv) age <60 years and receiving tamoxifen or rosuvastatin with postmenopausal levels of FSH and estradiol.
(2) Patients with histologically confirmed primary invasive estrogen receptor- and/or progesterone receptor-positive carcinoma of the breast (stage I-III).
(3) Patients who are currently taking a third-generation AI for at least 1 month and are scheduled to continue taking the AIs during the study period.
(4) Patients who reported ongoing pain and/or stiffness in one or more joints, which started or worsened after initiation of AIs therapy.
(5) Patients scored at least 3 on the Brief Pain Inventory Worst Pain (BPI-WP) item (score range, 0-10; higher scores indicate greater pain) within the past week.
(6). Patients with an Eastern Cooperative Oncology Group performance status (ECOG PS) scored 0 to 2.
(7). Patients who consent voluntarily to participate in this study.
Exclusion criteria
Patients who meet at least 1 of the following are excluded:
(1) Having a history of fracture or surgery in the previous 6 months.
(2) Suffering from inflammatory arthritis, such as rheumatoid arthritis or ankylosing spondylitis.
(3) Intake of non-steroidal anti-inflammatory drugs (NSAIDs), opioid analgesics, or any other medical therapy for the treatment of joint pain or stiffness within 1 month prior to registration.
(4) The presence of contraindications to ATE, such as allergy to the materials and severe bleeding tendency.
(5) Metastatic breast cancer or combined with other primary malignant tumors.
(6) Patients complicated with severe heart, brain, kidney, and other organ dysfunctions, infectious diseases, and mental and behavioral abnormalities are not easy to observe.
(7) Having received acupuncture for joint symptoms due to AIs or having received acupuncture within 1 month prior to study entry.
Randomization and Allocation Concealment
Randomization will be conducted using a central web-based central randomization system (http://crk.sdwgem.com/shiyan-jiuyao/login.php). An independent statistician generated a blocked randomization sequence using the statistical program SAS 9.3 (SAS institute Inc, Cary, NC). The study will follow a randomized block design, with the block sizes changing dynamically. The clinical research coordinator will input the patient information on a computer and will be given a random number and grouping.
Blinding
Outcome assessors and statisticians who perform the statistical analyses will be blinded to group assignment.
Intervention
Pathogenesis of TCM
Deficiency of the spleen and kidneys is a common cause of bone and joint symptoms, as well as breast cancer. Strengthening the spleen and kidneys not only treats bone and joint symptoms but also inhibits the development of breast cancer. Therefore, the treatment should follow the principles of tonifying the kidney and strengthening the spleen, preventing arthralgia and dredging collaterals.17,27
Treatment group
ATE combined with Wenshen Bugu Decoction was used for treatment. ATE therapy will be applied to patients fortnightly, a total of 6 sessions during 12 weeks. All ATE treatment sessions will be performed by an acupuncturist who has at least 3 years of clinical experience in acupuncture practice. The Wenshen Bugu Decoction will be made in the decoction room of Yueyang Hospital and dispensed to the patient.
(1) ATE: The selection of acupoints was developed from clinical practice and literature review and includes 4 obligatory acupoints and 2 adjunct acupoints. The obligatory acupoints include qihai (CV6), guanyuan (CV4), pishu (BL20), and shenshu (BL23). Two adjunct acupoints will be chosen from the following combinations based on the patient’s most painful joint areas: jianyu (LI15) with jianliao (SJ14), shousanli (LI10) with quchi (LI11), zusanli (SP10) with yanglingquan (GB34), yaoyangguan (DU3) with dachangshu (BL25), huantiao (GB30) with zhibian (BL54), and sanyinjiao (SP6) with xuanzhong (GB39). The specific anatomical locations are described in Table 1 and shown in Figure 3.
Locations of the Obligatory and Adjunct Acupoints for ATE Treatment.

Abbreviations: ATE, acupoint thread embedding; BL, bladder channel; CV, ren channel; LI, large intestine channel; SP, spleen channel; GB, gallbladder channel; GU, governor channel.
1 cun (≈20 mm) is defined as the width of the interphalangeal joint of the thumb.

Acupoints for ATE therapy: (A) obligatory acupoints for ATE therapy (CV4, CV6, bilateral BL20, and bilateral BL23), (B) adjunct acupoints for shoulder joint (bilateral SJ14 and bilateral LI15), (C) adjunct acupoints for finger/wrist joint (bilateral LI10 and bilateral LI11), (D) adjunct acupoints for knee joint (bilateral SP10 and bilateral GB34), (E) adjunct acupoints for waist (bilateral GU3 and bilateral BL25), (F) adjunct acupoints for hip (bilateral GB30 and bilateral BL54), and (G) adjunct acupoints for ankle joint (bilateral SP6 and bilateral GB39).
The operated apparatus is disposable. The obligatory acupoints will be embedded with 3-0 medical catgut threaded into the No.7 needle (0.7 mm × 60 mm), and the adjunct acupoints will be embedded with 3-0 medical catgut threaded into the No.6 needle (0.6 mm × 60 mm). The disposable embedding needles and absorbable catgut are shown in Figure 4. The reason for choosing this size was based on clinical practice, specifically, those previous patients who received this size of suture reported better treatment response and less discomfort sensation. Subsequently, the acupuncturist will scrub with 75% alcohol disinfectant at the selected acupoints, and the needle was rapidly inserted into the acupoints and pushed forward. The needle will be inserted at a depth of 0.5 to 2.0 cun into the acupoints. The depth was consistent with routine acupuncture insertion depth in previous trials. Until arriving at the right depth while the patients get de qi sensation, the needle tubing will be withdrawn and the suture segments were left. Finally, the pinhole will be pressed with a sterilized cotton ball for a while, and cover the needle hole with infusion tape to prevent infection, hemorrhage, and bruising. The participators will be required to keep the pinholes away from water within 24 hours.
(2) Wenshen Bugu Decoction: The drug composition of decoction includes Astragalus (Huangqi), Codonopsis (Dangshen), Atractylodes (Baizhu), Poria cocos (Fuling), Adenophora tetraphylla (Nanshashen), Epimedii (Xianlingpi), Morinda Officinalis (Bajitian), Cistanche (Roucongrong), Curcuma zedoaria (Ezhu), Salvia chinensis (Shijianchuan), Fructus Psoraleae (Buguzhi), Rhizoma Drynariae (Gusuibu), Achyranthes bidentata (Huainiuxi), Himalayan Teasel Root (Xuduan), Anemarrhena (Zhimu) and Cortex Phellodendri (Huangbai). The herbs are boiled and extracted into 200 mL water per dose. Orally administered daily, twice a day for 12 weeks.
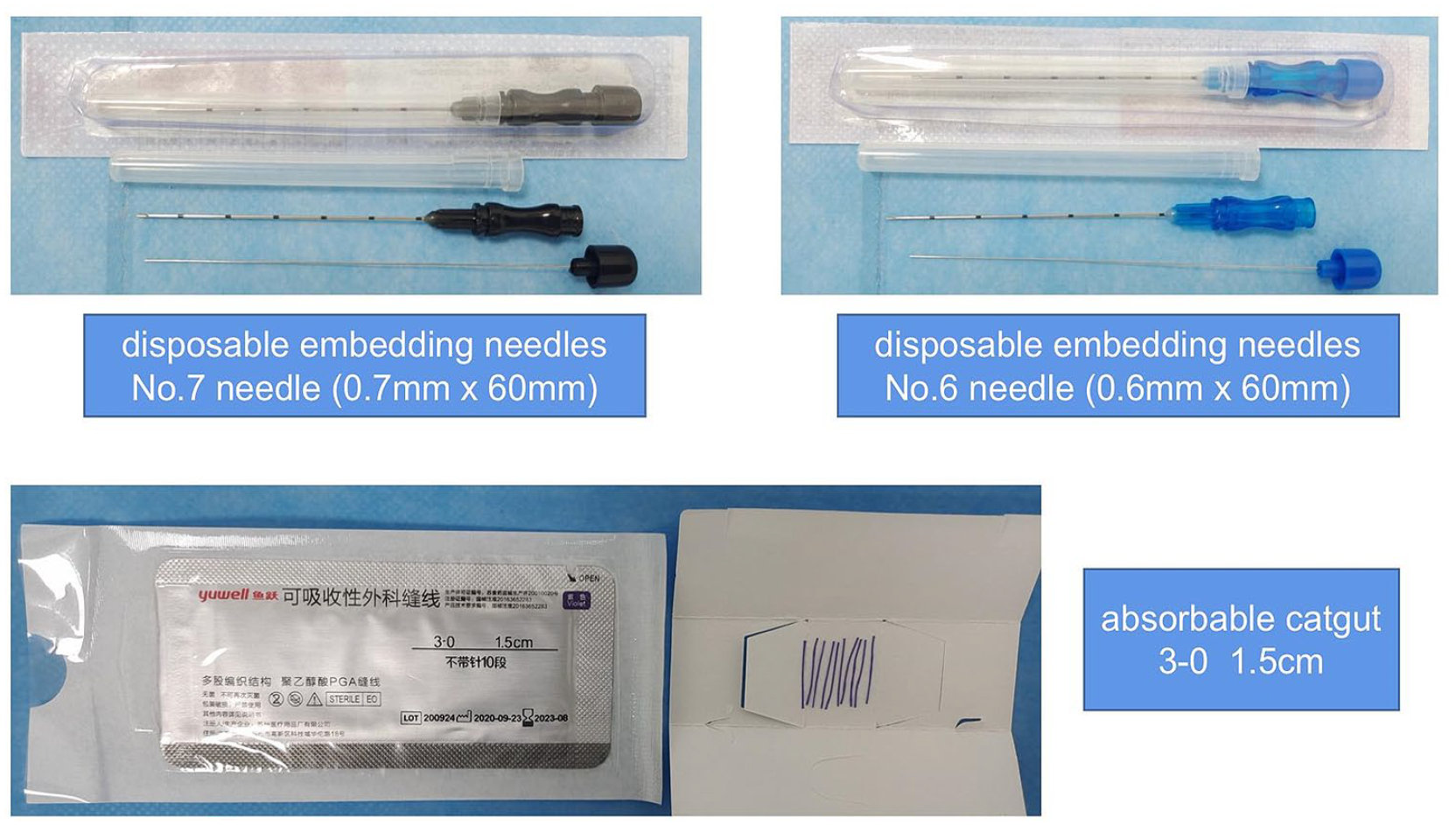
Disposable embedding needles and absorbable catgut.
Control group
Similarly, following a 12 week treatment with Wenshen Bugu Decoction alone, the remaining procedure is identical to that of the treatment group.
Concomitant medication
All subjects will be required not to use other TCM therapies for AIMSS, as well as NSAIDs and opioid analgesics during the study period. Participants will be allowed to take antiosteoporotic drugs throughout the trial, and details should be recorded on the concomitant medications form if used.
Outcome Measurement
Primary outcome
The primary outcome will be subjective pain as assessed by the BPI-WP at 12 weeks after randomization, with scores ranging from no symptoms (0 point) to worst (10 points).
Secondary outcomes
The response rate
Two points on the BPI-WP are identified as a minimal clinically important improvement (MCII) for a patient. 28 The response rate was defined as the proportion of patients with BPI-WP decreased by more than 2 points from baseline to treatment endpoint (at week 12).
Brief Pain Inventory-Short Form (BPI-SF)
The BPI-SF is a self-report questionnaire comprising 9 items with 15 questions designed to measure multiple aspects of pain and pain interference with daily activities. 29 The outcome will be assessed at baseline, week 2, week 4, week 6, week 8, week 10, week 12, week 14, week 16, week 18, week 20, week 22, and week 24. This means that the BPI-WP score as part of the BPI-SF questionnaire will also be evaluated every 2 weeks until 24 weeks after randomization.
Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC)
The WOMAC is a 24-item questionnaire with 3 subscales: pain is addressed in 5 questions (WOMAC-A), morning stiffness in 2 (WOMAC-B), and physical condition in the remaining 17 questions (WOMAC-C). 30 The score of each item is ranging from 0 to 5, with higher scores indicating greater symptom severity. The WOMAC will be measured at baseline, week 6, week 12, week 18, and week 24.
Functional Assessment of Cancer Therapy-Endocrine Symptom (FACT-ES)
The overall levels of menopausal symptoms will be assessed at baseline and weeks 6, 12, 18, and 24 after randomization using the FACT-ES. FACT-ES, a 46-item endocrine symptom scale, measures endocrine symptoms as well as psychological, social/family, emotional, and functional well-being. 31 Each item is graded on a five-point Likert scale. Responses for each item will be scored with points on a five-point Likert scale as follows: “not at all” (score 0), “a little” (score 1), “some” (score 2), “quite a bit” (score 3), and “very much” (score 4). A lower score indicates fewer perimenopausal symptoms.
Functional Assessment of Cancer Therapy-Breast (FACT-B)
The FACT-B will be assessed at baseline and weeks 6, 12, 18, and 24 after randomization. FACT-B (version 4, 1997) assesses breast cancer-specific health-related quality of life (HRQOL) through a 37-item self-report instrument which includes a 27-question FACT-General (FACT-G) survey as well as 10 questions asking about additional concerns. 31 The total FACT-B score ranges from 0 to 108, with higher scores indicating a better quality of life.
Bone marrow density (BMD)
BMD of the lumbar spine (L1-L4) and the femoral neck will be measured by dual-energy X-ray absorptiometry at baseline and 24 weeks after randomization to compare changes in BMD. According to the World Health Organization, osteoporosis is defined as a T-score ≤ −2.5, osteopenia between −2.5 and −1.0, and normality is greater than −1.0. 32
Bone metabolic markers
Serum levels of bone metabolic markers, such as propeptide of type I procollagen (PINP), beta C-terminal cross-linked telopeptides of type I collagen (β-CTX), bone alkaline phosphatase (B-ALP), parathyroid hormone (PTH), 25 hydroxy vitamin D (25-OH-VD), serum calcium and phosphate, and 6 sexual hormones (including the follicle-stimulating hormone [FSH], luteinizing hormone [LH], estradiol [E2], progesterone [P], testosterone [T], prolactin [PRL]), will be measured in all patients at baseline and then at week 12 and 24 to elucidate the possible mechanisms.
Morisky Medication Adherence Scale-8 (MMAS-8)
AIs medication adherence will be assessed using the MMAS-8, an 8-item generic self-report questionnaire. 33 MMAS-8 scores range from 0 to 8, with 8 indicating high adherence, 6 to <8 indicating medium adherence, and <6 indicating low adherence. 34 Assessment evaluations will be conducted at baseline, week 6, week 12, week 18, and week 24.
Survival follow-up
The 1 and 3-year DFS and overall survival (OS) of all participants will be tracked.
Credibility and expectancy
The treatment will last for 12 weeks, and the patient’s expectations and judgments of credibility will be measured using the Credibility/Expectancy Questionnaire at the end of the first and last scheduled treatment, respectively. 35
Safety Assessment
Patient safety will be monitored continuously during the course of this study and will be summarized at the end of the study. Safety endpoints involve laboratory indexes (blood routine tests, liver functions, renal functions, and electrocardiograms), adverse events (AEs), and serious adverse events (SAEs). AEs and SAEs will be recorded and graded with the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE, version 5.0). 36 Possible AEs related to ATE include allergy, infection, subcutaneous hematoma, bleeding, and so forth. The occurrence, duration, severity, measures adopted, and outcomes of the AEs should be recorded in case report forms (CRFs) and electronic case report forms (eCRFs). SAEs will be reported to the Data and Safety Monitoring Board (DSMB) within 24 hours as directed.
Data Management and Monitoring
Data entry and data management will be implemented using CRFs and eCRFs through the electronic data capture (EDC) system, developed by the Beijing Huajing Technology Co., LTD. The researchers will be trained on how to collect data, record data, enter data electronically, and manage data using the system before study initiation to standardize the procedures. To ensure that no one will ever see the participant’s information, it will be held in the EDC system with restricted access. As soon as the study is complete, EDC’s system will be locked. After publication, the study files will be kept securely for at least 5 years. Readers and reviewers can ask the relevant author for the original data if they are still uncertain about our trial.
A DSMB, composed of authorities in acupuncture, breast surgery, oncology, methodology, and statistics, has been organized to oversight and monitor clinical trial conduct, and review data quality. If necessary, they will recommend modifications or termination of the trial at their discretion.
Sample Size
The PASS 15.0 was applied to calculate the sample size in this study. The sample size was calculated based on the BPI-WP scores of the treatment and control groups in week 12. The primary study hypothesis is that ATE combined with Wenshen Bugu Decoction is more effective than Wenshen Bugu Decoction alone in reducing BPI-WP score at week 12 in postmenopausal breast cancer survivors. Regarding the preliminary study and literature,21,37 the BPI-WP score of the treatment group and the control group are expected to be (3.85 ± 2.23) points and (4.80 ± 1.41) points, respectively. With α = 0.05 (2-sided), and β = 0.20, the goal was to enroll 58 participants in each group. Assuming a 10% dropout rate, the final sample size was calculated as 128 (64 in each group).
Statistical Analyses
All outcomes will be extracted for the modified intention-to-treat (mITT) population, defined as all randomly assigned participants who are exposed to scheduled therapy without protocol violations throughout the study. Continuous variables will be expressed as means and standard deviation or medians (interquartile range), and categorical variables will be reported as counts and percentages. The baseline demographics, tumor characteristics, and treatment characteristics were compared using variance and chi-squared tests. For group comparisons, continuous and categorical variables were compared using t-tests, Wilcoxon rank-sum tests, or chi-square tests, as appropriate. A log-rank test was used to compare time-to-event variables based on the Kaplan-Meier method.
The primary analysis will be a difference in the mean pain score from BPI-WP between the groups. The secondary analysis will be to compare the mean score from BPI-SF, the WOMAC, the FACT-ES, the FACT-B, the MMAS-8, and the Credibility/Expectancy Questionnaire, and the changes in BMD and bone metabolic markers, and the proportion of the response rate, and the distribution of DFS and OS between the groups. A per-protocol (PP) analysis of the primary outcome will be performed restricted to participants receiving the 12 week treatment as specified in the protocol and accomplishing the outcome measures to test sensitivity. We conducted multiple imputations for the primary outcome variable to ensure that participants with missing outcomes did not skew our conclusions. The missing values for secondary outcomes and safety outcomes will be not replaced. Statistics will be performed with IBM SPSS Statistics software (version 22.0), with statistical significance being set at P < .05.
Discussion
Currently, the third-generation AIs have become the drug of choice for postoperative adjuvant treatment of postmenopausal patients with HR+ breast cancer.38,39 Patients taking AIs are often forced to discontinue their treatment by AIMSS. In general, therapeutic strategies in Western medicine are symptomatic offering a limited amount of clinical benefit. TCM (CHM or ART) is widely regarded as a safe alternative to western medicine, and patients are increasingly seeking effective TCM treatments for their ailments. 40 Due to the limited efficacy of single therapy, we will conduct the present randomized controlled trial to evaluate the benefits of ATE combined with CHM (Wenshen Bugu Decoction) for the treatment of AIMSS in postmenopausal breast cancer survivors, which is the first known randomized controlled trial of ATE in combination with the decoction.
In a previous study, we observed the effects of Wenshen Bugu Decoction on the bone loss caused by AIs of postmenopausal women with breast cancer. 17 The findings indicated that Wenshen Bugu Decoction can, in comparison to no treatment, decrease the bone loss, and lower the risk of fracture. The alleviation of AIMSS was not assessed because this trial was designed to address bone loss in postmenopausal breast cancer patients. In clinics, breast cancer survivors with AIMSS were treated with Wenshen Bugu decoction alone, but some patients failed to respond robustly to treatment. No single treatment has proven to be effective and efficient for AIMSS, whether in TCM or western medicine. ATE is more effective than traditional acupuncture due to the persistent stimulation of acupoints by the embedded catgut. Several studies have shown that ATE therapy can be applied to a variety of diseases with significant efficacy, including shoulder and neck pain, lumbar and leg pain, cancer pain, postoperative pain, postmenopausal osteoporosis (PMOP), etc.41-43 Also, the time costs of treatment for outpatients are greatly reduced due to the small number of ATE treatments and the long time interval between treatments, which helps to improve subject compliance and facilitates the development and implementation of clinical studies. Owing to its long-lasting efficacy and time advantages, we could consider utilizing ATE in combination with decoction to treat AIMSS. The only published paper on ATE in the treatment of AIMSS reported that Calcium Carbonate-Vitamin D3 in combination with ATE can effectively treat AI-induced osteopenia in breast cancer patients, as well as have a certain effect on the estrogen levels. 44 Evidence quality of this study is limited by insufficient sample size and poor methodological rigor. Instead, we will conduct a methodologically rigorous, high-quality randomized controlled trial to compare the efficacy of ATE combined with Wenshen Bugu decoction on AIMSS in relation to decoction alone among postmenopausal breast cancer survivors. The significance of this study is to improve the quality of life and prolong survival time by improving AIMSS and thus their medication compliance. In contrast to previous studies, one additional strength of our study is that we will additionally assess AI medication adherence, as well as DFS and OS at 1 and 3 years.
Several limitations are important to note. Firstly, this is a single-center study, and the study population deriving from a single hospital, which may not represent the wider population of postmenopausal breast cancer patients with AIMSS. This is due to the COVID-19 environment, and we plan to conduct multi-center studies in the future to further verify the results. Secondly, there was no further survival follow-up after 3 years after randomization. For most postmenopausal women with HR+ breast cancer, adjuvant endocrine therapy is recommended for 5 to 10 years. Therefore, a follow-up of 5, 10, or even longer survival might be necessary. However, limited by research project funding, we are only able to carry out this part of the survival follow-up, record, and analyze the data by ourselves.
Together, we sincerely hope that the results of this rigorous randomized controlled will provide an objective, high-quality evidence base to support the application and promotion of ATE combined with Wenshen Bugu Decoction in postmenopausal breast cancer patients with AIMSS.
Supplemental Material
sj-docx-1-ict-10.1177_15347354231188679 – Supplemental material for Acupoint Thread Embedding Combined With Wenshen Bugu Decoction for the Treatment of Aromatase Inhibitor-Associated Musculoskeletal Symptom Among Postmenopausal Breast Cancer Patients: Study Protocol of a Randomized Controlled Trial
Supplemental material, sj-docx-1-ict-10.1177_15347354231188679 for Acupoint Thread Embedding Combined With Wenshen Bugu Decoction for the Treatment of Aromatase Inhibitor-Associated Musculoskeletal Symptom Among Postmenopausal Breast Cancer Patients: Study Protocol of a Randomized Controlled Trial by Xuan Zou, Zi-Yi Chen, Yu-Han Yang, Yu Qiao, Shu-Jin He, Qiong Li, Wei-Li Chen, Xin-Yue Zhang, Si-Yu Li, Shan-Yan Sha, Min-Hao Hu, Xi-Yin Zhang, Ming-Ju Yang, Rui-Ping Wang, Huan-Gan Wu, Yin Shi, Xiao-Hong Xue and Ya-Jie Ji in Integrative Cancer Therapies
Footnotes
Acknowledgements
We express our deepest appreciation to each employee of Beijing Huajing Technology Co., LTD for generating the EDC system and assisting in the monitoring and verification of the accuracy of data. We thank all researchers for their joint effort to promote the future success of the study. We would like to thank in particular Shanghai Biochip Co., and Cinoasia Institute for their excellent technical support.
Author Contributions
YJJ, XHX, HGW, and YS conceived the idea behind the trial. YJJ, XHX, and XZ contributed to the design. QL, XYZ, SYL, SYS, and MHH were responsible for recommendation participants. YHY, YQ, SJH, WLC, XYZ, and MJY helped with the implementation of the study. ZYC was responsible for collecting and recording the data. RPW is responsible for the statistical analysis. YJJ and XZ draughted and strictly revised the manuscript for important intellectual content. XHX sought funding. YJJ and XHX obtained ethical approval. All authors revised and approved the final manuscript.
Availability of Data and Materials
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shanghai Committee of Science and Technology, China (No. 22Y11923000). The sponsors have no roles in the design of the study; collection, analysis, and interpretation of data; and writing of the manuscript.
Ethics and Dissemination
Ethics approval was obtained from the Ethical committee of Yueyang Hospital of Integrated Chinese and Western Medicine, Shanghai University of Traditional Chinese Medicine (No. 2022-005). The results will be disseminated through peer-reviewed publications.
Ethics Approval and Consent to Participate
The study protocol (version 2.0, 10 February 2022) has been approved by the IRB of Yueyang Hospital of Integrated Chinese and Western Medicine, Shanghai University of TCM (No. 2022-005). All participants will provide oral informed consent and written informed consent before participating in this study.
Trial Status
The study started in October 2022 (October, 25th: the first participant enrolled) and is currently ongoing.
Trial Registration
Chinese Clinical Trial Registry, ChiCTR2200064982. Registered on 25 October 2022.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
