Abstract
Aim:
Quxie capsule (QX), a compound of 21 kinds of Traditional Chinese Medicine (TCM) herbs, has been used to treat patients with metastatic colorectal cancer (mCRC) and could suppress the growth of colon cancer. However, the mechanisms of QX inhibiting colorectal cancer remain unclear. In current study, we attempted to determine the anti-colorectal cancer (CRC) effects of QX and the mechanisms of QX in alleviating colorectal cancer.
Methods:
A colitis-associated colon cancer (CAC) model was established by intraperitoneally injecting mice with AOM followed by 3 cycles of 2% DSS in water. During establishment of CAC model, we orally gavaged mice with QX. Hematoxylin and eosin (H&E) and immunohistochemistry were performed to assess lesion of the colonic tumors. The expression of pro-inflammatory cytokines in colonic tumors was measured by qPCR. The proportion of immune cells in colonic tumors was analyzed by flow cytometry. Internal transcribed spacer (ITS) sequencing and 16S rRNA gene sequencing were performed to detect intestinal microbiota. The expression of glycolytic related enzymes, lactate production, and extracellular acidification rate (ECAR) were used to assess the level of aerobic glycolysis.
Results:
QX markedly inhibited intestinal tumorigenesis by decreasing the expression of pro-inflammatory cytokines and the proportion of myeloid-derived suppressor cells (MDSCs), and increasing the proportion of CD8+ T cells in colon tumors. Fecal microbiota sequencing revealed that QX increased the relative abundances of intestinal symbiotic probiotics, such as, Lactobacillus, Bifidobacterium and Faecalibacterium genera. What’s more, opportunistic pathogens, Bacteroides genera and Aspergillus-Aspergillus fumigatus, exhibited remarkably reduced abundances in mice treated with QX compared with untreated CAC mice. Further experiments showed that QX significantly reduced glycolysis of colon tumor and suppressed A. fumigatus-induced glycolytic metabolism of colon cancer cells.
Conclusions:
QX alleviates the development of CRC at least in part through modulating intestinal microbiota and reducing A. fumigatus-induced aerobic glycolysis of colon cancer cells.
Introduction
Colorectal cancer (CRC) incidence rates rank third and mortality rank second worldwide. CRC incidence rates are about threefold higher in transitioned versus transitioning countries. 1 CRC incidence and mortality rates have been increasing, especially in younger adults. 2 Recurrence and metastasis are a major cause of increased CRC mortality.3,4 Although a number of therapeutic strategies, such as targeted therapy, chemotherapy and immunotherapy have been improved, they have limited efficacy in the control of CRC. 5 Therefore, there is an urgent need to develop novel therapeutic strategies for CRC.
Quxie capsule (QX) is a TCM herbal medicine which was developed by Yang et al of Xiyuan Hospital, China Academy of Chinese Medical Sciences, on the basis of the “Yinyanggongji pill” contained in the ancient book “Yizong Bidu,” and Yang et al used QX for the first time in the clinical treatment of digestive tract malignant tumors.6-8 The clinical use of QX follows the principle in the original text of “Yizong Bidu.” QX has been used to treat advanced colorectal cancer. A double-blind randomized placebo controlled trial has demonstrated that QX showed good efficiency and safety, and could reduce the risk of death and prolong the overall survival (OS) of patients with metastatic colorectal cancer (mCRC).9,10 In addition, QX could inhibit colon tumor growth in CT26 mouse colon cancer model and exert antitumor immune response. 11 A randomized controlled clinical trial suggested that QX could modulate the gut microbiome in mCRC patients and enhance the amount of intestinal anticancer bacteria. In the meantime, QX could increase T helper cells level in patients with mCRC. 12 Therefore, QX plays an anti-tumor role through various functions. However, the molecular mechanisms by which QX inhibits colorectal cancer remain largely unclear.
In recent years, an increasing number of studies have indicated that gastrointestinal microbiota plays an important role in the tumorigenesis and development of colorectal cancer. 13 Gastrointestinal microbiota is involved in maintaining gastrointestinal homeostasis.14-17 When this homeostasis is broken, resident opportunistic pathogenic microbes, such as fungus and bacterium, can promote colorectal cancer by inducing inflammation, enhancing cell proliferation and so on. 18 Previous studies have shown that the pathogenic fungi Candida tropicalis and Candida albicans aggravated colitis-associated cancer (CAC).19,20 Another pathogenic fungus, Aspergillus fumigatus, is closely associated with the development of colorectal cancer, 21 and it also induces myeloid-derived suppressor cells (MDSCs) which functionally suppress T cell responses. 22 Many experimental results have confirmed that pathogenic bacteria, such as pks+ Escherichia coli, Fusobacterium nucleatum, and Bacteroides fragilis promote development of colon cancer. 23 Bifidobacterium and lactobacillus can promote antitumor immunity and protect against colorectal cancer.16,24 Therefore, manipulating the gastrointestinal microbiota may alleviate colorectal cancer.
Under the condition of adequate oxygen supply, malignant tumor cells show enhanced aerobic glycolysis to meet the metabolic requirements of their own energy supply and cell proliferation, which is known as the Warburg effect. 25 The transcription factor hypoxia-inducible factor-1α (HIF-1α) plays a key role in the Warburg effect. In cancer cells, HIF-1α enhances aerobic glycolysis by inducing the expression of glycolysis-related enzymes and promoting their activity. 26 Targeting aerobic glycolysis has been reported as a promising approach for cancer therapy.27-29
The present study aimed to explore the role of QX in ameliorating colorectal tumorigenesis and investigate the mechanisms of QX protecting against development of CRC. This study hypothesized that QX could alleviate CRC development and the mechanisms were related to the modulation of intestinal microbiota composition and reduction of A. fumigatus-induced aerobic glycolysis of colon cancer cells by QX.
Materials and Methods
Animal Experiments
Pathogen-free 10-week-old female C57BL/6 mice were housed in a conventional and pathogen-free facility at Medical School of Nanjing University. These mice were fed for 12/12 hours in a cycle of day and night, with humidity (50 ± 5%) and temperature (22 ± 2℃) being controlled and fed daily with basic maintenance feed and sterilized water. Mice diet and research conformed to all aspects of the ARRIVE guidelines checklist. 30 All mouse handling and in vivo experiments were performed according to the NIH “Guide for the Care and Use of Laboratory Animals.” All animal procedures and experiments were performed following a protocol reviewed and approved by the Institutional Animal Care and Use Committee at Nanjing University (SCXK-Jiangsu-2019-0056).
As shown in Figure 1A, to induce CAC model, female C57BL/6 mice (10 weeks old) were intraperitoneally injected with a dose of 10 mg/kg azoxymethane (AOM) (Sigma, # A5486, USA) on day 0 (n = 6-8, each group). After 5 days, mice were treated with 3 cycles of 2% dextran sodium sulfate (DSS) (36-50 kDa, MP Biomedicals, #160110, USA) in drinking water for 7 consecutive days. During the induction of the CAC model, the mice in QX-treated groups were gavaged with QX [QX-Low dose (QX-L): 40 mg/kg; QX-High dose (QX-H): 80 mg/kg] for 14 consecutive days and then QX treatment was stopped for 7 days, which represents a cycle of treatment. Three cycles of QX treatment were used. Mice in the control group (vehicle) were not treated with QX. Mice were weighed daily. After 100 days, the mice were euthanized.
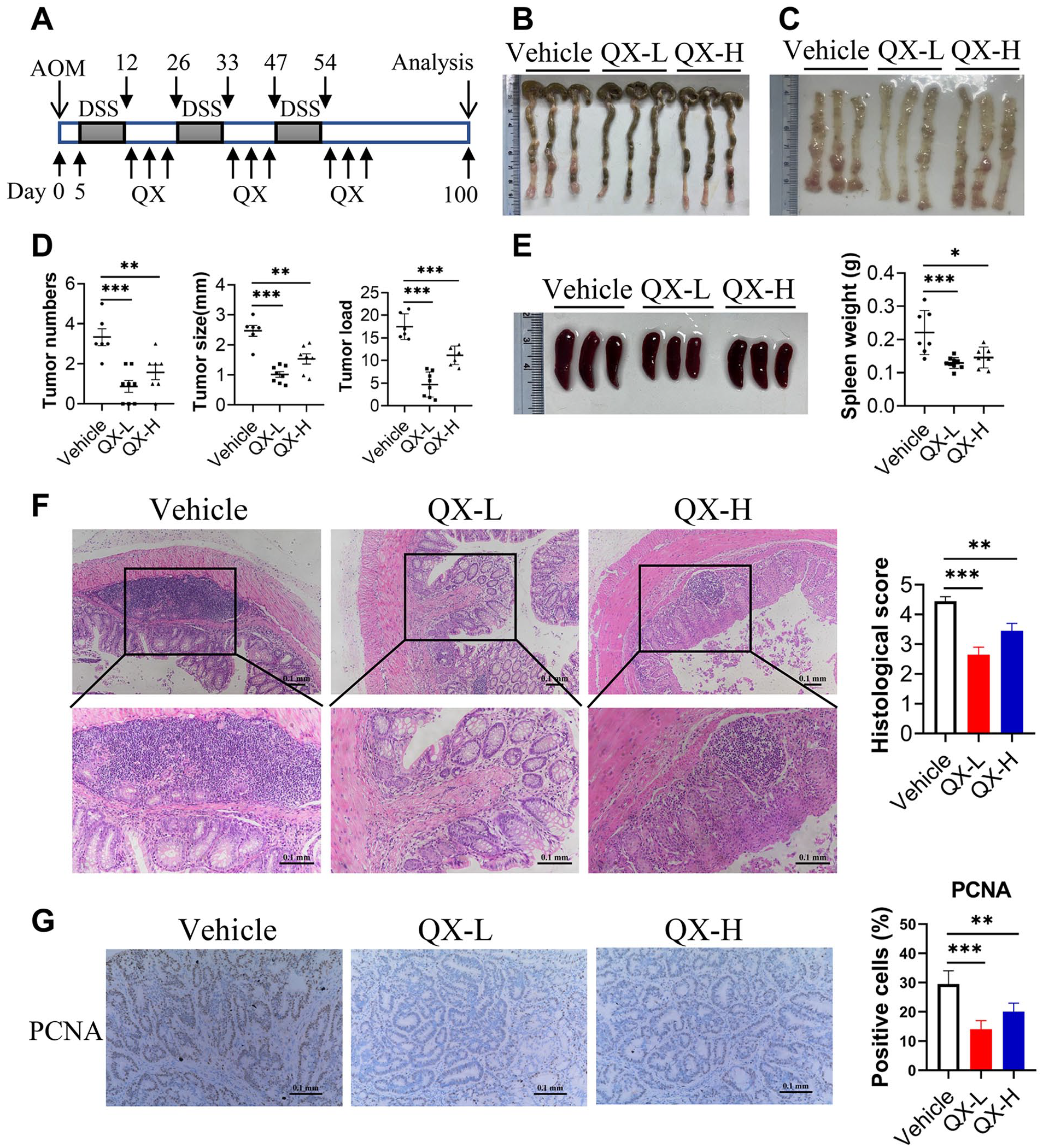
QX prevents colorectal tumorigenesis in an AOM/DSS-induced CRC model. (A) Schematic showing the experimental design and timeline of AOM/DSS mouse model. (B) Representative images of colonic tumors. (C) Representative images of the inside of the colon. (D) The numbers, size and load of tumors in the mouse colon tissues were measured. (E) Representative images of spleens. And the weight of the spleen was measured. (F) Representative histological images of colonic tumors were indicated by hematoxylin and eosin (H&E) staining. Scale bars, 0.1 mm. Histological score was evaluated by a pathologist. (G) The expression of PCNA was examined by immunochemistry in colonic tumors. The percentages of PCNA-positive tumor cells were quantified. Scale bars, 0.1 mm. Data are presented as the mean ± SEM. n = 6-8. *P < .05, **P < .01, ***P < .001 were calculated by one-way ANOVA.
Cell Lines and Cell Culture
Human colorectal cancer cell line SW480 was obtained from the Type Culture Collection of the Chinese Academy of Sciences (Shanghai, China), which was cultured in DMEM (Gibco, USA) supplemented with 10% FBS (Gibco, USA), 1% penicillin, and 1% streptomycin (Gibco, USA) at 37℃ in a 5% CO2-humified atmosphere. For QX treatment experiment, QX was first dissolved in the medium with final concentrations of 2.5%, 5%, 10%, 20%, and 40%. Then SW480 cells were seeded onto 12-well plates or 6-well plates at a concentration of ~5 × 105 cells/well or ~1 × 106 cells/well. A. fumigatus (A.F.) (MOI = 1) was used to stimulate cells in absence or presence of different concentrations of QX dissolved in the medium.
Preparation of QX
QX was kindly provided by Professor Yufei Yang of Xiyuan Hospital, China Academy of Chinese Medical Sciences. QX consists of 21 kinds of TCM herbs: Evodiae fructus, Zingiberis rhizoma, Cortex cinnamomi, Radix aconiti, Coptis chinensis, Pinelliae rhizoma, Citri grandis exocarpium, Poria, Arecae semen, Magnoliae officinalis, Aurantii fructus immaturus, Acori tatarinowii rhizoma, Corydalis rhizoma, Panax ginseng, Lignum aquilariae resinatum, Radix platycodonis, Succinum, Crotonis fructus, Galli Gigerii endothelium corneum, Hordei fructus germinatus, and Gleditsiae fructus abnormalis, at a ratio of 10:10:10:10:8:8:8:8:8:8:8:8:8:8:8:8:8:5:5:5:60, respectively. 11 All herbs were powdered and mixed into capsules by the Laboratory of Chinese Medicine Preparation of Xiyuan Hospital under the quality control of the institution. QX was manufactured by the Laboratory of Chinese Medicine Preparation of Xiyuan Hospital (batch number 20210801, Beijing, China) and was dissolved in filtered (0.22 μm) water for the animal study. 12 QX used in this study was first proposed by Professor Yufei Yang and used in clinical treatment, so there is no concern about infringing the intellectual property rights of the original creator.
Quality control data: Packing volumes differences: ±10%; Disintegration time: 20 minutes; Water content: 6.7%; Total number of aerobic bacteria: <10 CFU/g; Total number of molds and yeasts: <10 CFU/g. It has been identified that QX capsule contains 21 kinds of plant species in the formula and the proportions of each plant species meet the standard. This product is tested in accordance with the fourth part of The Chinese Pharmacopoeia 2020 edition, and the results meet the requirements.
Histopathological and Immunohistochemical Analysis of Colon Tumors
Mice colon tumors were fixed in 4% paraformaldehyde, and embedded in paraffin. Then sections were stained with hematoxylin and eosin (H&E). For immunohistochemical analysis, the sections were stained with PCNA (CST, #13110T, USA).
Quantitative PCR
Total RNA was isolated from colonic tumors and cells using Trizol reagent according to the manufacturer’s instructions. Then total RNA was reverse transcribed into cDNA. qPCR was performed using SYBR Green PCR master mix solution on a Step One sequence detection system (Applied Biosystems, MA, USA). The relative expression level of genes was calculated using the 2−ΔΔCT method and normalized to the expression of β-actin.
Western Blot Analysis
Total protein was extracted from colonic tumor tissue and cells. Protein concentration in the extracts was measured by the BCA assay (Beyotime P0010S, China). 30 ug protein of each sample were separated on 8% or 10% SDS–PAGE and then transferred onto PVDF membranes, which were subsequently blocked with 5% (w/v) BSA for 1.5 hour, and incubated with specific primary antibodies overnight at 4℃ and secondary antibodies for 1.5 hour at room temperature, respectively. Finally, the membranes were exposed in the ECL plus western blotting detection reagents (Millipore, MA, USA). Following antibodies were used: β-actin (CST, #8457, USA), HIF-1α (Immunoway, #YT2133, USA), GLUT1 (CST, #12939, USA), HK2 (CST, #2867, USA), GAPDH (Proteintech, #10494-1-AP, China), PGK1 (Proteintech, #17811-1-AP, China), PKM2 (CST, #4053, USA) and LDHA (CST, #2012, USA).
Flow Cytometry Analysis
Spleen and colonic tumor were isolated into single-cell suspensions. After washing, the single cells were pre-incubated with the fluorescence-conjugated primary antibody for 30 minutes at 4°C in the dark. Then, we performed flow cytometry on a FACS Calibur flow cytometer (BD Biosciences, USA). The results were analyzed by FlowJo software. The following antibodies were used: FITC-conjugated anti-mouse/human CD11b (BioLegend, #101206, USA), APC-conjugated anti-mouse Ly-6G/Ly-6C (Gr-1) (BioLegend, #108412, USA), APC-conjugated anti-mouse CD3 (BioLegend, #100235, USA), FITC-conjugated anti-mouse CD4 (BioLegend, #100405, USA), APC-conjugated anti-mouse F4/80 (BioLegend, #157306, USA), FITC-conjugated anti-mouse CD11c (BioLegend, #117305, USA), APC-conjugated anti-mouse I-A/I-E (BioLegend, #107614, USA).
Internal Transcribed Spacer (ITS) Sequencing and Analysis
Fungal DNA was isolated from the feces of mice using a MagPure Soil DNA LQ Kit (Magen, Guangdong, China, #D6356-02) following the manufacturer’s instructions. PCR was initially performed using the primer set ITS1F (5′-CTTGGTCATTTAGAGGAAGTAA-3′) and ITS2 (5′-GCTGCGTTCTTCATCGATGC-3′). The PCR products were purified with Agencourt AMPure XP beads (Beckman Coulter Co., U SA) and quantified using Qubit dsDNA assay kit. The concentrations were then adjusted for sequencing. Sequencing was performed on an Illumina NovaSeq6000 with 2 paired end read cycles of 250 bases each. (Illumina Inc., San Diego, CA; OE Biotech Company; Shanghai, China).
Clean reads were subjected to primer sequences removal and clustering to generate operational taxonomic units (OTUs) using VSEARCH software with 97% similarity cutoff. The representative read of each OTU was selected using the QIIME package. All representative reads were annotated and blasted against Silva database (Version 132) using RDP classifier (confidence threshold was 70%). The fungal diversity of the feces of mice was estimated using the alpha diversity that includes the Shannon index and Simpson index. The Unifrac distance matrix performed by QIIME software was used for unweighted Unifrac Principal coordinates analysis (PCoA) and phylogenetic tree construction. The internal transcribed spacer (ITS) sequencing and analysis were conducted by OE Biotech Co., Ltd. (Shanghai, China).
16S rRNA Gene Sequencing and Analysis
Bacterial DNA was isolated from the feces of mice as described above. PCR amplification of the V3-V4 hypervariable regions of the bacterial 16S rRNA gene was carried out in a 25 μL reaction using universal primer pairs (343F: 5′-TACGGRAGGCAGCAG-3′; 798R: 5′-AGGGTATCTAATCCT-3′). Library construction, sequencing and bioinformatic analysis were performed as described above. The 16S rRNA gene sequencing and analysis were conducted by OE Biotech Co., Ltd. (Shanghai, China).
Measurement of Extracellular Acidification Rate (ECAR)
The extracellular acidification rate (ECAR) of SW480 cells was analyzed using Seahorse XF Glycolysis Stress Test Kit (Agilent Technologies, USA). The measurement of ECAR was performed according to the XF Glycolysis Stress Test protocol through the Seahorse XFe 96 Extracellular Flux Analyzer (Seahorse Bioscience, USA).
Lactate Production Assay
The lactate production in serum and culture medium supernatant were determined by Lactate Assay Kit (Eton Bioscience, #1200012002, USA) according to the instructions of manufacturer.
Statistical Analysis
Microbiome analysis was conducted by OE Biotech Co., Ltd. (Shanghai, China). The OTU number of microbial diversity was analyzed by Venn analysis and the rank curve. By using principal coordinates analysis (PCoA), beta diversity was shown. Chao1, Observed species, PD whole tree, Shannon, and Simpson indices were used to assess alpha diversity at the Operational Taxonomic Units (OTU) level. The study of species differences between groups was evaluated at the genus and species levels. One-way ANOVA was used to compare the statistical significance, and Tukey’s multiple comparisons test was used for post-hoc test. Statistical significance was defined as a P value <.05.
Statistical analysis was conducted using GraphPad Prism 8 (GraphPad Software, La Jolla, CA). Before performing statistical analyses, we first performed an analysis of variance and checked the normality of data. Analysis showed that the normality of data was parametric and the data accorded with homogeneity of variance. All data were presented as means ± SEM and statistical significance was determined by one-way ANOVA. And Tukey’s multiple comparisons test was used for post-hoc test. Values of P < .05 were considered to be statistically significant.
Results
QX Protects Against Colorectal Tumorigenesis in a CAC Model
To investigate the effect of QX on the development of CRC, a CAC model was established according to the experimentation timeline of Figure 1A. We found that QX significantly decreased tumor number, tumor burden and tumor size of CAC mice (Figure 1B-D). Also, the size and weight of the spleens were markedly reduced in QX-treated tumor-bearing mice (Figure 1E). Histopathological examination indicated that QX ameliorated the mucosal lesions and reduced the infiltration of inflammatory cells in colonic tumors of CAC mice (Figure 1F). Next, immunohistochemistry staining was performed to detect the expression of proliferating cell nuclear antigen (PCNA), which could participate in cell proliferation. The results showed that QX significantly inhibited the expression of PCNA in colonic tumors of CAC mice (Figure 1G). Taken together, these results indicate that QX inhibits colorectal tumorigenesis in a colitis-associated cancer model.
QX Modulates Tumor Immune Microenvironment to Suppress Colorectal Tumorigenesis
Next, we investigated the mechanisms of QX inhibiting colorectal tumorigenesis. So, we performed qPCR to analyze the expression of inflammatory cytokines in colonic tumors from QX-treated CAC mice and control CAC mice. QX markedly reduced the expression of pro-inflammatory cytokines, IL-1β, IL-6, and TNF-α and upregulated the expression of anti-inflammatory cytokines IL-10 (Figure 2A). Increasing evidence indicates that immune cells play a key role in the tumor microenvironment. And the tumor microenvironment can transform myeloid cells, such as myeloid-derived suppressor cells (MDSCs), macrophages and dendritic cells (DCs) into immunosuppressive cells. 31 Hence, we measured the proportion of MDSCs, macrophages and DCs in spleen and colonic tumors of CAC mice by flow cytometry. We observed that compared with CAC mice, the proportion of MDSCs (Figure 2B and C) was significantly reduced and the proportion of CD8+ T cells (Figure 2D and E) was markedly increased in spleen and colonic tumors of QX-treated CAC mice. However, QX administration did not affect the proportion of macrophages and DCs in spleen and colonic tumors of CAC mice (Figure 2F-I). Collectively, these results suggest that QX affects a large number of immune cells, particularly immunosuppressive MDSCs and CD8+ T cells.

QX alters tumor immune microenvironment to promote colorectal cancer. (A) qPCR was performed to detect the mRNA expression of indicated genes. (B-I) Flow cytometry was performed to determine the proportion of MDSCs (CD11b+Gr1+), CD8+ T cells (CD3+CD8+), macrophages (CD11b+F4/80+) and DCs (CD11c+MHC II+). Data are presented as the mean ± SEM. n = 6 to 8. *P < .05, **P < .01, ***P < .001 were calculated by one-way ANOVA.
QX Alters the Intestinal Fungal Microbiota in CAC Mice
To further confirm whether QX can modulate the intestinal microbiota in mice with CAC, we first performed internal transcribed spacer (ITS) sequencing to detect fungal microbiota in feces of mice with CAC. Compared with CAC mice, the number of operational taxonomic unit (OTU) in QX-treated CAC mice was significantly increased (Figure 3A and B). A comparison between the fecal fungal microbiota of CAC mice and QX-treated CAC mice by principal coordinate analysis (PCoA), demonstrated that QX treatment altered the principal component of fecal fungus (Figure 3C). We also found lowered alpha-diversity of fungi in feces of QX-treated CAC mice compared with CAC mice (Figure 3D). Further analysis showed that QX treatment significantly changed Ascomycota and Basidiomycota in intestinal tract of CAC mice (Figure 3E and F), in which the abundance of Aspergillus, an opportunistic pathogenic fungus, was significantly reduced in QX-treated CAC mice (Figure 3G). Aspergillus fumigatus is proved to be closely related with colorectal cancer. 21 To identify specific species of Aspergillus, we quantified Aspergillus fumigatus in feces by qPCR. We observed that Aspergillus fumigatus was decreased in QX-treated CAC mice (Figure 3H). In a word, QX significantly changes the intestinal fungal microbes of CAC mice.

The intestinal fungal microbiota is altered in QX-treated tumor-bearing mice. (A) The operational taxonomic unit (OTU) number of fungal diversity in the feces was shown by Venn diagram. (B) Fungal diversity was determined by the OTU rank curve. (C) Principal coordinate analysis (PCoA) of β-diversity based on fungal ITS gene sequence abundance. (D) Fungal alpha-diversity was analyzed by Shannon and Simpson indices. (E) Fungal-taxon-based analysis at the phylum level. (F) Heat map showing relative abundances of the top 10 fungal phyla. (G) Box plot showing relative abundances of the top 10 fungal genera in feces. (H) Specific fungal burden of Aspergillus fumigatus was determined by qPCR. Data are presented as the mean ± SEM. n = 6-8. *P < .05, **P < .01, ***P < .001 were calculated by one-way ANOVA.
QX Modulates the Gut Bacteria Microbiome of CAC Mice
Recent studies show that, in addition to fungi, dozens of bacteria could regulate or contribute to cancer. 23 To explore the effects of QX on the gut bacteria microbiome, we used 16S rRNA gene sequencing on stool samples of CAC mice and QX-treated CAC mice. We found that QX treatment increased the number of operational taxonomic unit (OTU) (Figure 4A and B) and principal coordinate analysis (PCoA) suggested that the principal component of fecal bacteria in QX-treated CAC mice was altered compared with CAC mice (Figure 4C). We also observed elevated alpha-diversity of bacteria in feces of CAC mice treated with QX (Figure 4D). Specifically, QX enhanced the relative abundances of commensal probiotics including Lactobacillus, Bifidobacterium and Faecalibacterium genera. Moreover, some cancer-associated bacteria, such as Bacteroides exhibited markedly reduced abundances in mice treated with QX compared to control groups (Figure 4E-G). Together, our data demonstrate that QX can enrich the relative abundances of commensal probiotics and decrease pathogens.
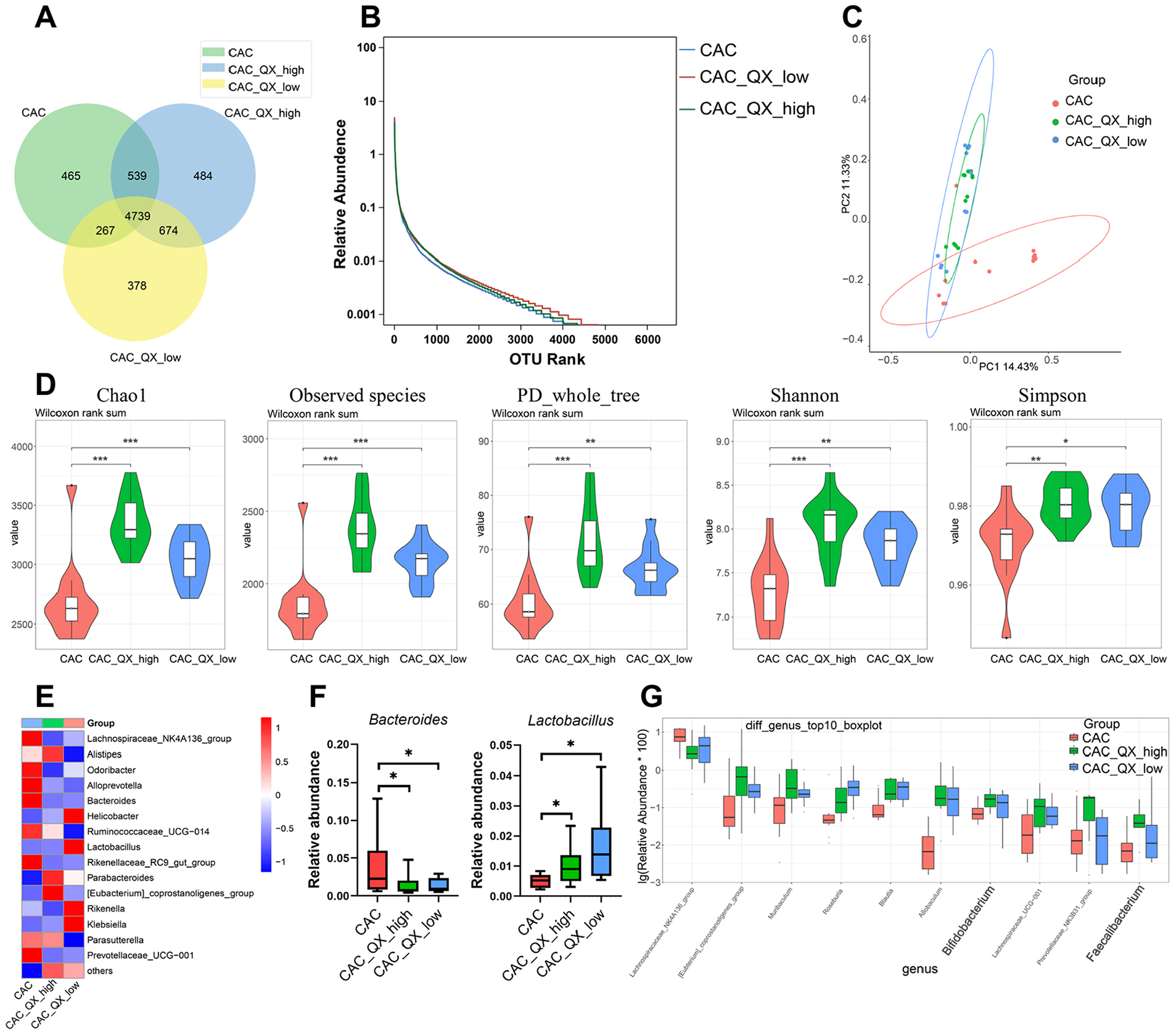
QX modulates the gut bacteria microbiome of CAC mice. (A) The operational taxonomic unit (OTU) number of bacterial diversity in the feces was shown by Venn diagram. (B) Bacterial diversity was determined by the OTU rank curve. (C) Principal coordinate analysis (PCoA) of β-diversity based on 16S rRNA gene sequence abundance. (D) The feces were tested for bacterial alpha-diversity measures, including Chao1, Observed species, PD_whole_tree, Shannon and Simpson indices. (E) Heat map showing relative abundancies of the top 15 bacterial genera. (F) Box plot showing relative abundances of Bacteroides and Lactobacillus. (G) Box plot showing relative abundances of the bacterial genera. Data are presented as the mean ± SEM. n = 6-8. *P < .05, **P < .01, ***P < .001 were calculated by one-way ANOVA.
QX Suppresses Glycolysis of Colonic Tumors in CAC Mice
The opportunistic fungal pathogen A. fumigatus has been reported to induce glycolysis. 32 Furthermore, previous studies have shown that Streptococcus thermophilus prevented CRC development through increasing the abundance of probiotics, including Bifidobacterium and Lactobacillus, which reduced the glycolysis of colonic tumors. 33 We also found that a preparation of 3 probiotic species, Bornlisy (BO) protected mice against intestinal tumorigenesis by inhibiting aerobic glycolysis of colon cancer cells. 34 Therefore, we hypothesized that QX affected glycolysis of colonic tumor in CAC mice. To test this hypothesis, we first detected the expression of glycolytic enzymes by Q-PCR analysis. The results indicated that QX dramatically decreased the mRNA expression of glycolytic enzymes including Glut1, Hk2, Gpi, Pfkfb3, Pfkl, Aldoa, Gapdh, Pgk1, Pgam1, Eno1, Pkm2, Ldha, and Pdk1 (Figure 5A). Hypoxia-inducible factor-1α (HIF-1α) is a key regulator of glycolytic enzyme expression. So, we also examined the expression of HIF-1α and Phd3 representing the transcriptional activity of HIF-1α. We observed that the mRNA expression of HIF-1α and Phd3 were significantly down-regulated in colonic tumors of QX-treated CAC mice compared to CAC mice (Figure 5A). Consistent with the above results, immunoblotting analysis also demonstrated that QX markedly reduced the expression of HK2, GAPDH, PGK1, PKM2, and LDHA (Figure 5B). Moreover, serum lactate production in CAC mice treated with QX was decreased compared with CAC mice (Figure 5C). All together, these data show that QX potentially restrains aerobic glycolysis of colon cancer to abrogate intestinal tumorigenesis.

QX suppresses glycolysis of colonic tumors in CAC mice. (A) The expressions of indicated genes in colonic tumors were detected using qPCR. (B) Immunoblotting was performed to determine the protein expression of glycolytic enzymes in colonic tumors. (C) Lactate production in serum was measured by Lactate Assay Kit. Data are presented as the mean ± SEM. n = 6 to 8. *P < .05, **P < .01, ***P < .001 were calculated by one-way ANOVA.
QX Reduces A. fumigatus-induced Glycolytic Metabolism in Colon Cancer Cells
To verify whether QX can inhibit A. fumigatus-induced glycolysis of colon cancer cells, colon cancer cells SW480 were stimulated with A. fumigatus in the presence or absence of QX. We found that A. fumigatus-induced mRNA expression of glycolytic enzymes including HIF-1α, Glut1, Hk2, Gapdh, Pgk1, Pkm2, and Ldha was markedly decreased in QX-treated SW480 cells (Figure 6A). Consistently, immunoblotting indicated that QX inhibited the expression of these glycolytic enzymes induced by A. fumigatus in a dose-dependent manner (Figure 6B). In addition, QX treatment significantly inhibited lactate production in A. fumigatus-stimulated SW480 cells in a concentration-dependent manner (Figure 6C). In order to find out whether QX directly affects glycolytic metabolism of A. fumigatus-stimulated SW480 cells, we used a Seahorse XF analyzer to measure the level of extracellular acidification rate (ECAR). The results indicated that QX significantly reduced the level of ECAR in A. fumigatus-stimulated SW480 cells (Figure 6D and E). Therefore, the results suggest that QX abolishes A. fumigatus-induced glycolytic metabolism of colon cancer cells.
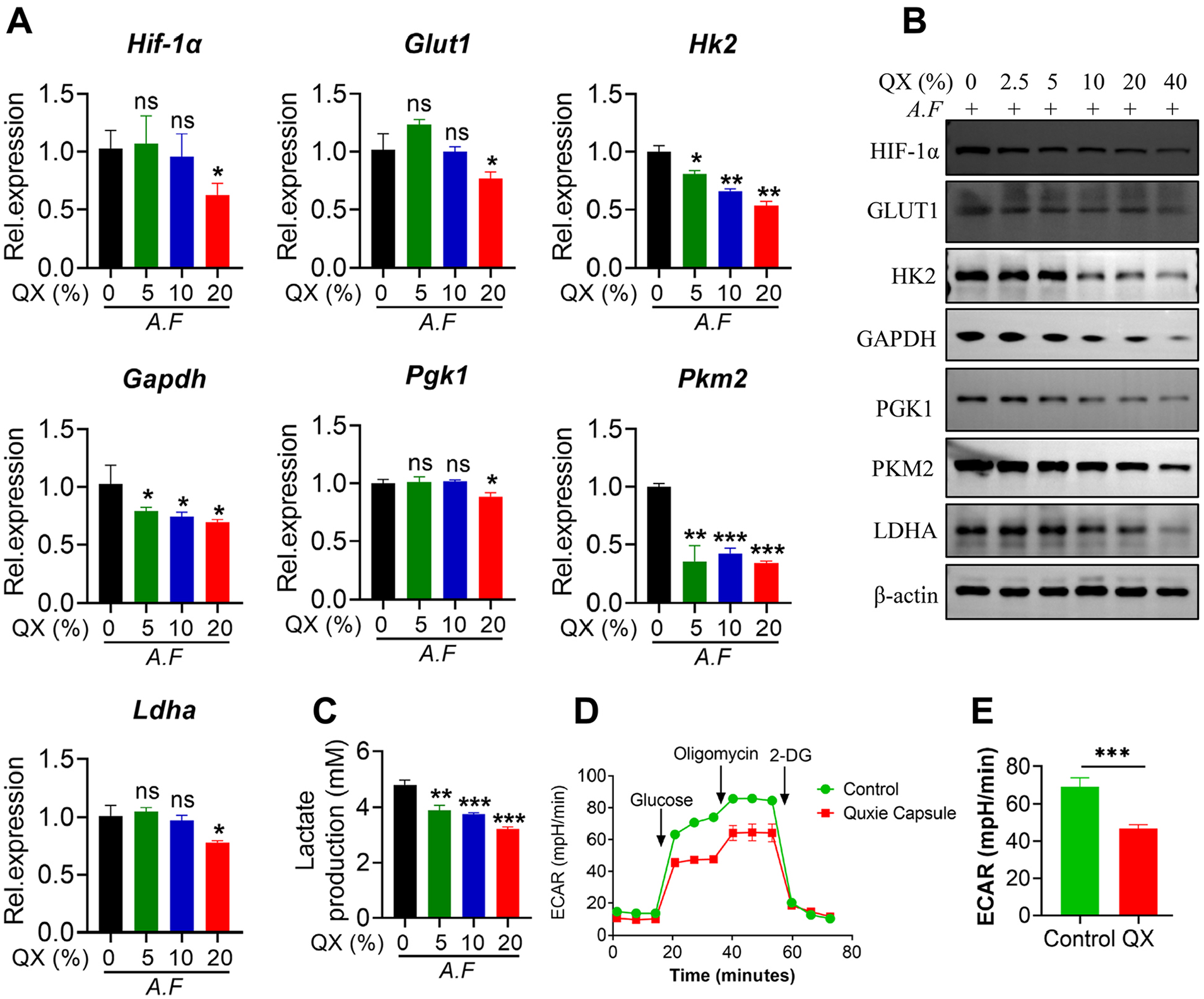
QX reduces A. fumigatus-induced glycolytic metabolism of colon cancer cells. (A-C) SW480 cells were stimulated with A. fumigatus (A.F) (MOI = 1) in combination with or without QX dissolved in medium (as indicated concentration) for 6, 12, or 24 hours. Then the mRNA expressions of the indicated genes were assayed by qPCR (A). Western blot was performed to detect the expression of the indicated protein (B). Cell culture supernatants were collected to detect lactate production (C). (D and E) SW480 cells were stimulated with A.F (MOI = 1) in combination with or without QX dissolved in medium (10%) for 3 hours. Then the ECAR level was assessed by Agilent Seahorse XFe96 Analyzer. Data are presented as the mean ± SEM. Results are representative of 3 independent experiments. n = 3. *P < .05, **P < .01, ***P < .001 were calculated by one-way ANOVA.
Discussion
The incidence and mortality of CRC are increasing year by year. 2 Many factors can aggravate the occurrence of CRC, such as intestinal inflammation, disturbance of tumor immune microenvironment, intestinal microbial dysbiosis, and increased glycolysis metabolism of colon cancer cells.23,29,35,36 However, there is a lack of effective intervention and therapeutic drugs for CRC. QX, a compound of 20 kinds of TCM herbs, has been reported to inhibit colon tumor growth, regulate immune cells, modulate gut microbiome and prolong the overall survival (OS) of patients with metastatic colorectal cancer (mCRC).2,11,12 Therefore, this suggests potential benefits of QX for the treatment and intervention of colorectal cancer. In the current study, we investigated the anti-tumor effects of QX and potential molecular mechanisms. We demonstrated herein that intragastric administration of QX observably protected mice against colitis-associated tumorigenesis, as evidenced by reduced intestinal tumor size, number, mucosal lesions, infiltration of inflammatory cells and proliferation of tumor cells. This further provides a solid theoretical basis for the clinical application of QX.
Chronic intestinal inflammation promotes the development of CRC, and many pro-inflammatory cytokines may play a vital role. It is noteworthy that pro-inflammatory cytokines IL-1β, IL-6 and TNF-α promote intestinal inflammation, which leads to the development of CRC.23,37-40 What’s more, an anti-inflammatory cytokine, IL-10, can inhibit intestinal inflammation. 41 Our results suggested that the expression of IL-1β, IL-6, and TNF-α were down-regulated in colonic tumors from CAC mice treated with QX compared with control CAC mice. Consistently, the expression of IL-10 was up-regulated in colonic tumors from CAC mice treated with QX. These results suggested that QX might play an anti-inflammatory role. In addition to intestinal inflammation, the tumor immune microenvironment plays a crucial role in the development of CRC. For example, Peptostreptococcus anaerobius promoted colorectal tumorigenesis by increasing the proportion of immune-suppressive MDSCs, tumor-associated macrophages (TAMs) and reducing the proportion of CD8+ T cells in colonic tumors of CAC mice. 35 Our previous study indicated that the pathogenic fungus Candida tropicalis aggravated colitis-associated colon cancer by increasing the accumulation of MDSCs and inhibition of CD8+ T cells. 19 Interestingly, we found a decreased proportion of MDSC and increased proportion of CD8+ T cells in colonic tumors from CAC mice treated with QX. However, QX has no obvious effect on other immune cells. So we speculated that QX may reduce the number of MDSCs, leading to increased number of CD8+ T cells. And CD8+ T cells then exert their antitumor effects. These data collectively suggest that QX may alleviate the development of CRC at least to a certain extent by inhibiting intestinal inflammation and increasing number of CD8+ T cells.
It is reported that certain probiotics can inhibit the development of CRC. For example, Lactobacillus gallinarum could suppress colitis-associated tumorigenesis by regulating gut microflora and producing anti-tumor metabolites. 24 Our previous findings suggested that Bornlisy (BO), a cocktail of 3 probiotics, Lactobacillus acidophilus, Lactobacillus bulgaricus and Bacillus subtilis, protected mice against intestinal tumorigenesis. 34 In the present study, we discovered that QX prominently enriched the relative the abundance of commensal probiotics, such as Lactobacillus, Bifidobacterium and Faecalibacterium, which resulted in decreased glycolytic metabolism in colon cancer. Increasing evidence demonstrates that the metabolites of probiotics have antitumor effects. A study has shown that cancer cells were selectively harmed by metabolites generated by Lactobacillus plantarum. 42 Therefore, we hypothesized that the antitumor effect of QX may be attributed to the metabolites produced by probiotics. This needs to be further studied. While a genus of pathogenic bacteria, Bacteroides, was markedly reduced in CAC mice treated with QX. Intestinal flora plays an important role in the development of CRC. We have previously reported that transplantation of the conditionally pathogenic fungi Candida tropicalis and Candida albicans aggravate intestinal tumorigenesis in CAC mice and germ-free mice.19,20 Another study showed that the pathogenic fungus Aspergillus fumigatus is closely associated with the progression of CRC. 21 These studies also demonstrated that intestinal fungal dysbiosis accelerated colon tumorigenesis. We also found that Aspergillus fumigatus was observably depleted in mice treated with QX. Therefore, QX ameliorated the development of CRC through enriching commensal probiotics and reducing pathogenic bacteria and fungi.
Cancer cells preferentially use glycolysis over oxidative phosphorylation (OXPHO) to produce energy, even when oxygen is available. This phenomenon is called “the Warburg effect” or aerobic glycolysis. Aerobic glycolysis provides intermediates for biosynthesis of highly proliferating tumor cells, which are used to support rapid growth, division and survival of tumor cells. It is reported that glycolysis can enhance progression of CRC. 36 Targeting lactate metabolism is a promising strategy for the treatment of tumors, 27 which highlights the prospective role of glycolysis as a therapeutic target. It has been reported that Aspergillus fumigatus can induce glycolysis. 32 Our present study also suggested that Aspergillus fumigatus promoted glycolysis in colon cancer cells. Recent studies have shown that Streptococcus thermophilus suppressed the Warburg effect of colon cancer through increasing the abundance of probiotics, including Bifidobacterium and Lactobacillus to protect against intestinal tumorigenesis. 33 And our previous study also indicated that probiotics could inhibit glycolysis of colon cancer cells. 34 In the current study, we demonstrated that QX significantly reduced aerobic glycolysis in colonic tumor at least in part by increasing the abundances of probiotics. Consistently, QX suppressed A. fumigatus-induced aerobic glycolysis of colon cancer cells SW480 in a concentration-dependent manner in vitro. Thus, the anti-tumor effect of QX could be attributed, at least in part, to its ability to reduce A. fumigatus-induced aerobic glycolysis of colon cancer cells. Furthermore, the molecular mechanism of QX inhibiting A. fumigatus-induced aerobic glycolysis of tumor cells requires further elucidation.
Conclusions
To sum up, we demonstrate for the first time that QX protects against colitis-associated colon cancer. The anti-tumor effect of QX is associated with inhibiting intestinal inflammation, modulating the tumor immune microenvironment, altering the intestinal microbiota and reducing A. fumigatus-induced aerobic glycolysis of colon cancer cells. These results may explain why QX reduces the risk of death and prolongs the overall survival of patients with mCRC, which further facilitates the significance of QX in clinical application.
Footnotes
Acknowledgements
We thank Xuan Zhou of OE Biotech Co., Ltd for helping with microbiome analysis and technical support.
Author Contributions
Tingting Wang, Yufei Yang, Geliang Yang: Conceptualization, Methodology, Software. Zhiyong Zhang: Data curation, Writing-Original draft preparation, Writing-review & editing. Yuxi Chen: Visualization, Investigation. Sunan Shen: Supervision. Yaojun Zheng, Lei Wang: Software, Validation. All authors have read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by grants from National Natural Science Foundation of China (82072648), the Natural Science Foundation of Jiangsu Province (BK20211508), the Fundamental Research Funds for the Central Universities (021414380500), National Administration of Traditional Chinese Medicine of China major difficult clinical collaboration pilot of Chinese and Western medicine project (2018) 275, Study on characteristics and diagnosis and treatment path of patients benefiting from integrated TCM and WESTERN Medicine program for Colorectal Cancer (Capital Development Fund 2022-1-171).
