Abstract
Objective
We examined the factors influencing hospitalization and prognosis among patients with non-small cell lung cancer receiving epidermal growth factor receptor tyrosine kinase inhibitor (EGFR-TKI) targeted therapy during the first wave of the coronavirus disease 2019 (COVID-19) pandemic.
Methods
In total, 267 patients diagnosed with NSCLC who were receiving treatment with third-generation EGFR-TKIs were included in our retrospective study. Data on patients’ demographics, clinical characteristics, and survival were collected and analyzed.
Results
Over a mean follow-up of 18 months, 80.5% (215/267) of the patients contracted COVID-19, and 12.6% (27/215) of these patients were hospitalized for COVID-19 treatment. Vaccinated patients, those with body mass index (BMI) ≥22.3 kg/m2, and those with no comorbidities had lower rates of infection and hospitalization than unvaccinated patients, those with BMI <22.3 kg/m2, and those with comorbidities, respectively. Continued NSCLC treatment in patients with COVID-19 was identified as a risk factor for patient survival.
Conclusions
NSCLC treatment can be continued for patients who received COVID-19 vaccines, those with higher BMI, and those without comorbidities during the COVID-19 epidemic, but treatment interruption might be required for patients during the active phase of infection.
Keywords
Introduction
The severe global pandemic triggered by coronavirus disease 2019 (COVID-19), which is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has spread globally. China has been in a post-epidemic era since the discontinuation of its “zero COVID” policy and the release of a new strategy for the prevention and control of COVID-19 outbreaks on 7 December 2022. Despite the decline in acute cases, individuals continue to experience recurrent infections and long-term effects on organ systems, 1 and the potential impact on patients with malignancy is of great concern. A large number of patients diagnosed with cancer are immunosuppressed by their underlying disease, malnutrition, and treatment-related side effects, and therefore, they are highly vulnerable during the COVID-19 pandemic. These patients are at increased risks of opportunistic infections, serious complications (including death), and intensive care unit admission. 2 Mounting evidence indicates that patients with COVID-19 and cancer have a worse prognosis than those with COVID-19 alone.3–8
Antitumor therapy, especially chemotherapy, increases the risk of severe disease and death among patients with coincident cancer and COVID-19. Surgery increases the risk of death, and targeted therapy increases the incidence of severe COVID-19. 9 Several studies investigated antitumor treatment as an additional risk factor for adverse outcomes in patients with severe and specific pneumonia and whether it is necessary to change treatment paradigms to reduce this risk.10–12 Previous research suggested that stable patients with lung cancer undergoing oral targeted therapy should maintain their original drug therapy regimen during the pandemic. 13 A study by Deben et al. identified a potential interaction between SARS-CoV-2 infection and outcomes in patients who received tyrosine kinase inhibitors (TKIs) for epidermal growth factor receptor (EGFR)-mutant NSCLC. 14 However, the risks of continued therapy in patients with NSCLC who are undergoing targeted therapy during a surge in COVID-19 cases and the prognosis for continuing or discontinuing therapy in patients in the infectious phase of the disease have not rigorously investigated. Therefore, a systematic analysis of the clinical characteristics of patients with COVID-19 receiving targeted therapy is urgently required, especially knowledge of the risk factors that affect the severity of COVID-19 and patient prognosis, to ensure patients’ access to effective treatment during and after the pandemic.
To address this uncertainty, we collected data and conducted retrospective analyses of patients who received targeted therapy with EGFR-TKIs at our hospital of the onset of China’s COVID-19 pandemic and underwent follow-up for 18 months. We aimed to describe the clinical characteristics of these patients after the onset of the COVID-19 pandemic, determine the COVID-19 rates, and identify factors influencing the hospitalization and prognosis of patients who were receiving targeted therapy following a diagnosis of COVID-19s.
Materials and methods
Study design and participants
This retrospective cohort study was conducted at the Second Affiliated Hospital of Nanchang University from 7 December 2022 to 5 January 2023. We enrolled patients with NSCLC who were receiving targeted therapy with third-generation EGFR-TKIs regardless of the date of diagnosis. The inclusion criteria included a diagnosis indicating pathology, such as malignancy, and laboratory confirmation of SARS-CoV-2 infection. Patients treated for less than 1 month and those with treatment interruption during the follow-up period were excluded from the analysis because we chose to focus on patients with NSCLC who received continuous EGFR-TKI targeted therapy. SARS-CoV-2 infection was confirmed by reverse transcription polymerase chain reaction using nasopharyngeal swab specimens. All patient details have been de-identified. The study’s endpoint was 1 June 2024.
The enrolled patients were grouped according to the results of a clinical assessment. Age, sex, body mass index (BMI), smoking status, alcohol use, education, vaccination, comorbidities, gene mutation, type of targeted therapy, type of EGFR-TKIs, and duration of treatment were used as matching factors in this study. The clinical outcomes of interest included the rates of hospitalization and SARS-CoV-2 infection. The clinical data collected included patients’ demographics, smoking and alcohol history, vaccination status, baseline clinical comorbidities, gene mutation type, drug type, types of targeted treatment, and the duration of treatment. Demographic data included age, BMI, sex, and education. The reporting of this study conformed to the STROBE guidelines. 15
We conducted our study in accordance with the Declaration of Helsinki of 1975 as revised in 2013. 16 The requirement for ethics approval was waived by the Ethics Committee of the Second Affiliated Hospital of Nanchang University at the beginning of the study. Informed consent was obtained from all subjects.
Data collection
We obtained data on the enrolled patients’ demographics, clinical presentation, cancer history, smoking history, alcohol history, treatment(s), and outcomes from electronic medical records and by patient interviews. Missing and uncertain data were collated and clarified through communication with relevant healthcare professionals or patients and their families. Detailed information on all patients’ demographics, comorbidities, symptoms, and disease severity were recorded on visit records or diagnosed for most outpatient visits and a few inpatient visits, and details of their clinical treatment were also included.
Statistical analysis
Patients with NSCLC undergoing EGFR-TKI therapy exhibited disparate prognoses and severities of SARS-CoV-2 infections during the subsequent follow-up visit, which we hypothesized was related to their demographic characteristics, clinical features, therapeutic regimens, cessation of medical intervention, and vaccination status. The primary outcomes of interest were survival and COVID-19 risk factors.
We used IBM SPSS Statistics 27.0 software (IBM Corp., Armonk, NY, USA) for all statistical analyses. Survival data were analyzed using the Kaplan–Meier method (log-rank test) and the Cox proportional hazards model. Hazard ratios (HRs) and 95% confidence intervals (CIs) were estimated using the Cox model and log-rank test. All p-values were based on two-tailed tests with statistically significant differences indicated by P < 0.05. Variables were selected according to their levels of significance in univariate logistic regression analyses (P < 0.05), and the correlations among the indicators and baseline clinical characteristics were examined.
Results
Patients with NSCLC who were receiving EGFR-TKI targeted therapy (N = 267) from 7 December 2022 to 10 January 2023 were recruited from the Second Affiliated Hospital of Nanchang University. In total, 80.5% (215/267) of the patients were infected with COVID-19 during the study period. Among the infected patients, 12.6% (27/215) were hospitalized to receive treatment for COVID-19, and 1.4% (3/215) died from the infection. As of 5 January 2023, the mean follow-up time was 30 days. None of the 267 patients was lost to follow-up.
Patients’ characteristics are presented in Table 1. The mean age of the sample was 62.6 years, 64.8% of the cohort was female, and the mean BMI was 22.6 kg/m2. In addition, 52.4% had a lower education level, 13.1% smoked cigarettes, and 10.1% consumed alcohol. Approximately 40.0% of the patients had comorbidities other than cancer, including hypertension (21.3%), diabetes mellitus (9.0%), chronic obstructive pulmonary disease (6.0%), and cardiovascular disease (3.7%). More than half of the patients (51.7%) had been vaccinated for COVID-19, with 3.0%, 27.3%, and 34.5% of patients receiving one, two, and three vaccine doses, respectively. All patients received the SARS-CoV-2 inactivated vaccine (Sinovac Biotech, Beijing, China). Conversely, 35.2% of the patients had never been vaccinated.. The rationale for treatment with EGFR-TKI targeted therapy was mainly palliative in 82% of the patients, with a lower proportion (18%) of patients receiving postoperative adjuvant therapy. The most prevalent gene mutation found in this cohort was EGFR 19Del (57.7%), followed by EGFR L858R (36.0%). The EGFR T790M mutation was detected in 6.3% of the patients. No significant differences were found in the COVID-19 infection rate or clinical outcomes of COVID-19 among patients receiving different therapeutic agents, and no correlations of infection rate or clinical course with age, sex, education, smoking status, alcohol use, gene mutation, type of targeted therapy, or duration of treatment were identified. Cox regression analysis of the aforementioned factors revealed that patients who were vaccinated, those with BMI ≥ 22.3 kg/m2, and those without comorbidities had higher rates of infection than those who were not vaccinated against COVID-19 (HR = 2.072, P = 0.059), those with BMI < 22.3 kg/m2 (HR = 2.287, P = 0.036), and those with comorbidities (HR = 3.439, P = 0.004), respectively (Figure 1). Nevertheless, targeted therapy during hospitalization (P = 0.139) was not significantly associated with COVID-19 infection in patients irrespective of the vaccination status, BMI, and comorbidities. Among the 215 patients infected with COVID-19, no significant association was found between the receipt of targeted therapy and COVID-19 infection in regression analysis (P = 0.637).
Demographics and characteristics of patients treated with third-generation EGFR-TKIs.

EGFR, epidermal growth factor receptor; TKI, tyrosine kinase inhibitor; BMI, body mass index; COVID-19, coronavirus disease 2019.

Kaplan–Meier curves of hospitalization-free survival and infection-free survival rates among patients categorized by vaccination status (vaccinated vs. unvaccinated), BMI (BMI < 22.3 kg/m2 vs. BMI ≥ 22.3 kg/m2), and the presence of comorbidities (comorbidities vs. no comorbidities). All patients in the analysis received targeted therapy. BMI, body mass index; HR, hazard ratio; CI, confidence interval.
Table 2 presents the results of our comparative analysis of patients’ clinical characteristics, educational level, vaccination status, comorbidities, type of mutation, type of treatment, infection rate, and clinical outcomes between those who recovered spontaneously after a diagnosis of COVID-19 and those who had severe disease or died. Among these patients, 87.4% recovered spontaneously without having to discontinue targeted therapy or receive any antiviral treatment, and 12.6% experienced progression to severe disease or died. Patients with BMI < 22.3 kg/m2 (70.4%) were more likely to have severe or fatal outcomes, whereas vaccinated patients (67.0%) and those without comorbidities (72.9%) were more likely to experience spontaneous recovery. The results of the analysis indicated that the outcomes of patients admitted to the study were associated with BMI (P = 0.020), the vaccination status (P = 0.021) and comorbidities (P = 0.010), whereas no significant association was found with age, sex, smoking, alcohol consumption, education, the type of mutation, the type of treatment, the treatment period, and type of comorbidity.
Relationships of patient characteristics with COVID-19 outcomes.
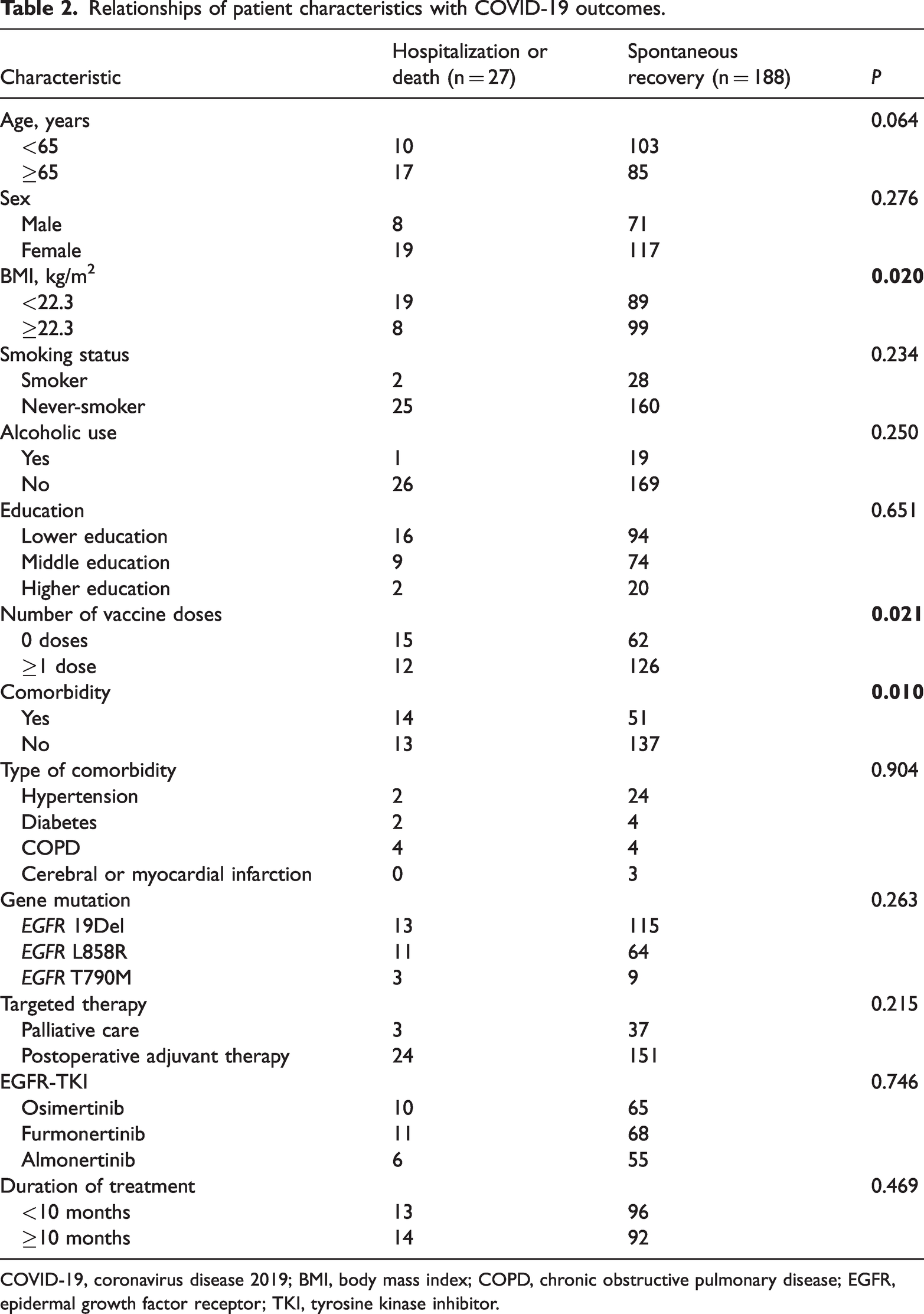
COVID-19, coronavirus disease 2019; BMI, body mass index; COPD, chronic obstructive pulmonary disease; EGFR, epidermal growth factor receptor; TKI, tyrosine kinase inhibitor.
The patient cohort underwent follow-up for up to 18 months. As presented in Table 3, SARS-CoV-2 infection was identified as an independent risk factor for prognosis in patients undergoing third-generation EGFR-TKI therapy (HR = 0.21, 95% CI = 0.09–0.49, P < 0.01). The cessation of targeted therapy during the course of infection was associated with a more favorable prognosis in this subset of patients (HR = 4.57, 95% CI = 1.41–14.87, P = 0.011). Age, sex, BMI, and comorbidities were not significantly correlated with survival.
Risk factors for survival among patients with non-small cell lung cancer receiving third-generation EGFR-TKIs during the first wave of COVID-19 in China.

EGFR, epidermal growth factor receptor; TKI, tyrosine kinase inhibitor; COVID-19, coronavirus disease 2019; BMI, body mass index; HR, hazard ratio; CI, confidence interval.
Discussion
The shift in China’s strategy for controlling the spread of COVID-19 and the dramatic increase in the number of patients with COVID-19 indicate the urgent need for research to guide treatment decisions for patients receiving targeted therapies. To the best of our knowledge, this is the first retrospective cohort study describing the clinical characteristics, outcomes, and risk factors for severe disease and survival among patients with NSCLC who were receiving targeted therapy during and after the first wave of the COVID-19 pandemic in China. We reviewed the clinical characteristics of patients with NSCLC who received EGFR-TKI targeted therapy after the liberalization of China’s policy for the prevention and control of COVID-19 and examined the associations of various risk factors with hospitalization and infection rates. Compared with their counterparts, unvaccinated patients, those with BMI < 22.3 kg/m2, and those with comorbidities had higher rates of hospitalization and increased risks of severe disease. Conversely, vaccinated patients, those with BMI ≥ 22.3 kg/m2, and those with no comorbidities had higher rates of spontaneous recovery and lower risks of severe disease without any antiviral intervention. It is noteworthy that SARS-CoV-2 infection represents a prognostic risk factor for patients with NSCLC. Conversely, the discontinuation of treatment during infection is associated with a more favorable prognosis.
The uncontrolled inflammatory response caused by COVID-19 can lead to severe organ damage, resulting in pulmonary and cardiac dysfunction, and therefore, the risk of severe disease is closely related to immune function. 17 Patients with cancer have a greater risk of severe COVID-19 outcomes and mortality. The risk factors identified in this study were previously highlighted in prior studies as significant risk factors for severe disease or mortality 30 days after a diagnosis of COVID-19 among patients with cancer (age ≥ 65 years) and comorbid underlying diseases.4,6,18–24 The negative effects of cancer treatments on the immune system are well known. Studies reported that patients who recently received cancer treatment had worse outcomes during the COVID pandemic.21,25,26 Another study has indicated that patients with lung cancer who are undergoing immunotherapy and experiencing immune-related adverse events might be at an elevated risk of developing COVID-19 pneumonia. 27 In our study, no increased risk of severe disease was observed in patients with NSCLC who were receiving targeted therapy, in line with the findings of Yekedüz et al. 28 The reasons for these findings could be related to the lower risk of immunosuppression (e.g., neutropenia or leukopenia) with targeted therapy compared with other antitumor treatments. 29 Patients with malignancies do not have similarly high risks of serious complications from COVID-19, as these risks commonly arise from uncontrolled viral proliferation or a subsequent viral-driven hyperinflammatory response.30–32 Targeted therapies did not pose safety concerns in the treatment of patients with cancer during the pandemic. Moreover, the European Society for Medical Oncology does not recommend discontinuation of targeted therapy. 33 Therefore, the potential for favorable or unfavorable effects of various conventional and novel therapies (generally used in cancer treatment) on the outcomes of SARS-CoV-2 infection warrant further exploration. The interaction between several targeted drugs and antiviral therapies should be considered when treating patients with cancer and COVID-19. 34 However, the sample size of our study was insufficient to perform such an analysis.
Cox regression analysis was used in our study to explore the factors associated with the severity of COVID-19 and survival. Previously reported risk factors among patients without cancer, such as older age and comorbidities, have been validated in patients with NSCLC. 35 Age is a significant independent predictor of mortality in persons with COVID-19. Advanced age was identified as a risk factor for serious adverse events in previous studies,36–38 but this was not observed in our study. This difference might be related to our inclusion of a cohort of older adults with underlying diseases (mean age = 62.6 years). Gao et al. reported a linear increase in the risk of severe COVID-19 when BMI exceeded 23 kg/m2; 39 however, relevant evidence has not been validated in Asian populations. The findings of our study complement the correlation between BMI and severe COVID-19 among patients of Asian ethnicity with NSCLC who were receiving targeted therapy. Previous studies reported an increased likelihood of progression to severe COVID-19 among underwent (BMI < 18.5 kg/m2) and obese patients (BMI ≥ 28 kg/m2).39,40 Our results validate the prediction of a higher risk of severe disease among patients with cancer and lower BMI; however, because of the small number of overweight patients in our study, we cannot conclude that overweight is a risk factor for severe disease among patients receiving targeted therapy. Common chronic non-communicable diseases, including cardiovascular disease, chronic respiratory diseases, metabolic diseases, and various types of cancer, are consistently associated with an increased risk of adverse clinical outcomes among persons infected with SARS-CoV-2.41–43 The consistency of these results supports our findings; therefore, the benefits and drawbacks of therapy should be weighed, and individualized treatment plans should be developed for patients with NSCLC and multiple comorbidities.
Vaccination is undoubtedly one of the most important and critical elements of a pandemic response. Recent studies comparing vaccinated and unvaccinated patients with cancer found that despite the protective effects of vaccination, vulnerable patients with cancer (i.e., those with risk factors such as lymphopenia, active and progressive cancer, and advanced age) can develop serious and fatal breakthrough infections.6,44 The number of affected patients in this study was too small to draw a definitive conclusion regarding the possible associations between targeted therapies and breakthrough infections. Despite the time-dependent improvements in outcomes reported during the Omicron (B.1.1.529) phase compared with that in the early phase of the pandemic, patients with cancer are highly susceptible to SARS-CoV-2 infection if they are not vaccinated. 45 Although a subset of patients with NSCLC responded poorly to a SARS-CoV-2 mRNA vaccine and they had low neutralizing antibodies in response to the B.1.1.529 Omicron variant, 46 our findings support universal vaccination among patients with cancer as a protective measure against COVID-19 morbidity and mortality. Among the individuals with breakthrough SARS-CoV-2 infections, partially vaccinated patients who were infected had a poor prognosis, whereas vaccination was associated with a lower risk of severe outcomes. Among patients with breakthrough infection, the presence of comorbidities was not associated with severe illness or death. 47 Our results confirm the clinical relevance of observations from previous studies.
COVID-19 is an endothelial disease, as the virus binds to angiotensin-converting enzyme 2 (ACE2) on vascular endothelial cells, after which it enters these cells to replicate. 48 The acute symptoms and long-term effects of infection with the virus in various organs could be related to capillary damage caused by the virus and to the widespread protein expression of ACE2 proteins on the endothelial and epithelial cells of capillaries.49,50 A study by Deben and colleagues identified a role for EGFR pathway activation in membrane-bound ACE2 expression in NSCLC cells, and EGFR pathway activation was associated with increased levels of soluble ACE2 (sACE2) in patients. Significant differences in sACE2 levels were observed in patients receiving targeted therapies, and the authors proposed a possible interaction between SARS-CoV-2 infection and TKI therapy in patients with EGFR-mutant NSCLC. 14 A study by Stewart and colleagues demonstrated that elevated ACE2 expression in SARS-CoV-2–infected cells was linked to augmented cellular sensitivity to EGFR-TKIs. Additionally, ACE2 expression was reduced in cells carrying the EGFR T790M mutation, indicating a potential correlation between SARS-CoV-2 infection and EGFR-TKI resistance. 51 The present study corroborates the correlation between SARS-CoV-2 infection and the prognosis of EGFR-TKI targeted therapy in patients with NSCLC, as evidenced by clinical data from long-term follow-up. It was previously proposed that patients with NSCLC who have achieved a stable state on oral targeted therapy with EGFR-TKIs should maintain their original drug regimen throughout a pandemic. 13 Our study further suggests that the decision to withhold therapy during the onset of COVID-19 could prove prognostically beneficial for patients. Our study further suggests that the suspension of therapy during the course of SARS-CoV-2 infection is favorable for patients.
Our study had several limitations. First, our study lacked dynamic clinical data because of the retrospective design, and it was susceptible to recall bias and missing data. The lack of an adequate case–control design (i.e., comparisons of patients without cancer or patients receiving other antitumor treatments) imposed limitations on our conclusions. Second, the data from our study were mostly from oncology inpatients in poor general health, which might have skewed the data toward higher hospitalization rates and a greater risk for severe disease in patients with COVID-19, thereby increasing the heterogeneity of the data. Third, univariate analysis might not reveal the true effect of targeted therapy on the severity of COVID-19 because of the presence of confounding factors, such as age and sex, which are critical to the prognosis of COVID-19. Most patients in our study who received targeted therapy had advanced lung cancer and they were receiving palliative care. No additional information on patients’ primary tumor progression, such as tumor stage or information about their treatment-related complications or deaths caused by tumor progression, was included in the study. The proportion of patients with advanced cancer might have influenced the severity and mortality of COVID-19. The current proportion of vaccinated patients with cancer who become severely ill or die from COVID-19 is unknown, as vaccination has been widely implemented and it is likely to protect most people from severe disease. Finally, this retrospective study was conducted in a single hospital. A more expansive investigation across a multitude of regions would reinforce the reliability of our findings and substantiate the generalizability of our conclusions.
The present study described the clinical characteristics and major risk factors for COVID-19 outcomes among patients with NSCLC who were receiving targeted therapy during the pandemic. The results might guide clinicians toward the early identification of patients at high risk for serious clinical outcomes. Confidence in the safety of targeted therapy can be assured using the risk factors of low BMI, a lack of vaccination, and severe comorbidities. For clinicians, the early assessment and identification of risks related to antineoplastic therapy and individualized management can help circumvent delays in treatment and reduce the risk of tumor progression and COVID-19 in the post-pandemic era.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241281907 - Supplemental material for Risk of COVID-19 infection in patients with NSCLC receiving EGFR-TKI targeted therapy during the first wave in China
Supplemental material, sj-pdf-1-imr-10.1177_03000605241281907 for Risk of COVID-19 infection in patients with NSCLC receiving EGFR-TKI targeted therapy during the first wave in China by Peng Huang, Ling-Ming Liao, Jia-li Zhao, Chen Luo, Yan-Ling Yi, Yu Chen and Long Huang: on behalf of the WEAR-Stroke Study Group in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605241281907 - Supplemental material for Risk of COVID-19 infection in patients with NSCLC receiving EGFR-TKI targeted therapy during the first wave in China
Supplemental material, sj-pdf-2-imr-10.1177_03000605241281907 for Risk of COVID-19 infection in patients with NSCLC receiving EGFR-TKI targeted therapy during the first wave in China by Peng Huang, Ling-Ming Liao, Jia-li Zhao, Chen Luo, Yan-Ling Yi, Yu Chen and Long Huang: on behalf of the WEAR-Stroke Study Group in Journal of International Medical Research
Footnotes
Acknowledgements
We thank Qi Zhang and Hong-Yan Xu for their help in data collection collect data and follow-up.
Author contributions
PH and LM: Data collection, statistical analysis, and manuscript writing and revision. JLZ and CL: Data collection, data summary, and statistical analysis. YLY and YC: Literature review and manuscript writing and revision. LH: Manuscript review and revision and writing guidance. All authors contributed to the article and approved the submitted version.
Data availability
The original data presented in the study are included in the Supplementary Material of this article, and further inquiries can be directed to the corresponding author.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work was supported by Jiangxi Provincial Natural Science Foundation (grant numbers: 20242BAB25550 and 20242BAB25518), Science and Technology Plan Project of Jiangxi Provincial Health Commission (grant number: SKJP_1220240037).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
