Abstract
Cancer-related fatigue (CRF) is a common and burdensome, often long-term side effect of cancer and its treatment. Many non-pharmacological treatments have been investigated as possible CRF therapies, including exercise, nutrition, health/psycho-education, and mind-body therapies. However, studies directly comparing the efficacy of these treatments in randomized controlled trials are lacking. To fill this gap, we conducted a parallel single blind randomized controlled pilot efficacy trial with women with CRF to directly compare the effects of Qigong (a form of mind-body intervention) (n = 11) to an intervention that combined strength and aerobic exercise, plant-based nutrition and health/psycho-education (n = 13) in a per protocol analysis. This design was chosen to determine the comparative efficacy of 2 non-pharmacologic interventions, with different physical demand intensities, in reducing the primary outcome measure of self-reported fatigue (FACIT “Additional Concerns” subscale). Both interventions showed a mean fatigue improvement of more than double the pre-established minimal clinically important difference of 3 (qigong: 7.068 ± 10.30, exercise/nutrition: 8.846 ± 12.001). Mixed effects ANOVA analysis of group × time interactions revealed a significant main effect of time, such that both groups significantly improved fatigue from pre- to post-treatment (F(1,22) = 11.898, P = .002, generalized eta squared effect size = 0.116) There was no significant difference between fatigue improvement between groups (independent samples t-test: P = .70 ), suggesting a potential equivalence or non-inferiority of interventions, which we could not definitively establish due to our small sample size. This study provides evidence from a small sample of n = 24 women with CRF that qigong improves fatigue similarly to exercise-nutrition courses. Qigong additionally significantly improved secondary measures of mood, emotion regulation, and stress, while exercise/nutrition significantly improved secondary measures of sleep/fatigue. These findings provide preliminary evidence for divergent mechanisms of fatigue improvement across interventions, with qigong providing a gentler and lower-intensity alternative to exercise/nutrition.
Introduction
Cancer-related fatigue (CRF) is a common and debilitating side effect of both cancer pathology and its treatment. As many as 45% of cancer survivors, regardless of cancer type, report moderate to severe fatigue years after treatment cessation. 1 Fatigue is considered more burdensome and disruptive to daily life than ongoing pain, nausea, and depression. 2 Unremitting fatigue further represents a major barrier to recovering a sense of autonomy and vitality following cancer treatment, and poses an increased risk of other poor health outcomes.3-5 As such, CRF represents a serious burden to both the patient and the healthcare system. Appropriate treatment of CRF is more pressing than ever as the incidence of cancer rises globally alongside improved survival rates of cancer patients.6,7
CRF is considered a multidimensional diagnosis, defined as “a distressing, persistent, subjective sense of physical, emotional, and/or cognitive tiredness or exhaustion related to cancer and/or cancer treatment that is not proportional to recent activity and interferes with usual functioning.” 3 Due to its inherently subjective nature, self-report is still the gold-standard method for measuring fatigue. 3 Many CRF patients report poorly managed fatigue symptoms that are often met with confusion and uncertainty by healthcare professionals.8-12 This complicates early identification and delivery of effective CRF treatments, further perpetuating the burden of CRF for the patient and health system.
While there is no gold standard treatment of CRF, 5 major categories of treatment have been consistently studied. These include: (1) Pharmaceuticals, 13 (2) Diet/nutrition,14-16 (3) Health/Psychoeducation approaches, 13 (4) Exercise,13,17 -19 and (5) Mind-body and meditative movement approaches.20 -23 Pharmaceuticals have shown limited efficacy in reducing fatigue, with the possible exception of stimulants such as methylphenidate, though effect sizes are considered small and inconclusive.13,24 Behavioral interventions and lifestyle modification such as diet/nutrition, health/psychoeducation, exercise, and mind-body approaches rather than pharmaceuticals currently show the best evidence for reducing fatigue. 13
Nutritional approaches have shown inconclusive results for reducing fatigue, though plant-based diets appear promising.14,15,19,25 However, long-term dietary changes can be difficult to accomplish and maintain when fatigued. 15 Psychoeducation has also been shown to be effective, either on its own or in combination with other exercise courses. 13 A recent meta-analysis indicates that exercise has the largest evidence base for fatigue reduction during and after cancer treatment. 24 However, there is considerable variation in the type of exercise utilized across these studies (eg, stretching vs strength training vs aerobic exercise). Additionally, standard exercise can also be prohibitively intense for a physically deconditioned cancer survivor struggling with fatigue, which limits adherence to exercise protocols in the short-term despite the long-term benefits.18,26,27 While providers often fail to adequately assess CRF in clinical settings,2,28 standard advice when any is offered usually includes some form of nutrition, psychoeducation, and exercise recommendations.13,15
Mind-body approaches, which include yoga,20,21,29,30 mindfulness,31,32 tai-chi and qigong,22,23,30,33,34 have also shown efficacy in reducing fatigue on meta-analysis and are increasingly studied for their multifaceted health effects across physical, emotional, and cognitive domains. Importantly, many of these practices include a gentle, low-intensity movement component which could recapitulate some of the salutary effects of standard exercise without requiring the same level of physical effort that can be difficult in a physically deconditioned cancer survivor. Non-Western mind-body movement traditions such as tai chi and qigong that engage the body in a specific form of gentle movement accompanied by a meditative form of attention/awareness are commonly referred to in the research literature as “meditative movement” practices. 35 The specific meditative movement practice of qigong has a rich history in Chinese medical traditions aimed at increasing a sense of vital energy, 36 and naturally incorporates movement with breath and meditative practice. Numerous different forms of qigong and related tai chi practice have been previously implemented in hospital settings and show promise for alleviating cancer-related fatigue and cancer-related symptoms.33,37,38 A recent review of 13 articles examining the efficacy of qigong for reducing cancer-related fatigue indicates that qigong has a small-to-medium efficacy for management of fatigue, but that many studies have been limited by bias or small sample size.22,39
While “standard” approaches to fatigue reduction emphasize adherence to a moderate-to-vigorous exercise intervention, it is increasingly understood that lower intensity exercise has been understudied but may be uniquely suited to a fatigued population. 40 Meditative movement practices such as tai chi or qigong may form an ideal version of low-intensity physical exercise that has added benefits for psychological and cognitive health due to the cultivation of meditative awareness. To this end, one recent study directly compared tai chi to standard moderate-to-vigorous exercise in older adults with cognitive decline and showed that both groups significantly improved cognitive function, 41 suggesting that such meditative movement practices can improve multiple health outcomes beyond physical function. A few studies in cancer survivors have directly compared qigong interventions to gentle non-meditative movements (eg, line dancing, light stretching, walking) in assessing multiple health outcomes including fatigue.42-45 However, to our knowledge none have directly compared qigong to a combination of the more “standard” fatigue interventions, including lifestyle changes in nutrition and exercise. Outcomes from such comparisons are needed to principally extend standard care to include the low-metabolic meditative movement practice of qigong.
To address this gap, in the current study we directly compare the meditative movement practice of qigong (n = 11) to a combined exercise/nutrition intervention (n = 13) over 10 weeks in female cancer survivors. We aimed to assess the magnitude of fatigue reduction generated by the very different physical intensity demands and explanatory frameworks for fatigue across the 2 courses. We report the results of a primary fatigue outcome endpoint as well as secondary analyses examining self-reported sleep, mood, stress, and quality of life. Though this study is underpowered to meet clinical trial significance, we provide evidence that CRF improved by similar amounts in both the qigong and exercise/nutrition interventions. The qigong group additionally reported significantly improved self-reported measures of mood, emotion regulation, and stress, while exercise/nutrition led to significant changes in sleep and fatigue measures. These findings may indicate both shared and divergent mechanisms of fatigue reduction in qigong versus exercise/nutrition, providing a basis for further investigation in future studies.
Methods
Study Design
This study was a single-blind, single-center randomized controlled pilot efficacy trial with a primary outcome of comparing changes in fatigue level in female cancer survivors randomized to 2 parallel 10-week movement-based intervention groups. CRF is common both during and after active cancer treatment, with different potential mechanisms underlying fatigue at these different stages. During treatment, immune and cytokine dysregulation resulting both from the cancer type and treatment (eg, chemotherapy, radiation) most likely interact with the central nervous system to produce the fatigue experience.24,46 After treatments have ended and the cancer pathology has been reduced or eliminated, central brain changes and complex psychological states may become more important in maintaining fatigue.24,46 In this study, we were specifically interested in assessing how different behavioral interventions would influence lingering post-treatment fatigue, regardless of cancer type or treatment. For that reason we included anyone with fatigue who had been out of treatment for at least 8 weeks (see Table 1, Inclusion/Exclusion criteria) to ensure that the fatigue was not potentially driven by the cancer’s activity or a response to ongoing treatments. With an average of 4.6 years since last treatment in our sample (see Table 2, Baseline Characteristics), it is unlikely that the fatigue measured in our study is a result of aberrant immune reactivity or signaling resulting from the specific type or stage of cancer or treatment regimen.
Eligibility Criteria.

Baseline Characteristics of Study Participants.

P-valuea denotes independent samples t-test for continuous variables.
All participants ranked their baseline fatigue as >3/10 in terms of its disruption of their normal daily activities or sleep quality. Participants were randomized to one of the following 2 intervention groups and all analyses presented include only those who completed treatment: (1) qigong (N = 11) and (2) the Pre-Train Exercise/Consolidated Health Improvement Program (CHIP) nutrition + lifestyle class (N = 13). Both classes met twice weekly for 2 hours and 15 minutes per class (total 4.5 hours in-class time per week) over 10 weeks. Participants engaged in two 3.5 hour testing sessions at 2 different time points: prior to the classes beginning (pre), and immediately after the classes concluded (post). This study was single-blinded as participants knew their assignment in order to actively engage with the class, but study staff working on randomization, data collection, and analysis were blind to group assignment. Enrollment began in June of 2017 and concluded in August 2017. All participants completed both testing sessions and their assigned class by December, 2017. The protocol was approved by the Lifespan Miriam Hospital Institutional Review Board, where the classes were conducted, on April 4, 2017 (#1040485). All participants gave written, informed consent prior to being randomized.
Participants (Table 1 for inclusion/exclusion criteria)
All participants were female cancer survivors (avg age 57.3 ± 9.0 years) who completed cancer treatment (including surgery, radiation, and/or chemotherapy) at least 8 weeks prior to the initial phone screening. All participants met study eligibility criteria (see Table 1).
Procedure
Intervention classes began in September and ended in November 2017. A variety of recruitment methods were utilized including posting flyers, placing advertisements in local radio shows and community magazines, outreach to local oncologists, oncology nurses, cancer support groups, and local organizations, and hosting stands at local events like parades and baseball games. Interested women called in, where an initial phone screen was conducted to describe the study’s goals, methods, and time requirements and to screen for eligibility. If eligibility was satisfied, the participant was scheduled for an in-person consent session, during which the participant was informed about both the qigong class and the exercise/nutrition healthy living class and told that they would be randomly assigned to either class. They were asked to indicate that they were willing to be randomized to either class and didn’t have a strong expectation of which class they would be in. If the participant consented to join the study, they were then scheduled for an initial 3.5 hours testing session.
Forty-eight hours prior to a scheduled testing session, the participant was emailed the packet of questionnaires to fill out online via REDCAP before their session (~45 minutes). If participants were not able to fill out questionnaires online, they were asked to come to their testing session 45 minutes early to do so then. Testing sessions included multiple assessments in the allotted 3.5 hours, including measures of back, leg and arm strength, a 6 minutes walk test, and venipuncture. Some participants also completed an EEG, EMG, ECG, laser doppler flowmetry, working memory task, and electrodermal skin conductance measures; all measures other than the questionnaires are beyond the scope of the current study. All testing sessions took place at the hospital clinic where the classes were conducted.
Randomization and Blinding
We generated a 1:1 computer-based randomization scheme using the blockrand package in R. The randomization was stratified by cancer recurrence versus no recurrence, to ensure an equal number of participants who had experienced multiple episodes of cancer and its treatment were assigned to each group. Therefore, for each stratum (cancer recurrence vs no recurrence), subjects were randomized to one of the 2 treatment groups within sequential blocks with random block sizes of 2, 4, and 6 using the blockrand function. Random block sizes were used to prevent treatment allocation anticipation. A pdf of randomization cards was created based on the output from blockrand using the plotblockrand function. To ensure blinding of the study personnel to group assignment despite having limited personnel, a data scientist generated multiple possible randomization sequences. A research assistant not otherwise involved in this study then randomly selected one of the sequences, and printed the randomization cards and sealed them in envelopes. At the completion of the baseline testing session for each new subject, the next envelope was opened and the subject was assigned to the corresponding treatment. Participants were not randomized to their intervention group until the end of their first testing session to ensure that both research assistants and participants were blind to their intervention group at baseline testing. Participants started their assigned intervention within 3 weeks of completing the baseline assessment.
Intervention Groups
The primary objective of the study was to compare the efficacy of qigong meditative movement to a standard healthy living program of exercise and plant-based nutritional/health education (described below) in reducing fatigue.
Both intervention groups met for a total of 4.5 hours per week (2 classes/week). Both intervention groups were asked to practice the movement-based component of class at least 30 minutes a day on days without classes. Both a morning and an evening class were offered to accommodate participants’ schedules for both intervention groups.
Qigong intervention
The qigong class was assembled and taught by an internationally recognized qigong instructor with 40 years of experience in teaching qigong in both China and the US. The focus of the class was on the qigong/Chinese medicine principles of “Nourishing Life” (Yang Sheng) and drew heavily from the Hun Yun system of qigong. Practices were selected by the instructor specifically for fatigued cancer survivors because of their prior use in China in improving energy levels and physical functioning in cancer patients. Movements were gentle, rhythmic, and repetitive and began with seated meditation, and progressed to standing movements in place. Gentle swinging of the arms and bending of the legs was the main source of exertion. A meditative focus was cultivated throughout as participants were instructed to focus on their breath and softening their muscles throughout each posture. Throughout the first 6 weeks of class, new qigong movements were sequentially introduced working up to a final sequence of 15 movements. The last 4 weeks of class were spent solidifying and refining the full sequence. Each class was roughly allocated into a first hour of learning a new movement, a relevant topic and discussion for the day about the purpose of the movement and related qigong principles of healing and energy cultivation. After a 15 minutes break, the second hour was spent practicing the sequence and answering any practice-related questions. Participants were given instruction sheets for each movement as it was introduced to facilitate home practice.
Exercise/Nutrition Group: Pre-Train Pilates & Complete Health Improvement (CHIP) Intervention (Pre-Train/CHIP)
This intervention group combined 2 pre-existing standard healthy living-focused classes incorporating both physical exercise and general health and nutrition education. Because the focus of the study was to examine how qigong compared to standardized health advice offered by physicians to combat fatigue—mainly exercise, dietary changes, and health education - this course included components of all 3. For the first hour of each class, participants were led in a program called Pre-Train, a Pilates-like exercise program designed by a local chiropractor to help improve core and overall strength. All participants were given a manual of exercises and asked to practice on their own at home 5 days a week for 30 minutes. Practices progressed throughout the 10 weeks to include both resistance and aerobic exercise. After a 15 minute break, the second hour followed the well-validated Complete Health Improvement (CHIP) program,47,48 a standardized nutrition and healthy living educational program which has been shown to improve multiple chronic disease metrics in patients for over 25 years. This hour consisted of instructional videos, and discussions led by a licensed hospital nutritionist. Participants received a manual and cookbook on plant-based eating and general health principles, and were encouraged to incorporate as many plant based foods into their diet as possible. Any differences in preferential engagement with either the nutrition or the exercise components of this course were not tracked in this study, but given that every class included 1 hour of physical exercise we know that attending class alone would result in 2 hours of weekly physical exercise per week even if no effort was made to engage in the nutritional or exercise recommendations outside of class. There was no discussion of any aspect of breathing or meditative focus in any part of this course.
Comparison of Qigong and Exercise/Nutrition groups
Each class in both the qigong and exercise/nutrition groups spent approximately 1 hour doing physical movement and approximately 1 hour in didactic learning and discussion. The main differences between the groups were the different physical demand intensities between the groups and differing explanatory frameworks for fatigue reduction. The qigong course consisted of 1 hour of gentle, low-intensity meditative movements while the exercise-nutrition group consisted of 1 hour of cardio and strength training with much higher physical intensity demands. During the hour of didactic learning and discussion, the qigong group focused on understanding fatigue in terms of regulating subtle energy (“qi”) in a traditional Chinese medicine framework, while the exercise/nutrition group focused on understanding fatigue reduction by improving metabolic and physical health through plant-based nutrition, exercise, and healthy lifestyle modifications in a standard biomedical model.
Both classes were matched in the total amount of in-class time per week, the number of classes offered per week, the use of expert instructors, the same physical location, the provision of materials for reference outside of class, and the supportive group setting.
Questionnaires
Baseline and follow-up testing sessions were conducted at the same hospital where the interventions took place. The online REDCap system was used to collect responses to all questionnaires as well as demographic information and background information related to age, race/ethnicity, socioeconomic status, and cancer history (see Table 2).
Primary outcome measures
The primary outcome measure was fatigue reduction. The Functional Assessment of Cancer Therapy-Fatigue (FACIT-F) is 41-item scale that measures overall well-being and health-related quality-of-life (QOL) in patients with cancer and cancer-survivors to determine frequency in the past week of statements concerning physical, emotional, social, and functional well-being in addition to fatigue. The “Additional Concerns” subscale was utilized as the primary outcome measure because it directly assesses fatigue using 13/41 items on the questionnaire. This fatigue scale was selected as the primary outcome measure given its widespread use for measuring fatigue in cancer populations and cancer studies, and because it has previously shown good reliability and validity in cancer populations. 49 For all scales, a higher score indicates improvement. 50
Secondary outcome measures
Eleven additional questionnaires were administered to assess different aspects of fatigue experience and intervention-related improvements. The wide range of questionnaires was selected based on commonly assessed scales that capture multidimensional aspects important in cancer survivorship, including changes in sleep, emotion regulation, mood, stress, and social support. Given the preliminary nature of this study, we included this range of questionnaires to help establish which commonly used scales were most responsive to change between the 2 groups. Information about the reliability and validity of the respective questionnaires can be found in the associated citation. These include:
Pittsburgh Sleep Quality Index (PSQI): The PSQI is a 9-item questionnaire that assesses sleep habits and quality over the last month. It includes different subscale measures of (1) subjective sleep quality, (2) sleep latency, (3) sleep duration, (4) habitual sleep efficiency, (5) sleep disturbances, (6) sleep medication, (7) daytime dysfunction as well as a (8) global score. Each subscale quantifies a different form of sleep-related dysfunction. Daytime dysfunction refers to difficulties experienced during the day as a result of poor sleep. A global score greater than 5 indicates poor sleep quality. 51
Fatigue Symptom Inventory (FSI): The Fatigue Symptom Inventory is a 14-item questionnaire that uses a Likert-style 1 to 11 scale to assess clinically meaningful fatigue, including (1) average fatigue, (2) fatigue interference, and (3) fatigue frequency and (4) severity in the last week. A higher score indicates worse fatigue. 52
Multidimensional assessment of interoceptive awareness (MAIA): The MAIA is a 32-item questionnaire that assesses the frequency of certain body sensations or body related thoughts in daily life using Likert-style items scored 0 to 5. It includes subscales relating to (1) noticing, (2) not distracting, (3) not worrying, (4) attention regulation, (5) emotional awareness, (6) self-regulation, (7) body awareness, and (8) trusting. A higher score is better. 53
Profile of Mood States (POMS): The POMS is a 65-item questionnaire that assesses mood “right now” using different words that are scored according to subscales assessing (1) tension, (2) depression, (3) anger, (4) fatigue, (5) confusion, (6) vigor, and (7) total mood disturbance on a Likert-style scale ranked 1 to 5. A higher score indicates a higher impact of that subscale. 54
Difficulties in Emotion Regulation Scale (DERS): The DERS is a 36-item questionnaire assessing general emotion regulation difficulties across 6 subscales including (1) non-acceptance of emotional responses; (2) difficulties engaging in goal directed behavior; (3) impulse control difficulties; (4) lack of emotional awareness; (5) limited access to emotion regulation strategies; (6) lack of emotional clarity. Each item is ranked Likert style from 1 to 5. Higher scores suggest greater difficulty with emotion regulation. 55
The Perceived Stress Scale (PSS): The PSS is a 10-item questionnaire that assesses thoughts and feelings related to stress over the last month. Each item is ranked Likert style from 0 to 4. Higher scores indicate more stress. 56
Multidimensional Scale of Perceived Social Support (MSPSS): The MSPSS is a 12-item questionnaire that assesses the amount of support someone feels from their friends, family, and significant others. Items are ranked likert style from 1 to 7. A higher score indicates greater social support. 57
Unmitigated Communion (UC): The UC questionnaire consists of 9 items ranked Likert style from 1 to 5 that assess the tendency to attend to and care for others to the exclusion of the self. A higher score indicates a greater tendency to care for others over one’s self.58,59
Brief Resilience Scale (BRS): the BRS is a 6-item questionnaire that examines the ability to bounce back after a stressor. Items are ranked Likert style from 1 to 5. A higher score indicates greater resilience. 60
Patient Health Questionnaire (PHQ): The PHQ is a 59 item questionnaire that assesses someone’s physical and psychological symptoms over the past week using a Likert-style scoring of 1 to 3 for each item. The PHQ-9 is a subscale of 9 questions that assesses depression symptom severity. The PHQ-15 is a subscale of 15 questions that assesses severity of somatic symptoms. A higher score indicates a greater severity of symptoms. 61
Short-Form-36 (SF-36): The SF-36 is a 36-item questionnaire that assesses perceived general health and vitality in the last month as well as expectations for health. Items are ranked with radio buttons from 0 to 100. It consists of 8 subscales including: (1) physical functioning, (2) role limitations due to physical health, (3) role limitations due to emotional health, (4) vitality (energy/fatigue), (5) emotional well-being, (6) social functioning, (7) pain, (8) general health. Higher scores indicate better health status. 62
Statistical Analysis
Our primary analysis was per-protocol, meaning we only analyzed participants who completed their assigned intervention. A traditional intent-to-treat (ITT) analysis was beyond the point of the study if the participants didn’t complete intervention, as the goal was to assess the treatment effect of the interventions in this pilot efficacy trial. 63 Further, the majority of people who dropped out declined to return for the second testing session making it difficult to conduct an ITT analysis (only n = 2 who dropped out during the intervention completed follow-up questionnaires, see Figure 1). The pre-specified primary outcome measure was fatigue reduction (see 2.6.1).

CONSORT diagram.
Sample size determination: Power analysis and effect size
The power analysis to determine sample size was completed for the FACIT-F “Additional Concerns” Fatigue subscale as the primary outcome measure. We estimated the sample size needed to achieve 80% power at the 2-tailed .05 significance level as 66 participants completing the study (33 in each group) with an effect size of 2.5 and standard deviation of 9, using a non-inferiority margin of 3. The effect size of 2.5 and standard deviation of 9 for treatment induced changes in fatigue were estimated based on the literature. 42 The non-inferiority margin of 3 represents the minimal clinically important difference in the FACIT-F score established in previous research. 64 Power analysis was performed in R using the package Trial Size (TwoSampleMean.NIS function). Though we planned to do a non-inferiority analysis to more conclusively establish that the improvement of fatigue from qigong practice is statistically not worse than improvement from exercise/nutrition, the low power from our small sample size per group prevented conducting this analysis in a meaningful way. We report the standard deviation and confidence intervals for all measures in Table 3 to inform other researchers in their estimation of sample sizes for future research with these measures/interventions.
Summary of Questionnaire Subscales that Showed Significant Main Effect of Time on ANOVA.

Summary statistics for each group at each time point that showed a significant main effect of Time on ANOVA (see Supplemental Table S1). T0 = Baseline (Pre-intervention), T1 = Post-intervention, T0-T1 = Change score between baseline and post-intervention. All P-values are reported as uncorrected. Any P-Value that is less than .05 is bolded in the table. If the uncorrected P-value was P < .05 it is denoted as*U. Any Uncorrected P < .05 is reported with an additional holm-corrected adjusted P-value that is denoted asH. If the holm-corrected value is <.05 it is denoted as*H. Anything that is significant at the P < .01 level is denoted with**.
Statistical tests
We used mixed-effects ANOVA to assess outcome measures, along with post-hoc Holm-corrected t-tests for significant differences in ANOVA outcomes. We used Holm correction to account for multiple comparisons because it is less conservative than Bonferroni and preserves more power in our already underpowered sample. 65 Post-hoc analyses for ANOVA measures that were significant at the Holm-corrected P-value below our pre-specified alpha of .05 were carried out using paired and independent samples t-tests for continuous variables. All P-values are reported as 2-sided tests. We report generalized eta squared effect sizes for significant ANOVA measures as this as been shown to be optimal for mixed designs, 66 computed as ηG 2 = SSeffect / ( δ × SS effect + ∑measuredSSmeasured)66,67. We report Cohen’s D effect size for the t-test analyses of the primary outcome measure, computed as d = Mean(X1) – Mean( X2)/pooled SD. 68 This approach of conducting Holm-corrected post-hoc analyses only on scales that first showed significant change in ANOVA was adopted in order to minimize the alpha inflation level introduced by the large number of questionnaires assessed in our study, many of which may not be fully independent of each other. Given our small sample size and the potential correlation between many of these measures, we further decided against running correlations or regressions between variables and rather report the baseline and follow up descriptive and inferential statistics in Table 3. For missing data in questionnaires, we followed the protocol of the designated questionnaire for correct handling of missing data. A custom R script was used to analyze questionnaire data.
Results
Demographics/Baseline Characteristics of Participants
Demographics, baseline characteristics, and cancer history for the overall participants and broken down into groups are listed in Table 2. Participant recruitment is detailed in Figure 1, with 41 out of the initial 72 eligible participants randomized to the intervention groups (Figure 1). There were no statistically significant differences between groups for any of the continuous variables. The class retention rate was 62% for the qigong group, and 65% for the exercise/nutrition group (Pre-train/CHIP), which was similar to prior studies with 8 to 10 week behavioral interventions.44,69 Additionally, there were no significant group differences in any of the questionnaires at baseline (Table 3).
The Primary Outcome Fatigue Measure Improved in Both Qigong and Pre-Train/CHIP Interventions, With No Between-Group Difference
The primary endpoint of fatigue was measured with the FACIT-F “Additional Concerns” subscale and analyzed using a 2 (Group: qigong vs exercise/nutrition, between-subjects) × 2 (Time: pre vs post, within-subjects) mixed-effects ANOVA. The main effect of Time was significant, (F(1,22) = 11.898, P = .002), with a generalized eta squared effect size of .116 (med-large). The main effect of Group was not significant (F(1,22) = 1.915, P = .180). The interaction between Group × Time was also not significant (F(1,22) = 0.149, P = .704).
Exploratory post-hoc pairwise comparisons reveal that both groups improved fatigue more than the predetermined minimal clinically important difference of 3 from pre-post intervention 64 : For the qigong group the mean improvement in fatigue was 7.068 ± 10.30, 95% CI [.148, 13.99] (t(10) = 2.28, Holm-adjusted p-value = .046, Cohen’s D = 0.686), while for the exercise/nutrition group the mean improvement was 8.846 ± 12.001, 95% CI [1.59, 16.11], (t(12) = 2.66, Holm-adjusted P-value = .042, Cohen’s D = 0.738) (see Table 3 for descriptive statistics including mean, median, SD, SE, CI, range, and IQR. See Figure 2 for a boxplot of individual fatigue change scores pre-post for each intervention). 73% of the qigong group experienced an improvement in fatigue of greater than 3 (n = 8/11) compared to 77% of the exercise/nutrition group (n = 10/13). This indicates that both groups had a clinically significant effect on fatigue improvement pre-post. Further, comparison of change scores across groups does not reveal a statistically significant difference in efficacy between groups (P-value between groups = .70, see Table 3). Our findings indicate that both groups changed in similar directions and magnitude from pre-post intervention, and we could not reject the null hypothesis that no difference exists between the two.
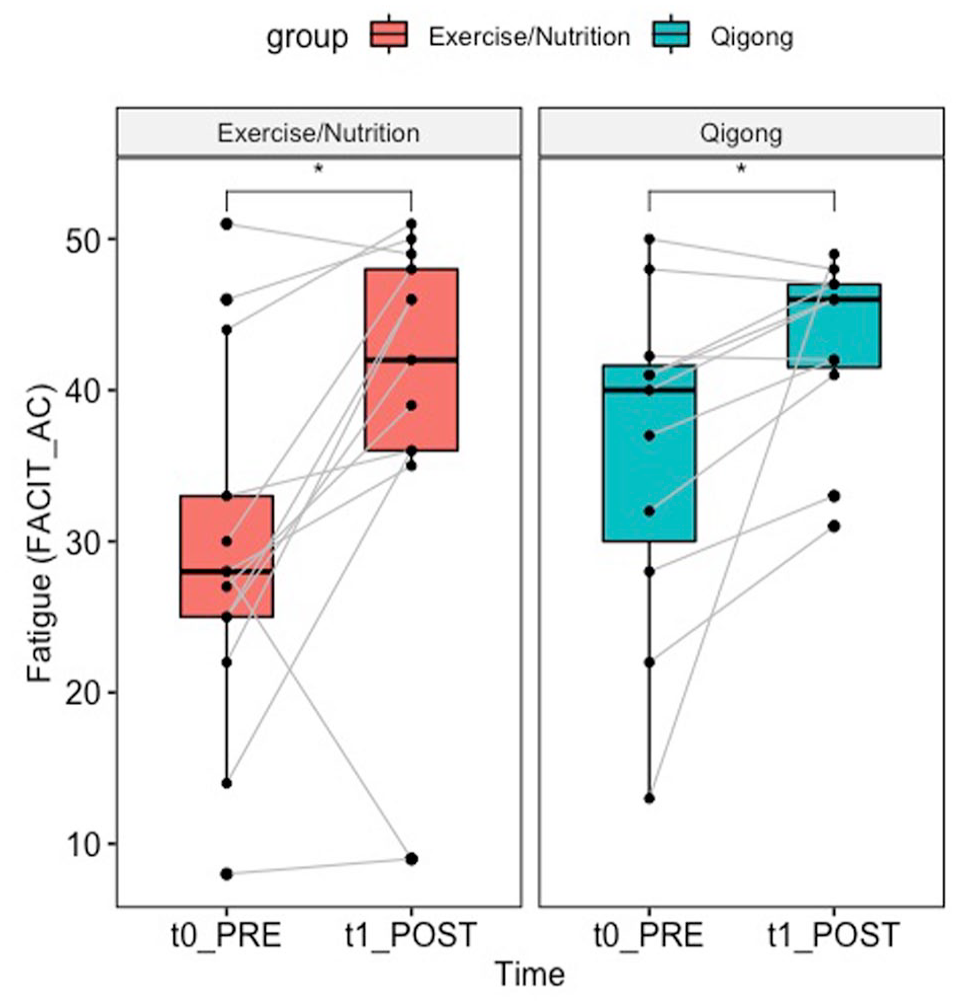
Boxplot of fatigue change by group. Results for fatigue change from pre-post for the primary outcome measure of fatigue, measured by the FACIT “Additional Concerns” Subscale. The change from pre-post for each subject is denoted by the gray lines. *Indicates a significant difference at the holm-corrected P < .05 level on paired t-tests.
Secondary Outcome Measures
Given the large quantity of questionnaires assessed, many of which contained multiple independent subscales, we conducted similar secondary analyses as described above for each questionnaire. Many of the scales measuring various quality of life, fatigue, sleep, functional, emotional, social, and stress related outcomes revealed significant main effects of “Time” changes in 2 × 2 mixed-effects ANOVA indicating improvement from pre-post across subjects regardless of intervention group. However, none of them showed any significant Group × Time interactions. Only 2, the DERS “Clarity” subscale and the SF-36 “emotional limitations” subscale, had a significant main effect for Group in addition to Time (See Supplemental Table S1). We provide a list of those scales with significant time effects along with the generalized eta squared effect size for each (Supplemental Table S1).
Post-hoc exploratory analyses
In service of further exploration of the numerous questionnaires collected, we conducted a post-hoc exploratory analysis of secondary outcome measures that had a significant “Time” effect (or “Group” effect) on ANOVA similarly to the primary outcome measure to establish if the significant time effect on ANOVA appeared to be statistically significantly present in both groups or primarily in one group. The descriptive statistics of each scale, per group, at each time point as well as the change scores (including mean, median, standard deviation, standard error, range, 95% confidence interval, and interquartile range) are provided in Table 3 for those scales that had a significant main effect of “Time” on ANOVA (Supplemental Table S1).
Though the post-hoc pairwise comparisons were exploratory given the lack of significant interaction terms on ANOVA, 2 patterns of note did emerge in pairwise t-tests of the change scores by group that suggest possible differences between the groups that should be tested in future studies (see Table 3). First, we note changes in sleep and fatigue that appeared to be driven more by the exercise/nutrition group than the qigong group. While the significant main effect of time on ANOVA indicated that both groups improved pre-post, the t-tests of change scores across secondary fatigue and sleep outcomes indicated a significant difference mainly for the exercise-nutrition group (see Table 3 for significant changes: FSI Avg Severity, FSI # of days fatigued, PSQI subjective sleep quality, PSQI daytime dysfunction. For trends toward significance see: FSI composite, PSQI global). Second, we note significant changes in emotionality, mood, emotion regulation, and stress pre-post primarily in the qigong group alone (see Table 3 for Significant Changes: POMS confusion, POMS fatigue, POMS tension, DERS Goals, DERS Impulse, DERS Total. For Trends toward significant changes see: POMS total mood disturbance, DERS Clarity, DERS Nonacceptance, PSS, PHQ-9). Both groups significantly changed pre-post on multiple measures related to overall physical sensations (see Table 3 for significant changes: MAIA “Body listening,” PHQ-15, and SF36-Vitality. For trends toward significance see: FACIT-F, SF-36 emotional well-being). Though these changes appear specific to the different groups, it is important to keep in mind that there are nuances across measurement of questionnaires that assess similar constructs. For example, while scales on the Difficulties in Emotion Regulation Questionnaire and the Profile of Mood States were significantly different pre-post for the qigong group, the measurement of emotions via the SF-36 emotional well-being score showed a trend toward significant change in both groups (see Table 3). However, these findings may form a preliminary motivation for future studies to investigate further.
Discussion
In this single-blind, parallel randomized controlled pilot efficacy trial we primarily demonstrated that 10 weeks of both qigong and exercise/nutrition training (Pre-train/CHIP) significantly improved fatigue pre-post intervention. Both groups showed improved mean fatigue scores pre-post more than double the pre-established minimal clinically important difference of three 64 (see Figure 2), with over 70% of participants in each group reporting a difference of 3 or more. Our results indicate that the exercise/nutrition group demonstrated significant improvements in self-reported secondary measures of sleep and fatigue, while the qigong group showed significant improvements in secondary measures of emotional health and stress. The lack of statistical difference between groups on ANOVA suggests a possible equivalence of the interventions across many domains, but requires a higher-powered study to definitively assess. Establishing equivalence would have important clinical consequences, as qigong constitutes a gentler, lower-intensity movement practice than vigorous exercise, and as such may be an effective alternative for those struggling with exercise or could be incorporated into polymodal fatigue interventions to help build stamina required for exercise adherence. Here, we provide effect sizes and descriptive statistics from our sample to help facilitate such future studies.
Advantages of the Study Design and Intervention Groups Compared to Prior Studies
A primary contribution of this study’s design was the direct comparison of qigong to exercise/nutrition. While a few prior studies have compared qigong and exercise, such studies utilized either light stretching (1 hour class, 2x/weeks, 12 weeks) 42 or line dancing (mild-moderate intensity, 90 minutes, 1x/week, 8 weeks) 44 or light stretching/strength training designed to match the same intensity as the qigong group 45 as parallel classes to the qigong intervention. In contrast, our study required significantly more in-person instruction time for each group (2.25 hours classes, 2x/week, 10 weeks) and included an exercise/nutrition group which incorporated not only moderate intensity aerobic exercise, which has previously been demonstrated to improve fatigue, 17 but also moderate-high intensity resistance training. This combination of exercise alongside a plant- based diet and overall health counseling, all of which have also shown previous evidence in improving fatigue,13,15 constituted an incredibly strong intervention group to compare qigong against. The fact that the qigong group improved fatigue levels by a similar amount as the exercise/nutrition group is therefore novel and provides an important baseline for future investigation. It further lends support to the finding of a recent meta-analysis of qigong’s efficacy in fatigue reduction that qigong may constitute an effective alternative to exercise for improving fatigue in cancer survivors.22,39
An important aspect of the current study was the large time demands required for participation, which had a few notable effects. First, it impacted the dropout rates across both classes, as the majority of participants who dropped cited too much time as their primary reason (See Figure 1, CONSORT flow chart). However, the retention rates in this study were still similar to those of other trials.44,69 Second, those who completed had a fairly large improvement in fatigue regardless of group assignment, and this improvement could be in part due to the large amount of face-to-face instruction time (4.5 hours, regardless of group, per week). This is much higher than many comparable studies, which typically include interventions meeting for 1.5 to 2 hours total in person (eg, McQuade et al, 45 Larkey et al 34 ).
A critical component of behavioral interventions is the role of the teacher. Both intervention groups were taught by experts of their specific area: the qigong group was taught by an internationally recognized master with more than 40 years experience and the exercise/ nutrition group was taught by a combination of behavioral change experts with licensing in nutrition and chiropractic medicine. The use of expert teachers, the large amount of in-person instruction, and social support provided from the group environment were therefore balanced across groups.
Other Factors to Consider When Directly Comparing Qigong and Exercise/Nutrition
In our small sample the exercise/nutrition group had lower mean values at baseline than the qigong group for all measures shown to have significant pre- to post- treatment differences. However, due to the large variance across subjects, none of the measures were significantly different between the 2 groups at baseline. In our case, the exercise/nutrition group was more fatigued at baseline (had a lower mean value), which is notable in light of a recent finding that the magnitude of fatigue reduction was largest for those most fatigued at baseline. 39 This could indicate that if the qigong group had been as fatigued at baseline as the exercise/nutrition, the qigong intervention may have yielded more fatigue reduction. Additionally there was a high degree of variability seen in the improvement scores in both groups (see Figure 2, Table 3), which indicates that while some people greatly benefited from their assigned intervention, others in the same group did not experience the same change in fatigue. Given this high variability was evident across both groups, it may suggest that some people may have done better in the other group. It is also notable that the average age of the qigong group (60.8 years ±5.7) was higher than the exercise/nutrition group (54.2 years ±10.3), and while this was not significantly different it had a trend toward significance (P = .062). Future work is needed to understand if subpopulations of fatigued patients may be more responsive to qigong versus exercise/nutrition. However, efforts toward personalization may be beyond what is feasible in a randomized controlled trial.
A considerable aspect of the current study is that qigong, a practice that derives from Chinese medical/philosophical theory, was compared to standard exercise/nutrition practices predominant in Western culture. Women who were raised in the United States and had no active practice or knowledge of qigong were the main participants. As such, the women randomized to the qigong group may have had more to learn about the overall framework of qi and how it related to concepts of health improvement than those randomized to exercise/nutrition, which are both commonly accepted to improve health. Women in the exercise/nutrition group may therefore have had a stronger expectation of fatigue improvement than those in the qigong group. Accordingly, cultural background and expectation of fatigue relief may have played a role in early adoption of the classes, though it seems to have been surmounted given the similar improvements in fatigue across groups. This further suggests that qigong’s practices are efficacious for fatigue relief even in people who were not raised in geographic regions where qigong is culturally endemic and widely recognized. However, this may be driven largely by having a highly trained teacher who was able to contextualize and translate the practices across the specific cultural backgrounds of the participants. Future work is needed to more directly explore these components.
Possible Shared and Divergent Mechanisms of Fatigue reduction across Qigong Versus Exercise/Nutrition training
Findings in our secondary exploratory post-hoc analysis revealed significant pre-post changes for the qigong intervention in emotional distress, mood regulation, and perceived stress (see Table 3 for Significant Changes in Qigong group: POMS total mood disturbance, POMS confusion, POMS fatigue, POMS tension, DERS Goals, DERS Impulse, DERS Total and PHQ-9. For Trends toward significant changes see: DERS Clarity, DERS Nonacceptance, DERS Total, PSS, PHQ-9), while the exercise/nutrition intervention was associated with significant improvements pre-post in fatigue and sleep metrics (see Table 3 for significant changes in Exercise/Nutrition Group: FSI Avg Severity, FSI # of days fatigued, PSQI subjective sleep quality, PSQI daytime dysfunction. For trends toward significance see: FSI composite, PSQI subjective sleep quality, PSQI daytime dysfunction, PSQI global). Such findings could be indicative of divergent mechanisms related to primary outcome of fatigue reduction (See Table 3 FACIT Additional Concerns) or overall quality of life improvement between the 2 interventions, which requires further study.
The physically fatiguing aspects of the exercise/nutrition class required from muscular exertion and increases in cardiorespiratory and metabolic demands during class may be partly responsible for the improvements in sleep/fatigue that showed significant change in this class. 70 It has been suggested that exercise interventions may promote fatigue recovery by improving muscular strength and metabolic and oxygen usage by the muscles. 71 The incorporation of a plant-based diet in this exercise/nutrition class may further have facilitated fatigue reduction by improving nutritional status and nutrients available for muscular exertion and recovery. 16 While this combined intervention was effective for fatigue reduction in our sample, moderate-vigorous intensity exercise can be prohibitively intense. Future work should explore the respective contributions of the exercise compared to nutrition components of the course on fatigue reduction.
While the qigong class did not focus explicitly on any components of psychological health or mental health education, it was effective in improving these metrics. The general practice typically emphasizes an intentional focus on linking breath and bodily movement to circulate qi in the body, while maintaining a meditative awareness on sensations generated by the body during breath and movement. 72 As such, qigong practice naturally incorporates movement with breath and meditation practice, potentially fostering a strengthening of physical form alongside a reduction of stress and clarity of the mind. This potentially indicates a more affective or stress-based mechanism of fatigue reduction from qigong training. This interpretation is consistent with prior findings in qigong and tai chi studies showing their efficacy in improving mood regulation, depressive symptoms, and stress,22,23,69,73-80 and cognitive function. 41 Importantly, qigong/taiji movement alone without the specific mindset/attentional quality (eg, sham qigong) failed to improve fatigue as much as standard qigong/taiji at post-intervention and 3 months follow-up. 34 This suggests the specific type of awareness cultivated in qigong training in addition to the movement is important in fatigue reduction. Given that these were self-reported changes, future studies should pay attention to potentially divergent physiologic mechanisms by which qigong versus exercise/nutrition interventions may alleviate fatigue and investigate potential objective measures associated with such mechanisms.
Study Limitations and Future Trials
Though one strength of this study was its single-blind randomized study design, which is notably absent in many studies of qigong,22,76 several limitations could be improved upon in future trials. One limitation is that fatigue was only assessed before and after the intervention, and long-term follow-up could be important for potentially capturing divergence in responses as a function of time. Additionally, this study compared 2 parallel intervention groups without including a treatment-as-usual group or no-treatment control, which would have been useful for testing the magnitude of fatigue change between intervention versus no intervention but was not feasible due to recruitment limitations. Further, given that exercise, nutrition, and health/psychoeducation were combined in one group in this study, future studies could compare the separate components’ efficacy for fatigue reduction against qigong or other mind-body meditative movement interventions.
A significant limitation of this study was the level of drop-out in randomized participants which further decreased our already small sample size. Across the 2 interventions, participants’ most common reason for dropping out was the classes taking up too much time. Future goals in refining the intervention delivery should therefore focus on minimizing the level of in-person class time as much as possible, though this may have been an important factor in the overall fatigue reduction effect size in those who did complete the intervention. Future studies should explore the optimal time requirements in balancing subject drop out and fatigue reduction. Another reason for drop-out could have been the large number of study questionnaires included at the assessment points that may have felt burdensome to some participants. While the broad array of questionnaires allowed us to assess what types of health outcomes were most susceptible to change in the respectives classes, future studies should strive to minimize questionnaires assessed to ease participant burden. We provide the outcomes of all these questionnaires in Table 3 for better identification of which questionnaires may be most relevant to select in future studies. Given the small sample size of this study (n = 24), our results should be interpreted cautiously given that we are underpowered to detect a true effect. Nevertheless, we believe this data provides important preliminary data for approaching direct comparison of non-pharmacological interventions for CRF in a randomized controlled trial format.
Due to the preliminary pilot nature of the study and the loss-to-follow up of participants who dropped out, we were able to carry out only a per protocol analysis of those who completed the intervention. While this allows us to estimate the effect of adherence to the intervention, it potentially introduces levels of bias that undercut the randomization efforts and overestimates the effect size, so future studies should aim to include both per protocol and intent-to-treat analyses. 63 A major limitation of this paper is the small sample size, which makes us underpowered to conduct non-inferiority or equivalence analyses and further limits our power to detect true effects. This could also be resolved in future larger scale trials.
Conclusion
In this parallel, single-blind pilot randomized controlled trial we directly compared the efficacy of the meditative movement practice of qigong (n = 11) to a combined exercise/nutrition intervention (n = 13) for improving fatigue in female cancer survivors. We found that both qigong and exercise/nutrition improved fatigue over the 10 weeks of the intervention by more than double the pre-established minimal clinically important difference of 3, despite having very different physical demand intensities and explanatory frameworks for fatigue reduction. The qigong group additionally showed significantly improved self-reported measures of mood, emotion regulation, and stress. This study therefore provides preliminary data in a small sample size that qigong may constitute an effective clinical therapy for CRF. This may be especially useful for those experiencing CRF who prefer a gentler and lower-intensity alternative to commonly suggested exercise/nutrition treatments, or for those who are struggling with the psychological components of CRF. This should be directly tested in future higher-powered randomized controlled trials.
Supplemental Material
sj-docx-1-ict-10.1177_15347354231162584 – Supplemental material for A Randomized Controlled Pilot Trial Comparing Effects of Qigong and Exercise/Nutrition Training on Fatigue and Other Outcomes in Female Cancer Survivors
Supplemental material, sj-docx-1-ict-10.1177_15347354231162584 for A Randomized Controlled Pilot Trial Comparing Effects of Qigong and Exercise/Nutrition Training on Fatigue and Other Outcomes in Female Cancer Survivors by Chloe S. Zimmerman, Simona Temereanca, Dylan Daniels, Cooper Penner, Tariq Cannonier, Stephanie R. Jones and Catherine Kerr in Integrative Cancer Therapies
Footnotes
Acknowledgements
We wish to thank Camilla Moore, DC, ART, SFMA for designing and delivering the Pre-Train exercise components of the Exercise/Nutrition class. We thank Greg Salguiero, MS, RD, LDN and Katie Lester MS, RD, LDN, CIC for delivering the CHIP components of the Exercise/Nutrition class. We thank Harrison Moretz for developing and instructing the Qigong intervention. We wish to thank Brandan Cullen, Helen Ding, Nikisha Vaghjiani, Sophie Sandweiss, Sam Fredericks, Danielle Silva, Christopher Black, Juan Santoyo, Uday Agrawal, Arison Than and Blake Mandell for their contributions to the data collection aspects of this study. We wish to thank Sara Lazar, PhD for her professional input at different stages of the study. We especially thank all the participants in this trail who made this study possible and wish to extend our gratitude for their engagement and interest.
Data Availability Statement
Materials, deidentified data and analysis code are available by emailing the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a private donation from the Berkman-Landis Family Fund to Brown University PI’s CK and SJ. CZ was funded by NIH training grant 2T22MH020068-21 during the analysis and writing of the manuscript.
Human Welfare Statement
The protocol was approved by the Institutional Review Board of the Miriam Hospital in Providence RI.
Trial Registration Number
This clinical trial was registered at clinicaltrials.gov with NCT# 03259438.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
