Abstract
Purpose:
Pegylated liposomal doxorubicin (PLD) is highly effective for treating multiple myeloma (MM). Hand-foot syndrome (HFS) is a dose-limiting adverse event of PLD that may reduce a patient’s quality of life or prevent certain patients from receiving PLD. Several researchers have discovered that pyridoxine, an activated form of vitamin B6, may prevent PLD-associated HFS. We designed a prospective randomized trial to examine whether prophylactic pyridoxine might prevent the incidence or delay the occurrence of PLD-induced HFS in patients with MM.
Methods:
Patients who met the trial’s eligibility requirements were randomized and then administered either pyridoxine 100 mg twice daily or no pyridoxine, in both cases accompanied by their PLD-containing chemotherapeutic agent. Follow-up of patients was performed until the completion of induction therapy, the development of HFS or disease progression.
Results:
Between January 1, 2017, and January 1, 2019, 105 patients were randomly assigned to the pyridoxine group (n = 52) or the no pyridoxine group (n = 53). In the pyridoxine and no pyridoxine groups, HFS developed after a median of 4 (range, 1-8 cycles) and 3 (range, 1-7 cycles) chemotherapeutic cycles, respectively. There were no grade 3 incidents recorded. Overall, 13.3% of patients experienced HFS. A 11 of 53 (20.8%) patients in the no pyridoxine group experienced HFS, compared to 3 of 52 (5.8%) patients in the pyridoxine group (P = .042); there was no difference in HFS grades (P = .725).
Conclusions:
The findings of benefit from prophylactic pyridoxine in this open-label trial have suggested its promise as a treatment for reducing HFS in MM patients. Further research with a placebo-controlled design is recommended.
Clinical Trial Registration:
ChiCTR2100050294.
Introduction
Multiple myeloma (MM) is a malignant aberration of plasma cells whose characteristics include an unregulated proliferation of the clonal plasma cells in the bone marrow. Among malignant hematological abnormalities, it is the second most prevalent disorder. The overall survival (OS) rates of MM patients have increased dramatically in the most recent decade, primarily due to the advent and advancement of innovative treatments. 1 Pegylated liposomal doxorubicin (PLD), a new version of an “old” drug, is effective in various cancers, and PLD-containing regimens are commonly used to treat MM in China. A liposome formulation incorporating polyethylene glycol (PLG) is used to deliver doxorubicin in the form of PLD. And PLD enhances the antitumor effect while simultaneously decreasing the harmful side effects on the myocardium compared with free doxorubicin. 2
However, unlike free doxorubicin, the use of PLD is associated with palmar-plantar erythrodysesthesia syndrome, commonly referred to as hand-foot syndrome (HFS). HFS is dose-limiting and is the most prevalent accumulated toxicity reported by patients undergoing PLD-containing chemotherapeutic agents; an occurrence rate of HFS induced by PLD varying from 18% to 49% has been reported in previous studies.3,4 HFS is characterized by dermatologic reactions that appear on the hand palms and/or the soles of the feet. 5 Clinical symptoms differ from person to person, and they generally progress to blistering, ulceration, desquamation, swelling, and pain. Even though HFS is not a life-threatening condition, the consequences of exposure to HFS might be severe. Even a minor case might well be accompanied by significant discomfort. It could progress to an unpleasant condition that inhibits functioning and negatively impacts patients’ quality of life (QoL). It may result in reductions in cancer therapy dosage, period, or intensity, which has the potential to alter treatment outcomes. When HFS can be avoided or reduced to a minimum level, it may not be necessary to reduce the PLD dose, thus enhancing the effectiveness of PLD and the prognostic status of MM patients receiving PLD-based treatment. 6 Because the pathophysiology of HFS has not been determined, a variety of methods have been tested for preventing or minimizing the disease occurrence rates.
Pyridoxine is a kind of activated vitamin B6, which has been suggested in many case series at various institutions for preventing PLD-induced HFS because HFS has been demonstrated to have a certain resemblance to the rodent disease acrodynia, which is induced by pyridoxine deficiency. Pyridoxine is inexpensive and relatively safe.7,8 Inconsistent findings have been reported from randomized studies on the prophylactic application of pyridoxine to protect against HFS. 9 The results of randomized, placebo-controlled studies of ovarian cancer patients and gastrointestinal cancer patients do not support the preventive use of pyridoxine. 5 Nevertheless, several case studies have shown that oral pyridoxine, at doses within the range of 50 to 800 mg daily, can be used successfully to manage a wide variety of medication-induced HFS. The different results in the previous studies may be related to the dosage of pyridoxine used, the type of disease, and the dosage of PLD used.10,11 However, in some case reports and clinical trials, it was found that pyridoxine 100 mg BID, is safe for the treatment and prevention of HFS, which was applied in relatively large samples. 5 There is no evidence that pyridoxine is efficacious for preventing PLD-associated HFS in MM patients. We conducted an open-label, randomized, prospective study to assess whether preventive pyridoxine (100 mg twice a day) could prevent HFS in MM patients receiving PLD (40 mg/m2 every 4 weeks).
Methods
Study Design
The First Affiliated Hospital of Sun Yat-sen University performed an open-label, randomized, prospective study investigating the clinical application of pyridoxine in the prevention of PLD-induced HFS in MM patients between January 1, 2017, and January 1, 2019. The Chinese Clinical Trials Registry received the registration of this trial (ChiCTR2100050294). The ethics committee approval number is [2017]126 (December 2016). The hospital’s clinical research and experimental animal ethics committees approved the study.
Patients
Before enrollment, all patients signed an informed consent form. Our study cohort consisted of newly diagnosed MM (NDMM) patients or relapsed/refractory MM (RRMM) who were set to begin induction therapy with PLD-based chemotherapy. Patients were treated at an initial PLD chemotherapy dosage of 40 mg/m2 every 4 weeks (weekly total divided into 2 days). The following criteria were used to determine inclusion eligibility: age above 18 years; performance status of 2 or less in the Eastern Cooperative Oncology Group (ECOG); willingness to participate; and no contraindication to chemotherapy (renal, cardiac, and liver functions; and adequate bone marrow). The patients who did not meet the inclusion criteria included those who had a previous history of treatment for HFS; those who were pregnant or lactating; those who were hypersensitive to pyridoxine; those who had been diagnosed with another type of cancer, a severe illness, or a medical problem; those who had a currently existing skin-related disorder negatively impacting the hands or feet, which might restrict the evaluation of the results; and those who had a background of hypersensitive responses to a traditional kind of doxorubicin or any of the constituents of PLD. Moreover, those patients with a history of cardiac disease or clinical symptoms of congestive heart failure were deemed ineligible for inclusion in the present research. Limb cooling and the usage of moisturizers were prohibited in our clinical trial. 12
Randomization
Patients who met the criteria were randomly classified into different categories at a ratio of 1:1, namely, PLD-based chemotherapy in the presence or absence of the administration of oral pyridoxine. The randomization sequence was created by an independent statistician using a computer program with a 1:1 allocation and it formed the basis for numbered sealed envelopes containing the randomization information. Trial participants, oncologists, data analysts, care providers, and outcome investigators had no access to the randomization sequence. The envelopes were opened only after the enrolled participants completed all baseline assessments and when it was time to allocate the intervention. After allocation, patients were not blinded to their group assignment, nor were their treating oncologists blinded, considering the study characteristics. The allocation sequence was blinded only to data analysts and outcome investigators. Our center had retrospectively analyzed unpublished data on 143 patients; 19.4% (21/108) had HFS in a group that received no pyridoxine and 5.5% (1/18) had HFS in a group receiving pyridoxine. Based on these data and previous clinical trials 13 of HFS in patients receiving PLD-containing chemotherapy, we estimated that the HFS rates of the no-pyridoxine and pyridoxine groups would be 20% and 5%, respectively. Calculation of the sample size was performed according to the accuracy of the estimation that the upper bound of the 90% confidence interval (CI) would not exceed 0.9 with 80% power for each group. The rate of losses due to failure to follow up was estimated to be 10%. With the aid of the above criteria, 127 patients were required for randomization for this study.
Procedures
PLD (40 mg/m2 intravenously, divided into 2 days, with a 4-week interval between treatments) was given, combined with dexamethasone (20 mg/day, day 1-day 4) plus lenalidomide (25 mg/day, day 1-day 21), bortezomib (1.3 mg/m2, day 1, day 4, day 8, and day 11), or vincristine (2 mg/day, day 1). In our study, steroid ointments were not prohibited. During the first morning before the scheduled PLD infusions and twice daily afterward, pyridoxine was administered in a 100 mg tablet delivered via oral ingestion twice daily for the next 7 days. Before administering PLD, the patients’ demographic details and clinical features were documented. The primary investigators were blinded to whether the patient was administered pyridoxine. Follow-up for the patients was carried out for 2 weeks after the respective groups completed the PLD infusion after each treatment duration. The investigators performed the CTCAE assessments and evaluated the severity of HFS. After each cycle, the oncologist completed an on-study sheet to provide all necessary background information so that investigators could conduct the CTCAE assessments and also completed a follow-up sheet to monitor the patients’ response to chemotherapy treatment. Patients were studied after the completion of PLD-containing treatment, after disease progression, or after HFS, whichever occurred first.
Assessment
The primary endpoints were the HFS grade, incidence, and time to HFS. Using the NCI Common Terminology Criteria for Adverse Events v4.0 grading scale, which ranges from 1 to 3, the HFS grade was assigned to each patient (Common Terminology Criteria, 2021). 14 Grade 1 is characterized by minor skin abnormalities (hyperkeratosis, edema, or erythema) that are not painful. Grade 2 comprises skin abnormalities (hyperkeratosis, edema, bleeding, blisters, or peeling) that are painful and restrict fundamental activities of daily living. Grade 3 is characterized by severe skin abnormalities (hyperkeratosis, edema, bleeding, blisters, or peeling) that are painful and restrict activities of daily living such as self-care.
Statistical Analysis
The statistical analysis of data was carried out utilizing SPSS (version: 22.0). When comparing ratios, Fisher’s exact test or Pearson’s chi-square test was utilized. When comparing measurement data between both groups, the t test was utilized. For the measurements obtained that did not follow a normal distribution, the nonparametric Mann-Whitney U test was applied. The time to HFS was analyzed with the aid of the Kaplan-Meier method, whereas the differences between the 2 curves were subjected to comparisons utilizing the log-rank test. P < .05 was established to denote statistical significance.
Results
From January 2017 to December 2018, a total of 127 participants were enrolled in the Department of Hematology at our hospital; 22 subjects were omitted from the study for various reasons (including not meeting inclusion criteria or declining to participate), and the final analysis set consisted of 105 subjects (Figure 1). Among those included, 52 subjects were randomly assigned to the pyridoxine cohort, and 53 subjects were allocated to the no pyridoxine cohort. Among the 105 subjects, the male-to-female ratio was 2:1 (75/30), ranging between 40 and 76 years old with a median age of 58 years old. By the R-ISS phases, 19 individuals were diagnosed with stage I, 69 with stage II, and 17 with stage III. In addition, patients exhibited a heavy burden of disease, as shown by the median rate of bone marrow plasmacytosis of 22% (range, 1%-89%), a hemoglobin concentration of 98.0 g/L on median (range, 57.0-169.0 g/L), a median creatinine concentration of 88.0 µmol/L (range, 37.0-691.0 µmol/L), and lactate dehydrogenase (LDH) concentration of 171.0 U/L (range, 86.0-831.0 U/L); 20% of patients had elevated LDH levels; LDH has a typical range of 240 U/L at the upper limit of normalcy. The disease statuses of the patients were as follows: 99 individuals (94.3%) had NDMM, and 6 individuals (5.7%) had RRMM. Among all patients, 96 (91.4%) were treated with bortezomib-based regimens (PAD, including bortezomib, dexamethasone, and PLD), 3 (2.9%) with conventional therapy (VAD, including vincristine, dexamethasone, and PLD), and 6 (5.7%) with immunomodulator-based regimens (RAD, including lenalidomide, dexamethasone, and PLD). There were no patients in the present study who had previously experienced HFS. The relative dose intensity (RDI) of pyridoxine was 98.3% in the pyridoxine-treated group. Table 1 shows the patients’ clinical features. The mean number of PLD chemotherapy cycles delivered was 4 (ranging, from 1 to 8 cycles), and there were no differences between the pyridoxine and no pyridoxine cohorts. The baseline demographic data were generally well-balanced (Table 1).

Patient eligibility and enrollment.
Baseline Clinical Characteristics of the MM Patients in the Pyridoxine Group and the No Pyridoxine Group.

Abbreviations: R-ISS: revised international staging system; DS: Durie-Salmon; NDMM: newly diagnosed multiple myeloma; RRMM: relapsed or refractory multiple myeloma; β2 MG: Beta2 microglobulin; PLT: platelet; HBsAg: Hepatitis B surface antigen; PAD: bortezomib, dexamethasone, pegylated liposomal doxorubicin; VAD: vincristine, dexamethasone, pegylated liposomal doxorubicin; RAD: lenalidomide, dexamethasone, pegylated liposomal doxorubicin.
In the pyridoxine and no pyridoxine cohorts, HFS appeared following a median of 4 (range, 1-8 cycles) and 3 (range, 1-7 cycles) chemotherapeutic cycles, respectively. Overall, 13.3% of patients suffered HFS (all grades), with 8.6% experiencing grade 1 incidents and 4.8% experiencing grade 2 incidents, according to the findings. As per the results, there were no cases of grade 3 incidents. HFS developed in 3 of 52 (5.8%) patients in the pyridoxine cohort, whereas it developed in 11 of 53 (20.8%) patients in the no pyridoxine cohort. The variation between the 2 cohorts was significant (P = .042; Figure 2). The rates of grades 1 and 2 HFS after randomization were 66.7% (2 patients) and 33.3% (1 patient) in the pyridoxine group and 63.6% (7 patients) and 36.4% (4 patients) in the no pyridoxine cohort, respectively. There were no substantial differences in HFS grades (P = .725).

Incidence of hand-foot syndrome in the pyridoxine and no pyridoxine groups.
The Kaplan-Meier curves for all-grade HFS showed a propensity toward pyridoxine effectiveness, as illustrated in Figure 3. The median number of cycles before the occurrence of HFS in the pyridoxine cohort was more significant than the median number of cycles until the onset of HFS in the no pyridoxine cohort [hazard ratio (HR) 3.8, 95% CI 1.19-10.23, P = .022]. The responses to initial treatment were also comparable between patients with MM in the presence or absence of pyridoxine. In the pyridoxine cohort, most patients (59.6%; 31/52) attained complete response (CR) or excellent partial response (VGPR), followed in number by patients who attained partial response (PR) or worse (40.4%; 21/52). In the no pyridoxine group, most patients achieved CR or VGPR (64.2%; 34/53), followed in number by patients who attained PR or worse (35.8%; 19/53). There were no statistically significant differences in response between the groups (P = .690).
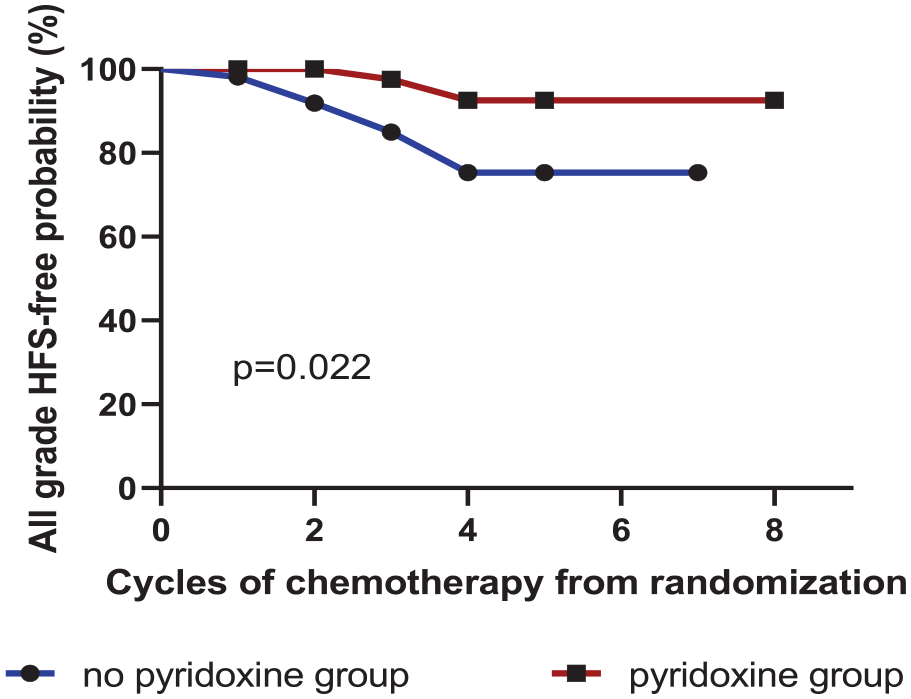
Cycles of chemotherapy to HFS occurrence between the pyridoxine group and the no pyridoxine group.
In total, 14 of the 105 patients developed HFS. A comparison profile between clinical and demographic variables among patients with and without HFS is shown in Table 2. In the univariate analysis of risk variables for HFS, regardless of pyridoxine prophylaxis, more cycles of PLD-containing chemotherapy and low albumin levels were significant risk variables for HFS (Table 2).
Comparison of Demographic and Clinical Characteristics Between Patients With and Without HFS Induced by PLD.

Abbreviations: R-ISS: revised international staging system; DS: Durie-Salmon; NDMM: newly diagnosed multiple myeloma; RRMM: relapsed or refractory multiple myeloma; β2 MG: Beta2 microglobulin; PLT: platelet; HBsAg: Hepatitis B surface antigen; PAD: bortezomib, dexamethasone, pegylated liposomal doxorubicin; VAD: vincristine, dexamethasone, pegylated liposomal doxorubicin; RAD: lenalidomide, dexamethasone, pegylated liposomal doxorubicin.
Discussion
MM is a common hematological cancer. The OS rate of individuals with MM has risen significantly in the previous decade, primarily due to the advent and advancements of innovative treatments. Consequently, given the possibility that cancer will progress to the stage of chronic illness, it is essential to pay close attention to the patient’s QoL. Medical oncologists and other healthcare providers who treat MM patients currently focus on ways to create a balance between effective tumor control and minimal toxicity. 15
PLD is one of the main therapeutic drugs used to treat MM in China. 13 According to the literature, HFS is the most prevalent complication of PLD-based chemotherapy, with an occurrence rate ranging from 18% to 45% in solid-tumor patients. In the present study, 13.3% of patients with MM who received PLD developed HFS, which is less than the reported incidence in the literature, and no grade 3 HFS occurred. Some studies suggest that patients with hematological malignancies have a reduced risk of HFS. 16 MM patients who were treated with PLD were all given dexamethasone. However, steroid use was an exclusion criterion in other studies, which could factor into the occurrence of HFS. 17 Since dexamethasone is an integral part of the chemotherapy regimen for MM patients, in our study, we could not exclude the influence of steroids.
While HFS is neither a life-threatening condition, HFS might reduce health-related QoL in many different ways due to the associated swelling, pain, loss of the capacity to participate in everyday activities, emotional impairment, and social isolation. Therefore, the prevention of HFS is of substantial clinical significance. Because there is a shortage of reliable data on the pathophysiology and management of HFS, therapeutic recommendations have relied primarily on professional judgment. The most effective management approach continues to be dosage titration or treatment discontinuation, both of which have the capacity for lowering the overall effectiveness of the chemotherapy treatment that is being planned. 18
Several studies and anecdotal evidence have proposed that diverse approaches should be used to reduce the occurrence rate of HFS. However, while pyridoxine, commonly referred to as vitamin B6, is routinely utilized as an oral medication to treat and prevent HFS, the evidence demonstrating that it has created advantages for patients is controversial and limited. Despite advances in science, the specific mechanism through which pyridoxine acts in HFS remains largely unknown. Denda et al 19 postulated that the mode of action of pyridoxine could be via the inhibition of the P2X purinergic receptor, which speeds up the restoration of the epidermal barrier once it has been disrupted and inhibits epithelial hyperplasia. Therefore, we conducted an open-label randomized investigation designed to provide information about the effectiveness of pyridoxine for preventing PLD-induced HFS in MM patients.
Our study suggests that pyridoxine prophylaxis effectively reduces the rate of HFS and delays the onset of HFS induced by PLD in MM patients. To the best of our knowledge, this is the first research report that offers a strong indication of the prophylactic efficacy of oral pyridoxine in MM patients. However, the findings of previous studies demonstrated conflicting findings on the clinical application of pyridoxine in preventing PLD-induced HFS. Several case reports and preclinical studies have suggested that pyridoxine is an effective therapeutic or preventative agent for PLD-induced HFS. 18 However, some contemporary double-blind, randomized studies demonstrated that pyridoxine was not satisfactory in preventing HFS in ovarian, breast, or endometrial cancer patients treated with PLD.20,21 Several factors could be at play in this difference, such as distinct cancer types, differences in statistical analysis methodologies, and differences in sample sizes. Furthermore, the data might deviate from ours due to variability in the sex ratio. It was demonstrated that women are more concerned than men are about the state of the skin on their feet and hands, in general. 8 Most of the patients included in these previous trials were women, but in our study, most patients were men. We hypothesize that this is among the s reasons for the differences between our results and those of earlier studies. It is also possible that other factors are at play, such as the change in pyridoxine dosage. Different research results were obtained at different doses of pyridoxine. Several case studies have shown that oral pyridoxine, at doses ranging from 50 to 800 mg daily, can be used successfully to manage HFS related to fluorouracil, docetaxel, etoposide, doxorubicin, and sorafenib, and the pyridoxine dose of >200 mg/day can provide better relief of HFS symptoms than that of <200 mg/day.10,11,22 However, the mechanism by which pyridoxine may exert a protective effect on HFS remains unknown and we do not have a preclinical model or pharmacodynamic data of the dose-response relationship of pyridoxine. In some case reports and clinical trials, pyridoxine 100 mg BID is safe for treatment and prevention of PLD and capecitabine-related HFS, which was applied in relatively large samples.5,23 Therefore, pyridoxine 100 mg BID was used in our clinical trial. And our study inferred that 200 mg of pyridoxine could be the optimum dosage for preventing HFS in MM patients. Another outcome of the present research is related to the HFS grade. The findings revealed the absence of a statistically significant difference between the 2 subgroups in terms of the reduction in HFS grade after the intervention (P = .725). This finding agrees with the assertions of a meta-analysis, which found that there was no overall correlation between prophylactic pyridoxine supplementation (oral usage) and HFS grade. 20 The mechanism is not precise. Our data indicate an obvious beneficial outcome of pyridoxine prophylaxis in preventing PLD-related HFS in MM patients. In specific trials, however, it was discovered that long-term usage of vitamin B6 affected the treatment responsiveness in some cancer patients.8,24 If high-dose pyridoxine hurts the effectiveness of chemotherapy drugs, this has not yet been proven. According to our findings, there were no statistically significant differences in response between the cohorts in the present study (P = .690). There is almost no significant concern regarding the potential of adverse effects related to prophylaxis pyridoxine for the preventative measures of PLD-induced HFS, as it is only administered for short durations.
Although the above results demonstrated that prophylactic pyridoxine reduced the rate of HFS, many studies have shown that prophylactic pyridoxine is not the only contributing factor.24,25 Our study found that in the absence of pyridoxine prophylaxis, more cycles of PLD chemotherapy (>3) and low albumin levels were significant risk factors for HFS.
In terms of the number of PLD cycles, according to related literature reports, the intensity of the dosage independently serves as a risk variable that influences the occurrence rate of HFS; the greater the intensity of the dosage is, the greater the possibility of developing HFS. 17 In our study, the PLD dose and schedule were the same among the different treatment regimens (40 mg/m2, day 1 and day 2), and PLD was used every 4 weeks; therefore, the number of PLD cycles influenced the dose intensity of PLD. More cycles of PLD chemotherapy (>3) are related to a high rate of HFS. Furthermore, in this study, the analysis results showed that the albumin level was another risk factor for the occurrence of HFS. The lower the albumin level was, the greater the probability of HFS was, which has not been reported in other studies. This may be because if a patient has a low albumin level, it can influence the proliferation of skin cells, resulting in a greater susceptibility to HFS. 25 Referring to baseline albumin-related biomarkers could be beneficial for preventing HFS and making appropriate modifications to the PLD dosage.
There are several drawbacks related to the present research. First, this was a single-center investigation. The research findings could be skewed and should be validated in multicenter, prospective investigations. Second, we did not evaluate QoL; if we had, we might have found other benefits of pyridoxine. Third, this study was designed without a placebo; hence, patients and clinicians who knew they were randomized to an extra pill to fight HFS might have reported findings differently. Nevertheless, to the best of our knowledge, this study is among the largest on PLD-related HFS in MM patients, and it. It identifies risk factors for HFS among MM patients instead of simply reporting the HFS incidence.
In conclusion, the present study is the first randomized, open-label clinical experiment to examine the effects of pyridoxine prophylaxis on HFS induced by PLD in MM patients. The administration of pyridoxine reduced the rate and delayed the occurrence of HFS. Nevertheless, prevention with pyridoxine is not the only variable affecting the occurrence and progression of HFS. The albumin level and the number of PLD cycles are also risk factors for HFS in MM patients, and the management of these factors may be helpful for the prevention of HFS.
Footnotes
Acknowledgements
The authors thank all the patients who participated in our research. The corresponding author has full access to all the data in the study and is solely responsible for the decision to submit this work for publication.
Author Contributions
Li Xiaozhe developed the protocol for the study and helped write the manuscript. Chen Meilan, Huang Beihui, and Liu Junru assisted with data collection. Gu Jingli and Kuang Lifen contributed to the statistical analysis. Li Juan had full access to all aspects of the research and writing process and took final responsibility for the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the 5010 Clinical Research Project of Sun Yat-sen University (2017005), Guangdong Natural Science Foundation (2021A1515011715) and the National Natural Science Foundation of China (No. 82070220).
Ethical Approval
This study was reviewed and approved by the First Affiliated Hospital of Sun Yat-sen University (IRB: [2017]126).
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Data Availability
The data sets used in the present research are accessible from the relevant author upon valid request if they are not available in the public domain.
Statement of Data Availability
The data sets analyzed in this study were available from the corresponding author upon reasonable request.
