Abstract
The ketogenic diet (KD) was initially used in 1920 for drug-resistant epileptic patients. From this point onward, ketogenic diets became a pivotal part of nutritional therapy research. To date, KD has shown therapeutic potential in many pathologies such as Alzheimer’s disease, Parkinson’s disease, autism, brain cancers, and multiple sclerosis. Although KD is now an adjuvant therapy for certain diseases, its effectiveness as an antitumor nutritional therapy is still an ongoing debate, especially in Neuroblastoma. Neuroblastoma is the most common extra-cranial solid tumor in children and is metastatic at initial presentation in more than half of the cases. Although Neuroblastoma can be managed by surgery, chemotherapy, immunotherapy, and radiotherapy, its 5-year survival rate in children remains below 40%. Earlier studies have proposed the ketogenic diet as a possible adjuvant therapy for patients undergoing treatment for Neuroblastoma. In this study, we seek to review the possible roles of KD in the treatment of Neuroblastoma.
Keywords
Background
Neuroblastoma (NB) is the most commonly occurring extra-cranial solid tumor in childhood. 1 It is responsible for approximately 6% to 10% of childhood malignancies in children under 15 and 15% of pediatric cancer deaths.2-4 Neuroblastoma has an average age of diagnosis of 19 months and is rarely diagnosed in patients older than 19 years. 5 The likelihood of NB metastasis at the time of presentation is as high as 50%. 6 NB tumor cells originate from primordial neural crest cells and sympathetic cells. 7 NB tumors develop in the fetal or postnatal life; 70% appear in the abdomen, 40% arise in the adrenal medulla, and 25% in the sympathetic ganglia and other sites.8,9
There are 2 staging systems for this disease: International Neuroblastoma Staging System (INSS) and International Neuroblastoma Risk Group (INRG). INSS classifies patients based on surgical resection findings while INRG classifies based on imaging features of the tumor before treatment. 10 Patients with NB are divided into 3 risk groups: low-, intermediate-, and high-risk. The high-risk patients compromise 50% of all new cases, for which very aggressive treatments should be performed. 5 Based on the stage of the disease, patient age, genetic abnormalities, tumor biology, risk group, and histological classifications, different treatments can be used either alone or in combination with each other, including surgery, chemotherapy, radiotherapy, autologous stem cell transplantation, immunotherapy, and novel targeted therapies. Despite the availability of these treatments, the chance of survival for children with high-risk NB is less than 50%; however, this number reaches 90% in patients with intermediate/low-risk NB.8,11-17 African-American and Native American patients are at a higher risk of unresponsiveness to treatment and are thus less likely to be cured. 18
Based on epidemiological analyses, lifestyle factors including diet play an important role in the initiation, promotion, and progression of cancers, bearing a direct effect on patient survival.19,20 NB is characterized by a marked reduction in aerobic energy metabolism. 21 Metabolic reprogramming at the cellular level has a direct effect on the energy demand and neoplastic transformation profile of cells. 22 For example, Warburg et al showed that tumor cells use more glucose as compared to other cells and convert glucose to lactate; they cannot do oxidative phosphorylation even when oxygen supply is sufficient, a phenomenon called the Warburg effect.23-26 On the contrary, the majority of tumor cells cannot use fatty acids or ketone bodies to produce energy even in abundance thereof.27-30 One exception for this fact is cancers with BRAF V600E mutation, whose growth is enhanced by ketones. 20 Therefore, every diet and nutrient that has a role in glucose metabolism, could potentially affect the physiology of NB cells and potentially be used for cancer therapy. 25 Another reason for using diet therapy is that most of the time the standard therapies (chemotherapy, surgery autologous hematopoietic stem-cell transplantation, and irradiation) cause some permanent side effects that develop over time, causing morbidity in NB survivors, later in their life. 21 These side effects include impaired pulmonary, auditory, cardiac, endocrine, psychological, cognitive, and nervous system function.31-34 Most patients with NB have difficulties in learning, memory, and motor skills. 35 To reduce these side effects, scientists have suggested using several special regimens along with the standard oncotherapy to make them more efficient and to reduce the time of exposure to standard therapies. 21 In this article, we will review the current literature on the role of the ketogenic diet in the treatment of NB.
The Ketogenic Diet and Its Therapeutic Potential
The ketogenic diet (KD) consists of high fat, medium protein, and very low carbohydrate. 36 Reduced carbohydrate consumption (below 50 g/day) depletes glucose reserves for both normal fat oxidation via the supply of oxaloacetate in the Krebs cycle and for the supply of glucose to the central nervous system (CNS). In an extended period of starvation (3.5 days+), the glucose energy consumption of the human brain reduces to 75% of control values while the remaining 25% is compensated from the overproduction of acetyl coenzyme A (CoA), a process called ketogenesis. 37 Ketogenesis is seen in prolonged fasting, type 1 diabetes mellitus, and high-fat/low-carbohydrate diets, producing high levels of ketone bodies (KBs), such as acetoacetate, β-hydroxybutyric acid, and acetone, predominantly from the liver mitochondrial matrix. 38
Diabetes mellitus type 1 induced ketosis is pathological, commonly known as diabetic ketoacidosis, and is characterized by elevated levels of blood glucose greater than 250 mg/dl, arterial pH less than 7.3, serum bicarbonate less than 15 mEq/l, and the presence of ketonemia or ketonuria. 39 Physiological ketosis occurs during starvation and intake of very-low-calorie ketogenic diets, producing a ketonemia that does not exceed 8 mmol/l (18 mg/dl) and with no change in pH. 38
The growing evidence of therapeutic benefits from physiological ketosis has led to the development of nutritional ketosis, best defined as a nutritionally induced metabolic ketotic state resulting in blood β-hydroxybutyrate concentrations of ≥0.5 mM. 40 KD was initially designed in 1920 for the treatment of epilepsy, removing the need for medications in certain instances. 41 The role of KD is also well established in the management of weight loss, cardiovascular disease, type 2 diabetes mellitus, central nervous system (CNS) pathologies such as Alzheimer’s disease, Parkinson’s disease, autism, and multiple sclerosis38,42-46 In recent years, the KD has been one of the suggested treatment regimens for CNS cancers including Neuroblastoma. In preclinical, clinical, and review studies KD has been reported to reduce tumor growth, increases survival rate, especially in low- and intermediate-risk NB, and enhances therapeutic effects, for example by increasing sensitivity of malignant glioma to radiation therapy and lung cancer to radio-chemotherapy.25,47-55 Although KD has been widely accepted as an effective therapy for epilepsy and other disorders, its mechanism of action remains an enigma, 56 requiring further exploration. Improving understanding of the mechanistic relations between cellular metabolism and growth control will eventually lead to better treatments for human cancer.
Mechanisms of Ketogenic Diet in Malignancies
The Warburg Effect
Two major metabolic pathways that provide energy for cells are glycolysis and oxidative phosphorylation (OXPHOS). 57 Glycolysis is the anaerobic and relatively inefficient way of ATP generation compared with OXPHOS. 58 Normal cells only use glycolysis in anaerobic conditions, while contrarily, malignant cells mainly depend on glycolysis, even in the presence of sufficient oxygen, leading to a state called “aerobic glycolysis.” This phenomenon, whereby cancer cells depend on glycolysis for ATP production, instead of OXPHOS, was first reported by Dr. Warburg in 1955 and is therefore known as the Warburg Effect. 23 Although not fully known, some types of cancer cells are thought to make use of aerobic glycolysis to allocate more metabolites toward the production of structural components for rapid cell growth, and as an adaptation method to decreased vascular supply in the early stages of cell development. 59 Moreover, some cancer-associated mutations have been found to enable cancer cells to acquire and metabolize nutrients in a manner conducive to proliferation rather than efficient ATP production. 60 These hypotheses illuminate the great need for aerobic glycolysis in cancer cell growth and proliferation, which in turn, denotes the great therapeutic potential of interventions targeting these glycolytic pathways in malignant cells, such as KD. The antitumor effects of KD emanate from its ability to create a tumor microenvironment deficient in glucose for glycolysis, forcing the cells to obtain their energy from fatty acid oxidation through OXPHOS. 21 This condition will kill or reduce the proliferation of NB cells since they are inefficient to use fatty acids for ATP production due to their respiratory chain deficiency compared to normal cells. 21
Angiogenesis Suppression
Earlier animal studies have found that KD directly or indirectly alters the expression of several proteins involved in malignant progression. These molecules include vascular endothelial growth factor receptor 2, matrix metalloproteinase-2, and vimentin. 61 Moreover, KD strengthens the anti-angiogenic effect of cyclophosphamide, which causes hypoxia in tumor cells. Normally, the carbonic anhydrase IX (CAIX) gene is expressed during hypoxia as a response to the low-oxygen environmental status, but KD combined with low-dose cyclophosphamide does not lead to such a response despite its angiogenesis inhibition. This is because KD reduces the expression of hypoxia-inducible factor-1 alpha (HIF-1α) and subsequently, the expression of CAIX, leading to reduction of the tumor microvasculature. 21 It has been shown that fasting can enhance therapeutic effects of chemotherapy at the same time reducing its side effects. Since KD induces a fasting-like state by ketosis in the body, it also enhance chemotherapeutic effects by the same mechanism. 21
Decreased Oxidative Damage in Normal Cells
In addition, KD reduces the expression of serine/threonine kinase Akt (Protein kinase B). Normally, Akt increases in human cancerous cells and leads to enhanced cell proliferation, more oncogenic mutation, and apoptosis inhibition.62,63 Akt stimulates reactive oxygen species (ROS) production and their accumulation; however, KD decreases ROS production while also increasing antioxidants in the blood. 36 Reduced ROS production by KD leads to a better mitochondrial function. 28 Cancerous cells are not able to use ketone bodies as their energy source, and since the amount of glucose in KD is low, these cells cannot produce energy. Also, low glucose levels associated with KD reduce the pentose phosphate pathway activity. 64 The pentose phosphate pathway generates NADPH for use in reductive biosynthesis reactions and reduced glutathione (GSH) generation; KD decreases this source of energy that is associated with increased generation of reactive oxygen species (ROS) and toxicity of cancerous cells. 64 Also, this energy depletion inhibits the cell cycle through the activation of the LKB1/AMPK pathway and the inhibition of the mTOR pathway. 64 Oxidative phosphorylation is the principal mechanism for energy production. This mechanism is interrupted in cancer cells due to their decreased mitochondrial efficacy. As a result, cancer cells try to reimburse this energy reduction by increasing glycolysis and activating the pentose phosphate pathway. Glycolysis produces energy from glucose, but the more important function of glycolysis is that most of the intermediates in this pathway are necessary for several biosynthesis pathways that are essential for cell proliferation. Thus, glucose is necessary for the production of proteins, nucleotides, and fatty acids, and KD decreases glycolysis by decreasing the availability of glucose.36,65,66
Decreased Insulin, IGF1 Signaling, and Plasma Amino Acids
Normal cells enter dormancy during starvation but cancer cells do not. This subjects cancer cells to severe catabolic undernourishment during starvation, making them more vulnerable to cancer treatment. A previous investigation on fasting showed reduced growth of acute lymphoblastic leukemia in animal models. 67 Moreover, a non-carbohydrate ketogenic diet (NCKD) has been shown to reduce insulin and insulin-like growth factor 1 (IGF1) in the blood and decrease the IGF1/IGF1 binding protein (IGFBP) ratio. IGF1 is essential for growth and thus, its reduction causes reduced tumor growth; also, increased IGFBP activates the pro-apoptotic pathway and leads to cancer cell apoptosis (Figure 1).55,68,69 KD may change the expression of genes related to oxidative stress and inflammation in tumoral cells, as such, the activity of ROS and expression of particular genes are altered to be within the ranges as for normal cells. 70

Overview of the mechanisms of KD in Neuroblastoma. KD inhibits cell proliferation, growth, and angiogenesis and promotes cell apoptosis by targeting specific genes and molecular pathways in neuroblastoma cells.
It is reported that KD decreases amino acids (AAs) in the plasma and within the tumor but a normal diet with low protein does not lead to this decrease. So, high ketone bodies maybe the reason for a decline in AAs. More precisely, KD leads to decreases in essential AAs and urea cycle metabolites but increases in serine, glycine, and glutamine plasma levels. KD also decreases AA catabolism, which could ultimately result in anti-cachexia effects. 21 Nevertheless, the molecular mechanisms of KD require further investigations. 71
Despite these potential advantages, KD is not an appropriate treatment option for all types of cancer. For example, it is reported that the proliferation of cancer cells increased in BRAF V600E-mutated melanoma cells with KD, while proliferation did not change in NRAS Q61K-mutated and wild-type melanoma cells by KD.20,72 Also, Liśkiewicz et al 73 showed that the proliferation of renal cancer cells increases with KD intervention in mice with tuberous sclerosis. It is also observed that KD intervention is not beneficial in renal cancers that have similar characteristics to Stauffer’s syndrome. 74
Types of Ketogenic Diets
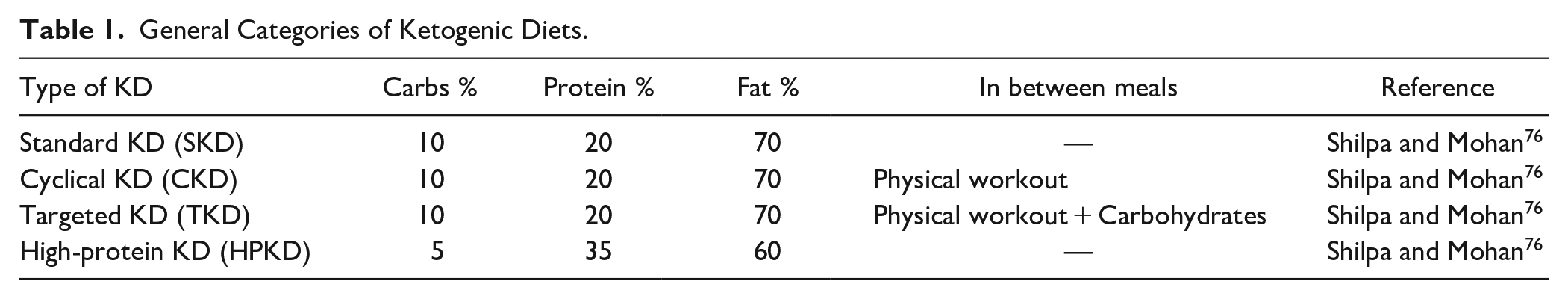
There are many types of ketogenic diets, with varying proportions of carbohydrates (carbs), fat, and protein. Typical KDs that are meant to induce nutritional ketosis, usually contain low carbohydrates below 50 g/day. 75 Standard ketogenic diets (SKD) have been widely used, most researched, highly recommended, and generally referred to as KD in this article unless/otherwise specified (Table 1).
General Categories of Ketogenic Diets.
Other Ketogenic Diets
Supplemented ketogenic diet
Recently, novel versions of the KD including the Mediterranean KD, the Paleolithic KD, the modified Atkins diet, and MCT-based KD have been investigated in the therapy of metabolic and chronic diseases.43,77,78 It is reported that ad libitum KD that is supplemented with 25% MCT8 (8-carbon medium-chain triglycerides) is as efficient as LCT (long-chain triglycerides)-based KD combined with calorie restriction in increasing the therapeutic effects of chemotherapy in NB and also, lowering the requisite dose of chemotherapy. 21 This murine study showed that MCT supplemented diet in comparison with LCT supplemented diet (with the same amount ratio) demonstrated a greater antitumor effect on NB. 21 Additionally, KD in combination with MCTs decreases both tumor growth and cachexia caused by the tumor. KD supplemented with omega-3/MCT delays and decreases gastric cancer proliferation. 21 Aminzadeh-Gohari et al 21 reported that a diet with 60% fat and 30% MCT added to low carbohydrate and medium protein imitates the effects of KD, decreases glioblastoma growth, and improves survival in mouse xenograft models just like normal KD (fat/carbohydrate plus protein ratio = 8). Another study showed that the 10-carbon MCT diet caused energy metabolism and mitochondrial functioning in SH-SY5Y cells. 79
Calorie-restricted ketogenic diet
Cancer patients experience cachexia as a result of cancer itself and also its treatments. Cachexia is a syndrome with symptoms of muscle and fat loss. 80 Cachexia is known to be a reason for morbidity and mortality during and after cancer treatment. Accordingly, calorie restriction is not a good option for some cancer patients. 21 Calorie-restricted KD (CR-KD) is a supplementary treatment for some brain cancers and NB; but, as mentioned above, it cannot be applied to all patients, especially those with cachexia. 21 KD decreases the catabolism of AAs, and enhance de novo amino acid synthesis of non-essential AAs, therefore it has anti-cachexia effects. 21 Studies have shown that KD without calorie restriction increases survival in animals and cancer patients who undergo dietary therapy.64,81,82 KD increases the chemotherapy effect but this increase is much more significant when it is combined with calorie restriction. 83 In this regard, metronomic cyclophosphamide (MCP) combined with CR-KD significantly decreases tumor size and inhibits the growth of NB xenografts. Studies also show that dietary intervention increases the sensitivity of tumor cells to the cytotoxic effects of therapeutic approaches.55,83-85 Nevertheless, although calorie-restricted KD can be helpful, more research is required.
The Ketogenic Diet as an Adjuvant Cancer Therapy: Clinical and Laboratory Studies
The monotherapy of KD and/or calorie restriction has been reported to significantly reduce tumor growth and prolong survival in preclinical studies of xenograft murine models.64,86 In a most recent murine study, the addition of a ketogenic diet to rodents with transplanted brain tumors increased survival in 9 of 11 animals to over 299 days compared to survival in untreated controls of 33 days and radiation only controls of 38 days. 87 Although KD monotherapy is a potential treatment option that can improve progression-free and overall survival for cancer patients, it may not be suitable for some cancer patients especially those with pre-existing cachexia, 21 therefore a combination of KD and other modalities is preferable.
Combination of Chemo-Radiotherapy and Ketogenic Diet
In the literature, KD has been used as an adjuvant treatment for chemotherapy, radiotherapy, and hyperbaric oxygen therapy. In a study by Allen et al, 48 KD has enhanced radio-chemo-therapy responses in lung cancer xenografts by a mechanism that may involve increased oxidative stress. An earlier phase 1 clinical trial has demonstrated that KD increases radiation sensitivity in a pancreatic cancer xenograft model, although locally advanced lung and pancreatic cancer patient receiving concurrent radiotherapy and chemotherapy showed suboptimal compliance to the oral ketogenic diet due to poor tolerance. 88 In a murine study, Aminzadeh-Gohari et al have reported that KD enhances metronomic cyclophosphamide therapy of neuroblastoma xenografts and invokes a robust anti-tumor effect. This combination significantly suppressed tumor growth, by reducing tumor blood-vessel density, intratumoral hemorrhage, and activation of AMP-activated protein kinase in Neuroblastoma cells. 21 In addition, Li et al 89 also reported that KD could increase the sensitivity of tumor cells to classic chemotherapy and radiotherapy when used in combination. This information suggests that KD can improve the efficacy of a multimodal treatment regimen of classic anti-NB therapy.
Integration of Ketogenic Diet and Immunotherapy
Immune checkpoint inhibitors have significantly improved the cancer treatment landscape in the past decade, becoming a first- and second-line modality across many tumor types, 90 and are even being tried in settings of immunosuppression. 91 The most successful immune checkpoint inhibitor antibodies are those targeting and inhibiting cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), programmed cell death 1 (PD-1), or programmed death-ligand 1 (PD-L1). 90 A recent investigation showed that in conditions where anti–PD-1 alone or in combination with anti–CTLA-4 failed to reduce tumor growth in mice receiving a standard diet, KD reestablished therapeutic responses. 92 Tumors cause overstimulation of the PD-1/L1 signaling pathway to reduce T cell activation, antigen-specific T cell immune response, and activate intrinsic cellular signals that enhance cancer cell survival and tumor resistance against pro-apoptotic stimuli such as interferons. 90 Mechanistically, KD prevents the immune checkpoint blockade–linked upregulation of PD-L1 on myeloid cells, while favoring the expansion of CXCR3+ T cells. 92 The energy stress induced by the ketogenic diet decreases PD-L1 protein expression on immune cells, therefore increasing their antigen-specific immune response against cancer. Conclusively, KD may enhances the efficacy of anti-CTLA-4 and anti–PD-1 immunotherapy against malignancies. 93
Ketogenic Diet and Other Nutritional Interventions
There has been an increasing interest in the potential benefits of adjuvant dietary interventions such as fasting, and KD in the past decade. Nutritional therapies target metabolic pathways of tumor cells and their tumor microenvironment hence inhibiting cancer progression. 94 Earlier research has demonstrated that metformin decreases the proliferation of breast cancer cells, 95 partly due to induction of fasting- and antifolate-mimicking modification of systemic host metabolism in breast cancer patients. 94 Metformin is a semi-synthetic oral hypoglycemic agent, used as a first-line oral treatment for type 2 diabetes mellitus, polycystic ovary syndrome, and adjuvant tuberculosis treatment, and has been linked with a decrease of cancer incidence, and cardiovascular diseases users. 96 A clinical trial is currently investigating the tolerability and effectiveness of KD in conjunction with metformin on glioblastoma patients (NCT04691960). In their in vitro and in vivo studies, Zhuang et al 84 observed that metformin-induced breast cancer cells’ cytotoxicity was enhanced when serum glucose levels were reduced via low carbohydrate ketogenic diets. Performing more metformin and KD pre/clinical trials for NB to explore their therapeutic potential is most likely to yield positive results.
Combination therapies of KD and other therapeutic nutritional interventions like (Vitamin D and Omega 3, 97 Genistein, 98 Vitamin A and E,99,100 Phytochemicals, 101 Camellia sinensis, 102 and Palmitic acid 103 ) could be investigated in the future. For example, the combination of high-dose vitamin D3 and the ketogenic diet has demonstrated negativization of HER2 expression and increased expression of the progesterone receptor, which can improve targeted therapy for breast cancer. 104
Effects of KD on survival in other Central Nervous System cancers
Central nervous system (CNS) tumors comprise 20% of all cancers observed in childhood, and are the most common solid malignancies in childhood, posing a significant pediatric cancer mortality. 105
In a mouse model of malignant glioma treatment with 4:1 KD, KD increased median survival by 22% from 23 to 28 days. Remarkably, treatment with KD given together with radiation resulted in complete tumor remission in 9/11 animals, for 101 days until they were sacrificed at 299 days. 55 Another case series by Smith et al 106 studied patients with glioma who received ketogenic metabolic therapy through dietary adherence and intermittent fasting obtained a mean (SD) progression-free survival 20.0 (14.4) months. A recent systematic review showed that KD offers a mean overall survival of 15.9 months. 107
An adult subject investigation by Klein et al 108 showed that treatment of glioblastoma patients with 4:1 KD using total meal replacement program with standardized recipes was well tolerated. These studies suggest the potential effectiveness, tolerability, and acceptable safety profile of KD among other central nervous system cancers, on top of Neuroblastoma.
Adverse Effects, Contraindications, and Issues of Concern When Using Ketogenic Diets
KD Cannot Be Used Against BRAFV600E Mutation-Positive Tumors
BRAF V600E mutation is a kinase-encoding gene from the RAS/RAF/MAPK pathway. 109 Earlier studies found out that KD aggravates human cancers by selectively enhancing BRAF V600E mutant-dependent MEK1 activation. 20 Another study reported that ketogenic enzyme 3-hydroxy-3-methylglutaryl-CoA lyase (HMGCL) expression is upregulated in BRAF V600E-expressing human primary melanoma and hairy cell leukemia cells which stimulate proliferation and tumor growth. 110 This limits the use of KD in malignancies with high rates of this mutation such as colorectal carcinoma, thyroid cancer, malignant melanoma, glioblastoma, and pleomorphic xanthoastrocytomas. 111 Although Neuroblastoma has a low frequency of this mutation, 2 cases were recently reported. 112
KD Cannot Be Used in Hypoglycemia Prone Patients
KD is a low carbohydrate diet, with high-fat, adequate protein, and calories. 82 This implies that the major source of glucose in a patient on KD is the liver’s gluconeogenesis. Therefore KD is contraindicated in patients with pancreatitis, liver failure, disorders of fat metabolism, primary carnitine deficiency, carnitine palmitoyltransferase deficiency, carnitine translocase deficiency, porphyrias, or pyruvate kinase deficiency. 113 Diabetic patients on insulin or oral hypoglycemic agents may suffer severe hypoglycemia if the medications are not appropriately adjusted before initiating this diet. 82
Side Effects of KD
The ketogenic diet is largely safe; however, several patients suffer minor side effects, especially in the short term (less than 2 years), sometimes referred to as keto flu. These include nausea, vomiting, headache, fatigue, dizziness, insomnia, difficulty in exercise tolerance, and constipation.113,114 These symptoms resolve in a few days to few weeks. Moreover, literature has reported hepatic steatosis, hypoproteinemia, kidney stones, gout, symptomatic hypoglycemia, and vitamin and mineral deficiencies as potential long-term adverse effects of KD.82,113 Renal cell carcinoma patients presenting with Stauffer’s syndrome, should not receive KDs because they potentially worsen the associated hepatic dysfunction. 74 To a greater extent, KDs are well tolerated especially when prescribed to the right candidates in right doses. KD, especially in the pediatric population, is a painless, readily available, tolerable, and complementary treatment strategy.115,116
KD and Cancer Cachexia
Cancer cachexia is an insidious syndrome of progressive weight loss, anorexia, and persistent erosion of host body cell mass in response to a malignant growth. 117 It is characterized by systemic inflammation, negative protein, and energy balance, an involuntary loss of lean body mass and is associated with poor responses to chemotherapy and decreased survival. 118 Although cachectin/tumor necrosis factor (TNF) has been implicated, the complete mediators and catabolic mechanisms of this common syndrome (>50% cancer patients) are not known.117,119 Under this background, dieting is of concern to cancer patients with or without weight loss, since KD significantly cuts out carbohydrates high in caloric value. A recent publication has reported that higher energy intake is necessary for cachexic patients treated for head and neck cancer to maintain skeletal muscle mass. 120 However, another study found KD to be more effective than insulin in reversing weight loss, reducing tumor size, and promoting weight gain. 121 We recommend further research on this matter.
Improvement of Ketogenic Diets by Managing Possible Side Effects
The classic 3:1 or 4:1 ketogenic diets have 3 weaknesses namely (1) they are complicated to prepare; (2) they are not palatable; and (3) they are prepared by individual patient caregivers, and thus diverges between patients in a study, and between studies. Innovative dietetics and nutritional methods are required to modify these parameters. For example Klein et al 108 developed a novel program of total meal replacement (TMR) of ready-made 4:1 and 3:1 KD meals to simplify preparation and standardize the diet across a studies and make it comparable between studies as well as improving adherence.
Considering the side effects that are associated with KD, several strategies have been recognized for the management of these side effects. For instance, symptomatic hypoglycemia can be treated by carbohydrate-containing beverages such as orange juice. 122 Gastrointestinal symptoms, which are not permanent, may need KD ratio reduction. 122 Potassium citrate administration also decreases the risk of kidney stone formation. 123 Using multivitamins and minerals such as calcium and vitamin D is recommended for mineral and vitamin deficiencies that are seen with KD administration. 124 Increasing the unsaturated to saturated fats ratio, decreasing KD ratio, using MCT oil, and also carnitine supplementation can reduce dyslipidemia risk. 125 Moreover, 3 measures can be used to maximize compliance and high ketosis in patients: (1) frequent individual counseling by a dietician experienced with KDs; (2) provision of ketogenic formulas and meals; (3) offering cooking classes. 126 Finally, patients should be closely monitored for KD side effects since these side effects can lead to the reduction or even discontinuation of KD. 127
Future Studies and Recommendations
Although KD may be used as a possible adjuvant therapy for Neuroblastoma, its therapeutic potential can be improved. Some of the strengths of KD which make it worth for further investigation are that KD is not expensive as traditional oncotherapy and has high safety profile and history of use in epilepsy management. Moreover, KD can be administered to both in and out-patients, as long as adherence is ascertained, otherwise the benefits will not be realized.
Understanding the mechanisms of action of KD at cellular and molecular levels is and will be paramount in future studies seeking improvement of its efficacy. For example, cancers with BRAF V600E mutation are worsened by KD, however, this knowledge can enable a potentially effective targeted therapy for this group of malignancies. A good example for this is vemurafenib (Zelboraf; Plexxikon/Roche), an inhibitor of the mutated serine-threonine kinase BRAF and BRAF-V600E kinase that was approved for the treatment of BRAF-mutated metastatic melanoma in the United States in August 2011 and the European Union in February 2012.128,129 Moreover, the coexistence of enhancer of zeste homolog 2 (EZH2) and BRAF gene aberrations has been described in many cancer types and offers an opportunity for combination therapy with EZH2 and BRAF inhibitors in malignancies with concurrent BRAF V600E mutation and EZH2 gain. 130 Research seeking understanding of molecular mechanisms of action KD in malignancies will also benefit prevention, early screening, diagnosis, treatment, and prognostic prediction of survival of cancer patients. Moreover, future studies should investigate the safety and efficacy of nutritional interventions for every single type/genetic subtype of cancer before they can be translated for clinical use. 94 Lastly, institutional collaborations in research should be fostered among different science disciplines and backgrounds including third world countries, which will improve retention of expert skills. 131
Conclusion
Recent advances in understanding the molecular and metabolic alterations in cancers have opened a new door for intensive research on therapeutic approaches such as KD. Ketogenic diets may enhance the antitumor effects of classic chemotherapy and radiotherapy with acceptable safety, tolerability, and quality of life. However, the cellular and molecular mechanisms underlying the anti-tumor effects of the ketogenic diet need further elucidation. Current knowledge strongly suggests that cellular metabolic modifications may be a highly effective therapeutic modality and may enhance the current standard of care for Neuroblastoma. Although the majority of studies support the use of the ketogenic diet in combination with standard therapies, more molecular studies, and randomized controlled trials are needed to investigate KD as a monotherapy and in combination with other nutritional therapies, immunotherapy, and novel targeted therapies. Finally, that KD is contraindicated in BRAFV600E mutation-positive tumors, and hypoglycemia-prone-patients like diabetes mellitus.
Footnotes
Abbreviations
NB: Neuroblastoma
KD: Ketogenic diet
INSS: International Neuroblastoma Staging System
INRG: International Neuroblastoma Risk Group
LOH: Loss of heterozygosity
MCT: Medium chain triglycerides
Author Contributions
RM, ZS, MK, SR, and NR designed the study; ZS and NK performed the literature search; RM and ZS drafted the manuscript; NK, AM, NR, MK, SR, and MM critically revised the manuscript; NK designed the figure; MM and NR supervised the study. All authors approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
