Abstract
Background:
Cisplatin is a widely used anticancer drug in clinic, but it has a damaging effect on skeletal muscle cells. Clinical observation showed that Yiqi Chutan formula (YCF) had a alleviating effect on cisplatin toxicity.
Methods:
In vitro cell model and in vivo animal model were used to observe the damage effect of cisplatin on skeletal muscle cells and verify that YCF reversed cisplatin induced skeletal muscle damage. The levels of oxidative stress, apoptosis and ferroptosis were measured in each group.
Results:
Both in vitro and in vivo studies have confirmed that cisplatin increases the level of oxidative stress in skeletal muscle cells, thus inducing cell apoptosis and ferroptosis. YCF treatment can effectively reverse cisplatin induced oxidative stress in skeletal muscle cells, thereby alleviating cell apoptosis and ferroptosis, and ultimately protecting skeletal muscle.
Conclusions:
YCF reversed cisplatin-induced apoptosis and ferroptosis of skeletal muscle by alleviating oxidative stress.
Introduction
According to a global cancer report, cancer is a leading cause of death worldwide, accounting for nearly 10 million deaths in 2020, or nearly 1 in 6 deaths. 1 Although advances have been made in targeted therapy and immunotherapy, platinum-based chemotherapy is still the standard treatment for most advanced cancers (such as lung cancer), with a large proportion.2-4
Cisplatin is the first generation of platinum-containing antitumor drugs, belonging to a broad spectrum of antitumor drugs. It has been approved for clinical use by the US Food and Drug Administration (FDA) since 1978, and is still widely used in clinical practice. Cisplatin has a simple structure and a clear mechanism of action. It can induce cell apoptosis, ferroptosis and pyroptosis along with the increase of intracellular reactive oxygen species (ROS), thus killing cells.5-8 Since cisplatin cannot act specifically on cancer cells, it will inevitably have a killing effect on normal cells as well, resulting in adverse reactions during treatment. Weight loss and are serious adverse reactions during chemotherapy that may be related to the damage of muscle cells by cisplatin and that seriously affect the quality of life of patients.9-12
Yiqi Chutan formula (YCF) is a traditional Chinese medicine prescription.13,14 Clinical observation and animal experiments have shown that it can reduce weight loss and fatigue in tumor patients and tumor-bearing mice receiving cisplatin chemotherapy,13,15-17 but its specific pharmacological mechanism remains unclear. Therefore, it is of great significance for clinical use to clarify the pharmacological mechanism of YCF in alleviating the side effects of cisplatin chemotherapy. In this study, through in vitro and in vivo research, we elucidated the mechanism of cisplatin damage to muscle cells, and revealed the pharmacological mechanism of YCF in antagonizing cisplatin damage to muscle cells.
Materials and Methods
Animal Ethics Statement
A total of 24 female C57BL/6 mice weighing between 17 and 19 g and aged 6 weeks (Beijing HFK Bioscience Co., Ltd., Beijing, China) were used in this investigation. All experiments were conducted according to the relevant laws and institutional guidelines. Mice were individually housed in independent ventilated cages at 24°C to 26°C under constant humidity with a 12-h light/dark cycle. The mice were divided into 3 groups (n = 8), namely, control, cisplatin, and cisplatin plus YCF groups in a random order. Permission for the experimental scheme from the laboratory animal ethics committee of Jinan University was granted (Approval No. IACUC-20200923-16).
YCF Preparation
According to previous reports13,14,YCF is composed of 8 traditional Chinese medicines, which are Ranunculus ternatus Thunb., Bombyx mori L., Sarcandra glabra (Thunb.) Nakai, Cremastra appendiculata (D.Don) Makino, Fritillaria thunbergii Miq, Pinellia ternata (Thunb.) Breit, Ganoderma lucidum (Leyss.ex Fr.) Karst, and Panax quinquefolium L. respectively, and the composition ratio is 3:2:2:2:2:2:2:2. The Chinese medicines materials were purchased from the first affiliated hospital of Jinan University (Guangzhou, China), and the naming and processing methods of all Chinese medicines materials were in accordance with the provisions of the pharmacopeia of the People’s Republic of China (2020). The traditional Chinese medicine was prepared into concentrated solution as previously reported 18 at a final concentration of 1 g/mL (traditional Chinese medicine weight/volume). According to the previously reported method, 13 the obtained YCF extracts were identified by liquid chromatography-mass spectrometry (LC-MS) fingerprint.
Chemicals, Reagents and Antibodies
Cisplatin (Cat. #H20040813) injection was purchased from Jiangsu Hausen Pharmaceutical Co., Ltd (Jiangsu, China). Phosphate-buffered saline (PBS, Cat. #10010023) was purchased from Gibco (NY, USA). Primary antibodies against Calmodulin (Cat. #35944S), Cytochrome c (Cat. #4280T), Cleaved Caspase-3 (Cat. #9664T), Cleaved PARP (Cat. #5625T), and β-Actin (Cat. #3700T) and rabbit (Cat. #7074P2 and 8889S) or mouse (Cat. #7076P2 and 8890S) secondary antibodies were purchased from Cell Signaling Technology (Danvers, MA, USA). Primary antibodies against GPX4 (Cat. #67763-1-Ig) were purchased from Proteintech (Wuhan, China).
Cell Culture
C2C12 myoblasts were purchased from Fuheng BioLogy (Shanghai, China). Lewis lung cancer (LLC) cells were acquired from the Guangzhou University of Chinese Medicine (Guangzhou, China). Dulbecco’s modified Eagle’s medium, High Glucose, L-Glutamine, Phenol Red (DMEM, Cat. #11965092), fetal bovine serum (FBS, Cat. #10270106), penicillin/streptomycin (Cat. #10378016), trypsin-EDTA, and 0.05% phenol red (Cat. #25200072) were purchased by Gibco (NY, USA). All cells were identified by short tandem repeat (STR). Cells were cultured in DMEM supplemented with 10% FBS, 100 U/mL penicillin, and 100 µg/mL streptomycin. Cells were placed in the 3 gas incubator (Thermo Fisher Scientific, Waltham, MA, USA) containing 5% CO2, and 37°C constant temperature and damp environment culture. The medium was changed every 72 hours, and the cells were routinely subcultured when the cells reached 90% confluence. Logarithmic growth phase C2C12 and LLC cells were used to conduct the experiments. If the experimental process needed to be treated with ROS scavenger N-acetyl-cysteine (NAC, Cat. #A9165, Sigma–Aldrich, Darmstadt, Germany), apoptosis inhibitor Z-VAD-FMK (Cat. #V116, Sigma–Aldrich, Darmstadt, Germany), and ferroptosis inhibitor ferrostatin-1 (Fer-1, Cat. #SML0583, Sigma–Aldrich, Darmstadt, Germany), the use methods are pretreatment for 4 hours. All other factors were co-treated for 48 hours.
Animal Experiment and Sample Collection
After the experiment began, the control group was intraperitoneally injected with normal saline every 7 days (0.1 mL each time, 5 times in total) and intragastric administration with normal saline every day (0.2 mL each time, 28 times in total), the cisplatin group was intraperitoneally injected with cisplatin every 7 days (2 mg/kg, 0.1 mL each time, 5 times in total) and intragastric administration with normal saline every day (0.2 mL each time, 28 times in total), and the cisplatin plus YCF group was intraperitoneally injected with cisplatin every 7 days (2 mg/kg, 0.1 mL each time, 5 times in total) and intragastric administration with YCF every day (4 g/kg, 0.2 mL each time, 28 times in total). The body weight of the mice was measured every 7 days. After 28 days of treatment, all mice were anesthetized with isoflurane inhalation (Cat. #R510-22, RWD Life Science Pharmaceutical Co., Ltd., Shenzhen, China). The bilateral gastrocnemius muscles of mice were immediately removed on ice for subsequent examinations when the mice were unconscious. After the samples were collected, the mice were sacrificed with an overdose of anesthetic.
MTT Colorimetric Assay
Cell viability was measured by the MTT colorimetric assay according to the published protocol. 19 3-(4,5-Dimethylthiazol-2-y1)-2,5-diphenyltetrazolium bromide (MTT, Cat. #V13154) was supplied by Gibco (NY, USA). This experiment was performed using 96-well plates, with 6 replicates per group.
Flow Cytometry
The concentration of intracellular ROS and the proportion of apoptotic cells were measured by flow cytometry. Prior to experiments, cells were digested with trypsin and centrifuged in order to produce a single-cell suspension. The concentration of intracellular ROS was measured following the instructions of the 2′,7′-Dichlorodihydrofluorescein diacetate (DCFH-DA) staining Kit (Cat. #MAK143, Sigma, USA) and the proportion of apoptotic cells was measured following the instructions of the Annexin V-FITC Apoptosis Detection Kit (Cat. #C1062L, Beyotime Institute of Biotechnology, Shanghai, China).
Cytological and Histological Analysis
Western blot
Cells and gastrocnemius tissue samples were lysed in RIPA buffer (Cat. #P0013B, Beyotime Biotechnology, Shanghai, China) containing 1 mM phenylmethylsulfonyl fluoride (PMSF, Cat. #ST505, Beyotime Biotechnology, Shanghai, China) and 1× protease and phosphatase inhibitor cocktail (Cat. #P1045, Beyotime Biotechnology, Shanghai, China) on ice. The lysates were then centrifuged at 12 000g for 15 minutes at 4°C, and the protein concentration in the obtained supernatant was determined using an Enhanced BCA Protein Assay Kit (Cat. #P0010S, Beyotime Biotechnology, Shanghai, China). Equal amounts of protein were separated via SDS–PAGE and transferred to polyvinylidene fluoride (PVDF) membranes (Cat. #IEVH00005, Merck KGaA, Darmstadt, Germany). Next, the PVDF membranes were blocked with Tris-buffered saline plus Tween-20 (TBST) containing 5% skim milk for 1 hour and incubated with the indicated primary antibodies, including rabbit anti-Calmodulin (1:1000), anti-Cytochrome c (1:1000), anti-Cleaved Caspase-3 (1:1000), anti-Cleaved PARP (1:1000), and anti-β-Actin (1:1000), mouse anti-GPX4 (1:2000), after which the membranes were incubated with the corresponding secondary antibodies (1:3000). Then, PVDF membranes were washed 3 times with TBST solution, and visualized by Beyotime’s hypersensitive ECL kit (Cat. #P0018S). Density values of the bands were captured and documented through a gel image analysis system (ChemiDox™, Bio-Rad, USA) and normalized to GAPDH; n = 3.
Immunofluorescence
Fresh gastrocnemius tissue was added with optimal cutting temperature (OCT) compound and then quickly frozen in liquid nitrogen and cut into serial sections (8 μm thick). The tissue ROS concentration was measured following the instructions of the Frozen Section Reactive Oxygen Species (ROS) detection kit (Cat. #BB-470516, Bestbio, Shanghai, China). Fluorescence microscopy (excitation wavelength 535 nm, emission wavelength 610 nm) was used to detect the intensity of red fluorescence, so as to determine the level of reactive oxygen species in the samples. In addition, these sections were immunostained with the indicated primary antibodies, including anti-Cleaved Caspase-3 (1:400), anti-Cleaved PARP (1:400), and anti-GPX4 (1:400), followed by incubation with the corresponding secondary antibodies (1:500). Then, sections were washed 3 times with TBST solution, and finally the red fluorescence intensity was measured using a fluorescence microscope (excitation wavelength 510 nm, emission wavelength 590 nm) to determine the protein content of the sample. All sections were stained with DAPI for the nucleus in the tissues.
Terminal deoxynucleotidyltransferase-mediated dUTP-biotin nick end labeling (Tunel)
Fresh gastrocnemius tissue was fixed with 4% paraformaldehyde and then paraffin embedded and cut into serial sections (10 μm thick). The tissue apoptosis level was measured following the instructions of the TUNEL apoptosis detection kit (Cat. #ATK00001, AtaGenix, Hubei, China). Fluorescence microscopy (excitation wavelength 495 nm, emission wavelength 515 nm) was used to detect the intensity of green fluorescence, so as to determine the level of apoptosis in the samples. All sections were stained with DAPI for the nucleus in the tissues.
Transmission electron microscope (TEM)
Fresh gastrocnemius tissue was fixed with 2.5% glutaraldehyde and then embedded, sectioned, and stained as previously reported. 13 The sections were photographed using the transmission electron microscope (HITACHI HT7700, Japan).
Biochemical Index Detection
Measurement of intracellular and gastrocnemius tissue glutathione (GSH) concentrations
GSH and GSSG assay kits (Cat. #S0053, Beyotime, Shanghai, China) were used to measure intracellular and tissue GSH concentrations. In brief, cells and fresh tissues were collected, and the experimental data were determined with a microplate reader according to the kit instructions. These experiments were performed in triplicate. The data were expressed as a percentage relative to the control group.
Measurement of intracellular and gastrocnemius tissue iron concentrations
An iron assay kit (Cat. #MAK025, Sigma, USA) was used to measure iron concentrations in cells and tissues. Cells (2 × 106), or fresh tissue (10 mg) was rapidly homogenized in 5 volumes of iron assay buffer. The samples were centrifuged at 16 000g for 10 minutes at 4°C to remove insoluble material. The assay was performed according to the kit instructions. A microplate reader was used to measure the absorbance at 593 nm (A593). These experiments were performed in triplicate. The data are expressed in nmol/L.
Measurement of intracellular lipid ROS concentrations
C11-BODIPY (Cat. #D3861, Thermo, USA) was used to measure intracellular lipid ROS concentrations according to the manufacturer’s instructions. Briefly, cells were cultured with 100 μmol/L C11-BODIPY for 30 minutes at 37°C and then samples were washed twice with PBS. Then, the fluorescence intensities were measured at an emission wavelength of 510 nm and an excitation wavelength of 488 nm using a fluorescence microplate reader. These experiments were performed in triplicate. The data are expressed as the percentage of the fluorescence intensity relative to that of the control group.
Measurement of intracellular and gastrocnemius tissue malondialdehyde (MDA) concentrations
A Lipid Peroxidation MDA Assay Kit (Cat. #S0131S, Beyotime, Shanghai, China) was used to measure MDA concentrations in cells or tissues. In brief, cells or fresh tissues were collected, and the experimental data were determined with a microplate reader according to the kit instructions. These experiments were performed in triplicate. The data are expressed in μmol/mg.
Statistical Analysis
The experimental data were analyzed using Student-Newman-Keuls (S-N-K) in ANOVA with SPSS version 13.0 software (SPSS Inc., IL, USA). GraphPad Prism 9 (GraphPad Software, LLC, California, USA) was used to graph the data. The results are presented as the mean values ± standard deviation (SD). A P value ≤.05 was considered statistically significant.
Results
YCF Can Reverse Cisplatin-Induced Myoblast Death In Vitro
The LC-MS primary ion current chromatogram of YCF extracts is shown in Figure 1A. By comparing with the secondary chromatograms in the database (mzCloud), we obtained 18 compounds (mzCloud Best Match ≥90, from 1 to 18, the best match decreases) in YCF extracts. Compounds 1 to 18 in Figure 1A are palmitic acid, cytidine, adenine, nicotinic acid, 6-hydroxynicotinic acid, (±)12(13)-diHOME, (±)9-hpODE, (±)-abscisic acid, 16-hydroxyhexadecanoic acid, stearic acid, hypoxanthine, astragalin, neochlorogenic acid, adenine, chlorogenic acid, neochlorogenic acid, 2-isopropylmalic acid, and ferulic acid, respectively. It should be noted that compound 12 and compound 16 should be the same compound, neochlorogenic acid. As shown in Figure 1B, cisplatin inhibited the activity of C2C12 myoblasts in a concentration dependent manner, while low concentrations of YCF had no significant effect on the activity of C2C12 myoblasts (Figure 1C). We selected 3.33 μg/mL cisplatin and 0.64 mg/mL YCF as the concentrations for subsequent experiments. At this concentration, the cell viability inhibition rate of cisplatin on C2C12 myoblasts was 56.2% ± 2.5%, while the concentration of YCF was half of the maximum concentration of no statistical significance for the viability of C2C12 myoblasts. As shown in Figure 1D, YCF, ROS scavenger NAC, apoptosis inhibitor Z-VAD-FMK and ferroptosis inhibitor Fer-1 all reversed cisplatin induced C2C12 myoblast death to varying degrees. There was no significant difference between YCF and NAC in the reversal effect, while apoptosis inhibitors and ferroptosis inhibitors were less effective than YCF. In addition, we tested whether YCF affected the anti-tumor effect of cisplatin, and the results showed that cisplatin combined with YCF did not affect the anti-tumor effect of cisplatin, while YCF alone had a slight inhibitory effect on tumor cells (Figure 1E).
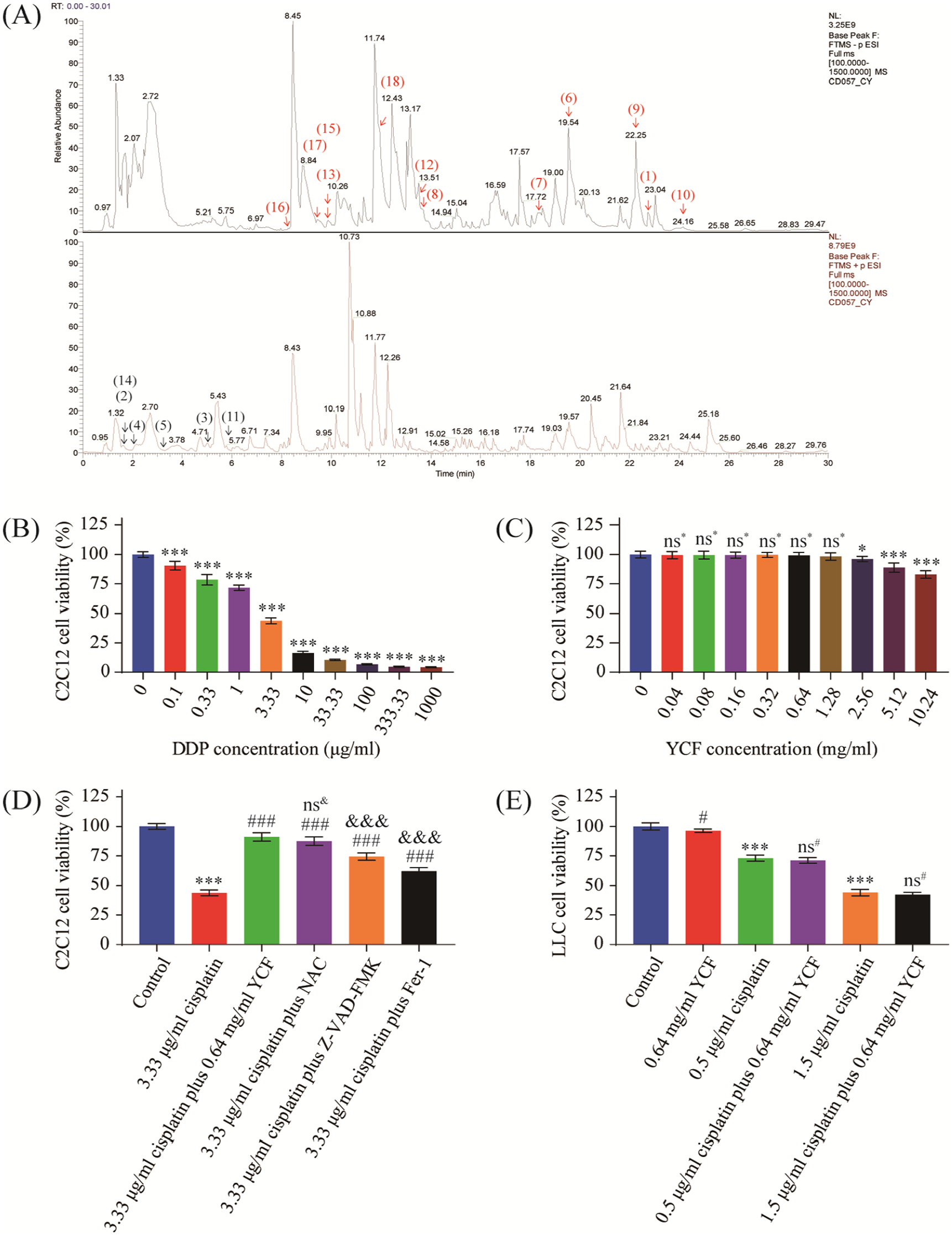
YCF reversed cisplatin-induced myoblast death in vitro. (A) Total ion current chromatogram of natural products identified by LC/MS; column 1 black is the total ion current diagram of negative ion mode and column 2 red is the total ion current diagram of positive ion mode. (B) C2C12 myoblast reactivity to cisplatin. (C) C2C12 myoblast reactivity to YCF. (D) Effect of YCF co-treatment and NAC, Z-VAD-FMK, and Fer-1 pretreatment on cisplatin inhibition of C2C12 myoblasts. (E) LLC cell reactivity to cisplatin and YCF. The data are presented as the means ± SD of triplicate experiments; (B, E) ns*P ≥ .05, *P ≤ .05, ***P ≤ .001 compared with the control group (0 μg or mg/mL); (D) *P ≤ .05, ***P ≤ .001 compared with the control group ns& P ≥ .05, &&&P ≤ .001 compared with the 3.33 μg/mL cisplatin plus 0.64 mg/mL YCF group; (E) ns# P ≥ .05, #P ≤ .05 compared with the groups with the same cisplatin concentration; n = 6.
YCF Can Alleviate Cisplatin-Induced Oxidative Stress and Reverse Myoblast Apoptosis In Vitro
As shown in Figure 2A, cisplatin increased intracellular ROS concentration in C2C12 myoblasts in a concentration-dependent manner, while YCF and ROS scavenger NAC could effectively antagonize cisplatin-induced ROS concentration increase. As shown in Figure 2B, cisplatin increased the apoptosis rate (Q2 + Q4) of C2C12 myoblasts in a concentration-dependent manner, while YCF and ROS scavenger NAC both effectively antagonized cisplatin induced apoptosis. Western blot results showed that cisplatin decreased the expression of calmodulin and increased the expression of Cytochrome c, Cleaved Caspase-3 and Cleaved PARP in C2C12 myoblasts in a concentration-dependent manner. YCF and NAC could reverse the expression changes of these proteins after cisplatin treatment (Figure 2C).
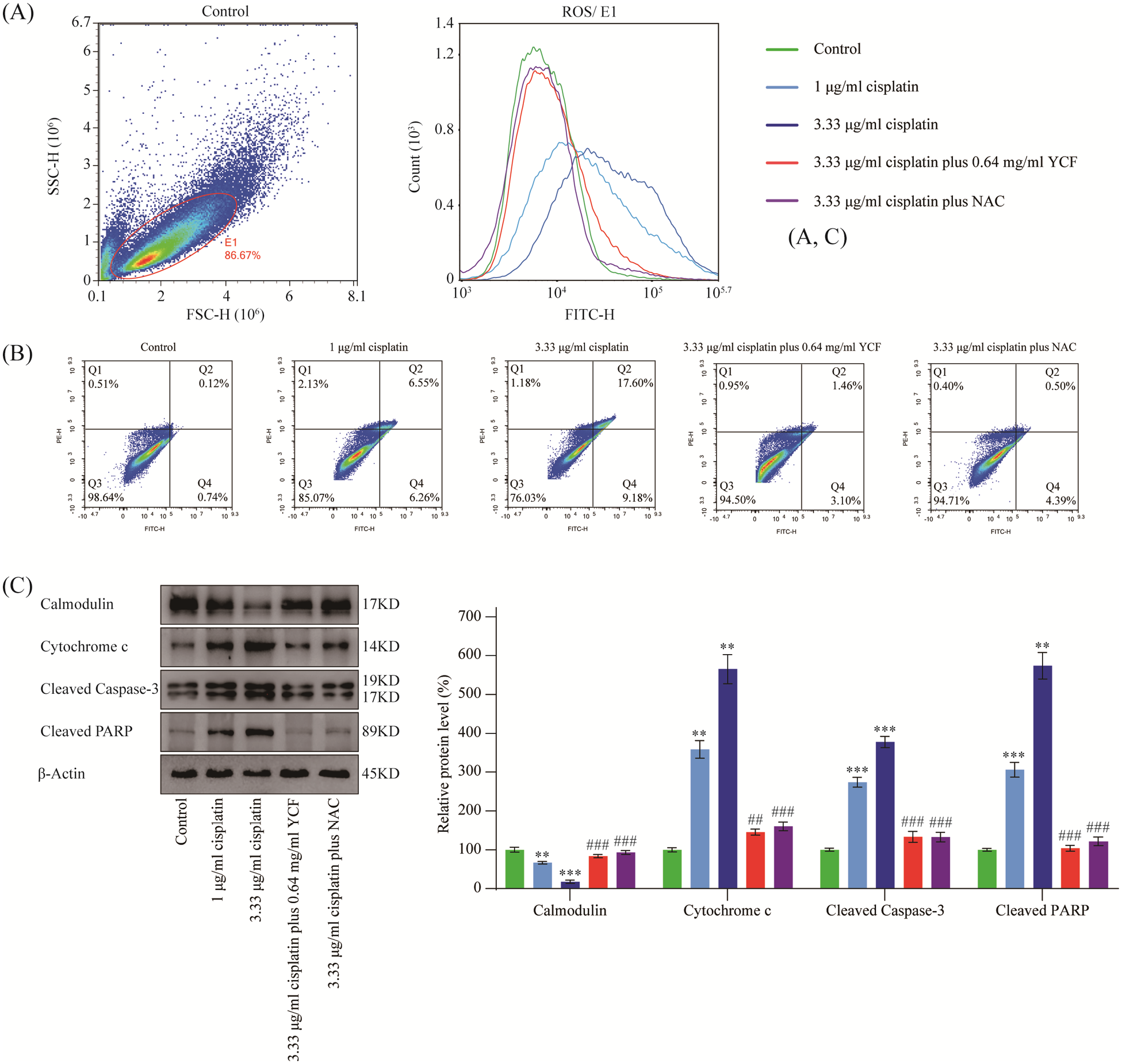
YCF alleviated cisplatin-induced oxidative stress and reversed apoptosis in vitro. (A) Determination of intracellular ROS levels. (B) Determination of cell apoptosis rate. (C) Determination and relative quantification of oxidative stress and apoptosis related proteins; the data are presented as the means ± SD of triplicate experiments; **P ≤ .01, ***P ≤ .001 compared with the control group; ## P ≤ .01, ### P ≤ .001 compared with the 3.33 μg/mL cisplatin group, n = 3.
YCF Can Reverse Cisplatin-Induced Ferroptosis in Myoblasts In Vitro
Based on the results in Figure 1D, we already know that the ferroptosis inhibitor Fer-1 can partially reverse cisplatin-induced C2C12 myoblast death. Therefore, we examined whether YCF can also reverse cisplatin-induced C2C12 myoblast ferroptosis. The results showed that cisplatin decreased the concentration of antioxidant index GSH and inhibited the expression of GPX4 protein in a concentration-dependent manner in cells. Both YCF and Fer-1 can effectively restore the expression of these 2 antioxidant indexes after cisplatin treatment (Figure 3A and E). In addition, after cisplatin treatment, ferroptosis related indicators such as intracellular iron ion concentration and lipid peroxidation indicators lipid ROS and MDA increase in a concentration dependent manner. Similarly, both YCF and Fer-1 were able to reverse these cisplatin-induced changes (Figure 3B-D).

YCF reversed cisplatin-induced ferroptosis in myoblasts in vitro. (A) Determination of GSH levels in cells. (B) Determination of iron levels in cells. (C) Determination of lipid ROS levels in cells. (D) Determination of MDA levels in cells. (E) Determination and relative quantification of ferroptosis related protein. The data are presented as the means ± SD of triplicate experiments; **P ≤ .01, ***P ≤ .001 compared with the control group; ## P ≤ .01, ### P ≤ .001 compared with the 3.33 μg/mL cisplatin group, n = 3.
YCF Can Alleviate Cisplatin-Induced Oxidative Stress in Skeletal Muscle In Vivo
Animal experiments have shown that cisplatin can cause significant weight loss in mice, and YCF can partially reverse the weight loss caused by cisplatin (Figure 4A). Consistent with in vitro experiments, cisplatin decreased the concentration of antioxidant index GSH in gastrocnemius muscle, while ROS and MDA concentrations, which reflect the degree of oxidation, increased. Similarly, YCF effectively reversed cisplatin-induced gastrocnemius oxidative stress (Figure 4B-D). In addition, cisplatin resulted in increased iron content in gastrocnemius muscle, which was significantly reversed by YCF (Figure 4D). Western blot results showed that cisplatin injection reduced the expression of Calmoduin protein and increased the expression of Cytochrome c protein in gastrocnemius of mice. YCF effectively reversed the cisplatin-induced protein expression changes (Figure 4F).

YCF alleviated cisplatin-induced oxidative stress in skeletal muscle in vivo. (A) The body weight of mice; n = 8. (B) ROS fluorescence staining of gastrocnemius tissues; scale bars = 100 μm. (C) Determination of GSH levels in gastrocnemius tissues. (D) Determination of iron levels in gastrocnemius tissues. (E) Determination of MDA levels in gastrocnemius tissues. (F) Determination and relative quantification of oxidative stress related proteins. The data are presented as the means ± SD of triplicate experiments; **P ≤ .01, ***P ≤ .001 compared with the control group; ### P ≤ .001 compared with the cisplatin group; n = 3.
YCF Can Reverse Cisplatin-Induced Apoptosis and Ferroptosis in Skeletal Muscle In Vivo
Immunofluorescence staining showed that the expression of apoptosis markers, Cleaved Caspase-3 and Cleaved PARP, and the number of Tunel positive cells were significantly increased after cisplatin treatment. In addition, the expression of antioxidant and anti-ferroptosis protein GPX4 was significantly down-regulated. Similarly, YCF can reverse cisplatin induced changes in apoptosis and ferroptosis markers (Figure 5A and B). The results of transmission electron microscopy showed that mitochondria in the cisplatin group were smaller, membrane density was increased, mitochondrial ridges were reduced or disappeared, and mitochondrial outer membrane was broken, while mitochondria in the control group and the cisplatin plus YCF group were normal (Figure 5C).

YCF reversed cisplatin-induced apoptosis and ferroptosis in skeletal muscle in vivo. (A) Immunofluorescence staining of proteins associated with apoptosis and ferroptosis in gastrocnemius; scale bars = 100 μm. (B) Tunel staining of gastrocnemius; scale bars = 100 μm. (C) Transmission electron microscope of gastrocnemius; scale bars = 2 μm (up) and 1 μm (down).
Discussion
As a well-established and highly effective chemotherapeutic drug, the use of cisplatin especially in the treatment of lung cancer tends to induce muscle injury and lead to weight loss and fatigue.9,10,12 YCF is a traditional Chinese medicine prescription commonly used in the adjuvant treatment of malignant tumors. Preliminary clinical observation has shown that it has good clinical therapeutic effect and can effectively reduce weight loss and fatigue caused by chemotherapy.15,16 In addition, tumor model animal experiments have also confirmed this point.13,17 This study confirms that YCF may achieve clinical therapeutic effect by alleviating cisplatin-induced skeletal muscle injury.
Cisplatin, as a broad-spectrum cell-nonspecific antitumor drug, can directly kill most cells in the body. Our data confirm that it can kill both tumor cells and normal myoblasts in a concentration-dependent manner. Previous studies have confirmed that the main mechanism by which cisplatin can inhibit tumor cells is that it can bind to DNA in tumor cells and cause DNA crosslinking, thus destroying the function of DNA and inhibiting cell mitosis, and ultimately inhibiting tumor progression. 20 However, for normal cells with slow division, the damage caused by cisplatin is mainly closely related to ROS.21,22 Our in vitro and in vivo experiments have demonstrated that cisplatin can lead to increased ROS levels in muscle cells, and this cisplatin-induced ROS can be effectively cleared by YCF and NAC. This may be related to the active ingredients in YCF. Active ingredients nicotinic acid, (±)12(13)-diHOME, astragalin, neochlorogenic acid, chlorogenic acid, 2-isopropylmalic acid, and ferulic acid in YCF have been proven to effectively remove ROS from cells and tissues, thereby preventing cells and tissues from being damaged by excessive ROS.23-29 Moreover, ferulic acid has been confirmed to directly reverse the excess ROS induced by cisplatin in normal cells, thus protecting normal cells. 29 Due to this difference between the antitumor effect of cisplatin and the mechanism of damage to normal cells, YCF can effectively relieve the adverse reactions of cisplatin to muscle without affecting its antitumor effect.
ROS in the physiological range is essential for life, but excessive ROS can cause serious damage to normal cells and tissues.30-33 ROS damages the mitochondria directly, causing the release of cytochrome C from the mitochondria into the cytoplasm. 34 In addition, ROS can damage the endoplasmic reticulum, resulting in decreased expression and function of calmodulin, which affects the normal regulation of intracellular calcium ions, and further promotes the release of cytochrome C from mitochondria into the cytoplasm.35-39 Cytochrome C will further promote the activation of Caspase-3, 39 and cleaved Caspase-3 will cleave PARP, resulting in the inactivation of PARP, 40 which will reduce DNA stability 41 and ultimately induce apoptosis. 42 Such chromosomal DNA double strand breaks or single strand breaks during apoptosis can be detected using Tunel. Our study confirmed that the ROS scavenger NAC could effectively inhibit the above process of changes induced by cisplatin and ultimately reverse cisplatin-induced apoptosis of C2C12 myoblasts. YCF also showed a similar effect to NAC, reversing cisplatin induced myoblast apoptosis.
In addition, previous studies have shown that cisplatin can directly enter cells and bind to GSH, leading to depletion of GPX4. 43 Both GSH and GPX4 are important antioxidant factors that reduce lipid peroxides to lipid alcohols to maintain intracellular redox homeostasis. 44 The inactivation of GSH and GPX4 leads to intracellular lipid peroxidation and ultimately to ferroptosis. 44 The typical characteristics of ferroptosis are smaller mitochondria, increased membrane density, reduced or disappeared mitochondrial ridges, and broken mitochondrial outer membrane. 45 This process can be inhibited by ROS scavenger NAC and ferroptosis inhibitor Fer-1. 46 Similarly, YCF showed a similar effect to Fer-1, reversing cisplatin induced ferroptosis in myoblasts.
Through in vitro and in vivo experiments, we confirmed that cisplatin leads to apoptosis and ferroptosis of skeletal muscle cells through oxidative stress, and ultimately leads to decreased myoblast activity and skeletal muscle injury. YCF is a commonly used prescription in clinical practice. In this study, we confirmed that YCF can effectively relieve cisplatin-induced muscle cell apoptosis and ferroptosis by alleviating cisplatin-induced oxidative stress, thus protecting skeletal muscle. This may be related to a variety of antioxidant compounds in the active components of YCF. Further experiments will be carried out for this hypothesis in the future.
The hypothesized mechanism by which YCF alleviates muscle cell apoptosis and ferroptosis by reducing cisplatin induced oxidative stress in skeletal muscle cells is shown in Figure 6.
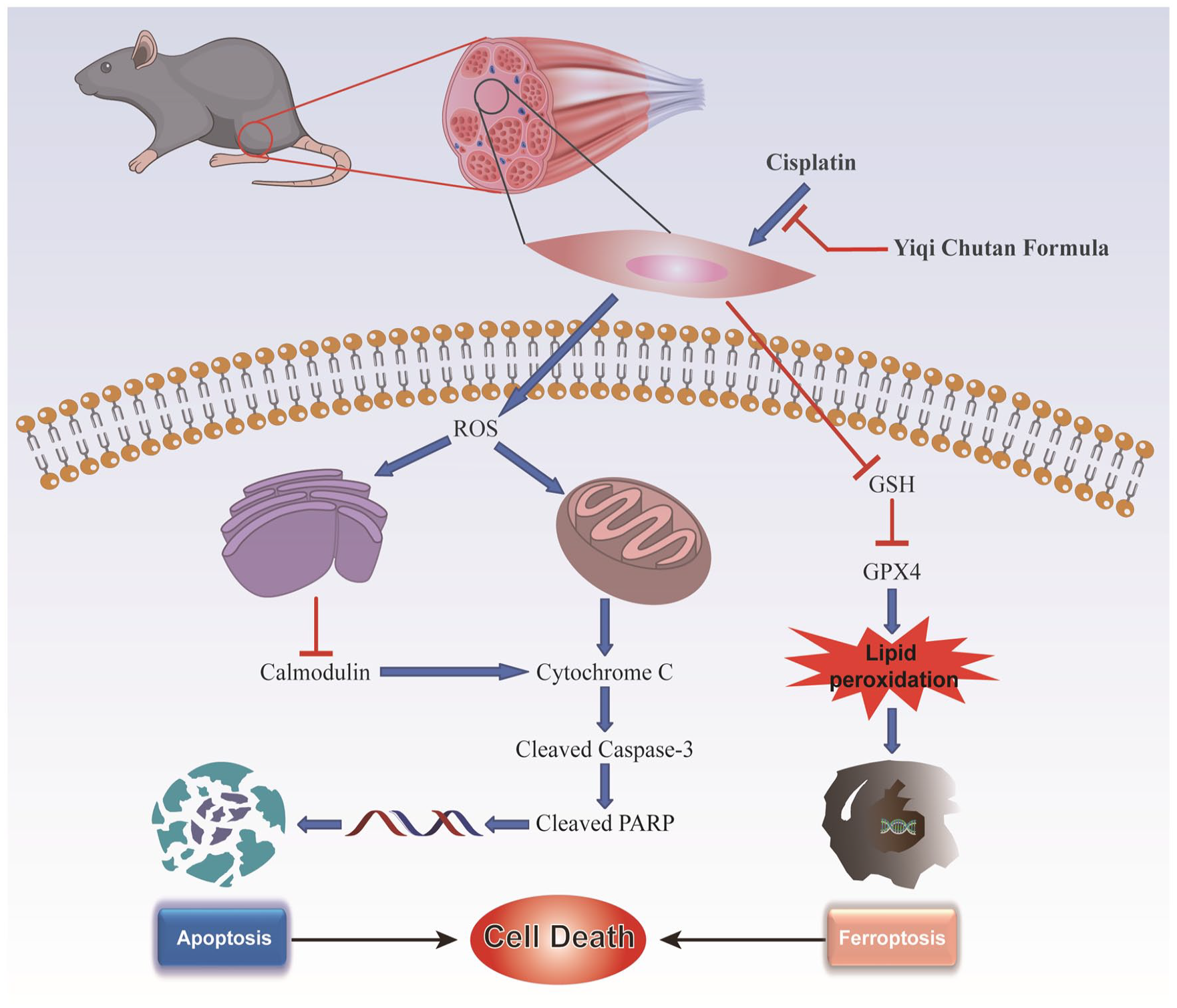
Schematic of the potential underlying mechanism by which YCF alleviates cisplatin-induced apoptosis and ferroptosis in skeletal muscle.
Conclusions
In this study, all results demonstrated that YCF could reverse cisplatin-induced apoptosis and ferroptosis of skeletal muscle by alleviating oxidative stress, thus protecting skeletal muscle cells.
Footnotes
Author Contributions
Yingchao Wu, Dajin Pi, and Shuyao Zhou are co-first authors. Yingchao Wu performed the in vitro experiments. Dajin Pi performed the in vivo experiment. Shuyao Zhou wrote the manuscript. Wuhong Wang and Huan Ye conducted the experiments and analyzed the data. Zhongjia Yi and Yiliu Chen conducted the experiments and completed the graphing of data and drawing of the figure. Mingzi Ouyang designed the study and provided the initial idea. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant Nos. 81873155 and 81403340), which supported the design, analysis, and interpretation of the data in this study. The Medical Scientific Research Foundation of Guangdong Province of China (Grant No. A2018006) and Project of Administration of Traditional Chinese Medicine of Guangdong Province of China (Grant No. 20191079) provided the animals, medicine, and other materials needed in the study.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
