Abstract
Objective:
The primary aim of the current study was to investigate the frequency of metabolic syndrome (MS) in early-stage breast cancer patients. Additionally, clinicopathological factors, such as anthropometric measurements and hormonotherapy, were examined for their roles as potential confounders of MS in these patients.
Patients and Methods:
In this retrospective cross-sectional study, all patients diagnosed with early breast cancer were included. Patients were divided into 2 groups with respect to MS diagnosis. Peripheral blood samples were obtained, clinical data were recorded, and body mass index (BMI) was calculated.
Results:
The study was completed with a total of 207 patients of which 128 (61.8%) had MS. MS was more frequent hormone receptor positive subgroup and in recipients of adjuvant hormonotherapy. The comparison of patients with and without MS revealed significant differences in age, BMI and estrogen/progesterone receptor status. There were no significant differences between groups in terms of cancer stage, inflammatory markers, basal insulin and LDL levels, and tumor markers.
Conclusion:
MS appears to be rather widespread among women with early-stage breast cancer, and lifestyle changes, which can improve obesity-related adverse outcomes, should be more emphasized in clinical practice.
Introduction
Metabolic syndrome (MS) is a combination of clinical pathologies and demonstrates a somewhat typical phenotype, including obesity of both central and abdominal origin, systemic hypertension, resistance to insulin (or type 2 diabetes mellitus), and atherogenic dyslipidemia. 1 It is a prothrombotic and proinflammatory condition characterized by an increase in cardiac and neurological disorders and has also been associated with various malignancies, especially breast cancer (BC). The most important common features in these conditions are abdominal obesity and increased visceral fat, which have been demonstrated to be independent risk factors for BC. 2 As a result of insulin resistance stemming from abdominal obesity, increased levels of extraglandular estrogen and insulin combined with decreased sex hormone-binding globulin and increased levels of estradiol, a mitogenic effect on mammary epithelial cells emerges. 3 Especially with aging and menopause, the increased frequency of MS in women has been accepted as one of the factors leading to an increased incidence of BC. 4
In the ORDET study, it was observed that the risk of BC was twice as high in patients diagnosed with MS. In addition, occult breast tumors were found in 40% of postmenopausal women followed for hormonal changes. There are studies which argue that MS cannot be evaluated as only a risk factor for BC since it also demonstrates prognostic features.5-7 Especially the concurrent presence of obesity and MS has been associated with an increase in BC-related mortality. All of these, as well as metabolic and inflammatory mechanisms, have been known to contribute to BC development and progression. Therefore, researchers have proposed that lifestyle changes minimizing the risk of MS and treatment of the components of MS might be effective in reducing BC risk. 8
The current study was planned because obesity, in particular abdominal obesity, had been noticed during the clinical follow-up of the patients early breast cancer (EBC). The primary aim of this study was to investigate the frequency of MS in BC patients to increase awareness of obesity as a risk factor. The secondary aim was to assess potential relationships between MS, biochemical variables and hormonotherapy in patients with breast cancer.
Patients and Methods
Stage I-III BC patients treated surgically and admitted to the Department of Medical Oncology, the Medical School of Mustafa Kemal University, Turkey, for adjuvant therapy between April 2014 and April 2020 were included in this retrospective study. All study procedures were conducted in compliance with the 1964 Helsinki Declaration and Ethical Standards. Mustafa Kemal University Institutional Ethics Committee approval (Number: 23-2020) were obtained prior to the study entry. Informed consent was obtained from all individual participants included in the study.
The blood pressure, height, weight, laboratory parameters and other comorbidities at the time of admission were recorded in the files of the patients. Waist circumference (WC) was collected and extracted from the pre-treatment medical records of the sample population at the initial presentation to the oncology outpatient clinic. These data were re-evaluated for the diagnosis of MS according to the NCEP ATP III (National Cholesterol Education Program Adult Treatment Panel III) criteria. The criteria of NCEP ATP III included hypertension (blood pressure ≥130/85 mmHg), hypertriglyceridemia (TG ≥ 150 mg/dL), fasting plasma glucose (FPG ≥110 mg/dL) or presence of diabetes mellitus, low HDL cholesterol (<40 and <50 mg/dL in men and women, respectively) and obesity of abdominal origin (waist circumference >102 and >88 cm in men and women, respectively). The patients who met at least 3 of the 5 criteria were diagnosed with MS. Patients who lacked records of relevant laboratory parameters, patients who were not evaluated by a dietitian, male breast cancer patients, and metastatic breast cancer patients were excluded from the study.
Data regarding demographics, comorbidities, oncologic staging, estrogen and progesterone receptor status, HER2/neu status, and the details of adjuvant endocrine therapy were obtained by questioning patients and reviewing patient files and hospital records.
The diagnosis of metabolic syndrome was made based on the measurement and laboratory results of the first admission to the oncology outpatient clinic with the diagnosis of breast cancer. During the evaluation of MS, serum insulin and tumor markers (CEA, CA15-3, CA125 levels) were conducted with the peripheral blood samples of patients using the Architect i2000 SR immunochemiluminescence assay (Abbott Diagnostics, USA), while the fibrinogen level was determined using the Coag L automated coagulometer (Diagon, Hungary). Serum glucose, cholesterol, triglyceride, HDL, and LDL levels were investigated using the Architect c8000 Chemistry System (Abbott Diagnostics, USA).
The neutrophil/lymphocyte ratio was calculated using the individual counts from peripheral blood. Patients’ weight (in kilograms) measured in light clothing and without shoes was divided by the square of height (in meters) using standardized instruments (Mewa Gmbtl/Schwerin-M318800) to calculate body mass index (BMI). The BMI classification of patients was made as determined previously by WHO using the following cut-off points: <18.5 kg/m2 for underweight, ≥18.5 to 24.9 kg/m2 for normal weight, ≥25.0 to 29.9 kg/m2 for overweight, and ≥30.0 kg/m2 for obesity.
Statistics
All statistical analyses were conducted using the SPSS version 22 (SPSS Inc., IBM, Chicago, IL, USA). The distribution normality of variables was checked using the one-sample Kolmogorov-Smirnov test. Mean ± standard deviation was used to express normally-distributed continuous data and these were compared by Student’s t-test, while the Wilcoxon rank-sum test was used when distributions were non-normal. Chi-square or Fisher’s exact tests were used to compare categorical variables that were expressed as ratios and percentages. P values throughout the study were 2-sided, and a value below .05 was considered to demonstrate statistical significance.
Results
In our study, 248 patients were evaluated. Thirty-two patients were excluded from the study because they did not have waist circumference measurements and 9 patients did not have lipid measurements. This retrospective study was completed with a total of 207 patients. The age of the patients ranged between 23 and 75 years. 54.6% (113) of the patients were postmenopausal.
The oncologic staging of the patients revealed that stage II BC was the most common (in 50.2% of patients), followed by stage III in 26.1% of the patients. In 79.3% of the patients, either the estrogen receptor or the progesterone receptor was positive with or without HER2 positivity. The frequency of patients who received adjuvant endocrine therapy was 79.3%. Anthropometric studies showed that mean height, weight, and waist circumference were 159 cm (range: 137-180), 77.5 kg (range: 45-134), and 102 cm (range:64-137) respectively. The BMI calculations showed that only 13% (n = 27) of the patients were within the normoweight definition, while the majority were either overweight (34.3%) or obese (52.7%). The details of anthropometric measurements and oncologic parameters are shown in Table 1.
Anthropometric Measurements and Oncologic Parameters.

Abbreviations: AI, Aromatase inhibitor; TMX, Tamoxifen.
We found that mean diagnostic age for BC in among patients with MS (53.2 ± 10.3 years) was significantly higher than that of BC patients without MS (48.2 ± 8.7 years) (P < .001). No significant difference was found in terms those with and without MS in terms of BC stage (P = .270).
When the relationships between the hormone receptor status in tumor tissue and the presence of MS were investigated, of the hormone receptor positive women; 43.9% (72) were premenopausal, and 56.1% (92) were postmenopausal. Of the hormone receptor negative women, 51.2% (22) were premenopausal and 48.8% (21) were postmenopausal (P: .492).
We observed that the frequency of MS was greater in ER or PR receptor positive (hormone receptor positive) subgroup compared to ER and PR receptor negative (hormone receptor negative) subgroup. The presence of metabolic syndrome was 59.8% in the hormone receptor positive subgroup, while the presence of metabolic syndrome was 30.2% in the hormone receptor negative group (P = .001). Waist circumference >88 was significantly higher in the hormone receptor positive subgroup than in the hormone receptor negative group (P ≤ .001). There was no significant difference between the 2 groups in terms of other parameters of the NCEP ATP III criteria. Table 2 show the distribution of metabolic syndrome parameters in all patients and by hormone receptor positivity.
Distribution of Metabolic Syndrome Parameters in All Patients and by ER and/or PR Receptor Positivity.
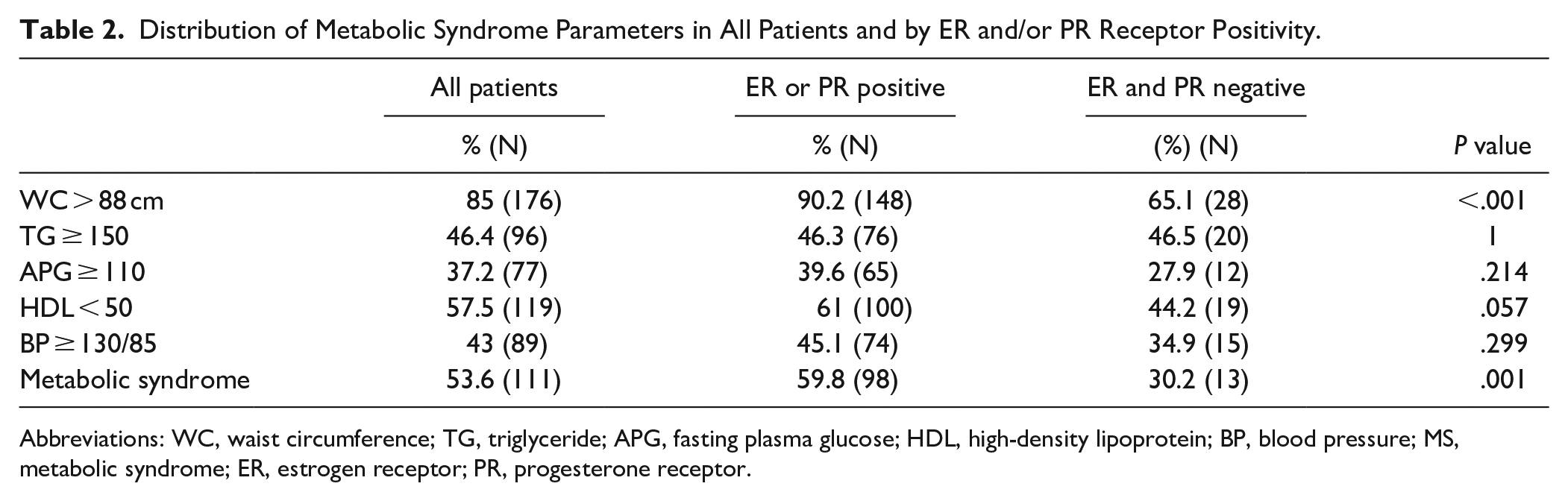
Abbreviations: WC, waist circumference; TG, triglyceride; APG, fasting plasma glucose; HDL, high-density lipoprotein; BP, blood pressure; MS, metabolic syndrome; ER, estrogen receptor; PR, progesterone receptor.
The clinical variables consisting of age at diagnosis, BC stage, hormone receptor status, and BMI classification were investigated by comparing patients with and without MS. We did not observe any significant relationships between BC stage, levels of inflammatory markers, basal insulin, tumor markers and the presence of MS. Table 3 show the distribution and comparison of clinical variables and laboratory parameters between patient groups with and without MS.
The Distribution of Laboratory Parameters in Patient Groups With and Without the Metabolic Syndrome.

Abbreviation: BMI, Body Mass Index.
Discussion
In our study, patients diagnosed with early-stage breast cancer were evaluated retrospectively. The frequency of MS was found to be significantly higher in the hormone receptor-positive disease subgroup.
According to different studies, the prevalence of MS in our country was found to be approximately 40% in women. 9 Sarici et al showed that 43.1% of patients undergoing surgery for BC had MS. 10 The incidence of BC has been found to be significantly increased, especially in postmenopausal women affected by the MS. 11 In the current study, the incidence of MS was 61.8% in all patients with BC.
BC can be considered as an entity with multiple distinct presentations that are due to constitutional molecular abnormalities, that is, Luminal A and B, human epidermal growth factor receptor 2 positive and Triple Negative (TN). 12 The reported data suggested that the potency of the MS and BC association changes according to the BC subtype. Hormone receptor positive tumors have greater associations with BMI. Capasso et al proposed that the possibility of an influence of MS was more on luminal type BC. 13 For premenopausal women, obesity has a protective effect on hormone receptor positive breast cancer but increases the risk of hormone receptor negative breast cancer. 14 For postmenopausal women, obesity is strongly associated with the risk of hormone receptor positive breast cancer but has a weak association with hormone receptor negative breast cancer.15,16 Obese patients’ adipocytes upregulate the expression of inflammatory factors such as fibroblasts, T cells, macrophages, leptin, TNF-α, IL-6, and IL-8 and decrease adiponectin. In the presence of obesity, estrogen levels increase in the body. The increased estrogen level is considered to be one of the mechanisms associated with breast cancer. Adipose tissue is the most important source of estrogen in postmenopausal Mediterranean women with a BMI above the acceptable range correlated positively with breast cancer risk. 17 Studies found that type 2 DM was strongly associated with hormone receptor positive breast cancer. 18 The main mechanism may be due to crosstalk between estrogen and the insulin/IGF-1 signaling pathway. The activation of ERα is influenced by the insulin/IGF-1 pathway, and estrogen levels may increase the expression and activity of the proteins in the pathways that promote signal transduction. 19
The impact of BMI solely has been demonstrated on Luminal A subtype, yet a slight tendency of association was present between Waist Circumference and insulin resistance and more aggressive BC types, such as HER2+ and Luminal B subtypes. 13 In our study, there was a significant relationship between MS and BC subtypes.
In 9 studies comprising a total of 6417 cancer patients which investigated relationships between MS and postmenopausal BC risk, a 52% increased risk for BC was found in the presence of MS. 20 In our study, the frequency of MS in BC patients was found to be more than the reported values in the literature.5,21 Wani et al exhibited higher prevalence of MS among women with breast cancer in postmenopausal compared to healthy women. 21 Metabolic syndrome is an insulin resistance syndrome, and several studies have implicated insulin in breast cancer development. 22 Insulin has a gonadotropic effect. Insulin stimulates the ovarian stroma for androgen production. Peripheral aromatization of androgen is the main source of estrogens after menopause. Insulin also increases aromatase activity. Abdominal fat is an important source of both androgens and estrogens. Increased hormone levels predispose to the formation of breast cancer.23-25
The prevalence of MS in the adult population of Western countries is estimated to be 20% to 25%. It is important to note that prevalence increases with advanced age, reaching 40% to 45% in people 50 years or older. 26 In accordance with the literature, we found a median diagnostic age of BC in the non-MS group of 48.2 ± 8.7 years, while it was 53.2 ± 10.3 years in the MS group. The risk of MS increases with aging; moreover, growing evidence shows the association of MS with both an increased incidence and poor prognosis of BC. 27 However, our study design did not include the investigation of prognosis as metastatic disease was one of the exclusion criteria for the study. Thus, no interpretations could be made for the disease prognosis.
Currently, there is enormous evidence that associates excessive body fat with increased risk for and poor prognosis of BC. 28 In a meta-analysis, a significantly worse prognosis of BC was associated with obesity in women. 29 Recently, obesity had been indicated as a negative and an independent factor for disease prognosis, that is, distant metastasis and mortality. 30 In our study, regardless of the menopausal status, the frequency of MS was increased with higher BMI.
Although there was no significant relationship between MS and BC stage in our study, it was noted that the incidence of MS was higher but non-significant in stage II and stage III patients compared to stage I (58.7%, 51.9%, and 44.9%, respectively) (P = .270).
Many cohort studies have shown a strong association of changes in glucose metabolism with cancer development. 31 The relationship between type 2 diabetes mellitus and BC is not fully understood. The common risk factors shared by type 2 diabetes and BC, such as obesity, sedentary lifestyle, excessive consumption of saturated fat and refined carbohydrates, add to the complexity of the relationship between the 2 diseases. 32 In our study, medical history revealed that 29.5% of all patients had type 2 diabetes, and FPG was found to be 110 and above in 37.2% of all patients and in 57.7% of MS patients. Although insulin levels were low patients without MS, the range was wide in the group with MS. We attributed this observation with the use of insulin-releasing drugs and insulin, both of which were not included in the study data.
Despite the strengths of the current study, some limitations should be taken into account when considering the results. First, although the menopausal status of the patients at the time of diagnosis was known, the menopausal status was not questioned again during the evaluation of MS. Our study group had a relatively higher proportion of patients with MS compared to the literature, but the study was designed for obesity and abdominal obesity in our region. Prospective and larger studies should be used to further validate and investigate our results.
Conclusion
In conclusion, the primary result of our study showed that MS is very common in patients with early-stage BC. The secondary result of our study demonstrated that BC was associated with the presence of MS, while further relationships were identified with respect to age, obesity, and adjuvant hormonotherapy. Thus, the current study provided important results that physicians could use when counseling patients with BC. Lifestyle changes, which can improve obesity-related adverse outcomes, should also be more emphasized in clinical practice.
Footnotes
Authors’ Contribution
M.R.D., S.M., and G.B. collected the patients data, A.O. and G.B. had done the statistical analysis and wrote the main manuscript text. All authors reviewed the manuscript.
Data Availability Statement
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Study was approved by Mustafa Kemal University Institutional Ethics Committee approval (Number: 23-2020) were obtained prior to the study entry.
Patients’ Consent
Informed consent was obtained from all individual participants included in the study.
