Abstract
Background:
Acute myeloid leukemia (AML) is a kind of hematopoietic malignancy with limited response and acquired resistance to therapy. Inducing apoptosis and inhibiting autophagy in tumor cells is a combinational strategy for the development of anticancer therapeutics. Tanshinone IIA (TAIIA) is one of the major ingredients in Salvia miltiorrhiza, which is the most prescribed herb for the treatment of AML in Taiwan. Therefore, this study aimed to delineate the anticancer effects of TAIIA and its effect when combined with an autophagy inhibitor to treat AML.
Methods:
The anticancer effects of a combination of TAIIA and the autophagy inhibitor 3-methladenine (3MA) on the human monocytic leukemia cell line THP-1 were explored. The apoptosis and cell cycle of the leukemia cells were examined by Annexin V and propidium iodide staining and analyzed by flow cytometry. The oxidative stress level was determined by a malondialdehyde (MDA) colorimetric assay, nitric oxide colorimetric assay and glutathione peroxidase (GPx) colorimetric assay. The expression of apoptosis-related proteins was determined by western blotting.
Results:
TAIIA treatment significantly induced apoptosis via increased p53, Bax/Bcl, PARP, and caspase-3 signals and oxidative stress by enhancing MDA and nitrate/nitrite production and reducing GPx activity in THP-1 cells in a dose-dependent and time-dependent manner. The combination of the autophagy inhibitor 3MA enhanced TAIIA-induced apoptosis via the p53, Bax/Bcl, PARP, caspase-3, and oxidative stress pathways in THP-1 cells.
Conclusion:
The results suggest that TAIIA and autophagy inhibitors have combined effects on the apoptosis of leukemia cells, thus representing a novel and effective combination with the potential for application as a clinical therapy for AML.
Keywords
Introduction
Acute myelogenous leukemia is also known as acute myeloid leukemia, and acute myeloblastic leukemia (AML) is caused by the abnormal clonal expansion of myeloid blast cells in bone marrow and peripheral blood. The majority of AML cases (approximately 80%) occur in adults, and this disease has a poor survival rate and prognosis relative to other forms of leukemia. 1 Conventional therapy for AML includes radiation therapy, chemotherapy, and allogeneic bone marrow (BM) transplantation. In addition to conventional therapy, targeted therapy, allogeneic stem cell transplantation, and immunotherapy should be considered for eligible candidates. 2 For many years, the combination of cytarabine and an anthracycline has been the standard induction regimen for intensive treatment of AML patients. 3 The major toxicity of the standard induction regimen for AML is myelotoxicity, resulting in death in at least 25% of cases during induction in non-selected patients and long-term cures of 30% to 40% among younger patients.4,5 Current treatments for AML patients of heterogeneous and different age groups combine novel targeted therapies (venetoclax, FLT3 inhibitors, and IDH inhibitors) with standard therapy, which can reduce the concentration of toxic drugs and their side effects. 5 However, new drugs to treat AML patients must be developed to reduce toxicity and improve efficacy, and additional research is needed to clarify the mechanism of action of these new drugs to ensure their safe use.
Over time, targeted therapies have been developed to better address the pathobiology and heterogeneity of AML. Dysregulation of apoptosis frequently occurs in cancer, and cancer cells escape apoptosis through different mechanisms, such as overexpression of anti-apoptotic proteins (Bcl-2 and Bcl-xL) or loss of expression of pro-apoptotic proteins (Bak/Bax). 6 Based on the importance of antiapoptotic proteins and proapoptotic proteins, many studies have targeted Bcl-2 and Bax to develop new drugs for the treatment of AML. For example, ABT-737 was shown to block Bcl-2, Bcl-xL, and Bcl-w and induce Bak/Bax-dependent apoptosis in AML cells, while being active in vivo. 7 Oblimersen was shown to decrease Bcl-2 mRNA expression and exert efficacy in a leukemia xenograft model. 8 Recently, abundant evidence has shown that autophagy has a critical role in the progression of AML and its response to targeted therapies. Thus, modulation of autophagy may enhance the therapeutic effect of AML. For example, inhibition of the essential autophagy-related protein Atg7 and autophagy-related signaling pathway SDF-1α–CXCR4 may improve the outcome of AML treatment by increasing chemosensitivity.9,10 However, these drugs still have limitations in the treatment of AML. For example, ABT-737 was resistant to many cell lines due to its inability to downregulate Mcl-1, while Oblimersen had no significant efficacy in older AML patients in clinical phase III trials.7,11
Reactive oxygen species (ROS) are products of the normal metabolism of oxygen in cells. Oxidative stress occurs when the redox balance within a cell is disrupted, resulting in damage to DNA, proteins, and lipids. 12 Several studies have confirmed a high rate of ROS production in AML. 13 Notably, due to the high rate of ROS production in AML cells, these cells have a lower buffer capacity for ROS destruction, which makes them more sensitive to pro-oxidative treatments than their normal counterparts. 14 In line with this concept, studies have indicated that myeloid leukemia cells are more sensitive to external pro-oxidant stimuli.15-17 Therefore, several therapeutic strategies are being developed to increase ROS levels to overwhelm the redox adaptations of these cells and induce oxidative stress that is incompatible with cell viability. 18 Moreover, previous studies have demonstrated that the tumor suppressor protein p53 can increase the level of intracellular ROS via induction of pro-oxidation genes and suppression of antioxidation genes, resulting in the promotion of cell apoptosis.19,20 Based on these studies, activation of the p53 and ROS pathways may represent a new therapeutic strategy for cancer by inducing a cytotoxic effect.
Over several decades, many Chinese herbal medicines and their compounds have been proven to have anticancer effects.21-24 Salvia miltiorrhiza is the most commonly prescribed single herb for the treatment of AML in Taiwan, 25 and tanshinone IIA (TAIIA) (molecular formula: C19H18O3; molecular weight: 294.34) is its well-known compound. TAIIA has been demonstrated to have therapeutic effects against various cancer cell lines, such as leukemia cells, 26 colorectal cancer cells, 27 breast cancer cells,28,29 and lung cancer cells,30,31 via multiple targets. In terms of the mechanism of inducing apoptosis in cancer cells, it is generally accepted that TAIIA can induce DNA damage to effectively activate p53 and act on apoptotic molecules, that is, Bax/Bcl2 and cleaved caspase-3, leading to apoptosis.32,33 However, the effects of TAIIA used combination with an autophagy inhibitor on p53 and ROS leading to apoptosis in leukemia cells are still unclear.
In this study, we found that a combination of TAIIA and an autophagy inhibitor had additive anticancer effects on AML cells. The mechanisms by which the combination of TAIIA and an autophagy inhibitor promote cancer cell apoptosis were studied and verified based on increases in p53, Bax/Bcl, cleaved caspase-3, cleaved PARP, and ROS.
Materials and Methods
Cell Culture
The human THP-1-cell line was obtained from the American Type Culture Collection (ATCC, Rockville, MD, USA). Cells were cultured in RPMI-1640 medium (Thermo Fisher Scientific, Waltham, MA, USA) containing 10% heat-inactivated fetal bovine serum (Cytiva, Washington D.C., USA) and 1% penicillin and streptomycin (Gibco, Carlsbad, CA, USA). The cell line was maintained in humidified air containing 5% CO2 at 37°C.
Chemicals
Tanshinone IIA (TAIIA) (purity > 98%) and 3-methladenine (3MA) (purity > 98%) were purchased from Sigma -Aldrich Co. (St. Louis, MO, USA). Stock solutions were generated by dissolving the compounds in dimethyl sulfoxide (DMSO) (Sigma,-Aldrich, St. Louis, MO, USA) and further diluting with RPMI-1640 medium to the desired concentrations.
Flow Cytometric Analysis of Cell Apoptosis
An Annexin V-FITC Apoptosis Detection Kit (BD Pharmingen, San Diego, CA, USA) was used to evaluate the percentage of apoptotic cells according to the manufacturer’s protocols. THP-1 cells (1 × 106 cells/mL) were seeded into a 24-well plate and incubated at 37°C overnight. Subsequently, the cells were incubated with TAIIA and/or 3MA for 72 hours. Cultured cells were centrifuged at 1200 rpm at room temperature for 5 minutes, followed by washing with ice-cold phosphate-buffered saline (PBS) 3 times. Afterward, pelleted cells were stained with Annexin V and propidium iodide (PI) (Sigma-Aldrich, Missouri, St. Louis, MO, USA) in apoptosis binding buffer for 15 minutes following the manufacturer’s instructions. Flow cytometric analysis was performed using a FACSVerse instrument (BD Bioscience, San Diego, CA, USA), and the data were analyzed using FlowJo software (Tree Star, Ashland, OR, USA).
Flow Cytometry Analysis of the Cell Cycle
Cells (1 × 106 cells/mL) were incubated with TAIIA and/or 3MA for 72 hours, washed with ice-cold PBS 3 times, and then fixed with 70% ethanol at 4°C for at least 2 hours. Subsequently, the cells were incubated with RNAase (Biokit, Miaoli, Taiwan) to degrade endogenous RNA at room temperature for 15 minutes. Afterward, the cells were centrifuged at 1200 rpm at 4°C for 5 minutes, followed by the addition of 300 µL of propidium iodide solution (Sigma–Aldrich, St. Louis, MO, USA). Cell cycle status was detected by flow cytometry.
Measurement of Oxidative Stress
THP-1 cells (1 × 106 cells/mL) were incubated with TAIIA and/or 3MA for 3, 6, and 24 hours. The level of glutathione peroxidase (GPx) activity and malondialdehyde (MDA) of the cells and total nitrate/nitrite of the cell suspensions were measured using commercial kits (BioVision, Milpitas, CA, USA) according to the manufacturer’s instructions. A colorimetric analysis was performed using a microplate reader (BioTek, Winooski, VT, USA) and calculated from the standard curve.
Flow Cytometry Analysis of Active Caspase-3
THP-1 cells (1 × 106 cells/mL) were seeded into a 24-well plate and incubated at 37°C overnight. After that, the cells were incubated with TAIIA and/or 3MA for 3, 6, and 24 hours. Cultured cells were centrifuged at 1200 rpm at room temperature for 5 minutes followed by washing with ice-cold PBS 3 times. Afterward, pelleted cells were stained with anti-active caspase-3-FITC (BD Pharmingen, San Diego, CA, USA) and PI in 500 μL apoptosis binding buffer for 15 minutes and analyzed by flow cytometry using a FACSVerse instrument, and the data were analyzed using FlowJo software.
Western Blot Analysis
Cells were washed with PBS twice and lysed in cell lysis buffer. Cell lysates were clarified by centrifugation at 12 000 rpm for 10 minutes at 4°C. The protein contents in the supernatant were measured by using a BCA Protein Assay Kit (Thermo Fisher Scientific, Waltham, MA, USA). The supernatants were mixed with loading buffer (Thermo Fisher Scientific, Waltham, MA, USA) and boiled for 10 minutes. The extracted cell total protein was loaded and separated by 10% SDS–PAGE, and the proteins were transferred onto polyvinylidene fluoride (PVDF) membranes. The membranes were blocked with PBST solution containing 5% FBS at room temperature for 1 hour. Next, the membranes were incubated with the following primary antibodies overnight at 4°C: anti-P53 (Cell Signaling Technology, Danvers, MA, USA) (1:2000), anti-Bax (Cell Signaling Technology, Danvers, MA, USA) (1:1500), anti-Bcl-2 (Cell Signaling Technology, Danvers, MA, USA) (1:1500), anti-Bcl-xL (Cell Signaling Technology, Danvers, MA, USA) (1:1500), anti-PARP (Cell Signaling Technology, Danvers, MA, USA) (1:2000), and anti-GAPDH (Proteintech, Rosemont, IL, USA) (1:10 000). After washing, the PVDF membrane was incubated with secondary antibody (Abcam; ab7090) (1:5000) for 1 hour. Finally, the PVDF membranes were incubated with ECL reagent (Santa Cruz Biotechnology, Dallas, TX, USA) to detect the blots. The intensity of the proteins was measured by Bio–Rad’s Image Lab software.
Statistical Analysis
GraphPad Prism 6.0 software (Graph Pad Software Inc., San Diego, CA, USA) was used for the statistical analysis. The results are expressed as the mean ± SD. Differences were assessed by Student’s t test. P < .05 was considered statistically significant.
Results
Effects of Tanshinone IIA on the Apoptosis of THP-1 Cells
THP-1 is a human monocytic leukemia cell line widely used as a model for AML. To investigate the effect of TAIIA on the apoptosis of THP-1 cells, Annexin V/PI staining was applied. TAIIA dose-dependently induced apoptosis in THP-1 cells compared with the vehicle control group. Treatment with 100 μM TAIIA increased the number of apoptotic cells (Annexin V+ cells) by approximately 14% and more than 2.6-fold compared to the vehicle control group (Figure 1A). Furthermore, we used PI staining and flow cytometry to analyze the cell cycle. The percentage of the cell cycle in the G2 phase was decreased and the percentage in the sub-G1 phase was increased in a dose-dependent manner after TAIIA treatment for 72 hours (Figure 1B). Moreover, we also examined the cytotoxicity of THP-1 cells by TAIIA treatment for 24 hours. The results showed us that only 100 µM TAIIA had a cytotoxic effect on THP-1 cells at 24 hours and without a dose-dependent manner (Supplemental Figure 1). Therefore, subsequent experiments analyzing the cytotoxicity of TAIIA treatment on THP-1 cells were all treated for 72 hours. These data suggested that TAIIA treatment significantly induced apoptosis in THP-1 cells.

Apoptosis and cell cycle progression of TAIIA-treated THP-1 cells. (A) THP-1 cells were treated with 10 to 100 μM TAIIA for 72 hours, and apoptotic cells were evaluated by Annexin V/PI double staining with flow cytometry. The bar charts show the percentage of Annexin V+ apoptotic cells. (B) Cell cycle distribution was assessed for THP-1 cells treated with 50 and 100 μM TAIIA for 72 hours and analyzed by flow cytometry. The bar charts show the percentage of cells in the indicated phases of the cell cycle, and the graph depicts the cell distribution in various phases of the cell cycle. The results are expressed as the means ± SD. *P < .05, **P < .01, ***P < .001 versus vehicle control (VC). The data shown are representative of 2 independent experiments.
Effects of Tanshinone IIA on Oxidative Stress in THP-1 Cells
Oxidative stress is an unbalanced state between the production of ROS and the efficiency of antioxidants. Therefore, we detected the levels of MDA, nitrate/nitrite and antioxidant defense enzymes (GPx) to determine the oxidative stress level after treating THP cells with TAIIA. MDA, an index of lipid peroxidation, was significantly increased after the 100 μM TAIIA treatment for 6 and 24 hours (Figure 2A). Nitric oxide (NO) is a type of ROS, and its high expression can induce oxidative stress in the body. Nitrite and nitrate are inert end products of NO metabolism, and they were significantly increased after the 100 μM TAIIA treatment for 3, 6, and 24 hours (Figure 2B). GPx activity, an enzyme family with peroxidase activity, was significantly decreased after the 100 μM TAIIA treatment for 24 hours (Figure 2C). These data indicated that TAIIA acted as an inducer of oxidative stress in THP-1 cells by enhancing MDA and nitrate/nitrite production and reducing GPx activity.

Oxidative stress in THP-1 cells after the TAIIA treatments. THP-1 cells were treated with 100 μM TAIIA for 3, 6, and 24 hours. The levels of malondialdehyde (MDA) (A) and glutathione peroxidase (GPx) activity (C) from cell lysates were determined by MDA colorimetric assay and GPx activity colorimetric assay. (B) Level of nitrate/nitrite from cell suspensions was determined by a nitric oxide colorimetric assay. The results are expressed as the means ± SD. *P < .05, **P < .01 versus vehicle control (VC). The data shown are representative of 2 independent experiments.
Effects of Tanshinone IIA Combined With an Autophagy Inhibitor on Apoptosis of THP-1 Cells
Several studies have demonstrated that autophagy supports leukemia cell proliferation in vitro and leukemic progression in vivo. 34 Moreover, traditional cytotoxic agents combined with autophagy inhibitor treatment could improve clinical outcomes and reduce drug toxicity in AML therapy.35,36 Therefore, we combined TAIIA with the autophagy inhibitor 3MA to identify whether this combination has higher cytotoxicity in leukemia cells. As shown in Figure 3A, TAIIA combined with 3MA treatment induced apoptosis of THP-1 cells in a dose-dependent manner and presented a significantly higher apoptosis level compared with the TAIIA treatment alone. Furthermore, the percentage of the cell cycle in the G2 phase was decreased and the percentage in the sub-G1 phase was increased in a dose-dependent manner after TAIIA treatment or TAIIA combined with 3MA treatment for 72 hours. The effect of TAIIA combined with 3MA treatment on the cell cycle was significantly greater than that of the TAIIA treatment alone (Figure 3B). These data suggest that TAIIA combined with 3MA treatment had an additive effect on the cytotoxicity of THP-1 cells.

Apoptosis and cell cycle progression of THP-1 cells treated with single or combined treatments. (A) THP-1 cells were treated with 50 and 100 μM TAIIA combined with or without 5 mM 3MA for 72 hours, and apoptotic cells were evaluated by Annexin V/PI double staining with flow cytometry. (B) Cell cycle distribution was assessed for THP-1 cells treated with 50 and 100 μM TAIIA combined with or without 5 mM 3MA for 72 hours and analyzed by flow cytometry. The right panels of Figure 3A and B show the statistical results of the triplicate. The bar charts of Figure 3A show the percentage of Annexin V+ apoptotic cells. The bar charts of Figure 3B show the percentage of cells in the indicated phases of the cell cycle. The results are expressed as the means ± SD. *P < .05, **P < .01, ***P < .001 versus vehicle control (VC). #P < .05, ##P < .01, ###P < .001 versus the same concentration of TAIIA treated alone. ++P < .01 versus the same concentration of 3MA treatment. The data shown are representative of 2 independent experiments.
Effects of Tanshinone IIA Combined With an Autophagy Inhibitor on Apoptosis-Related Proteins in THP-1 Cells
To further elucidate the combined effects on apoptosis of THP-1 cells induced by TAIIA and 3MA, the protein levels of the apoptosis-related proteins p53, Bax, Bcl-xL, Bcl-2, PARP/cleaved PARP, and caspase-3/cleaved caspase-3 were investigated by immunoblotting or flow cytometry. Compared with the TAIIA treatment alone, the combination of TAIIA and 3MA resulted in higher p53 and Bax protein levels after 30 minutes of treatment. Although there was no significant difference in Bcl-xL and Bcl-2 protein levels between the TAIIA treatment alone and TAIIA combined with 3MA treatment for 1 hour, the ratios of Bax/Bcl-xL and Bax/Bcl-2 were higher in the combined treatment group (Figure 4A and B). Furthermore, the downstream proteins of apoptosis, that is, cleaved caspase-3 (active caspase-3) and cleaved PARP, were higher in the combined treatment compared with TAIIA alone at 24 hours (Figure 5A-D). These results suggested that inhibition of autophagy could enhance TAIIA-induced apoptosis via the p53, Bax/Bcl, PARP and caspase-3 apoptotic pathways in THP-1 cells.
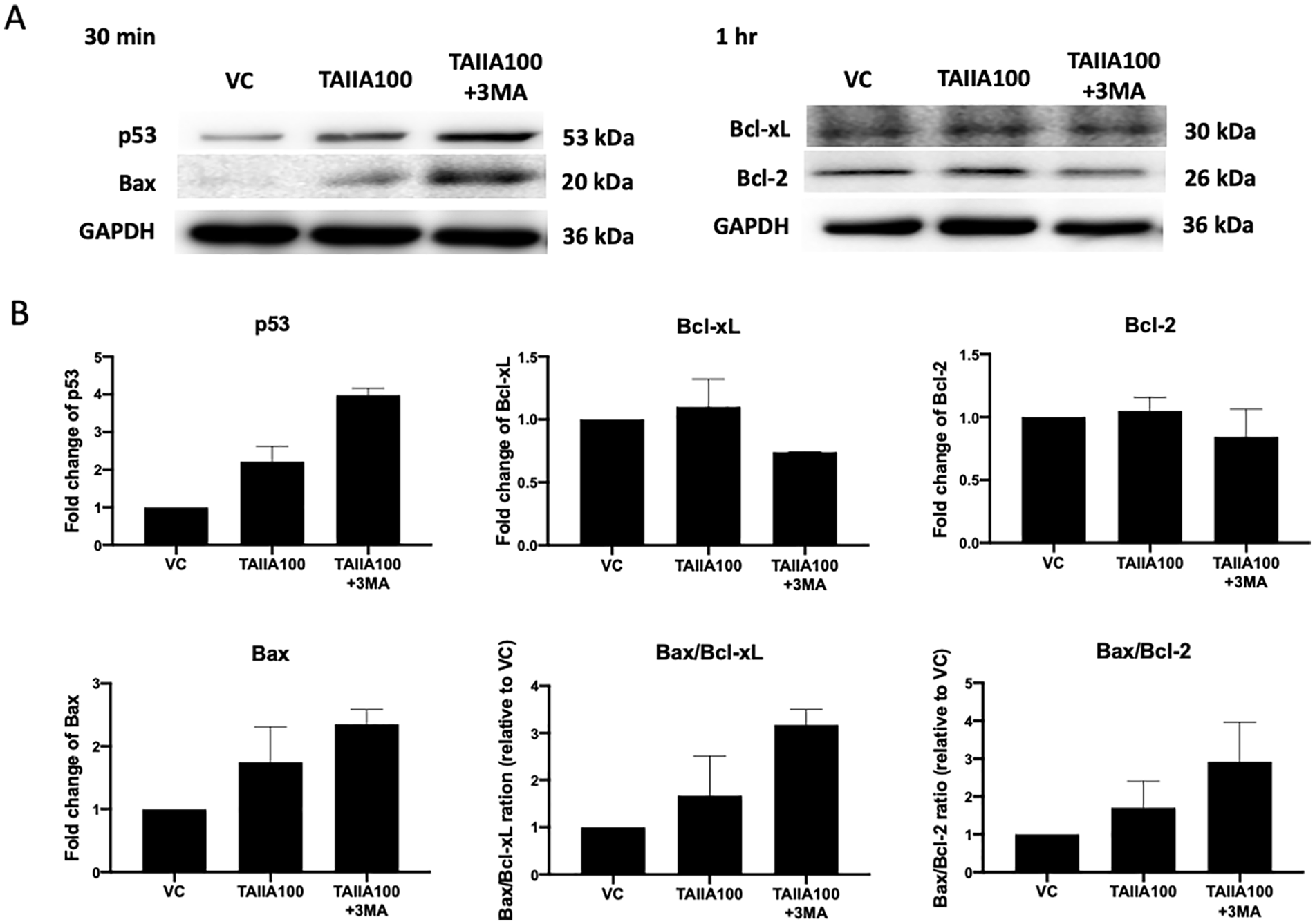
Apoptosis-related protein expression after single or combined treatments in THP-1 cells. Lysates were collected from 100 μM TAIIA combined with or without 5 mM 3MA-treated cells at the indicated time points. (A) Expression levels of p53, Bax, Bcl-xL, Bcl-2, and GAPDH were determined by western blotting. Western blotting for GAPDH was used as a loading control. (B) Intensities of p53, Bax, Bcl-xL, and Bcl-2 were normalized to that of GAPDH, and the results are reported as the fold change over the vehicle control (VC). The results are expressed as the means ± SD. The data shown are representative of 2 independent experiments.

PARP and caspase-3 expression after single or combined treatments in THP-1 cells. Cells were collected from treatments with 100 μM TAIIA combined with or without 5 mM 3MA at the indicated time points. (A) Level of active caspase-3 was determined by flow cytometry. (B) Bar charts show the percentage of active caspase-3 + cells. (C) Expression levels of PARP, cleaved PARP, and GAPDH were determined by western blotting. (D) Intensities of PARP and cleaved PARP were normalized to that of GAPDH, and the results are reported as the fold change over the vehicle control (VC). The results are expressed as the means ± SD. *P < .05, **P < .01, ****P < .0001 versus the vehicle control (VC). The data shown are representative of 2 independent experiments.
Effects of Tanshinone IIA Combined With an Autophagy Inhibitor on Oxidative Stress in THP-1 Cells
Previous studies have shown that overexpression of p53 leads to the upregulation of pro-oxidant enzymes as well as oxidative stress and consequently to apoptosis.37,38 We demonstrated that TAIIA treatment can increase p53 expression and oxidative stress and showed that the levels of p53 expression and apoptosis were higher upon combination with 3MA (Figures 3 and 4). Furthermore, we confirmed whether treatment with TAIIA combined with the autophagy inhibitor 3MA could enhance oxidative stress in THP-1 cells. When THP-1 cells were treated with 3MA alone (VC+3MA), significant differences were not observed in the levels of MDA (Figure 6A), nitrate/nitrite (Figure 6B), or GPx activity (Figure 6C). However, the levels of MDA, nitrate/nitrite, and GPx activity were increased significantly over different time courses after TAIIA treatment alone. Moreover, TAIIA combined with 3MA promoted higher MDA (Figure 6A) and nitrate/nitrite expression levels (Figure 6B) and lower GPx activity (Figure 6C) in THP-1 cells. These data suggested that TAIIA combined with 3MA had an additive effect on the cytotoxicity of THP-1 cells, which may be caused by the enhancement of p53 expression and oxidative stress.

Oxidative stress in THP-1 cells induced by single or combined treatments. THP-1 cells were treated with 100 μM TAIIA combined with or without 5 mM 3MA for 3, 6, and 24 hours. The levels of malondialdehyde (MDA) (A) and glutathione peroxidase (GPx) activity (C) from cell lysates were determined by MDA colorimetric assay and GPx activity colorimetric assay. (B) Level of nitrate/nitrite from cell suspensions was determined by a nitric oxide colorimetric assay. The results are expressed as the means ± SD. *P < .05, **P < .01 versus the vehicle control (VC). #P < .05 versus the same concentration of TAIIA treated alone. The data shown are representative of 2 independent experiments.
Discussion
In AML patients, the 5-year estimated overall survival for patients (measurable residual disease) was 34% and disease-free survival was 25%. 39 Cytotoxic chemotherapy and hypomethylating agents are 2 of the most commonly used induction therapies in AML. 40 In recent years, the outcomes have improved with the addition of various targeted drugs to conventional chemotherapy. Venetoclax is an oral targeted drug that is a highly selective inhibitor of the anti-apoptotic protein Bcl-2. The outcomes of venetoclax combined with hypomethylating agents were better than those of hypomethylating agents alone. 41 In these previous studies, the combination therapy significantly improved the historical median survival of approximately 10 months compared with the single drug. 42 In our previous study, we identified Salvia miltiorrhiza as the most commonly prescribed single herb for the treatment of AML in Taiwan. 25 In the present study, we further identified that treatment with TAIIA, one of the ingredient compounds of Salvia miltiorrhiza, alone could induce cancer cell apoptosis, enhance apoptosis-related protein Bax, and increase Bax/Bcl-xL and Bax/Bcl-2 ratios, but there was no difference in Bcl-xL and Bcl-2 protein levels. However, TAIIA combined with autophagy inhibitor treatment not only induced higher Bax protein levels and Bax/Bcl-xL and Bax/Bcl-2 ratios but also decreased Bcl-xL protein levels. Therefore, the combination therapy had a higher cytotoxic ability against cancer cells than the TAIIA treatment alone.
Several studies have shown that chemotherapeutic drugs widely used in the treatment of AML, such as cytarabine and daunorubicin, can induce autophagy as a survival mechanism against cytotoxic stress and counteract the therapeutic effects of these drugs.35,43 Therefore, autophagy has an important role in AML therapy and targeting autophagy may be a viable strategy against AML. Accordingly, we analyzed the effects on autophagy after applying a combination of TAIIA and 3MA. We found that the TAIIA treatment alone increased the expression of the autophagy-related proteins Beclin-1 and LC3 I and that the combined TAIIA and autophagy inhibitor 3MA treatment reversed the protein levels of Beclin-1 and LC3 I (Supplemental Figure 2). This result also confirms our finding that TAIIA combined with autophagy inhibitor indeed increased apoptosis in THP-1 cells compared to the TAIIA treatment alone.
Notably, high ROS levels are a common hallmark of AML cells, which makes them more sensitive to pro-oxidative treatments than normal cells. 14 Our study found that MDA and nitrite/nitrate were significantly increased after 6 hours of TAIIA treatment while GPx activity was significantly decreased after 24 hours of TAIIA treatment. Moreover, TAIIA combined with 3MA treatment promoted higher expression levels of MDA and nitrate/nitrite and lower GPx activity in THP-1 cells. p53 has been demonstrated to induce Bax and PUMA to enhance mitochondrial uncoupling, thus leading to the production of ROS. 44 Our results indicated that the expression of p53, Bax and ROS was enhanced and a higher incidence of apoptosis occurred after THP-1 cells were treated with TAIIA and 3MA in combination. However, p53 can affect cellular ROS production and ROS can also regulate p53 expression. Based on the chronological order of the observed effects in our study, the results suggest that p53 may be upstream of ROS. Therefore, an approach using a combination of therapeutic agents that simultaneously activate p53 and ROS has the potential to be applied in clinical anticancer therapy. The proposed mechanisms of action of the combined TAIIA and 3MA treatment in THP-1 cells are shown in Figure 7, and they are arranged in chronological order after drug treatment (Figure 7).
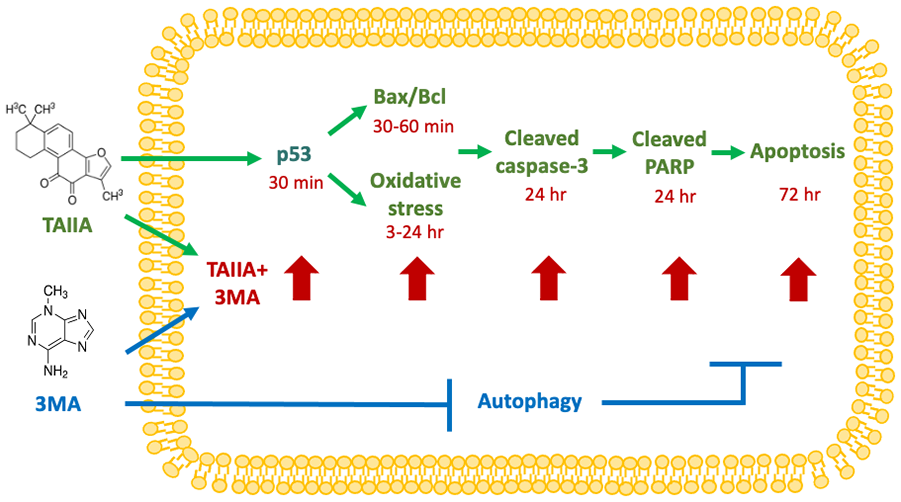
Proposed mechanism of action of TAIIA and 3MA treatment in THP-1 cells.
Many studies have confirmed that TAIIA has toxic effects on some cancer cells, mainly related to the activation of caspase-3, the downregulation of anti-apoptotic proteins Bcl-2 and survivin, and the up-regulation of pro-apoptotic protein Bax.45,46 We also confirmed a similar mechanism in THP-1 cells by TAIIA treatment and further demonstrated the combined effect of autophagy inhibitor 3MA and TAIIA treatment of THP-1 cells. Moreover, a recent study also found that the combination of autophagy inhibitor 3MA and TAIIA could enhance the induction of apoptosis in U937 cells. 47 However, although both studies aimed to treat leukemia, we used different cell line. U-937 cells were obtained from the pleural effusion of a 37-year-old, white, male patient with histiocytic lymphoma. THP-1 cells were derived from the peripheral blood of a 1-year-old male with acute monocytic leukemia. At present, the treatments for AML patients of heterogeneous and different age groups are different, which result in different efficacies. 5 In addition to the difference in cell lines, we newly discovered that p53 could be affected by the combination of autophagy inhibitor 3MA and TAIIA.
Conclusions
Our study demonstrated that the combination of TAIIA and the autophagy inhibitor 3MA had an additive effect on the cytotoxicity of leukemia cells. Compared with TAIIA treatment alone, the combination treatment resulted in higher levels of apoptosis and apoptosis-related proteins (Bax, Bax/Bcl, cleaved PARP, and cleaved caspase-3). Meanwhile, TAIIA had additive effects on p53 expression and oxidative stress when applied in combination with 3MA. Therefore, the combination of TAIIA and the autophagy inhibitor 3MA could potentially be developed into a clinical anticancer therapy.
Supplemental Material
sj-tiff-1-ict-10.1177_15347354221117776 – Supplemental material for Combined Effects of Tanshinone IIA and an Autophagy Inhibitor on the Apoptosis of Leukemia Cells via p53, Apoptosis-Related Proteins and Oxidative Stress Pathways
Supplemental material, sj-tiff-1-ict-10.1177_15347354221117776 for Combined Effects of Tanshinone IIA and an Autophagy Inhibitor on the Apoptosis of Leukemia Cells via p53, Apoptosis-Related Proteins and Oxidative Stress Pathways by Ying-Chyi Song, Cheng-Chieh Kuo, Chuan-Teng Liu, Tsai-Chen Wu, Yi-Ting Kuo and Hung-Rong Yen in Integrative Cancer Therapies
Supplemental Material
sj-tiff-2-ict-10.1177_15347354221117776 – Supplemental material for Combined Effects of Tanshinone IIA and an Autophagy Inhibitor on the Apoptosis of Leukemia Cells via p53, Apoptosis-Related Proteins and Oxidative Stress Pathways
Supplemental material, sj-tiff-2-ict-10.1177_15347354221117776 for Combined Effects of Tanshinone IIA and an Autophagy Inhibitor on the Apoptosis of Leukemia Cells via p53, Apoptosis-Related Proteins and Oxidative Stress Pathways by Ying-Chyi Song, Cheng-Chieh Kuo, Chuan-Teng Liu, Tsai-Chen Wu, Yi-Ting Kuo and Hung-Rong Yen in Integrative Cancer Therapies
Footnotes
Acknowledgements
Experiments and data analyses were performed in part through the use of the Medical Research Core Facilities, Office of Research & Development at China Medical University, Taichung, Taiwan. None of the funders and institutions listed had a role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by grants from China Medical University to YCS (CMU108-MF-65) and HRY (CMU107-TU-04), China Medical University Hospital to YTK (DMR-111-173), Ministry of Science and Technology to YCS (MOST 110-2320-B-039-036-) and HRY (MOST 108-2320-B-039-024-MY3), and Ministry of Science and Technology to CCK and HRY (107-2813-C-039-277-B) as well as a grant from “Chinese Medicine Research Center, China Medical University” from the Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education to HRY (CMRC-CHM-2).
Ethics Approval and Consent to Participate
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
