Abstract
Background:
Non-cancer drugs are currently being repurposed for cancer treatment. Mounting evidence highlights the influence of calcium channels on tumorigenesis and progression. Hence, inhibition of calcium signaling may be a promising cancer treatment strategy.
Objective:
In this study, we aimed to examine whether calcium channel blockers (CCBs) affect the efficacy of epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs) in patients with non-small cell lung cancer (NSCLC).
Design:
We conducted a retrospective analysis.
Methods:
In this study, conducted between January 2009 and June 2021, patients with NSCLC treated with erlotinib, or gefitinib for at least 1 week were enrolled and divided into 2 groups: CCBs-/EGFR-TKIs+ and CCBs+/EGFR-TKIs+, depending on whether they received CCB therapy. Progression-free survival (PFS) and overall survival (OS) were determined as the primary and secondary endpoints, respectively.
Results
: The estimated median PFS and OS for the CCBs-/EGFR-TKIs+group were 7.70 and 12.17 months, respectively, and they were significantly different from those of the CCBs+/EGFR-TKIs+ group (10.43 and 18.07 months, respectively). CCB use was associated with improved PFS (adjusted hazard ratios [HR] 0.77, 95% confidence interval [CI]: 0.61-0.98; P = .035) and OS (adjusted HR 0.66, 95% CI: 0.51-0.84; P < .001).
Conclusion:
Calcium channels have been implicated in cancer pathogenesis. Our findings revealed the potential additive anticancer effects of CCBs when used concomitantly with EGFR-TKIs. However, study limitations, including the retrospective nature and small number of patients, necessitate large-scale prospective studies on the therapeutic potential of CCB as an adjunctive therapy with EGFR-TKIs in patients with NSCLC.
Keywords
Introduction
Globally, lung cancer is the leading cause of cancer-related deaths among men and women and is expected to have killed around 132 000 people in the United States in 2021. 1 The prognosis of lung cancer detected at an advanced disease stage is considerably poor. 2 Among patients with advanced lung adenocarcinomas, 51.4% in Asia and 15% to 22% outside Asia have epidermal growth factor receptor (EGFR) mutations.3,4 EGFR tyrosine kinase inhibitors (TKIs) have increased efficacy in patients with lung adenocarcinoma who harbor sensitive EGFR mutations.5,6 The EGFR-TKIs that have been approved by the Food and Drug Administration for treating metastatic non-small cell lung cancer (NSCLC) include gefitinib, erlotinib, osimertinib, and afatinib. Patients with NSCLC frequently present with EGFR exon 19 deletions and exon 21 mutations (L858 R) in tumor tissue. Thus, EGFR mutations can be used to predict the effectiveness of EGFR-TKIs. 7
Calcium channel blockers (CCBs), such as diltiazem, amlodipine, verapamil, and nifedipine, are commonly used for the treatment of cardiovascular disorders. Voltage-gated Ca2+ channels are classified into at least 5 different subclasses (L-, N-, P-, Q-, and R-types). 8 CCBs inhibit the influx of calcium through calcium channels as well as calcium release from intracellular stores. 9 Previous studies have revealed an association of calcium channels with cancer pathogenesis. Calcium channels are involved in signal transduction pathways associated with cellular activities such as growth, proliferation, and programmed cell death. 10 NSCLC cell lines have been used to assess the important function(s) of calcium channels in lung cancer. 11 Thus, inhibiting calcium channels could potentially alter cellular signal transduction and affect the progression of lung cancer. Nevertheless, conflicting results have been reported. For instance, a deficiency in the expression of gene encoding CACNA2D2, which is a subunit of the Ca2+-channel complex, implies a potential association between this gene and Ca2+ signaling in the etiology of not only lung cancer but also other types of cancer. 12 The discrepancy in the outcomes could be attributed to the distinct subunits of L-type Ca2+-channels that are expressed by tumor cells, which determine whether Ca2+ contributes to tumor cell proliferation or apoptosis. The activation of calcium signaling is widely implicated in cancer progression by promoting the malignant behaviors of cancer cells, contributing to the further activation of several oncogenes and oncogenic pathways, and also suppressing immune function. Therefore, the inhibition of calcium signaling may be an effective cancer treatment strategy. 13 Several non-cancer drugs have the potential to alter the tumor microenvironment and increase the efficacy of cancer drugs when concurrently administered.14,15 To the best of our knowledge, no prospective trial to date has demonstrated the effects of CCBs on survival outcomes of patients with cancer.
Proton pump inhibitors (PPIs), the most widely prescribed gastric acid suppressants, are used by up to 33% of patients cancer. They decrease gastric acid secretion by inhibiting H+/K+-ATPase, which can relieve stomach syndrome associated with anticancer drugs. Currently, information on the safety of using PPIs concurrently with anticancer drugs is limited. It has been reported that PPIs can increase gastric pH, which in turn can decrease the solubility of EGFR-TKIs, resulting in reduced absorption. 16
In the present study, we aimed to investigate the effect of CCBs on EGFR-TKI treatment in patients with NSCLC. Furthermore, we assessed the effects of CCBs and PPIs, either alone or in combination, on the effectiveness of EGFR-TKIs in patients with NSCLC.
Materials and Methods
Study Design and Patient Recruitment
We retrospectively screened the files of patients with NSCLC who were admitted to a regional teaching hospital in Central Taiwan and were taking EGFR-TKIs between January 2009 and June 2022. The inclusion criteria were as follows: (1) patients with NSCLC who had the International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10-CM) code C34.0–C34.9; (2) patients aged >20 years; (3) patients with cytological or histological confirmation of stage IIIA, IIIB, or IV NSCLC who presented with EGFR mutations based on the Study of Lung Cancer (8th edition) of the Tumor Node Metastasis Staging classification; and (4) patients who were prescribed EGFR-TKIs (gefitinib or erlotinib) and treated for at least 1 week before enrollment. Patients with incomplete electronic medical records were excluded. The index date of patients was defined as the day when EGFR-TKI therapy was initiated. The study was performed in accordance with the Declaration of Helsinki and approved by the Research Ethics Committee of [BLINDED FOR REVIEW]. The consent to participate was waived owing to the retrospective nature of the study.
CCB Exposure
The period of exposure to CCBs was calculated from the index date till the end of the study. Patients were divided into the following 2 groups: CCBs-/EGFR-TKIs+ and CCBs+/EGFR-TKIs+. The CCBs-/EGFR-TKIs+ group consisted of patients who were not on CCB therapy (control group). The CCBs+/EGFR-TKIs+ group consisted of 113 patients, to whom CCBs (amlodipine, felodipine, lercanidipine, nifedipine, diltiazem, or verapamil) were administered, whose EGFR-TKI therapy overlapped by at least 70% and the mean drug ratio (CCB/EGFR-TKI) was 86.7% (95% CI: 81.0-92.4).
Statistical Analysis
The primary outcome of this study was progression-free survival (PFS), which was defined as the interval between the initiation of EGFR-TKIs and disease progression. The secondary outcome was overall survival (OS), which was defined as the interval between the initiation of EGFR-TKIs and the death of a patient. The day of EGFR-TKI treatment initiation was designated as the index date. Patients were followed up until the clinical endpoint or June 30, 2022.
Patients’ data before treatment, including CCB use status, age, sex, smoking history, Eastern Cooperative Oncology Group (ECOG) performance status, number of metastasis, presence of brain metastasis, and PPI use, were obtained and employed for subsequent univariable and adjusted analyses.
In this study, categorical variables were compared using the chi-squared test, and continuous variables were compared using the independent t-test. The Cox proportional-hazards model was employed to analyze all selected variables related to the clinical outcomes. Results are reported as hazard ratio (HR) with 95% confidence interval (CI). All statistical analyses were conducted using SAS v9.3 (SAS Institute Inc., Cary, NC, USA) and SPSS v28 (IBM Corp., Armonk, NY, USA) statistical software. Results with P < .05 were considered statistically significant for all analyses. In this study, the Cox regression model was devised with a 0.93 overall probability of death based on a sample of 345 (CCB- plus CCB+) observations, which achieved an 82% power to detect a regression coefficient of .71 at a significance level of .05.
Results
Between January 2009 and October 2021, 376 patients were diagnosed with NSCLC based on the ICD-10-CM system and categorized as C34.0–C34.9. Ultimately, 345 patients (136 male and 209 female patients) who met the study’s inclusion criteria were enrolled. The mean age of the participants was 65.1 ± 11.7 years (Figure 1). The CCBs-/EGFR-TKIs+ and CCBs+/EGFR-TKIs+ groups comprised 232 (67.2%) and 113 (32.8%) patients, respectively. A majority of the patients (n = 312, 90.43%) had stage IV cancer at baseline, whereas the rest had stage IIIA or IIIB NSCLC. Brain metastases were not observed in 61.7% (n = 213) of the patients, and 78.0% (n = 269) of the patients had an ECOG performance status score of 0 to 1 at the start of treatment with EGFR-TKIs. Adenocarcinoma was the most common histopathologic subtype of NSCLC (n = 330, 95.7%), followed by squamous cell carcinoma (n = 15, 4.4%). Exon 19 deletions were detected in 142 (41.2%) patients, whereas 203 (58.8%) patients had exon 21 (L858 R) mutations. PPIs were used by 178 (51.6%) patients. Age was the only baseline characteristic that exhibited significant differences between the groups of patients (Table 1).
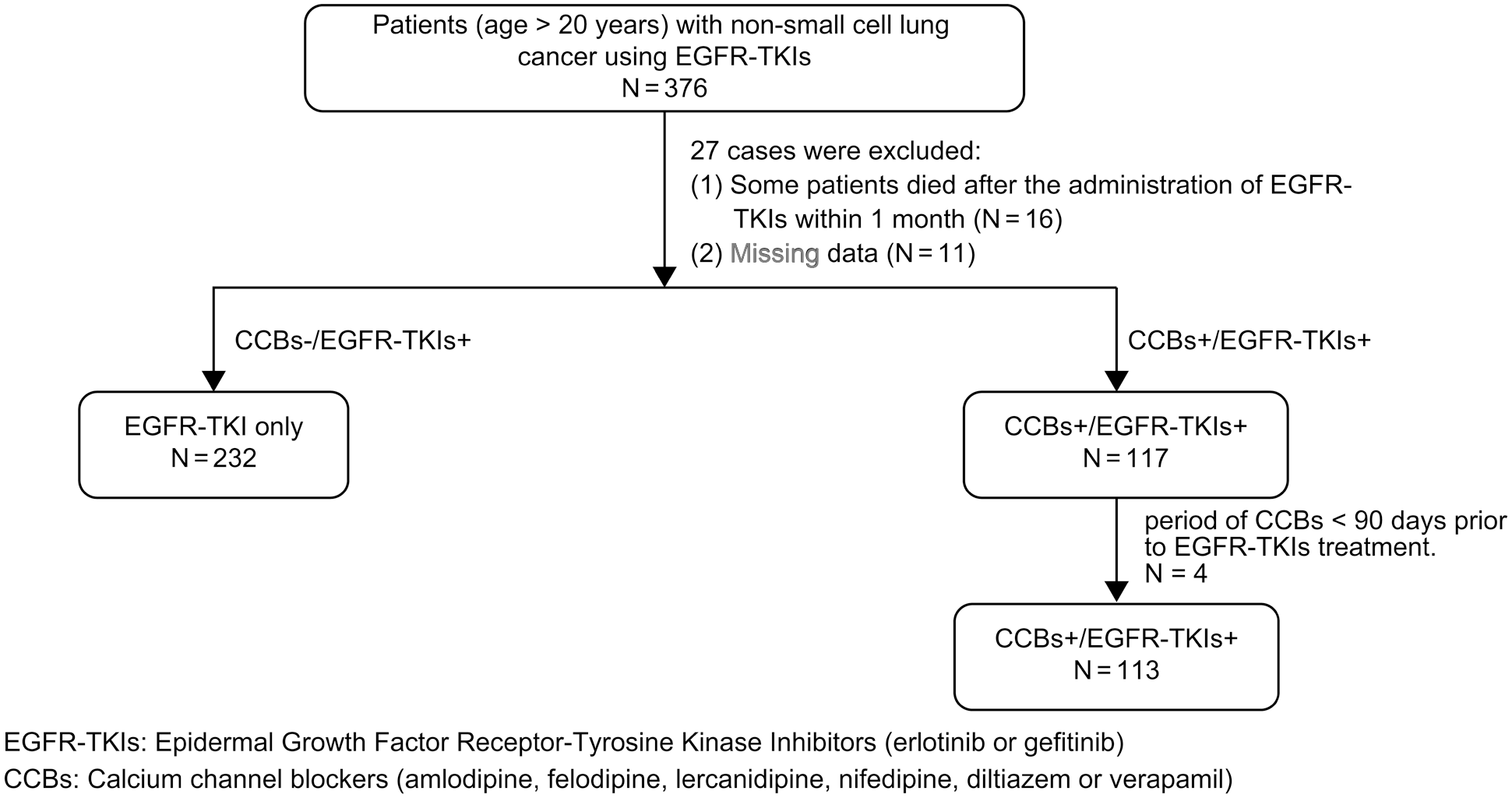
Flow diagram of the study.
Characteristics of the Study Population.

Abbreviations: SD, standard deviation; CCB, calcium channel blocker; PPI, proton pump inhibitor; ECOG, Eastern Cooperative Oncology Group; EGFR-TKI, epidermal growth factor receptor tyrosine kinase inhibitor.
P < .05.
Primary Endpoints
A total of 331 (95.9%) patients among the 345 patients exhibited disease progression or died at the end of the study. The estimated median PFS of the entire study cohort was 8.68 (95% CI: 7.77-9.56) months. Moreover, the estimated median PFS of patients in the CCBs-/EGFR-TKIs+ and CCBs+/EGFR-TKIs+ groups was 7.70 (95% CI: 6.56-8.84) and 10.43 (95% CI: 8.03-12.84) months, respectively. The Kaplan–Meier cumulative rate for PFS differed significantly (log-rank test: P = .011) between the groups (Figure 2). The multivariate analysis data were analyzed using Cox proportional-hazards models, which yielded the HR of PFS-related variables such as age, sex, CCB use (yes vs. no), smoking history, ECOG performance status score, histology findings, brain metastases, presence of EGFR gene, and PPI use. The univariate analysis results for the association of PFS with sex, CCB use, smoking, ECOG performance status score = 1, ECOG performance status score ≥2, brain metastases, and PPI use were 1.40 (95% CI: 1.12-1.75; P = .003), 0.74 (95% CI: 0.59-0.94; P = .012), 1.39 (95% CI: 1.09-1.76; P = .008), 1.86 (95% CI: 1.41-2.44; P < .001), 2.78 (95% CI: 2.00-3.87; P < .001), 1.98 (95% CI: 1.56-2.51; P < .001), and 1.49 (95% CI: 1.20-1.86; P < .001), respectively. After adjusting for sex, smoking, ECOG performance status score, brain metastases, and PPI use, CCB use was associated with improved PFS (adjusted HR 0.77, 95% CI: 0.61-0.98; P = .035) (Table 2). To elucidate the effect of the 2 drugs (CCBs and PPIs), we divided the patients into 4 subgroups based on their treatment regimen: CCBs-/PPIs-, CCBs+/PPIs-, CCBs-/PPIs+, and CCBs+/PPIs+. The estimated median PFS of the entire study cohort was 8.67 (95% CI: 7.77-9.56) months. Moreover, the estimated median PFS of patients in the CCBs-/PPIs-, CCBs+/PPIs-, CCBs-/PPIs+, and CCBs+/PPIs+ groups was 9.50 (95% CI: 7.76-11.24), 13.53 (95% CI: 9.42-17.64), 6.87 (95% CI: 5.75-7.99), and 8.47 (95% CI: 6.74-10.20) months, respectively. The Kaplan–Meier cumulative rate for PFS differed significantly (log-rank test: P < .001) among the 4 groups (Figure 3). Compared to the CCBs-/PPIs- group in the adjusted Cox model, only the CCBs-/PPIs+group demonstrated significance (HR: 1.37, 95% CI: 1.04-1.81; P = .028) (Table 3).
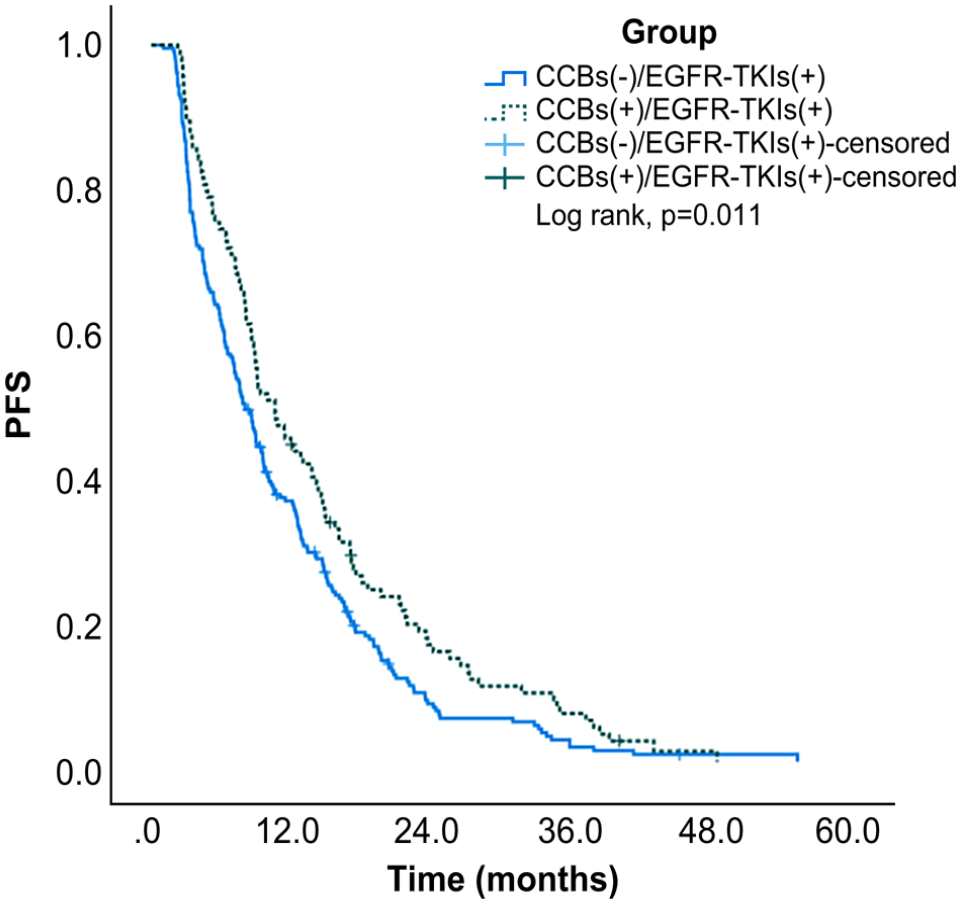
Kaplan–Meier curves of PFS for the CCBs-/EGFR-TKIs+ and CCBs+/EGFR-TKIs+ groups. CCB, calcium channel blocker; EGFR-TKI, epidermal growth factor receptor tyrosine kinase inhibitor; PFS, progression-free survival.
Cox Proportional Hazards Model for Evaluating the Effect of the Clinical Variables on Progression-Free Survival.

Abbreviations: CCB, calcium channel blocker; HR, hazard ratio; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; PPI, proton pump inhibitor.
P < .05.
Adjusted for sex, smoke, ECOG, brain metastasis, and PPI use.
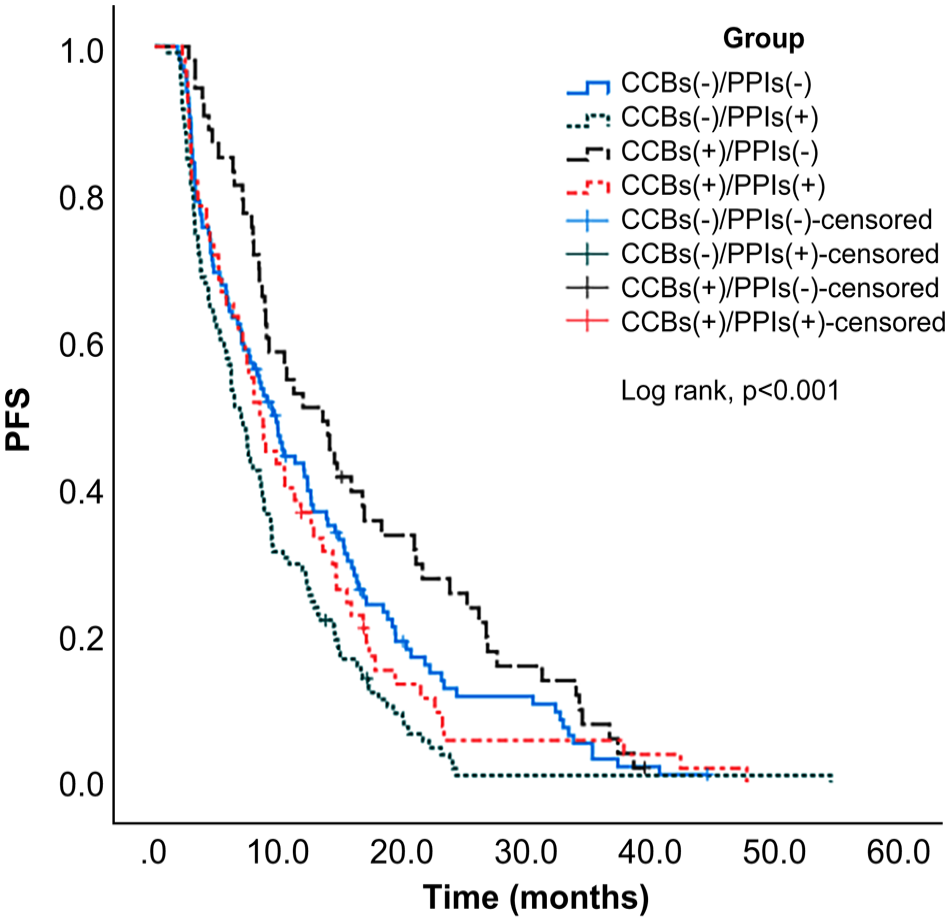
Kaplan–Meier curves of PFS according to CCB and/or PPI use. CCB, calcium channel blocker; PFS, progression-free survival; PPI, proton pump inhibitor.
Hazard Ratio and 95% Confidence Interval of Subgroups.

Abbreviations: CCB, calcium channel blocker; HR, hazard ratio; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; PPI, proton pump inhibitor.
P < .05.
Adjusted for age, sex, ECOG score, and brain metastasis.
Secondary Endpoints
Eventually, 322 (93.3%) of the 345 patients enrolled in this study died. The estimated median OS of the entire study cohort was 13.13 (95% CI: 11.11-15.16) months. Moreover, the estimated median OS of patients in the CCBs-/EGFR-TKIs+ and CCBs+/EGFR-TKIs+ groups was 12.17 (95% CI: 10.14-14.19) and 18.07 (95% CI: 12.37-23.77) months, respectively. The Kaplan–Meier cumulative rate for OS differed significantly (log-rank test: P = .005) between the groups (Figure 4). The univariate analysis showed a significant association of OS with age, sex, CCB use (yes vs. no), ECOG performance status score = 1, ECOG performance status score ≥2, brain metastases, and PPI use, with results of 1.01 (95% CI: 1.00-1.02; P = .028), 1.31 (95% CI: 1.04-1.64; P = .019), 0.71 (95% CI: 0.56-0.90; P = .005), 2.15 (95% CI: 1.62-2.85; P < .001), 3.87 (95% CI: 2.74-5.47; P < .001), 2.21 (95% CI: 1.73-2.83; P < .001), and 1.64 (95% CI: 1.32-2.05; P < .001), respectively. After adjusting for age, sex, ECOG performance status score, brain metastases, and PPI use, CCB use demonstrated an association with improved OS (adjusted HR 0.66, 95% CI: 0.51-0.84; P < .001) (Table 4). To understand the effect of the drugs (CCBs and PPIs), we divided the patients into 4 subgroups based on their treatment regimen: CCBs-/PPIs-, CCBs +/PPIs-, CCBs-/PPIs+, and CCBs+/PPIs+. The estimated median OS of the entire study cohort was 13.13 (95% CI: 11.11-15.16) months. Moreover, the estimated median OS of patients in the CCBs-/PPIs-, CCBs+/PPIs-, CCBs-/PPIs+, and CCBs+/PPIs+ groups was 14.33 (95% CI: 11.76-16.91), 25.60 (95% CI: 22.27-28.93), 9.27 (95% CI: 7.27-11.26), and 12.20 (95% CI: 8.72-15.68) months, respectively. The Kaplan–Meier cumulative rate for OS differed significantly (log-rank test: P < .001) among the 4 groups (Figure 5). A subgroup analysis with the adjusted Cox model demonstrated a lower HR in the CCBs+/PPIs- group (HR 0.66, 95% CI: 0.46-0.94; P = .019), and a higher HR in the CCBs-/PPIs+group (HR: 1.38, 95% CI: 1.04-1.83; P = .025) (Table 3).

Kaplan–Meier curves of OS for the CCBs-/EGFR-TKIs+ and CCBs+/EGFR-TKIs+ groups
Cox Proportional Hazards Model for Evaluating the Effect of Clinical Variables on Overall Survival.

Abbreviations: CCB, calcium channel blocker; HR, hazard ratio; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; PPI, proton pump inhibitor.
Adjusted for age, sex, ECOG, brain metastasis, and PPI use.
P < .05.

Kaplan–Meier curves of OS according to CCB and/or PPI use. CCB, calcium channel blocker; OS, overall survival; PPI, proton pump inhibitor.
Discussion
The findings of this retrospective study revealed an improvement in PFS and OS following concomitant CCB and EGFR-TKI therapy. Furthermore, PPI use was found to be associated with worsened PFS and OS.
Increasing evidence indicates the contribution of calcium channels to tumorigenesis and cancer progression. However, the precise mechanisms and pathways that are involved in signal transduction remain to be elucidated. Voltage-gated calcium channels (VGCCs) and non-VGCCs are the major types of calcium channels. Accumulating evidence highlights the crucial role of VGCC inhibitors in treating cancer. In clinical practice, CCBs such as dihydropyridines (nifedipine), phenylalkylamines (verapamil), and benzothiazepines (diltiazem and tetrandrine) are mainly used to block the effects of VGCCs. A VGCC known as CACNA1B (CaV2.2) facilitates the progression of NSCLC. CCBs exert an alternative anticancer action by possibly altering the tumor microenvironment. 17 An in vitro study published in 2020, which tested 4518 existing drugs against 578 cancer cell lines, showed unexpectedly high anticancer activity of non-oncology drugs, highlighting the antitumor effect of CCBs. 18
A retrospective cohort study published in 2021 demonstrated the additive anticancer effect of CCBs when combined with erlotinib. The 1-year OS rate for CCB users and nonusers was 78.3% and 39.7%, respectively, whereas the 2-year PFS rate was 44.4% and 8.3%, respectively. 19 Our study showed similar results, with the estimated median PFS and OS for the CCBs-/EGFR-TKIs+group being 7.70 and 12.17 months, respectively; the results were significantly different from those of the CCBs+/EGFR-TKIs+ group (10.43 and 18.07 months, respectively). Therefore, concomitant use of CCBs and EGFR-TKIs can improve PFS and OS.
PPIs have been reported to increase the gastric pH, which eventually decreases the solubility of EGFR-TKIs, leading to decreased absorption. In 2018, the effect of gastric acid inhibitors on the clinical outcomes of patients with NSCLC treated with EGFR-TKIs was retrospectively investigated. The findings revealed that concomitant use of gastric acid inhibitors and TKIs adversely affects PFS outcomes in patients with NSCLC irrespective of the type of gastric acid inhibitor used. 20 Our study showed that PPI use was associated with worse PFS (adjusted HR 1.30, 95% CI: 1.04-1.64; P = .024) and worse OS (adjusted HR 1.39, 95% CI: 1.10-1.75; P = .007). Therefore, the concomitant use of PPIs and EGFR-TKIs may adversely affect the PFS and OS of patients.
To the best of our knowledge, no study has analyzed and explored the effect of the concomitant use of CCBs and PPIs on the PFS or OS of patients receiving EGFR-TKI therapy. Our study revealed that the estimated median PFS and OS were 9.50 and 14.33 months for the CCB-/PPI- group, respectively, and 8.47 and 12.20 months for the CCB+/PPI+ group, respectively, with no significant differences between the groups. Concomitant use of EGFR-TKIs, CCBs, and PPIs decreases the PFS and OS compared to EGFR-TKI therapy alone, albeit insignificantly.
This retrospective study has several limitations. First, clinical data and medical records were obtained from hospital information systems, which may have resulted in incomplete records owing to physician-related personal factors. Second, the small study cohort, especially the small number of patients in the CCBs+/EGFR-TKIs+ group, did not allow for an accurate assessment of the treatment response. Further prospective studies with larger cohorts are necessary to validate our findings.
Conclusions
Calcium channels have been implicated in cancer pathogenesis. In our study, we found an additive anticancer effect of CCBs when concomitantly used with EGFR-TKIs. Conversely, the concomitant use of EGFR-TKIs and PPIs decreased the PFS and OS. However, the effect of the combined use of EGFR-TKIs, CCBs, and PPIs on the PFS and OS was similar to that of EGFR-TKI therapy alone. Therefore, future prospective studies with larger cohorts are warranted to elucidate the efficacy of the combination of EGFR-TKIs and CCBs in NSCLC treatment.
Footnotes
Acknowledgements
Author Contributions
Conceptualization: H-H.H, C-H.C., M-J.H.; Data curation: H-H.H, Y-H.K.; Formal analysis: Y-H.K.; Investigation: M-J.H.; Methodology: H-H.H, M-J.H.; Project administration: M-J.H.; Software: Y-H.K.; Supervision: T-Y.W., M-J.H.; Validation: H-H.H; Visualization: Y-H.K.; Writing – original draft: H-H.H, M-J.H.; Writing – review & editing: T-Y.W., C-H.C., M-J.H.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article. Data sharing is not publicly available due to the institutional restriction but request with permission of the Taichung Tzu Chi Hospital.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Consent to Participate
The consent to participate was waived due to the retrospective nature of the study.
Consent for Publication
Not applicable.
Ethics Approval
The study has been performed in accordance with the Declaration of Helsinki and approved by the Research Ethics Committee of Taichung Tzu Chi Hospital (REC111-59).
