Abstract
Background:
According to recent research, breast cancer has become the most commonly diagnosed non-skin cancer type among the entire population. Meanwhile, Chinese herbal medicine (CHM) has become an important therapy to improve the survival rate and quality of life in metastatic cancer patients, as part of a wider trend toward more personalized medicine. However, there is a lack of research in terms of the association between stage IV breast cancer and CHM. Therefore, this study aimed to investigate the association between CHM and the survival rate in breast cancer patients at different cancer stages, with a particular focus on patients diagnosed with stage IV.
Methods:
Patients with an initial diagnosis of breast cancer identified from the Taiwan Cancer Registry Database (TCRD) and China Medical University Hospital (CMUH) database were included in this study. Demographic characteristics including gender, age, and comorbidities were evaluated. Between-group differences for continuous and categorical variables were evaluated by the Student’s t-test and Chi-square test, respectively. Patients diagnosed with breast cancer were recruited and divided into cohorts of CHM users and non-users with a 1:1 propensity score matching. The survival of breast cancer patients was assessed by the Cox proportional hazard model. The cumulative incidence of survival was assessed by Kaplan–Meier analysis.
Results:
The adjuvant CHM treatment was associated with a higher survival rate in stage IV breast cancer patients (HR: 0.45, 95% CI: 0.2853-0.7044]. Additionally, the use of CHM had a positive impact on survival in stage IV breast cancer patients who had received surgery (P: .0273, HR: 0.3406, 95% CI: 0.1309-0.8865), chemotherapy (P: .0004, HR: 0.3893, 95% CI: 0.231-0.656), and hormone therapy (P: .0013, HR: 0.3491, 95% CI: 0.1836-0.6636). In terms of the specific CHM associated with survival, Zhi-Gan-Cao-Tang (ZGCT), Phellodendron chinense Schneid. (huang-bai), and Paeonia lactiflora Pall. (chi-shao) were the 3 most commonly prescribed herbal medicines correlated with a higher survival rate among stage IV breast cancer patients.
Conclusions:
CHM associated with conventional management achieved significant survival benefits in patients with stage IV breast cancer. Additional randomized controlled trials for prospective study are recommended for further validation.
Keywords
Introduction
According to recent research, breast cancer has become the most commonly diagnosed non-skin cancer type among the entire population. 1 According to the latest Taiwan Cancer Registry annual report, breast cancer is the most diagnosed cancer site and the second leading cause of cancer deaths among females in Taiwan. As breast cancer is a heterogeneous disease, patients may undergo various clinical management modalities according to the cancer stage and molecular phenotype, including surgery, radiotherapy, chemotherapy, hormone therapy, and target therapy.
Despite the therapeutic effects of conventional approaches, the cancer-related symptoms and treatment-induced side effects often negatively impact patients’ quality of life, which may lead to the discontinuation of cancer therapies. In Taiwan, many patients seek Chinese herbal medicine (CHM) as an adjuvant therapy to relieve symptoms and therapy-related discomforts, such as nausea and vomiting, diarrhea, paresthesia, fatigue, and chronic pain. 2
As previously reported, CHM may be considered as an adjuvant therapy throughout the entire course of cancer treatment, with effects including the suppression of cancer proliferation and migration, relief of surgical complications, increased sensitivity to chemotherapy, and improved immune function noted among breast cancer patients.3,4 CHM has been reported to decrease the incidence of congestive heart failure in breast cancer patients who received conventional chemotherapy. 5 In addition, Wang et al reported improved short-term treatment efficacy and an increased 5-year survival rate in breast cancer patients after mastectomy. 6 Moreover, CHM has been shown to alleviate aromatase inhibitor-related musculoskeletal symptoms in breast cancer patients and improve quality of life. 7
In previous studies, however, the absence of details regarding pathological staging and concurrent and previous treatments associated with CHM use, indeed limited the interpretation of the results. Meanwhile, accurate evaluations of the efficacy of CHM treatment have been further restricted by the relatively small study cohorts. Thus, the aim of the current study was to evaluate the effects of adjuvant CHM treatment on the survival rate of breast cancer patients at different stages of the disease, particularly those patients diagnosed with stage IV, by mining the integrated data of the Taiwan Cancer Registry Database (TCRD) and China Medical University Hospital (CMUH) database.
Materials and Methods
Study Design and Data Source
The data in the present study was retrieved from China Medical University Hospital, the largest Cancer Center, and Chinese Medicine Department in central Taiwan. All diagnoses in the TCRD are coded according to the International Classification of Disease, Ninth Revision, Clinical Modification (ICD-9-CM). Patients’ privacy is protected via the encryption of identification numbers prior to the release of the database. The consistency of the encryption procedure ensures the linkage of claims belonging to the same patient between the hospital database and the TCRD. Based on the anonymous nature of all personal information in the database, patients’ informed consents were waived. The Research Ethics Committee of CMUH in Taichung, Taiwan approved this study (CMUH-107-REC2-023).
Subjects
Patients with an initial diagnosis of breast cancer (ICD-9-CM code 174) between January 1, 2006 and December 31, 2017 at CMUH were included in this study. The index date was set as the first diagnosis date of each breast cancer patient in the registration data. The follow-up period was defined as the time from the index date to the date when patients withdrew from the TCRD (the last contact date for the information was September 10, 2019). Patients having received CHM treatment, other than acupuncture or traumatology therapies, after the index date and having taken CHM continuously for more than 14 days were defined as CHM users, while those without any records of CHM use were defined as non-CHM users. Figure 1 demonstrates the flow diagram of the enrollment process for this study.

Flow chart of the study enrollment process.
Covariates
Demographic characteristics evaluated in this study included gender, age, and comorbidities. The Charlson Comorbidity Index (CCI) for the assessment of comorbidities among breast cancer patients was applied. The comorbidities which appeared in medical records at least twice and within 1 year prior to the diagnosis of breast cancer were included. The higher the CCI score, the greater the risk of death caused by the disease, further indicating that the case may have multiple comorbidities, thereby negatively influencing the prognosis of breast cancer and related diseases.
Matching
We applied a 1:1 propensity-score matching to reduce selection bias between the 2 groups. The propensity score was calculated using logistic regression analysis including the variables of gender, age, comorbidities, and clinical cancer stage. The patients without a complete record of the cancer stage were defined as the “other clinical stage” group.
Statistical Analyses
Between-group differences for continuous and categorical variables were evaluated by the Student’s t-test and Chi-square test, respectively. A Kaplan-Meier analysis plotted the cumulative survival rates between the matched study groups.
The mortality risks in the 2 cohorts were identified using hazard ratios (HRs) and 95% confidence intervals (CIs). We applied multivariable Cox proportional hazard models and adjusted for covariates such as diagnostic age, molecular phenotype, and CHM use to evaluate the survival of breast cancer patients. For the herbal prescription analysis, we divided all CHM into 2 subgroups: single herb or herbal formula. The Chi-square test was used to evaluate the association between these subgroups and the survival rate among breast cancer patients. All statistical analyses were performed using the SAS System for Windows statistical software, version 9.4 (SAS Institute Inc., Cary, NC, USA) and R software (version 3.6.1). The significant criterion was set at less than 0.05 for 2-sided testing of a P-value.
Results
A total of 2860 patients with breast cancer were analyzed in this study, consisting of matched 1430 CHM users and 1430 non-CHM users. There were no significant differences in gender, diagnostic age, comorbidities, or cancer stage between the 2 groups (Table 1).
Characteristics of Breast Cancer Patients.
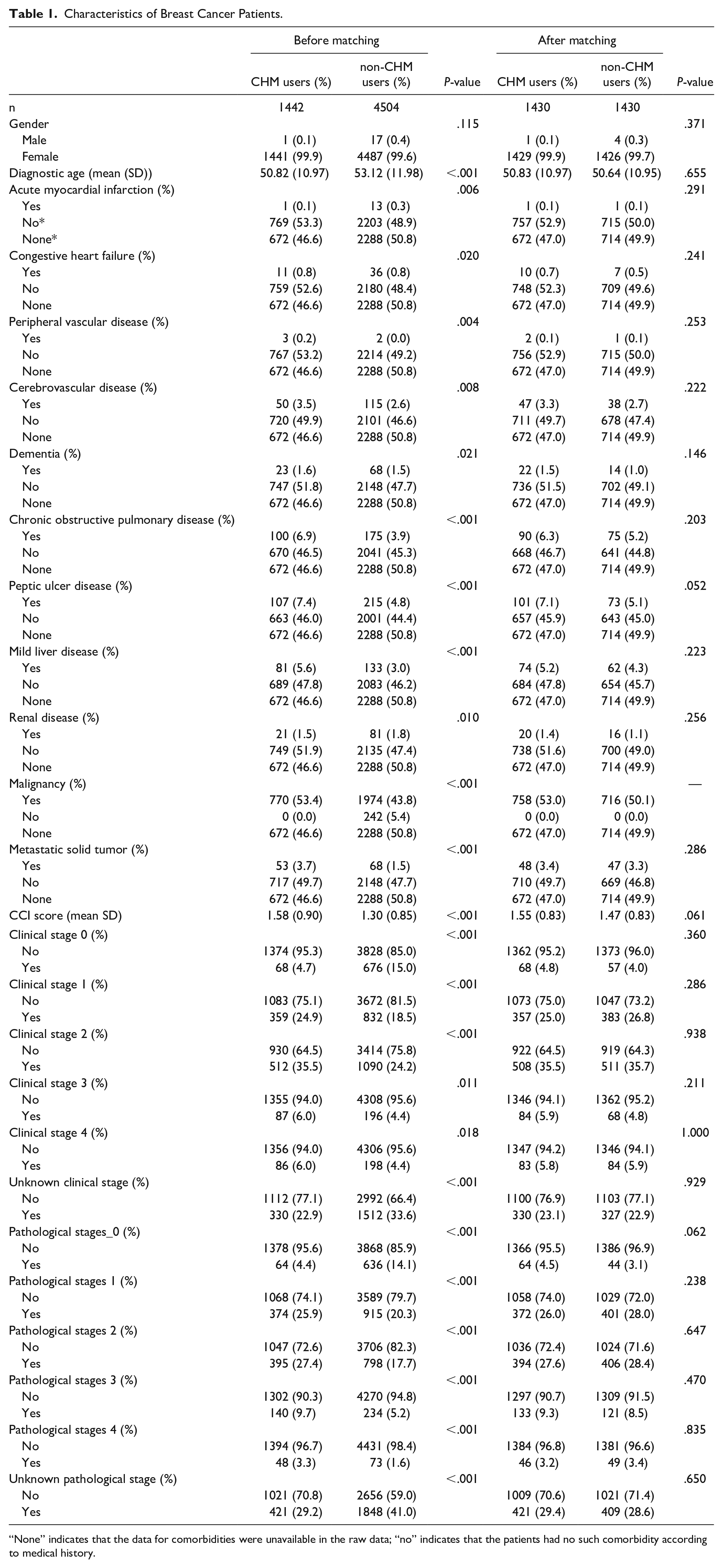
“None” indicates that the data for comorbidities were unavailable in the raw data; “no” indicates that the patients had no such comorbidity according to medical history.
In terms of the association between CHM use and the survival rate among patients with different cancer stages (Table 2), stage IV breast cancer patients demonstrated a higher survival rate with CHM use than those without CHM use ( 65.1%vs 73.9% in CHM users and 34.5%vs 26.1% in non-CHM users, P < .001) under both clinical and pathological staging evaluations. As shown in the Kaplan-Meier analysis (Figure 2), the adjuvant CHM treatment was associated with a higher survival rate as compared to non-CHM use among all stage IV breast cancer patients (P-value: 00035, HR: 0.45, 95% CI: 0.2853-0.7044).
Survival of Breast Cancer Patients With Different Clinical or Pathological Stages Associated With CHM Use.

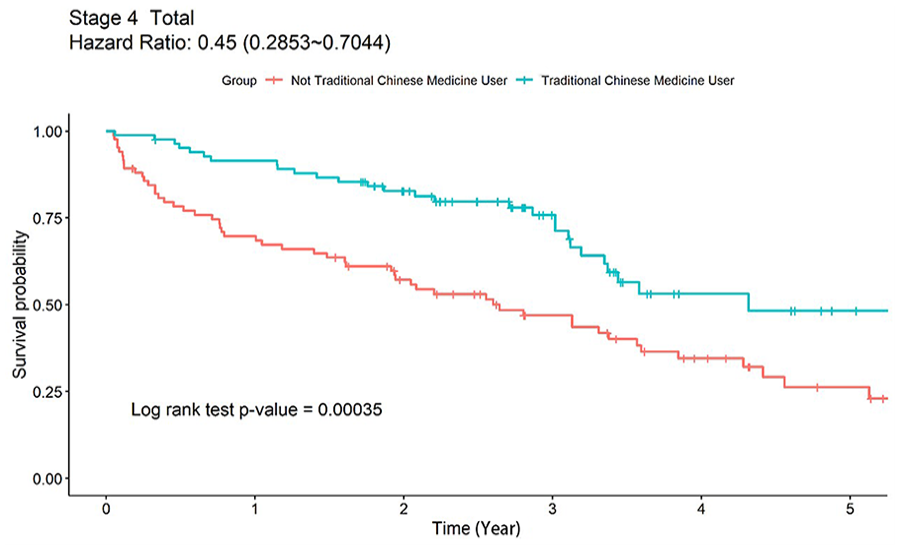
Kaplan–Meier analysis showing cumulative incidence of survival events in all stage IV breast cancer patients of both CHM and non-CHM cohorts during a 5-year follow-up.
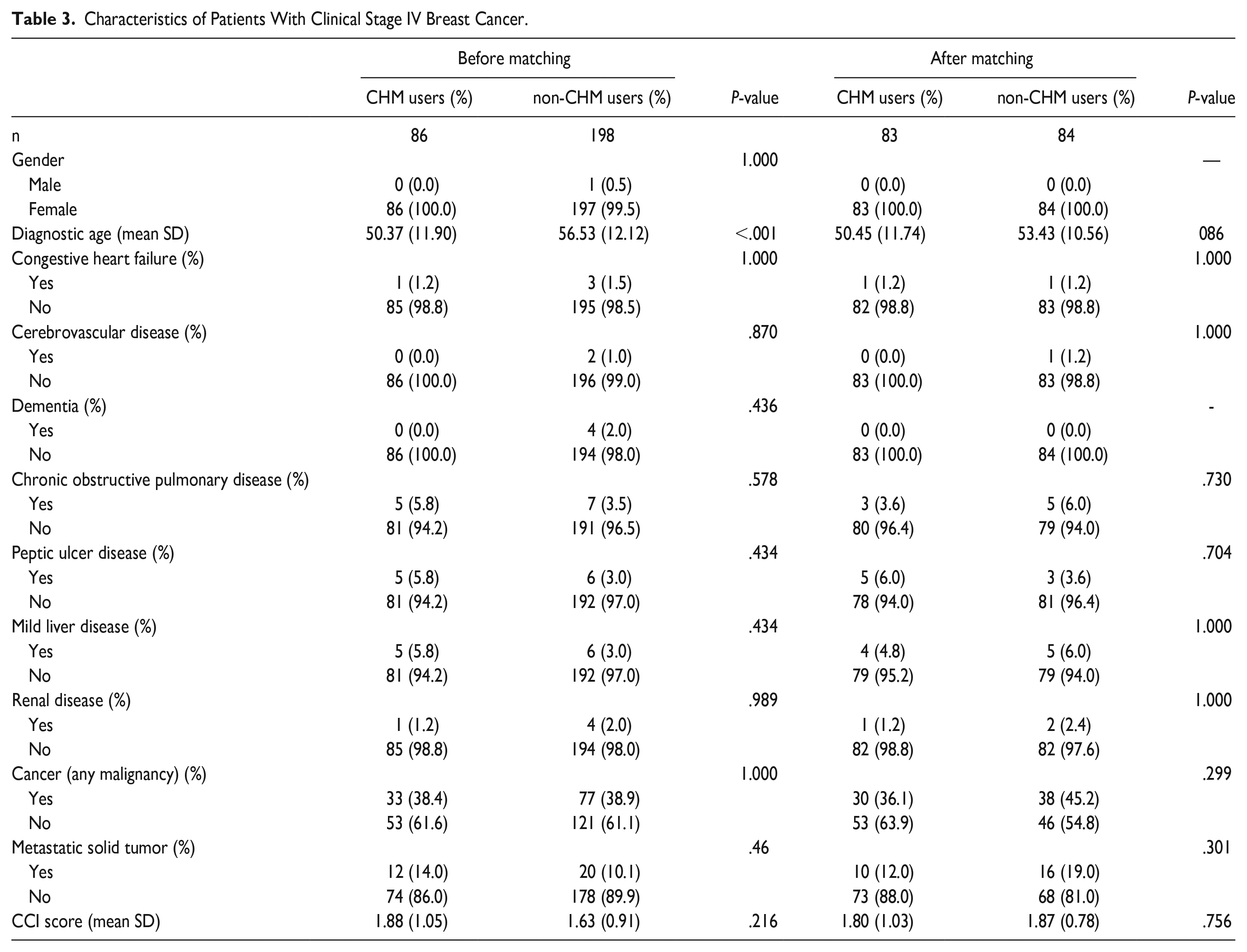
Since CHM use only demonstrated a significantly higher survival rate among stage IV breast cancer patients, we further matched these patients (including 83 CHM users and 84 non-CHM users) for gender, diagnostic age, and comorbidities with no significant differences, as shown in Table 3. There were no significant differences in gender, diagnostic age, or comorbidities between the 2 groups.
Characteristics of Patients With Clinical Stage IV Breast Cancer.
We further analysed the correlations between stage IV breast cancer patients having received a single therapy or combined therapies and CHM use. The analysis revealed that adjuvant CHM treatment was associated with a higher survival rate among stage IV breast cancer patients who had received certain interventions, such as surgery (p: 021, HR: 0.34, 95% CI: 0.1309-0.8865), chemotherapy (P: 00024, HR: 0.39, 95% CI: 0.231-0.656), and hormone therapy (P: 00079, HR: 0.35, 95% CI: 0.1836-0.6636), as shown in the Kaplan-Meier analysis (Figure 3A, C and D). Among stage IV breast cancer patients who had received radiotherapy (P: 064, HR: 0.43, 95% CI: 0.1675-1.0799) or targeted therapy (p: 4, HR: 0.71, 95% CI: 0.3088-1.6143), the CHM use did not have a significant impact on the survival rate (Figure 3B and E).

Kaplan–Meier analysis showing cumulative incidence of survival events including (A) surgery, (B) radiotherapy, (C) chemotherapy, (D) hormone therapy, and (E) target therapy for both CHM and non-CHM cohorts during a 5-year follow-up.
As shown in Table 4, both univariate and multivariate Cox regression analyses were performed to investigate the survival outcomes of stage IV breast cancer patients. The results revealed a significant association between the use of CHM and improved survival among total stage IV breast cancer patients (P: .0015, aHR: 0.466, 95% CI: 0.290-0.746) in the multivariate Cox regression model. Further multivariate analysis indicated that among patients who underwent surgery (P: .0278, aHR: 0.299, 95% CI: 0.102-0.877), chemotherapy (P: .0012, aHR: 0.397, 95% CI: 0.227-0.693), or hormone therapy (P: .0012, aHR: 0.305, 95% CI: 0.149-0.624), the use of CHM was associated with longer survival. However, multivariate Cox regression analysis revealed that CHM use had little impact on the survival rate of patients who had received radiotherapy (P: .1672, aHR: 0.473, 95% CI: 0.163-1.369) or targeted therapy (P: .5603, aHR: 0.776, 95% CI: 0.330-1.823).
The Cox Regression of Survival With Different Treatment Modalities Among Clinical Stage IV Breast Cancer Patients.

Univariate represented relative hazard ratio; Multivariate represented adjusted hazard ratio: Model was adjusted by age, income, area, comorbidities, medications, and CCI scores in Cox proportional hazard regression.
In terms of the association between CHM use and the survival rate of stage IV breast cancer patients, the top 10 most frequently prescribed single herbs and herbal formulas are listed in Table 5. Zhi-Gan-Cao-Tang (ZGCT), Phellodendron chinense Schneid. (huang-bai), and Paeonia lactiflora Pall. (chi-shao) were the 3 most frequently prescribed CHM correlated with an improved survival rate. The herbal constituents of the top 10 most common formulas prescribed for patients with stage IV breast cancer are shown in Supplemental Table 1.
The Top 10 Most Frequently Used Single Herbs And Herbal Formulas And Death Events in Patients With Stage IV Breast Cancer.

Discussion
The prognosis of breast cancer patients depends on a variety of factors, including age at diagnosis, cancer stage, tumor size, nodal status, and estrogen receptor status. 8 As for metastatic breast cancer, studies have shown that the relapse-free interval,9-12 location of metastasis,11-13 amount of circulating tumor cells, 13 patient performance status and weight loss12,14 are of additional concern. In this study, patients with stage IV breast cancer demonstrated a higher survival rate with the combination of CHM treatment. The attenuation of therapy-related side effects and the direct therapeutic effects of certain compounds in herbs are possible factors associated with an improved prognosis for breast cancer patients. Despite the lack of clinical studies evaluating the efficacy of herbal prescriptions in stage IV breast cancer patients, several herbal combinations have demonstrated anti-tumor, anti-metastatic, and bone-protective activities in metastatic breast cancer animal models. 15 The herbal medicines target various mechanisms, such as epithelial mesenchymal transition, reactive oxygen species, and angiogenesis. 16 The synergistic effects of CHM treatment in combination with conventional treatment act to extend the lifespan of stage IV breast cancer patients.
Patients with stage IV breast cancer having received either surgery, chemotherapy, hormone therapy, or adjuvant CHM treatment demonstrated higher survival rates. Our results are consistent with a previous report indicating that CHM treatment may act to suppress tumor progression, relieve post-operative symptoms, increase the sensitivity of chemotherapy, and lessen the damage caused by conventional therapies, 3 as stage IV breast cancer patients may receive various treatment modalities which differ from those received by early-stage breast cancer patients.
With regards to the specific CHM herbs and formulas, Zhi-Gan-Cao-Tang (ZGCT), Phellodendron chinense Schneid. (huang-bai), and Paeonia lactiflora Pall. (chi-shao) are the 3 most frequently prescribed CHM that correlate with the higher survival rate among stage IV breast cancer patients. ZGCT is traditionally used for patients with palpitations, and has been reported to exert effects against anthracycline-induced cardiac toxicity. 17 Meanwhile, glycyrrhetinic acid (GA), the main compound of Glycyrrhiza uralensis Fisch. in ZGCT, has demonstrated an anti-tumor effect in various cancer cells. As shown in a previous study, the combination of GA with doxorubicin not only exhibited a synergistic effect against MCF-7 breast cancer cells but also reduced doxorubicin-induced cardiotoxicity both in vitro and in vivo. 18 As for basal-like breast cancer, the combination of GA with etoposide has been shown to achieve a synergistic effect in regulating the MAPK and PI3 K/AKT pathways. 19 Glycyrrhizic acid has also been reported to exert anti-angiogenesis activities and reduce the production of reactive oxygen species in mice. 20 It is reasonable to speculate that the higher survival rate associated with the use of ZGCT among stage IV breast cancer patients may be due to its anti-cancer and anti-inflammatory properties. Additionally, berberine, the main compound of Phellodendron chinense Schneid., exerts anti-cancer effects by inhibiting cell proliferation and inducing cell apoptosis.21,22 As shown in a previous study, berberine could increase the sensitization of basal-like breast cancer to specific chemotherapeutic agents via XRCC-1-mediated base excision DNA repair. 23 Berberine could also interact with the over-expressed Eph receptor family in breast cancer cells to inhibit cancer proliferation and migration.24,25 Our study further demonstrates that patients having received treatment with Paeonia lactiflora. also demonstrated a higher survival rate. As reported by a previous study, total glycosides of Paeonia lactiflora Pall. exhibited anti-tumor effects in vivo. 26 It should be noted that despite the positive therapeutic effects demonstrated by our study and previous investigations, drawing definitive conclusions regarding the mechanisms responsible for the association between the use of CHM and a higher survival rate is challenging due to the limited number of studies involving CHM and breast cancer patients. The pharmacological effects associated with the possible molecular mechanisms of the top 10 single herbs and formulas noted in this study are listed in Supplemental Tables 2 to 3.
There are indeed several challenges related to conducting large-scale clinical trials involving CHM, however the mining of incorporated data from the TCRD and hospital database as used in the present study presents an efficient method of evaluating the efficacy of CHM. As such, this is the first study to assess the impact of CHM use on the survival rate of stage IV breast cancer patients.
There are several limitations to this study. First, evaluation of the association between CHM dose-response and the survival rate of breast cancer patients may be limited by variable prescriptions, dosage, duration, and frequency of CHM use. However, the integrated databases used in this study contained no relevant records in terms of the dosage or frequency of CHM use. Second, the data in the present study may suffer from selection bias since it was retrieved from a single medical center. In the present study, we have only matched age, comorbidities, and cancer stage which could affect the survival rate among breast cancer patients in our dataset. However, other possible confounding factors such as lifestyle, smoking, alcohol use, social status, and non-pharmacological therapies were unavailable for analysis in the present database. In addition, restricted by the insufficient number of samples in a single hospital database, we were unsuccessful at establishing an effective statistical model for stage IV breast cancer patients with only1 therapy or a combination of 2 therapies. Conducting further investigations with larger study populations is thus recommended.
Conclusions
Our study reveals that patients with stage IV breast cancer who received CHM as an adjuvant therapy exhibited an improved survival rate, particularly when combined with surgery, chemotherapy or hormone therapy. The most effective CHM herbs and formulas for patients with stage IV breast cancer identified in our study were Zhi-Gan-Cao-Tang (ZGCT), Phellodendron chinense Schneid., and Paeonia lactiflora Pall. Additional randomized controlled trials in future prospective studies are recommended to further investigate the beneficial effects of CHM for cancer patients.
Supplemental Material
sj-docx-1-ict-10.1177_15347354231178898 – Supplemental material for Chinese Herbal Medicine Improved Survival in Stage IV Breast Cancer Patients: Data Mining of the Incorporated Taiwan Cancer Registry Database and Hospital Database
Supplemental material, sj-docx-1-ict-10.1177_15347354231178898 for Chinese Herbal Medicine Improved Survival in Stage IV Breast Cancer Patients: Data Mining of the Incorporated Taiwan Cancer Registry Database and Hospital Database by Po-En Chen, Hao-Hsiu Hung, Wei-Te Huang, Chen-Hsuan Yeh, Yi-Wei Kao, Ben-Chang Shia and Sheng-Teng Huang in Integrative Cancer Therapies
Supplemental Material
sj-docx-2-ict-10.1177_15347354231178898 – Supplemental material for Chinese Herbal Medicine Improved Survival in Stage IV Breast Cancer Patients: Data Mining of the Incorporated Taiwan Cancer Registry Database and Hospital Database
Supplemental material, sj-docx-2-ict-10.1177_15347354231178898 for Chinese Herbal Medicine Improved Survival in Stage IV Breast Cancer Patients: Data Mining of the Incorporated Taiwan Cancer Registry Database and Hospital Database by Po-En Chen, Hao-Hsiu Hung, Wei-Te Huang, Chen-Hsuan Yeh, Yi-Wei Kao, Ben-Chang Shia and Sheng-Teng Huang in Integrative Cancer Therapies
Supplemental Material
sj-docx-3-ict-10.1177_15347354231178898 – Supplemental material for Chinese Herbal Medicine Improved Survival in Stage IV Breast Cancer Patients: Data Mining of the Incorporated Taiwan Cancer Registry Database and Hospital Database
Supplemental material, sj-docx-3-ict-10.1177_15347354231178898 for Chinese Herbal Medicine Improved Survival in Stage IV Breast Cancer Patients: Data Mining of the Incorporated Taiwan Cancer Registry Database and Hospital Database by Po-En Chen, Hao-Hsiu Hung, Wei-Te Huang, Chen-Hsuan Yeh, Yi-Wei Kao, Ben-Chang Shia and Sheng-Teng Huang in Integrative Cancer Therapies
Footnotes
Acknowledgements
The authors would like to thank James Waddell for the critical reading and revision of our manuscript.
Author Contributions
PEC and HHH equally wrote the draft and interpreted the data. WTH, YWK, YCL, CHH collected, assembled the data. WTH, YWK analyzed the data. BCS provided methodological support and rectified all analyzed data. STH designed, conceived the study, and edited the manuscript. All of the authors approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported and funded by Ministry of Science and Technology of Taiwan [MOST 110-2320-B-039-40 -], An-Nan Hospital, China Medical University [ANHRF-110-25] and China Medical University Hospital [DMR-110-189, DMR-111-005, DMR-111-013, DMR-111-016, and DMR-111-195].
Ethical Approval
This study was approved by the Research Ethics Committee of China Medical University Hospital (CMUH-107-REC2-023) and was in compliance with the Declaration of Helsinki.
Informed Consent
The requirement of informed consent signed by patients was waived in lieu of anonymized data.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
