Abstract
Introduction
Saliva, which consists of water (99%) and many electrolytes, immunoglobulins, proteins, enzymes, mucins, and nitrogenized products, 1 serves important functions in maintaining the health of the oral cavity, speech, ingestion, and swallowing.2,3 Xerostomia is the subjective feeling of oral dryness, which is usually associated with insufficient saliva secretion.1,2,4 Although there are differences among individuals, normal unstimulated salivary flow rate (USF) is 0.3 to 0.4 mL/min and stimulated salivary flow rate (SSF) is 1.5 to 2.0 mL/min.5,6 If USF is less than 0.1 mL/min and/or SSF is less than 0.5 to 0.7 mL/min, salivary hypofunction is considered.5,7 Common causes of xerostomia are medications, radiation therapy, systemic diseases, and pathological changes of the salivary glands.2,4,8 Severe xerostomia increases the potential for oral infections and affects the quality of life. 9
Because cancer therapies including radiotherapy can induce salivary hypofunction, xerostomia is one of the most common complaints in cancer patients. Radiation therapy in the head and neck region has damaged salivary glands, resulting in altering the volume, consistency, and pH of saliva. 3 According to a recent systematic review, about 93% of head and neck cancer patients suffered dry mouth during radiation therapy, and 73.6% to 85.3% complained of xerostomia after radiotherapy ended. 10 Also, chemotherapy can induce xerostomia due to its propensity to damage salivary glands. 10
The present treatment of xerostomia can be divided into general supportive measures, salivary substitutes, salivary stimulants such as pilocarpine and cevimeline, salivary gland protectors such as amifostine, hyperbaric oxygen therapy, and acupuncture.1,2,4,11 These treatments have been shown to be effective for treating xerostomia, especially cholinergic agonists, which are known to be more effective than hyperbaric oxygen therapy, acupuncture, and salivary substitutes.4,11,12 However, pilocarpine and amifostine also have a potential risk of undesirable adverse effects such as sweating, nausea, diarrhea, hypotension, and dizziness.12,13
In traditional East Asian medicine (TEAM), herbal medicine has been developed based on the theories of Yin and Yang, the 5 elements and visceral manifestation theory. Herbal medicine is prescribed from pattern identification (or syndrome differentiation, pattern classification) based on the aforementioned theories and patients’ conditions.14,15 According to TEAM, xerostomia is considered Yin and thin-fluid deficiency, Yin deficiency and dry heat, Qi deficiency, internal blockage of blood stasis, an accumulation of damp heat, and phlegm accumulation with blood stasis.9,16 Based on these TEAM theories and pattern identification, herbal medicine and acupuncture therapy have been used to treat xerostomia. To our knowledge, some systematic reviews have assessed the efficacy of acupuncture for xerostomia,17-19 while a systematic review assessing herbal medicine for xerostomia has not yet been conducted. Therefore, we aimed to assess the efficacy of herbal medicine for xerostomia in cancer patients.
Methods
Search Strategy
The protocol of this systematic review has been registered in PROSPERO (CRD 42016046420; available from https://www.crd.york.ac.uk/PROSPERO/printPDF.php?RecordID=46420&UserID=23230). A systematic literature search was performed in the following electronic databases regardless of publication date or language: MEDLINE, the Cochrane Central Register of Controlled Trials (CENTRAL), EMBASE, Allied and Complementary Medicine database (AMED), the China National Knowledge Infrastructure (CNKI), the Wanfang database, and 6 Korean medical databases (Korean Studies Information, DBPIA, Korea Institute of Science Technology Information, Research Information Center for Health Database, Korean Medline, and National Assembly Library). In addition, we searched Google Scholar (http://scholar.google.co.kr/).
To identify studies, search strategies were developed and modified for each database based on the search term for MEDLINE (see Appendix A) and CNKI (see Appendix B). Studies were searched until September 12, 2016.
Eligibility Criteria for This Review
All randomized controlled trials (RCTs) testing herbal medicine for xerostomia in cancer patients were included in this review without restrictions on time or language. All adult cancer patients who were diagnosed by histological or clinical evaluation were eligible. Xerostomia was diagnosed by clinical assessment or additional investigation such as scintigraphy, USF, SSF, and sialography. All forms of herbal medicine were included. There were no limitations on the number, administration methods, dosage, or duration of treatment. The comparators were conventional therapeutic agents, placebo, or no treatment. We excluded trials using other types of herbal medicines as a comparison or those that did not use the same baseline therapy.
Types of Outcome Measures
The primary outcome measures were the objective measurement of salivary flow rates, such as USF, SSF, or other standards, and salivary gland scintigraphy. Subjective measurements with observer-based grading or patient self-reported scoring were also included.
Secondary outcome measures were the patients’ quality of life assessed by objective instruments or adverse events.
Data Collection and Assessment of Risk of Bias
Two authors (BP and HN) independently assessed the titles and abstracts of researches retrieved from electronic databases and determined their eligibility for inclusion. Hard copies of the relevant researches were retrieved, and then 2 authors (BP and HN) independently extracted the data using a standard data extraction form. The form included methodology, participants, intervention, duration of treatment, outcomes, and conclusions. The 2 authors (BP and HN) assessed the risk of bias using the Cochrane Risk of Bias Tool. 20 The following items were assessed: random sequence generation (selection bias), allocation concealment (selection bias), blinding of participants and personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective outcome reporting (reporting bias), and other bias. The evaluated domains were assessed as “Yes,” “No,” or “Unclear” according to the criteria. A consensus was reached through discussion in the event of a discrepancy.
Data Synthesis
Data analysis was performed by using Review Manager (RevMan) 5.3 software. If there was sufficient data to pool, a meta-analysis was performed using a random effects model. We used the mean difference (MD) with 95% confidence intervals (CIs) for continuous outcomes. In the case of binary outcomes, a risk ratio (RR) with 95% CI was used. In the case of publication bias and sensitivity analysis, data analysis was not performed since the studies did not meet the qualifications.
Results
Description of Studies
From the 11 electronic databases, 698 articles were identified (Figure 1). After excluding duplicate studies and articles that did not meet the inclusion criteria on the basis of reading the title and abstract, full texts of 47 articles were retrieved and evaluated. By browsing full text articles, 22 articles were excluded; 5 were duplicates, 8 did not concern xerostomia, 1 was not herbal medicine, 5 were not randomized, and 3 had ambiguous outcomes. As a result, a total of 25 trials were included for this review process and data analysis (see Tables 1 and 2).21-45

Flow diagram of study selection.
Characteristics of the Included Randomized Controlled Trials of Herbal Medicines (Preventative Effect) for Xerostomia in Cancer Patients.

Abbreviations: MD, mean difference; CI, confidence interval; n.r., not reported; R-Tx., radiation therapy; RTOG, Radiation Therapy Oncology Group; C-Tx., chemotherapy; bid, twice a day; VAS, Visual Analogue Scale; USF, unstimulated salivary flow rate; SSF, stimulated salivary flow rate; qd, once a day; qod, every other day; UR, uptake rate; ER, excretion rate; DDP, cisplatin; tid, 3 times a day; 5-FU, 5-fluorouracil; NRS, Numerical Rating Scale; Rh-EGF, recombinant human epidermal growth factor; KPS, Karnofsky Performance Scale; LENT, late effects of normal tissue; SOMA, subjective, objective, management criteria with analytic laboratory and imaging procedure.
Characteristics of the Included Randomized Controlled Trials of Herbal Medicines (Therapeutic Effect) for Xerostomia in Cancer Patients.

Abbreviations: MD, mean difference; CI, confidence interval; n.r., not reported; RTOG, Radiation Therapy Oncology Group; tid, 3 times a day; SSF, stimulated salivary flow rate; R-Tx., radiation therapy; VAS, Visual Analogue Scale; USF, unstimulated salivary flow rate.
Characteristics of Included Studies
A total of 1586 cancer patients were included, and 22 patients dropped out. Nine trials tested patients with head and neck cancer, 15 trials studied nasopharyngeal cancer, and 1 trial investigated lung and esophageal cancer. Two trials did not report that baseline characteristics were comparable between the intervention group and the control group.36,44 The mean sample size was 63.44 patients (range = 28-140 patients), but no trial reported the calculation sequence of the sample size. The duration of treatment ranged from 2 weeks to 6 months.
Twenty-four formulations in 25 trials were investigated. Some form of decoction was used in 22 trials (Table 3). Of the 22 trials, 17 trials applied decoction orally, 2 trials applied herbal medicine through gargling, and 3 trials used nebulization. Each granule, powder, liquid, capsule, and gel was tested in 3 trials. Standardized formulations were tested in 23 trials, one trial chose 1 of 3 herbal formulas based on the individuals’ pattern identification, 43 and the other trial used a standardized formula with herbs added according to the individuals’ pattern identification. 36 Of the 24 formulations, Jiaweizengye decoction37,39 and Sarcandra glabra decoction28,35 were tested in 2 trials each.
Preparation of Herbal Formulas for Xerostomia in This Review.

Symptomatic treatments, which were artificial saliva,24,42 Dobell’s solution,22,23 vitamin B12, 21 vitamin C,40,41 and gargling with boric acid, lidocaine, dexamethasone, and chymotrypsin 25 were used as comparators. Nineteen trials investigated preventive effects for xerostomia during radiotherapy with or without chemotherapy.21-39 Of the 19 trials, 2 trials compared herbal medicine with symptomatic treatments,21,22 3 trials compared herbal medicine plus symptomatic treatments with symptomatic treatments,23-25 and 14 trials compared herbal medicine with water supplementation or no treatment.26-39 Six trials studied the therapeutic effects of herbal medicine in cancer patients with xerostomia.40-45 Of them, 3 trials compared herbal medicine with symptomatic treatments,40-42 and 3 trials compared herbal medicine with water supplementation or no treatment.43-45
Regarding outcome measurements, 9 trials reported salivary flow rate,23,25,27,33,37,39-41,45 3 trials reported the uptake rate (UR) and excretion rate (ER) of scintigraphy,24,32,33 all trials reported the grade of xerostomia or clinical improvement, and 1 trial reported the patients’ quality of life. Only 4 trials reported adverse events.24,26,32,33
Risk of Bias in Included Studies
Four trials mentioned the appropriate generation of a random sequence.22,32,37,45 Two trials reported inadequate randomization methods.21,25 Six trials reported concealment using envelopes; however, only one of these mentioned using a sealed, opaque envelope. 45 All studies had a high risk of bias in the blinding of participants and personnel, because no details were provided about a double-blind test. Regarding incomplete outcome data, 4 trials reported dropouts.32,34,42,45 Because dropouts were not clearly reported, we assessed 21 trials as “unclear risk of bias” in spite of the inclusion of outcome data. Additionally, one trial conducted “as-treated analysis,” despite substantial dropouts in the experimental group. 42 Since none of the trials were found to have preregistered protocols in the electronic databases, we basically assessed “selective reporting” as “unclear risk of bias.” By checking the methods and results sections from each trial, we assessed that 2 trials reported inappropriate outcomes that were not prespecified in the methods,26,34 and 2 trials did not report the results that were mentioned in the methods or reported inaccurate results.30,45 In addition, 2 trials did not report the participants’ characteristics, which were sex, age, and type of cancer36,44 (Figure 2).

Risk of bias summary: review authors’ judgements about each risk of bias item for each included study. “+”: low risk of bias; “?”: unclear risk of bias; “-”: high risk of bias.
Effects of Interventions
The analyzed data from the 25 included trials were divided into 2 categories:
Preventative category: Herbal medicine was investigated to prevent xerostomia in cancer patients receiving radiation therapy with or without chemotherapy (19 RCTs; Table 1).
Therapeutic category: Herbal medicine was investigated as a therapeutic agent in cancer patients diagnosed with xerostomia (6 RCTs; Table 2).
The 19 trials that investigated herbal medicine as preventive agents against xerostomia were analyzed as 3 subgroups: herbal medicine versus symptomatic therapy; herbal medicine plus symptomatic therapy versus symptomatic therapy; and herbal medicine versus no treatment or water supplementation. In addition, the 6 trials testing herbal medicines as therapeutic agents were analyzed as 2 subgroups: herbal medicine versus symptomatic therapy and herbal medicine versus no treatment or water supplementation. We performed meta-analysis for the trials investigating Jiaweizengye decoction,37,39 while the other 23 trials were evaluated descriptively since different herbal formulas or different outcome measures were used.
Preventive Effects: Herbal Medicine Versus Symptomatic Therapy
Two trials investigated herbal medicine in comparison with symptomatic therapy during radiation therapy with or without chemotherapy.21,22 Two trials reported the grade of dry mouth according to Radiation Therapy Oncology Group (RTOG) criteria.21,22 Yangyinqingre decoction and gargling with an herbal decoction were shown to have a significant effect on improving the severity of xerostomia, compared to vitamin B12 and Dobell’s solution, respectively. The salivary flow rate, scintigraphy, and the quality of life were not reported.
Preventive Effects: Herbal Medicine Plus Symptomatic Therapy Versus Symptomatic Therapy
Three trials were conducted to evaluate the preventive effects of herbal medicine plus symptomatic therapy in comparison with symptomatic therapy while receiving radiotherapy with or without chemotherapy.23-25 Two trials reported on salivary flow rate.23,25 Houxueshengjin decoction (mL/min, MD 0.09; 95% CI 0.03 to 0.15) and Shennongbaijie decoction (mL/min, MD 0.14; 95% CI 0.09 to 0.19) were found to have a significant effect on improving USF when compared with the control group. In addition, Houxueshengjin decoction was found to improve the secretion of salivary amylase (µ ×103/min, MD 50.1; 95% CI 19.21 to 80.99). One trial reported the UR and ER of 99mTcO4 scintigraphy. 24 The results showed that the UR and ER were significantly higher in the Xuanchaishaodihuang decoction group than in the control group 1 year after radiation therapy (UR: MD 0.63, 95% CI 0.40 to 0.86; ER: MD 1.33, 95% CI 1.06 to 1.60). Three trials reported the grade of dry mouth using different standards.23-25 Houxueshengjin decoction, Xuanchaishaodihuang decoction, and Shennongbaijie decoction were found to have a significant effect on lowering the severity of xerostomia in the intervention group compared with the control group. The quality of life was not reported.
Preventive Effects: Herbal Medicine Versus No Treatment or Water Supplementation
Fourteen trials tested the preventive effects of herbal medicine compared to no treatment or water supplementation during radiation therapy with or without chemotherapy.26-39 Administered herbal formulas were Shenqifanghou decoction, Xuanmaizengyehuadu decoction, Sarcandra glabra decoction, nebulization of Houxueshengjin decoction, Fuzhengjiandu granule, Zengye decoction, Yunnanbaiyao capsule combined with Niancianchuanqipipa gel, nebulization of Kangfuxin liquid, and Jiaweizengye decoction. Four trials reported on the salivary flow rate.27,33,37,39 The meta-analysis of 2 trials comparing Jiaweizengye decoction with no treatment showed a significant effect in improving SSF (mL/min, MD 0.30; 95% CI 0.24 to 0.35; Figure 3).37,39 Yunnanbaiyao capsule combined with Niancianchuanqipipa gel was found to significantly improve SSF (mL/min, MD 0.56; 95% CI 0.45 to 0.67), while no significant difference was found in the USF compared to no treatment. In addition, Xuanmaizengyehuadu decoction significantly decreased salivary flow time compared to mineral water (seconds, MD −93.00; 95% CI −114.30 to −71.70). Two trials presented the UR and ER of 99mTcO4 scintigraphy.32,33 One trial testing Zengye decoction 32 reported that the UR and ER were significantly higher in the intervention group compared to no treatment (UR: MD 0.39, 95% CI −0.53 to 1.31; ER: MD 0.10, 95% CI 0.00 to 0.20). The other trial investigating Yunnanbaiyao capsule combined with Niancianchuanqipipa gel did not show a significant difference in the UR and ER when compared to no treatment. Fourteen trials reported the grade of dry mouth using RTOG criteria; the Late Effects of Normal Tissue (LENT)–subjective, objective, management criteria with analytic laboratory and imaging procedure (SOMA) criteria; the Visual Analogue Scale (VAS); the Numerical Rating Scale (NRS); and the trials’ own standards.26-39 All trials reported a significant effect of lowering the severity of xerostomia in the intervention groups compared to the control groups. Regarding the quality of life, no significant difference was found between the 2 groups on the Karnofsky performance scale for Fuzhengjiandu granule. 31
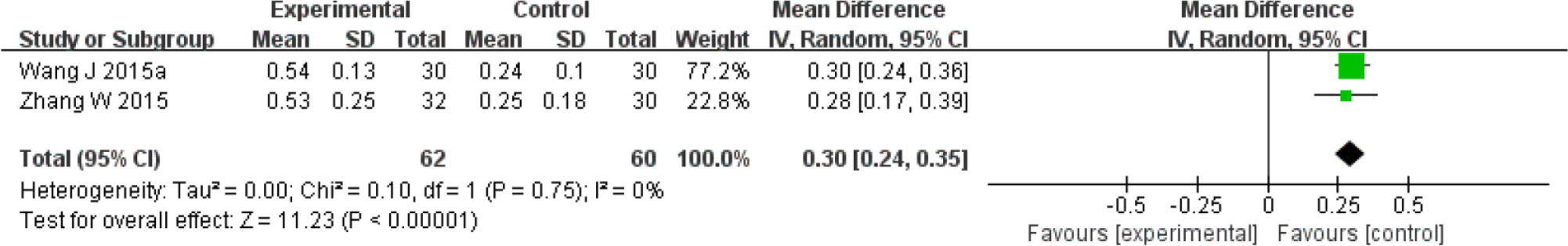
Forest plot for stimulated salivary flow rate: Jianweizengye decoction versus no treatment.
Therapeutic Effects: Herbal Medicine Versus Symptomatic Effects
Three trials investigated the therapeutic effects of herbal medicine compared to symptomatic therapy in cancer patients with xerostomia.40-42 Regarding the salivary flow rate, Jiaweishengmai decoction was tested in comparison with vitamin C, and a significant improvement of SSF was found (mL/min, MD 0.22; 95% CI 0.15 to 0.29). 41 Three trials reported on the grade of dry mouth using RTOG or VAS, with the severity of dry mouth significantly reduced.40-42 Scintigraphy and the quality of life were not reported.
Therapeutic Effects: Herbal Medicine Versus No Treatment (or Water Supplementation)
Three trials investigated the therapeutic effects of herbal medicine compared to no treatment or water supplementation in cancer patients with xerostomia.43-45 One trial reported on the salivary flow rate. 45 A gargling solution of Dark Plum with normal saline was found to make no significant difference in the USF and SSF compared to gargling with normal saline. Three trials reported on the grade of dry mouth using RTOG or VAS.43-45 Herbal formulas based on pattern identification and Liriopes Radix tea were shown to have a significant effect on reducing the severity of dry mouth, while a gargling solution of Dark Plum with normal saline did not. Scintigraphy and the quality of life were not reported.
Adverse Events
Four trials reported adverse events.24,26,32,33 One trial reported 5 cases of leukopenia in the intervention group compared to 7 in the control group, 32 while 3 trials reported no adverse events. The remaining 21 trials did not provide information about adverse effects.
Discussion
We identified 25 trials that included 1586 cancer patients on the preventive or therapeutic effectiveness of herbal medicines against xerostomia, used either individually or combined with symptomatic therapies. Four main outcomes were analyzed to assess the effectiveness of herbal medicines compared to different comparators. From this review, 5 formulas, namely, Shennongbaijie decoction, Xuanmaizengyehuadu decoction, Yunnanbaiyao capsule with Niancianchuanqipipa gel, Jiaweizengye decoction, and Sanganhuayin decoction, were shown to significantly improve the salivary flow rate compared to comparators. In addition, Xuanchaishaodihuang decoction, Zengye decoction, 32 and Yunnanbaiyao capsule with Niancianchuanqipipa gel were found to significantly affect, either partially or entirely, the UR and ER of scintigraphy. Regarding the grade of xerostomia, all formulas, with the exception of a gargling solution of Dark Plum with normal saline, showed significant effectiveness for reducing the severity of dry mouth. Adverse events were reported in 4 trials, with one reporting adverse effects from herbal medicine. The adverse events were relatively rare; however, we cannot conclude that the use of herbal medicines for xerostomia is safe since 21 of the 25 included trials did not report on adverse events. The documentation of adverse effects should be fully compliant to draw conclusions on safety.
Symptomatic therapies used for comparison in this review were artificial saliva, Dobell’s solution, vitamin B12, vitamin C, and gargling with boric acid, lidocaine, dexamethasone, and chymotrypsin. As already known, conventional therapies for xerostomia consist of salivary substitutes, salivary stimulants such as pilocarpine and cevimeline, and salivary gland protectors such as amifostine. To assess more accurately the effects of herbal medicine, it is necessary to use other conventional medicines that are known to be more effective than the symptomatic therapies used in this review.
For the number of trials included in this review, relatively many formulas were investigated. In fact, individualized herbal medicine is a representative feature of TEAM. Based on pattern identification, diverse herbal medicines are prescribed for the same disease. Furthermore, modifying the herbs in a particular formula is very common. These features, which are diverse formulas, forms, and medication methods, made it difficult to conduct meta-analyses. Thus, we assessed the evidence in accordance with each formula individually, instead of conducting an integrative analysis of all the herbal medicines.
From this review, we can assume that herbal medicines used individually or in combination with other therapies potentially have preventive or therapeutic effects for xerostomia in cancer patients. However, some limitations make it difficult to draw clear conclusions. First, most of the included trials had methodological limitations. The biggest problem was that most trials provided insufficient data to assess methodological quality. Information on randomization sequencing, allocation concealment, blinding, and dropouts was deficient. Consequently, we thought that potential biases might have occurred in selection, performance, attrition, and reporting. Second, due to clinical heterogeneity induced by the different herbal formulas tested in each trial, it was not possible to draw conclusions for global herbal medicine. Furthermore, the sample size was too small to draw conclusions about each herbal formula for clinical practitioners.
Conclusions
Herbal medicines were found to potentially improve salivary function and to reduce the severity of dry mouth in cancer patients, and they were shown to be relatively safe. However, methodological limitations and a relatively small sample size reduced the strength of the evidence. In the future, more high-quality trials reporting sufficient methodological data, more clinically homogeneous trials, and further evidence of safety are warranted to draw definitive conclusions concerning the effectiveness of herbal medicines.
Footnotes
Appendix A
Appendix B
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
