Abstract
Background
Celastrus orbiculatus ethyl acetate extract (COE) has shown a strong anti-gastric cancer effect, but the understanding of its mechanism is still lacking. The results of previous studies indicated that COE may be able to inhibit the stemness of gastric cancer stem cells (GCSCs) by regulating PDCD4 and EIF3H expression.
Aims
To explore if COE could inhibit the stemness of GCSCs by regulating PDCD4 and EIF3H expression in vitro and in vivo.
Procedure
The GCSCs model was established by stem cell-conditioned culture. Spheroid formation and flow cytometry assays were used to detect the effect of COE on the spheroid formation ability of GCSCs and the percentage of CD44+/CD24+ and ALDH+ cell subpopulations. Western blot analysis was applied to measure the expression of GCSCs biomarkers (Nanog, Oct-4, and SOX-2), PDCD4, and EIF3H in GCSCs treated with COE; and RT-PCR was performed to investigate the effect of COE on PDCD4 mRNA expression in GCSCs. An in vivo tumorigenicity experiment was also conducted to evaluate the effect of COE on tumor-initiating ability of GCSCs in vivo; and the expression of PDCD4 and EIF3H in xenograft tissues was examined by immunohistochemistry (IHC) staining.
Results
After culture in stem cell-conditioned medium, SGC7901 cells manifested significantly enhanced spheroid formation ability, upregulated Nanog, Oct-4, and SOX-2 expression and increased percentages of CD44+/CD24+ and ALDH+ cell subpopulations, indicating successful establishment of the GCSCs model. COE treatment significantly inhibited the spheroid formation ability of GCSCs and reduced the percentage of CD44+/CD24+ and ALDH+ cell subpopulations. The western blot analysis showed a significant decrease of Nanog, Oct-4, SOX-2, and EIF3H expression and an increase of PDCD4 expression in GCSCs after COE treatment in a concentration-dependent manner. COE treatment also significantly upregulated the mRNA expression of PDCD4 in GCSCs. In addition, COE displayed a strong inhibitory effect on the tumor-initiating ability of GCSCs in vivo and upregulated PDCD4 and downregulated EIF3H expression in xenograft tissues.
Conclusion
COE may be able to inhibit GC growth by suppressing the stemness of GCSCs via regulating PDCD4 and EIF3H expression.
Introduction
Gastric cancer (GC) is one of the principal diseases seriously threatening human health. According to statistics, in 2018, over 1 million new GC diagnoses were made, ranking it as the fifth most common cancer, and about 800,000 patients died from GC, making it the third leading cause of cancer death worldwide. 1 Due to its occult onset, most GC patients are already at an advanced stage when diagnosed. 2 Despite the rapid development of antitumor therapy in recent years, the curative effect of advanced GC is still far from satisfactory. The 5-year survival rate for patients diagnosed with advanced GC remains less than 30%. 3 Therefore, the demand for the exploration and development of new therapeutic targets and drugs is very urgent.
In recent years, researchers have identified a small fraction of neoplastic cells with stem cell properties, defined as cancer stem cells (CSCs), as a new promising therapeutic target. It was posited that only CSCs within tumors have the ability to self-renew and to produce all the kinds of cells necessary to repopulate a tumor, inducing and sustaining tumorigenesis.4,5 The theory also suggests that tumors containing more CSCs tend to be more refractory and recur more easily after initial remission, and the dissemination and migration of CSCs may be responsible for metastases of the tumor.6-8 Unlike normal cancer cells, CSCs have several unique properties. For example, different from the monolayer flattened growth pattern of normal cancer cells, CSCs grow in a non-adherent, three-dimensional pattern and show a strong sphere-forming capacity. In addition, several CSC-related proteins including Nanog, Oct-4, SOX2, CD44, CD24, and ALDH are overexpressed in the membrane, perinuclear, or cytoplasm of CSCs. 9 Based on the expanding understanding of CSCs biology characteristics, multiple new CSCs-targeted agents have been developed and have shown synergy with various therapies in clinical trials.10-14 However, efforts to develop more suitable therapeutic targets and anti-CSCs agents are still needed to better apply the therapeutic strategy designed based on this new knowledge to cancer therapy.
Results of several studies have indicated that programmed cell death 4 (PDCD4), an mRNA translation regulator with highly conserved amino acid sequences, might function as a stemness suppressor in cancer. Under normal conditions, PDCD4 serves as an mRNA translation inhibitor by inhibiting the activity of eukaryotic initiation factor 4A (eIF4A), an ATP-dependent RNA helicase which is critical for translation initiation, or by directly binding with mRNA to suppress its translation. 15 Besides, since PDCD4 works as a tumor suppressor, overexpression of PDCD4 could inhibit tumor promotion, growth, invasion, metastasis, and induced apoptosis in numerous types of cancers including gastric, lung, colorectal and liver cancers.16-20 Downregulation of PDCD4 expression levels has been found in various tumor tissues and is associated with poor prognosis in patients. 21 It was also reported that loss of PDCD4 may be related to the acquiring and maintaining of stem-cell-like characteristics in certain cancer cells.22-25 Moreover, PDCD4 could blockade the changes in cell phenotype from epithelial to mesenchymal states, termed the epithelial-mesenchymal transition (EMT), a cell-biological program, the activation of which has been demonstrated to be essential for the induction and maintenance of CSCs properties.17,26-28 These findings indicate that restoration or stimulation of PDCD4 expression may impair the stem cell-like characteristics of CSCs.
Eukaryotic initiation factor 3 subunit H (EIF3H), the overexpression of which was correlated with the progression and poor prognosis of patients in several cancers, has been found to be a target gene of PDCD4 in lung cancer.29-31 EIF3H is one of the core subunits of eukaryotic initiation factor 3 (EIF3) and takes part in several steps in the initiation of protein synthesis. 32 It has also been reported that EIF3H could function as an oncogene by activating EMT signaling.30,33
In a previous research, it has been reported that Celastrus orbiculatus ethyl acetate extract (COE), a natural compound extracted from the stem of Celastrus orbiculatus, could reverse the precancerous lesions induced by Helicobacter pylori infection through inhibiting the EMT program via enhancing PDCD4 expression in gastric epithelial cells. 28 Celastrus orbiculatus Thunb has been widely used in traditional Chinese medicine (TCM) to treat various diseases including cognitive dysfunction, joint pain, muscle pain, and dyspepsia for thousands of years. Investigators have so far isolated hundreds of extracts (mainly including β-dihydroagarofuranoids, diterpenoids, triterpenoids, flavonoids) from different parts of celastrus (roots, stem, leaves and seeds). These extracts have shown strong effects, such as antitumor, anti-inflammatory, cytotoxic, and insecticidal activities. 34 Among them, COE has showed remarkable antitumor bioactivity by inducing tumor cell apoptosis mainly through the mitochondrial pathway, or by reducing tumor cell migration and invasion via inhibiting EMT process.35-37 However, the functional role of COE in gastric cancer stem cells (GCSCs) remains unknown.
In this study, we found that COE impaired the spheroid-forming ability of GCSCs and suppressed the expression of Nanog, Oct-4, and SOX-2 in GCSCs. The percentage of CD44+/CD24+ and ALDH+ cell subpopulations was reduced after COE treatment. Further study revealed that COE also inhibited the tumor-initiating ability of GCSCs in vivo. Moreover, western blot and immunohistochemistry (IHC) analysis showed that COE upregulated PDCD4 and downregulated EIF3H expression both in GCSCs and xenografts. Our results indicate that COE could inhibit the stemness of GCSCs by regulating the expression of PDCD4 and EIF3H.
Materials and Methods
Cell Lines and Cell Culture
SGC7901 human GC cells were purchased from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China) and were maintained in Dulbecco’s modified Eagle’s medium (DMEM) (Gibco-BRL, Grand Island, NY, USA) containing 10% fetal bovine serum and 1% penicillin at 37℃, 5% CO2.
Spheroid Colony Formation Assay
Spheroid colony formation culture were performed in a 96-well ultra-low attachment plate (Corning, Inc., Tewksbury, MA, USA) (1 × 103 cells per well) supplemented with low-glucose (1 g/L) DMEM with addition of Sodium Pyruvate (1%), l-Glutamine (1%), Penicillin (1%), bFGF (20 ng/mL), EGF (20 ng/mL) and B27 (20 ng/mL) (Invitrogen, Grand Island, NY, USA), at 37℃, 5% CO2. Every 5 days, the spheroids were collected and dissociated into single cells by trypsin for passaging. After passaging the spheroids into the third generation, the spheroid-derived cells were treated with COE for 24 hour in a 96-well ultra-low attachment plate (200 cells per well). Then all cells were harvested and were resuspended in serum-free stem cell culture media. After culture for 14 days, the number of spheroids (>100 μm in diameter) was counted. The spheroid-formation rate was calculated as spheroids/input cells × 100%.
Preparation of COE
The Celastrus orbiculatus stem species were purchased from Guangzhou Zhixin Pharmaceutical Co, Ltd. (Guangzhou, China). The preparation procedure of COE has been reported in a previous study. 38 The resultant COE micropowder was dissolved in dimethyl sulfoxide (DMSO, Sigma-Aldrich Co., St Louis, MO, USA) and diluted to 80 μg/ml for subsequent research.
Flow Cytometry Analysis
To examine whether the amount of ALDH+ and CD44+CD24+ cells were increased in spheroid-forming cells, flow cytometry analysis was performed. Dissociated cells were washed twice with PBS and were resuspended in PBS before labeling with CD44 (1:200; BD Biosciences, San Jose, CA) and CD24 (1:200; Biomeda, Foster City, CA) or ALDH-1 (1:1000; BD Biosciences, San Jose, CA). After adding antibodies, the cells were incubated in an Eppendorf tube for 15 minutes at 4℃, then washed 3 times, resuspended in PBS and centrifuged for 5 minutes at 1000 rpm. First, an acquisition gate was established based on forward scatter and side scatter (FSC/SSC) to exclude dead cells and cell debris (Figure 1C-i). Then, a gate for sorting ALDH+ cells was drawn and combined with the SSC gate for the selection of GCSCs (Figure 1C-ii). We also established another gate to sort CD44+CD24+ cells for additional selection of GCSCs in separate analyses (Figure 1C-iii). The results of flow cytometry were analyzed on a BD FACS Calibur platform (BD Biosciences).

The GCSCs model was established by stem cell-conditioned culture (A) SGC7901 cells formed anchorage-independent and self-renewal spheroid colonies after stem cell-conditioned medium culture; (B) Nanog, Oct-4, and SOX-2 were overexpressed in spheroid-forming cells (SFCs); the statistical results are shown (n = 3); (C) The percentage of CD44+/CD24+ and ALDH+ cell subpopulations were significantly higher in SFCs than parental cells (PCs); i: dead cells and debris were excluded by SSC/FSC gate selecting; ii: ALDH+ cells were sorted by SSC/ALDH+ gate; iii:CD24+CD44+ cells were selected by CD24+/CD44+ gate.The statistical results are shown (n = 3). *P < .05 versus PCs. **P < .01 versus PCs.
Western Blot Analysis
Cells were lysed with cold radioimmunoprecipitation assay (RIPA) lysis buffer (Beyotime Institute of Biotechnology, Haimen, China) and protein concentrations were determined by Bicinchoninic Acid Protein Assay Kit (Beyotime Institute of Biotechnology). Proteins (50 μg) were separated by 10% SDS-PAGE and then were transferred to PVDF membranes (Millipore, Billerica, MA, USA). Then, the PVDF membranes were blocked in 5% nonfat milk for 2 hours at room temperature and incubated with the following monoclonal antibodies at 4℃ overnight: Nanog (1:500; Santa Cruz Biotechnology, Santa Cruz, CA), Oct-4 (1:500; Cell Signaling Technology, Danvers, MA), SOX2 (1:500; Abcam, UK), PDCD4 (1:1000; Abcam, UK), EIF3H (1:1000; Cell Signaling Technology, Danvers, MA ), and β-actin (1:500; Santa Cruz Biotechnology). Membranes were then washed 4 times in TBST, followed by incubation with goat anti-mouse HRP secondary antibody (1:2000; Millipore, Billerica, MA, USA) for 1h at room temperature. Membranes were washed again and were visualized with the DAB reagent (Millipore, Billerica, MA, USA). The protein bands were analyzed by GeneSnap software from SynGene (Cambridge, UK).
RT-PCR
Total RNA was isolated from cells using Trizol reagent (Life Technologies, USA) according to the manufacturer’s instructions. Primers specific for PDCD4 was as follows: forward primer 5ʹ-GCAAAAAGGCGACTAAGGAAAAA-3ʹ, reverse primer 5ʹ-TAAGGGCGTCACTCCCACT-3ʹ. β-actin was applied as internal normalization control (forward primer 5ʹ-CATGTACGTTGCTATCCAGGC-3ʹ, reverse primer 5ʹ-CTCCTTAATGTCACGCACGAT-3ʹ). RT-PCR was performed using the ABI 7500 Real-Time PCR System (Applied Biosystems). The results were calculated using the 2−ΔΔCt method. 39
Animals
In the present study, 6-week-old male BALB/c thymic nude mice with SPF grade, weight 20 ± 2 g, were selected for in vivo experiments. The animals were provided by Vitong Lihua Laboratory Animal Technology Co. Ltd (Beijing, China) and were kept in a laminar flow cabinet in a barrier system in the laboratory animal center of Anhui Medical University. All animal experiments were approved by the Animal Ethics committee of Anhui Medical University.
In Vivo Tumorigenicity Experiments
To examine whether COE treatment could inhibit the tumorigenicity of GCSC in nude mice xenograft model, 1 × 106 GCSCs were harvested and suspended in 100μl PBS before being inoculated subcutaneously at the right flank region of each mouse. One week after transplantation, masses could be palpated in the inoculated site, indicating that xenograft model was successfully established. Then, mice were divided into 5 groups (5 per group). Non-treated mice served as controls. The remaining mice were divided into the DMSO group, COE-L (COE low dose, 10 mg/kg/day) group, COE-M (COE medium dose, 20 mg/kg/day) group and the COE-H (COE high dose, 40 mg/kg/day) group, and were administered with different doses of COE intragastrically every day. The tumor size was measured and recorded every 7 days and tumor volume was calculated as the longest diameter × shortest diameter 2 /2. A 50 days after transplantation, all mice were sacrificed by cervical dislocation and tumor tissues were removed and paraffin-embedded for immunohistochemical staining.
IHC Staining Analysis
The paraffin sections were deparaffined and dehydrated, rehydrated, and endogenous peroxidase activity blocked. Subsequently, the sections were incubated with primary antibodies against PDCD4 (1:200; Abcam, UK) and EIF3H (1:1000; Cell Signaling Technology, Danvers, MA) overnight at 4°C. After PBS washing, the sections were incubated with horseradish peroxidase (HRP)-conjugated secondary antibody, and the staining was developed with DAB substrate chromogen (Maixin Biotech. Co., Ltd., Fuzhou, China) followed by hematoxylin counterstaining.
Statistical Analysis
All data in this text are shown as mean ± standard deviation (SD). Student’s t-test was applied to evaluate the significance of the difference between data and two-sided P < .05 was considered as statistical difference. All statistical analyses were conducted using the SPSS 16.0 software (SPSS, Inc., Chicago, IL, USA).
Results
The Establishment of the GCSC Model
The SGC-7901 cells cultured in stem cell-conditioned medium showed a non-adherent, three-dimensional growth pattern and formed spheroid colonies. The cell spheroids were maintained in culture for more than 15 days and were passaged 3 times (Figure 1A). The results of western blot analysis showed that the protein expression of Nanog, Oct-4, and SOX-2 were significantly higher in spheroid-forming cells than in parental cells (Figure 1B). The flow cytometry assay showed that the percentage of CD24+/CD44+ and ALDH+ cell subpopulations were significantly higher in spheroid-forming cells than in parental cells (Figure 1C). These results indicated that the spheroid-forming cells obtained CSC-like properties, and the model of GCSCs was successfully established.
COE Suppressed Sphere Formation and CSC Markers Expression in GCSC-like Cells via Regulating PDCD4 and EIF3H Expression
The spheroid colony formation assay showed that COE treatment reduced the spheroid-formation rate of GCSCs in a concentration-dependent manner (Figure 2A). The results of western blot analysis showed a significant decrease in the protein expression of Nanog, Oct-4, SOX-2 and EIF3H and an upregulation of PDCD4 in the GCSCs treated with COE (Figure 2B). RT-PCR analysis showed that COE could significantly upregulate mRNA expression of PDCD4 in GCSCs in a dose-dependent manner (Figure 2C). The results of flow cytometry analysis showed that COE treatment significantly reduced the percentage of CD24+/CD44+ and ALDH+ cell subpopulations (Figure 2D).
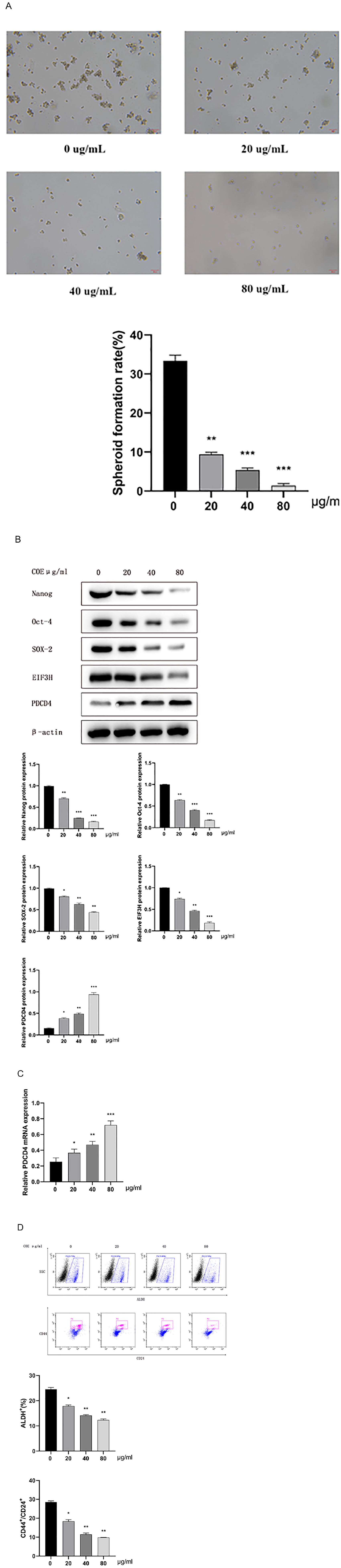
COE inhibited the stemness of GCSCs via regulating PDCD4 and EIF3H expression (A) COE inhibited the spheroid-formation ability of GCSCs; The statistical results are shown (n = 3); (B) COE inhibited the protein expression of Nanog, Oc-4, and SOX-2 in GCSCs, and the statistical results are shown (n = 3); (C) COE upregulated PDCD4 mRNA expression in GCSCs, and the statistical results are shown (n = 3); (D) COE significantly reduced the percentage of CD44+/CD24+ and ALDH+ cell subpopulations, and the statistical results are shown (n = 3). *P < .05. **P < .01. ***P < .001.
COE Inhibited the Tumorigenicity of GCSCs In vivo
The results of the in vivo tumorigenicity experiments showed that the volume of subcutaneous tumors in the COE-treated groups was significantly smaller than that in the control group (Figure 3A).The IHC analysis revealed that the expression of PDCD4 was significantly higher, while the expression of EIF3H was significantly lower in COE-treated groups (Figure 3B).

COE inhibited the tumor-initiating capacity of GCSCs. (A) The growth curves of xenografts; (B) COE upregulated PDCD4 expression and downregulated EIF3H expression in xenograft tissue. COE-H: COE high dose; COE-M: COE medium dose; COE-L: COE low dose. *P < .05. **P < .01.
Discussion
Natural compounds extracted from plants have drawn much interest in the development of anticancer agents owing to their wide pharmacological applications, rich sources, multitarget activity, and low side effects. Nevertheless, the clinical application of these extractions has been hampered seriously by an insufficient understanding of the mechanisms of how they work. In this study, we demonstrated for the first time that COE could inhibit gastric cancer growth by suppressing the stemness of GCSCs via regulating PDCD4 and EIF3H expression.
Gastric cancer cell lines could be divided into 2 types: the differentiated type (similar to intestinal metaplasia mucosa) and undifferentiated type (similar to intrinsic gastric mucosa). The SGC-7901 cell line is a undifferentiated gastric cancer cell line with high metastatic capacity. 40 In our pre-experiment, spheroid-forming cells (SFCs) were successfully isolated from the SGC-7901 cell line and showed a series of CSC-like properties, including a higher tumorigenicity, colony-forming capacity, and upregulation of CSC biomarkers expression (Nanog, Oct-4, and SOX-2). We also found that expression of PDCD4 in SFCs was significantly downregulated compared with normal SGC-7901 cells, indicating that GCSCs may obtain CSC-like properties by downregulating PDCD4 expression. Therefore, referring to relevant literatures and the results of previous research, we selected SFCs isolated from SGC-7901 cell line for further studies.28,41,42
Spheroid body culture has been widely applied as a method for enrichment of CSCs from primary tumor cell lines according to their property of anchorage-independent growth.43,44 Therefore, in the present study we developed spheroid-forming cells by cultivating the SGC7901 cell line in defined serum-free medium. The cells derived from spheroid bodies showed great spheroid formation ability and were passaged 3 generations, indicating these cells acquired substantial self-renewal capacity which is an important characteristic of CSCs. 9 To further examine the CSC properties of these cells, we measured the expression of Nanog, OCT-4, SOX-2, CD44, CD24, and ALDH, which have been shown to be biomarkers of GCSCs. 45 The western blot and flow cytometry analysis showed an overexpression of Nanog, Oct-4, and SOX-2, and the percentage of CD44+/CD24+ and ALDH+ cell subpopulations were significantly increased in these cells. These data indicate the GCSC model has been successfully established. To assess the effect of COE on GCSCs, we treated GCSCs with COE in different concentrations. After treatment with COE for 24 hour, the spheroid formation ability of GCSCs was impaired and the expression of Nanog, Oct-4, and SOX-2 in GCSCs was significantly downregulated; the percentage of CD44+/CD24+ and ALDH+ cell subpopulations were reduced. The results of western blot and RT-PCR assay showed that both protein and mRNA level of PDCD4 were upregulated significantly after COE treatment, indicating that COE may be able to impair the stemness of GCSCs by enhancing PDCD4 expression in vitro. The high tumorigenicity is another critical property of GCC, thus the in vivo tumorigenicity experiment was conducted to evaluate the effect of COE on tumor-initiating ability of GCSC. The results showed that the volume of subcutaneous tumors was significantly smaller in COE-treated groups than in the control group, indicating that COE may be able to inhibit the tumorigenicity of GCSC. In addition, we found that PDCD4 was upregulated and EIF3H was downregulated after COE treatment both in GCSCs and xenograft tissues. These results indicate that COE could inhibit the stemness of GCSCs and the mechanism may be related to its regulation of PDCD4 and EIF3H expression.
PDCD4 is a tumor suppressor, localized in the nucleus of cells, and is responsible for the inhibition of the growth, invasion, and metastasis of GC. Downregulation of PDCD4 is closely related to the progression and development of GC. It was reported that Helicobacter pylori infection, which is recognized as the most important risk factor for gastric carcinogenesis, can downregulate PDCD4 expression by enhancing DNA methylation and inducing EMT in GC cells.28,46,47 MicroRNAs (miRNAs) are short non-coding RNAs consisting of 21 to 25 nucleotides that negatively regulate the expression of target genes. It has been found that a number of miRNAs can contribute to GC by targeting PDCD4 directly, including miR-21, miR-23a/b, miR-93, and miR-183.48-51 Shi et al 52 showed that the blockade of miR-21 maturation could reverse EMT and suppress growth and progression of GC via upregulating PDCD4 expression. In suppressing the uncontrolled proliferation of GC cells, PI3K/Akt signaling pathway is a potential downstream target of PDCD4. Overexpression of PDCD4 could induce apoptosis in GC cells by inhibiting the PI3K/Akt pathway. 53
Although no study has investigated the effect of PDCD4 overexpression on CSC-like properties, downregulation of PDCD4 has been found to be correlated with the acquiring of CSC-like properties in several cancer cell lines.17,26-28 Some studies also have reported that PDCD4 can regulate certain signaling pathways or target genes whose abnormal activation is vital for obtaining or maintaining the CSC phenotype. Cyclin-dependent kinases (CDKs) are a family of serine/threonine protein kinases which play an important role in cell cycle regulation, and the inappropriate activation of CDKs is closely related to the self-renewal of CSCs. 54 Researchers found that PDCD4 can inhibit the expression of CDK1, CDK2, CDK4, and CDK6 in multiple tumors, and the mechanism may be related to the induced effect of PDCD4 on the expression of p21Waf1/Cip1, a CDK inhibitor.55,56 The Wnt pathway is a self-renewal and differentiation-related pathway in stem cells and is normally under tight control. Its aberrant activation is vital for obtaining and maintaining the CSC phenotype. 57 The study of Kakimoto et al showed that PDCD4 may inhibit gastric carcinogenesis at least partly by blocking the canonical WNT pathway.23,58 Moreover, PDCD4 could also target multiple genes that are critical for regulating the CSC phenotype like c-Myc, Bcl-2, CCND1.59,60 All these results suggest that PDCD4 may be an inhibitor of CSCs.
EIF3H is one of the non-conserved subunits of the EIF3 complex which is significantly upregulated in GC and is strongly associated with rapid proliferation of GC. Silencing of EIF3H arrested the cell cycle in the G0/G1 phase and led to apoptosis of GC cells. 31 It has been reported that overexpression of PDCD4 in lung cancer reduced EIF3H expression by inhibiting c-Jun-induced EIF3H transcription. 30 Chu et al 61 found that EIF3H was significantly upregulated in melanoma stem cells than cancer non-stem cells. It was also reported that EIF3H could enhance the Hippo signal by stabilizing YAP, and that it promotes breast cancer invasion and metastasis. 62 Given the impact of the Hippo-YAP pathway in the renewal of CSCs, we suggested that upregulation of EIF3H may contribute to the maintenance and renewal of CSCs.
In this study, we performed a preliminary exploration of the effect of COE treatment on CSC properties of GCSCs, and observed changes of PDCD4 and EIF3H expression in GCSCs after COE treatment. Our results may be able to provide reference for further research. However, a non-negligible limitation is that only one cell line (SGC-7901) was utilized in this study, which may dampen the enthusiasm of generalizing the results to different types of gastric cancer. Fortunately, multiple GC cell lines (MGC-802, BGC-823, AGC) have been applied in previous research to investigate the effect of COE treatment on the growth, invasion, and metastasis of GC.63-65 Relevant results indicated that COE could effectively inhibit the growth, invasion, and metastasis of GC in different GC cell lines, which may be able to partially compensate the lack of study. At present, our research group is isolating and identifying CSC-like cells using the HGC-27 cell line to explore the effect of COE on the differentiation of GCSCs. The results will be reported in another article that will be published later.
In conclusion, COE may be able to inhibit the stemness of GCSCs by regulating PDCD4 and EIF3H expression.
Footnotes
Author contribution
Yao-dong Zhu, Ping Li and Mei Zhang designed the study and guided the project, and He Ba performed the experiments and wrote the manuscript, and Jie Chen analyzed data and prepared figures. All authors have reviewed and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Natural Science Foundation of the Anhui Higher Education Institutions of China (KJ2020A0215); Cancer research fund of CSPC PHARMA (Y-SY201901-0067); Special fund for clinical research of Wu Jieping Medical Foundation (320.6750.2020-10-25)
Data Availability Statement
The data that support the findings of this study are openly available.
