Abstract
Objectives:
Spinal accessory nerve dysfunction is one of the complications of neck dissection in patients with oral cancer. This study aimed to explore the effects of long-term scapular-focused exercises and conscious control of scapular orientation on scapular movement and quality of life (QoL).
Methods:
This study was a randomized controlled trial with concealed allocation, assessor blinding, and intention-to-treat analysis. Thirty-six patients with oral cancer were randomly allocated to the motor-control group (scapular-focused exercise + conscious control of scapular orientation) or the regular-exercise group (scapular-focused exercises only). Both groups received conventional physical therapy after neck dissection for 3 months. Shoulder pain intensity, active range of motion (AROM) of shoulder abduction, scapular muscle strength and activity under maximal voluntary isometric contraction (MVIC), scapular muscle activity when performing scapular movements, and QoL were measured at baseline, 1 month after the start of the intervention, and the end of the intervention.
Results:
Both groups showed significant improvement in all outcomes except shoulder pain intensity. After the 3-month intervention, the motor-control group had more significant improvement in AROM of shoulder abduction with a 19° difference (95% CI: 10-29, P < .001), muscle strength of upper trapezius with an 11 N difference (95% CI: 2-20; P = .021), and QoL than the regular-exercise group. When performing shoulder horizontal adduction and flexion, the relative value (%MVIC) of serratus anterior was smaller in the motor-control group with a 106%MVIC difference (95% CI: 7-205, P = .037).
Conclusions:
Scapular-focused exercises have promising effects on spinal accessory nerve dysfunction. Combining scapular-focused exercises with conscious control of scapular orientation has more remarkable benefits on AROM of shoulder abduction, UT muscle strength, and muscle activation pattern than the scapular-focused exercises alone. Conscious control of scapular orientation should be considered to integrate into scapular-focused exercises in patients with oral cancer and scapular dyskinesis.
Trial registry name and URL, and registration number: ClinicalTrials.gov (URL: https://clinicaltrials.gov; Approval No: NCT03545100)
Keywords
Introduction
Spinal accessory nerve dysfunction following neck dissection results in trapezius paralysis, 1 leading to scapular dyskinesis.2,3 The trapezius muscle is functionally divided into 3 compartments: the upper trapezius (UT), middle trapezius (MT), and lower trapezius (LT). During arm elevation, the serratus anterior (SA) initiates scapula upward rotation and generates a force couple with the MT to make the axis of movement migrate to the acromioclavicular joint. 4 Once the scapula upwardly rotates, the UT elevates and upwardly rotates the scapula. The LT depresses and retracts the scapula to resist scapular elevation and protraction generated by the UT and SA.1,4,5 At the end range of arm elevation, the SA and rhomboid work with the trapezius muscle to form a force couple to produce scapula posteriorinstitution tilt and external rotation. 5 Therefore, the active range of motion (AROM) of shoulder abduction is the most affected movement after spinal accessory nerve injury.6-9
In addition to spinal accessory nerve dysfunction (eg, shoulder droop, shoulder pain, decreased AROM of the shoulder joint, and reduced scapular muscle strength),6-8,10 impairment of health-related quality of life (QoL)10-12 was reported in patients with head and neck cancer who underwent neck dissection. Additionally, abnormal activity of the scapular muscles, including the UT, MT, LT, SA, and rhomboid, was reported in subsequent studies.13-15 In particular, for the trapezius muscle, decreased activity was observed even 9 months after neck dissection. 14 The UT and MT muscle activities on the affected side were lower than those on the unaffected side when patients performed scapular exercises (eg, shoulder shrug, overhead press, shoulder adduction and flexion, and 1-arm row).13-15 In contrast to the decreased trapezius muscle activity, higher SA and rhomboid activity was observed on the affected side than on the unaffected side when performing the overhead press and 1-arm row. 15 This increased SA activity was suggested to be a compensatory effect for the insufficient strength of the trapezius muscle. 13 However, the SA protracts the scapula during arm elevation. 5 The compensation of the SA could not have the benefit to elevate the arm at the frontal plane.8,9
Many studies have shown that scapular-focused exercise is effective in rehabilitating scapular dyskinesis 16-21; additionally, motor control by conscious correction of scapular orientation is considered necessary to stabilize the scapula in both the static position and dynamic movements.22-24 Conscious correction of scapular orientation during arm movement could increase trapezius muscle activity, 24 and motor-control training could improve scapular kinematic parameters such as posterior tilt and upward rotation during arm movement.22,25 Furthermore, early exercise intervention has been suggested to be effective in restoring or improving shoulder joint range of motion (ROM) in patients with shoulder dysfunction.26,27 Short-term effects (1 month) of combined scapular-focused exercises and motor-control techniques on the AROM of shoulder abduction and compensatory scapular muscle activities were identified in patients with oral cancer and with spinal accessory nerve dysfunction. 28 However, long-term scapular training effects (3 months) on muscle strength are lacking in patients with oral cancer.
This study aimed to estimate the long-term effects of scapular-focused exercises combined with conscious control of scapular orientation on shoulder pain, the AROM of the shoulder joint, scapular muscle strength, and upper extremity function in patients with oral cancer and scapular dyskinesis. The secondary aim was to explore the training effects of the combined intervention on scapular muscle activity and QoL. We hypothesized that long-term scapular-focused exercises combined with conscious control of scapular orientation would be more effective than scapular-focused exercises alone in releasing shoulder pain and improving shoulder AROM, muscle strength, and upper extremity function. Furthermore, we expected that the addition of conscious control of scapular orientation would lead to better muscle activity patterns and QoL.
Methods
Participants
This study is a double-blinded randomized controlled trial. The participants were enrolled from the rehabilitation center of a hospital from June 2018 to July 2019. The inclusion criteria were (1) newly diagnosed oral cancer with clinical signs of spinal accessory nerve dysfunction (eg, shoulder droop, limited AROM of shoulder abduction, and insufficient muscle strength of the shoulder abductor against gravity), (2) the presence of scapular dyskinesis by observation, 29 and (3) an age between 20 and 65 years. The participants were excluded if they (1) were pregnant or breastfeeding, (2) had distant metastasis or recurrence, (3) could not communicate or comprehend the questionnaires, (4) had a history of shoulder pain in 1 year prior to neck dissection, (5) had any disorder that could influence movement performance, or (6) had bilateral neck dissection.
The sample size was determined using G*Power 3.1.9 based on shoulder pain data from a previous study. 17 The test family and statistical tests used were F tests and “ANOVA: Repeated measures, between factors.” A prior analysis demonstrated that at least 12 participants in each group were required to provide 80% power for a significance level of 0.05. Considering a 10% dropout rate, a sample size of approximately 26 participants was required.
All participants were randomly allocated into the motor-control group who received scapular-focused exercises combined with conscious control of scapular orientation or regular-exercise group who received scapular-focused exercises only. A researcher who was not involved in the intervention or evaluation sessions conducted block randomization (4 participants in 1 block) for allocation. Each participant was blinded to the intervention allocation and participated in 12 sessions over 3 months, with each session lasting 60 minutes. The interventions were conducted by 2 physical therapists with 7 years of clinical experience. Before the study, the physical therapists underwent 1 month of training for the intervention procedures and motor-control techniques. This study was approved by the Chang Gung Medical Foundation Institutional Review Board (approval Nos: 201800026A3 and 201800026A3C502) and registered in ClinicalTrials.gov (NCT03545100). Informed consent was obtained from each participant.
Interventions
Both groups received conventional physical therapy, including pain management; scar massage; stretching; active and passive ROM exercise of the shoulder joint; and scapular-focused exercises for the UT, MT, and LT. Scapular-focused exercises were based on previous studies,14,23,27 and the details of the exercises are shown in Supplemental Table 1. All participants were instructed and underwent scapular-focused exercises with supervision by the physical therapist. Participants in the regular-exercise group were instructed to perform the scapular-focused exercises without any description of the involved muscles or alignment of the scapula. In contrast, participants in the motor-control group received anatomical education about the scapular muscles, including the individual muscle functions for scapular alignment, before the intervention. Additionally, the physical therapists provided manual contact and verbal cues when the participants performed scapular-focused exercises to enhance conscious control of the scapular position and movement for the motor-control group.
Primary Outcomes
The AROM of shoulder abduction was measured with a universal goniometer in degrees, and shoulder pain was measured at rest with a 10-cm visual analog scale (VAS). The internal reliability of the 2-arm goniometer is 0.58 to 0.99, 30 and the minimal detectable change (MCD) of the AROM of shoulder abduction is 11° to 16° with good intra-rater reliability (0.91). 31 The test-retest reliability of the VAS is 0.94, 32 and the minimal clinically important difference (MCID) is 1.4 to 1.6 in the shoulder condition. 33 The strength of the maximum voluntary isometric contraction (MVIC) of the UT, MT, LT, and SA was measured with a hand-held dynamometer (MicroFET®3; Hoggan Scientific, USA) in newtons (N), and the testing position was based on Ekstrom’s study (Supplemental Table 2). 34 The intraclass correlation coefficient (ICC) for test–retest reliability of the hand-held dynamometer is excellent (0.85-0.96), 35 and the ICC value for the MVIC of each muscle is 0.84 to 0.98. 34 The participants were asked to contract against manual resistance provided by the physical therapist in each testing position for 5 seconds. Each MVIC task was repeated 3 times, with a 30-second rest between each repetition. A 60-second rest was applied between different MVIC tasks. The Disabilities of the Arm, Shoulder, and Hand (DASH) score was used to assess upper extremity function. The disability/symptom section contains 30 items that are scored from 1 to 5, with 1 indicating “no difficulty” and 5 indicating “unable” to perform the task. At least 27 items must be completed, and scores are consequently transformed into a total scale from 1 to 100. Higher scores indicate greater disability. The ICC for test-retest reliability is 0.91 in patients with head and neck cancer after neck dissection. 36 A change in the DASH score exceeding 10.83 points represents the meaningful change score for discriminating between improved and unimproved states for DASH. 37
Secondary Outcomes
The muscle activity of the UT, MT, LT, and SA were recorded under the 4 MVIC conditions and 3 scapular exercise tasks with surface EMG electrodes (Ambu® BlueSensor NF-50-K; Malaysia) and an AC amplifier (gain: 5000; cutoff frequency: 10-450 Hz; sampling rate: 1000 Hz; model: QP511, GRASS, USA). Before applying the EMG electrodes, the participants’ skin was cleaned with alcohol and shaved if needed. Placement of the EMG electrodes was in accordance with previous studies.24,38 The 3 scapular exercise tasks were the shoulder shrug with a 1 kg weight, shoulder horizontal adduction and flexion, and 1-arm row. Because the participants could not maintain a prone position due to tracheostomy, the scapular exercises were performed in an upright posture.15,28 Details of the placement of the EMG electrodes, MVIC tasks, and scapular exercises are presented in Supplemental Table 2.
The root mean square (RMS) values of the EMG data from the second to fifth second were analyzed during each MVIC task. For the scapular exercises, the participants were asked to remain in the target position for 10 seconds, and the task was repeated 3 times. The RMS values of the EMG data from the third to sixth second were analyzed for each scapular muscle. The RMS values for the scapular exercises were normalized by the RMS values for the MVIC and are presented as %MVIC. The ICC value for test–retest reliability of the EMG under MVIC is good for the scapular exercises in healthy subjects (0.81-0.94) 39 and the participants with shoulder pain (0.89-0.96). 40
We used the European Organization for Research and Treatment of Cancer Core Quality of Life Questionnaire (EORTC QLQ-C30) to evaluate QoL in the present study. The EORTC QLQ-C30 is considered a reliable and valid assessment of QoL among patients with head and neck cancer. 41 The EORTC QLQ-C30 contains 30 questions and is divided into a global health status, 5 functional scales, and 9 symptom scales. We used the global health score, physical functioning score, role functioning score, emotional functioning score, cognitive functioning score, and social functioning score to represent the health-related QoL in the present study, with higher scores indicating higher QoL or function. 42 The ICC values for these scores range from 0.33 to 0.82. 41
All evaluations but the DASH questionnaire were conducted at baseline (T0), 1 month after the start of the intervention (T1), and at the end of the intervention (T2) by another senior physical therapist with 24 years of clinical experience was blinded to the subject allocation. The DASH questionnaire was conducted at T1 and T2 as the result of post-operation immobilization before T0.
Statistical Analyses
The generalized estimating equations (GEE) procedure was conducted to analyze repeated-measures outcome variables over time. Compared with repeated measures ANOVA, the GEE procedure provides higher power with a small sample size for repeated measurements with complete or missing data.43-45 We used the GEE model with an exchangeable working correlation matrix. Separate models were run for all outcome measures with T0 as the reference group, and each muscle and each task were analyzed separately. The level of significance was set at P < .05. Statistical analyses were completed using SPSS version 21 (SPSS Inc., USA).
Results
Thirty-six participants were analyzed in the present study. The CONSORT flow diagram is shown in Figure 1. No significant difference in demographic data or clinical characteristics was found between the 2 groups (Table 1).
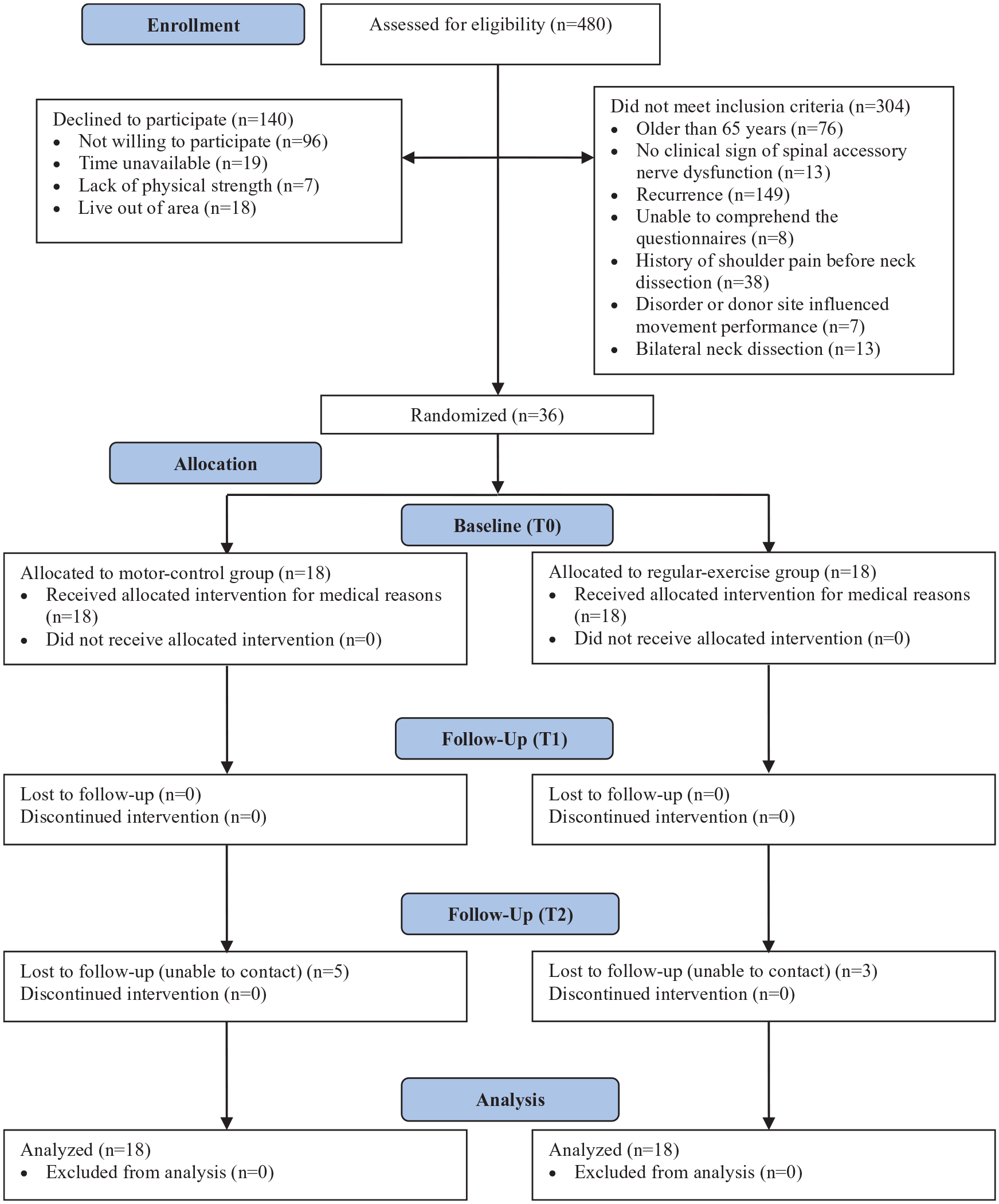
Design and flow of participants through the trial.
Demographic and Clinical Characteristics of the Study Participants.

Abbreviation: n, number of participants.
Primary Outcomes
No time, group or interaction effect on the VAS pain score and DASH score were found in either group. Relative to that at T0, the VAS score decreased from 1.5 to 0.1 (95% CI: 0.4-2.5; P = .006) in the regular-exercise group and from 1.2 to 0.5 (95% CI: −0.4 to 1.8; P = .211) in the motor-control group at T2. Only the regular-exercise group reached the MCID in the pain score. Comparing T1 with T2, the DASH score decreased from 15.8 to 12.5 (95% CI: −2.2 to 8.9; P = .232) in the regular-exercise group, and 12.2 to 12.0 (95% CI: −5.7 to 6.0; P = .955) in the motor-control group. However, significant group and time effects on the AROM of shoulder abduction without an interaction effect were found (Figure 2). Comparing T0 with T2, the improvement of the AROM of shoulder abduction was 18° (95% CI: 10-25; P < .001) in the motor-control group and 9° (95% CI: 3-16; P = .007) in the regular-exercise group. Additionally, greater shoulder abduction values at T2 were found in the motor-control group than in the regular-exercise group, with a 19° difference (95% CI: 10-29; P < .001) between the values in the 2 groups. Only the motor-control group reached the MCID of the AROM of shoulder abduction after a 3-month intervention.

Effects of intervention on the AROM of shoulder abduction for the motor-control and regular-exercise groups.
Concerning the strength of the MVIC, significant group and time effects on the UT without interaction effects and significant time effects on the MT, LT, and SA were observed (Figure 3). Relative to that at T0, the UT muscle strength of the motor-control group had an immediate intervention effect at T1 (95% CI: 7-21; P < .001) and a 3-month intervention effect at T2 (95% CI: 15-30; P < .001). The UT muscle strength increased by 14 and 22 N at T1 and T2, respectively, compared to that at T0 in the motor-control group. Although the UT muscle strength of the regular-exercise group increased by 13 N at T2 compared to T0 (95% CI: 5-20; P = .001), the improvement in UT muscle strength at T2 was smaller in the regular-exercise group than in the motor-control group with an 11 N difference (95% CI: 2-20; P = .021). Regarding the MT muscle strength, only the motor-control group showed significant improvement at T2 (95% CI: 7-24; P < .001). Regarding the LT and SA, the intervention effects on MVIC strength were similar between the motor-control and regular-exercise groups. Relative to T0, both groups showed immediate intervention effects at T1 and 3-month intervention effects at T2.
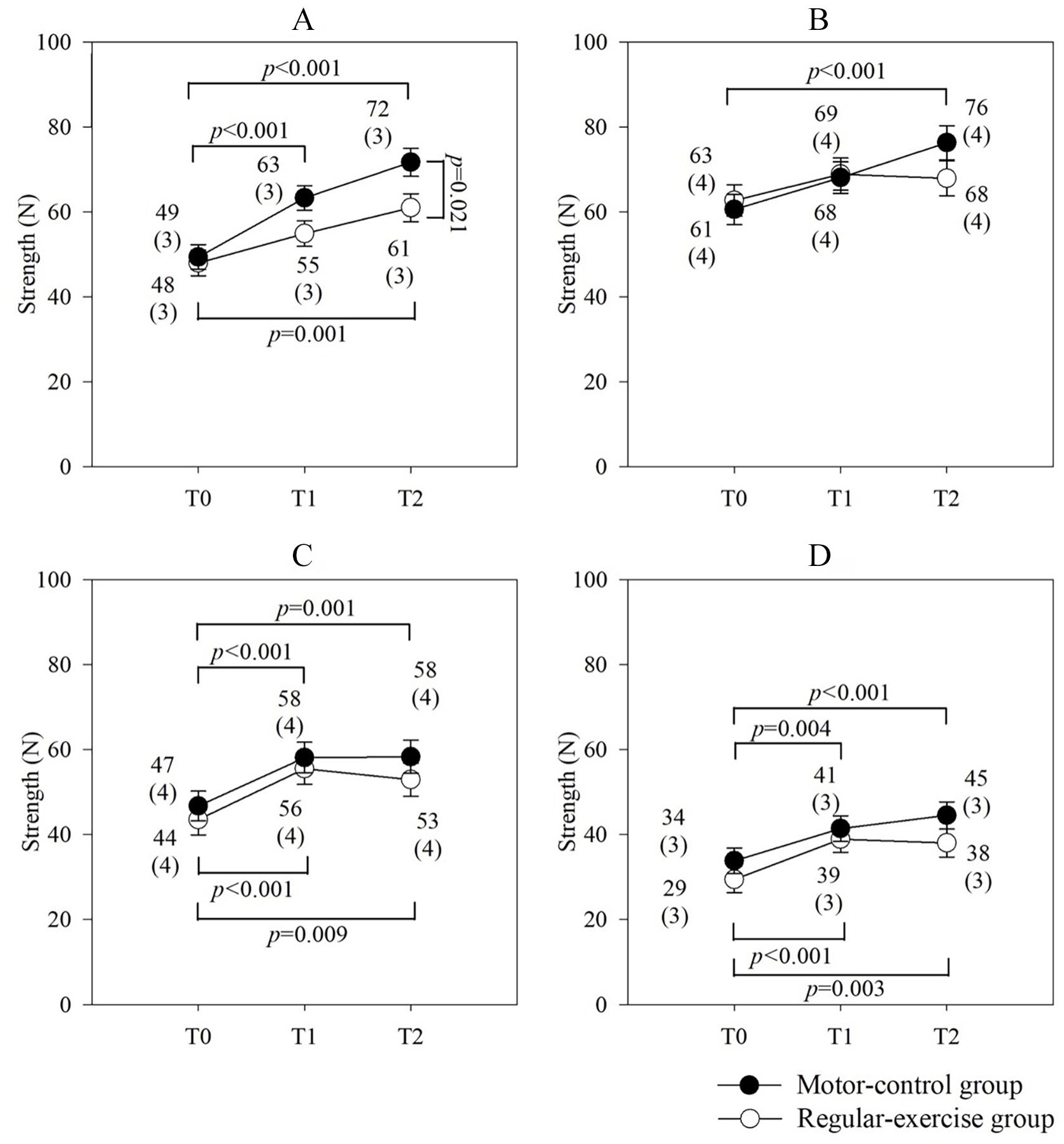
Effects of intervention on muscle strength of the (A) Upper trapezius. (B) Middle trapezius. (C) Lower trapezius. (D) Serratus anterior under MVIC conditions in the motor-control and regular-exercise groups.
Secondary Outcomes
Figure 4 shows the results of EMG activity analysis under the 4 MVIC conditions. Although no group or interaction effect on the EMG activity of the MVIC in any muscle was found, significant time effects were observed in each muscle. Immediate intervention effects at T1 were shown on the UT muscle activity (95% CI: 0.035-0.129; P = .001) and SA muscle activity (95% CI: 0.012-0.330; P = .036) of the regular-exercise group. Both groups showed increased EMG amplitude at T2 relative to T0 for UT muscle activity (motor-control: 95% CI: 0.003-0.106, P = .039; regular-exercise: 95% CI: 0.067-0.165, P < .001), MT muscle activity (motor-control: 95% CI: 0.047-0.164, P < .001; regular-exercise: 95% CI: 0.031-0.142, P = .002), and SA muscle activity (motor control: 95% CI: 0.112-0.464, P = .001; regular-exercise: 95% CI: 0.228-0.562, P < .001). For the LT muscle activity, only the motor-control group showed greater EMG amplitude at T2 than at T0 (95% CI: 0.024-0.103; P = .002).

Effects of intervention on EMG activity of the (A) Upper trapezius. (B) Middle trapezius. (C) Lower trapezius. (D) Serratus anterior under MVIC conditions in the motor-control and regular-exercise groups.
Regarding the scapular exercise tasks, no group, time, or interaction effects on muscle activity were observed when the participants performed the shoulder shrug with a 1 kg weight and 1-arm row tasks. In contrast, for the shoulder horizontal adduction and flexion task, a time effect on the muscle activities of the MT and LT in both groups and a group effect on the muscle activity of the SA were found (Table 2). Relative to that at T0, the muscle activity of the MT and LT were smaller at T2 in the motor-control group (MT: 95% CI: 9-37, P = .001; LT: 95% CI: 3-22, P = .013) and regular-exercise group (MT: 95% CI: 20-47, P < .001; LT: 95% CI: 5-24, P = .002). Additionally, the regular-exercise group had greater SA activity at T2 than the motor-control group (95% CI: 7-205; P = .037).
Mean (SE) EMG Activity of the 3 Scapular Exercises in the Groups.

Abbreviations: T0, baseline; T1, after 1 month of intervention; T2, after 3 months of intervention; MVIC, maximum voluntary isometric contraction; UT, upper trapezius; MT, middle trapezius; LT, lower trapezius; SA, serratus anterior.
Significant difference between groups (P < .05).
Significant difference between T0 and T1 (P < .05).
Significant difference between T0 and T2 (P < .05).
Figure 5 shows the global health scores from the EORTC QLQ-C30 after a 3-month intervention. Although no group or interaction effects were observed between the groups, significant time effects on the global health scores, physical functioning score, and role functioning score were found. Both groups showed significant improvement in the global health score (motor-control: 95% CI: 8-34, P = .001; regular-exercise: 10-36, P = .001) at T1 compared to T0; however, this improvement was maintained at T2 for only the motor-control group (95% CI: 4-33; P = .011). Regarding the physical functioning score, both groups showed improvement at T1 (motor-control: 95% CI: 0-20, P = .046; regular-exercise: 4-24, P = .008) and T2 (motor-control: 95% CI: 2-24, P = .017; regular-exercise: 6-27, P = .002). Regarding the role functioning score, only the motor-control group showed significant improvement at T2 compared to T0 (95% CI: 2-35; P = .025). No group, time, or interaction effects on the emotional functioning score, cognitive functioning score, or social functioning score were found between the 2 groups.

Effects of intervention on the EORTC QLQ-C30. (A) Global health score. (B) Physical functioning score. (C) Role functioning score. (D) Emotional functioning score. (E) Cognitive functioning score. (F) Social functioning score in the motor-control and regular-exercise groups.
Discussion
This study is the first randomized controlled trial to investigate the short-term (1 month) and long-term (3 months) training effects of scapular-focused exercises combined with conscious control of scapular orientation on spinal accessory nerve dysfunction in patients with oral cancer. The present study shows that, except for shoulder pain intensity and upper extremity function, scapular-focused exercises alone or combined with conscious control of scapular orientation benefit the AROM of shoulder abduction, muscle strength, muscle activity when performing MVIC and QoL. Scapular-focused exercises combined with conscious control of scapular orientation are more effective than scapular-focused exercises alone in the primary outcomes of AROM of shoulder abduction and UT muscle strength. However, there was no superiority for the other primary outcomes of MT, LT, and SA muscle strength, shoulder pain, and the DASH. Furthermore, the motor-control group had less SA muscle activity than the regular-exercise group when the participants performed shoulder horizontal adduction and flexion, indicating that the addition of conscious control of scapular orientation to scapular-focused exercises led to superior scapular control by reducing the compensatory phenomenon of the SA muscle.
For shoulder pain intensity, we found no significant change in either group. The present study implemented scapular-focused exercises early after neck dissection, which might have affected scapular kinematics to prevent the induction of shoulder pain. 46 There were only 31% of the participants who had shoulder pain initially. Although previous studies showed that scapular-focused exercises could reduce shoulder pain in patients with shoulder impingement syndrome,16-20 evidence in oral cancer patients with neck dissection is lacking because the intervention is not specific to accessory nerve-related muscles. 47 The VAS score reached the MCID in the regular-exercise group but not in the motor-control group. In addition, there were no between-group differences in DASH scores, and any changes did not reach the MCID. These results may reflect that the interventions were offered early, and both shoulder pain intensity and disability were low at baseline. More studies are needed to evaluate the effects of early interventions and the best type of intervention for shoulder pain in patients with spinal accessory nerve dysfunction.
Scapular-focused exercises are effective for shoulder abduction in patients with scapular dyskinesis.18,19 Although the AROM of shoulder abduction reached functional shoulder ROM (128° ± 7.9°) 48 in both groups after a 3-month intervention, only the improvement in the motor-control group achieved the MCID (11°-16°). 31 The trapezius muscle is a more effective scapular upward rotator in abduction,1,5 and the AROM of shoulder abduction is most affected by spinal accessory nerve dysfunction.6-8 The sensorimotor system (eg, proprioception) is affected in patients with shoulder problems.49-51 In the present study, the therapist used manual contact and verbal cues to instruct the participants to consciously control scapular alignment, through which they provided proprioception awareness 50 and neuromuscular control 51 during scapular-focused exercises in the motor-control group. The addition of conscious control of scapular orientation to scapular-focused exercises could effectively increase the AROM of shoulder abduction in patients with scapular dyskinesis after neck dissection.
The results showed a maximal strength increase in each muscle with respect to the baseline in the motor-control group after a 3-month intervention. Remarkably, the MVIC strength of the MT increased in the motor-control group, a finding that was not observed in the regular-exercise group. Furthermore, the motor-control group showed greater improvement in the MVIC strength of the UT than the regular-exercise group after the 3-month intervention. Although scapular-focused exercises improve the strength of the scapular muscles in scapular dyskinesis, 16 sensorimotor training is recommended to improve proprioception and neuromuscular control.3,50,51 Some studies have shown that motor-control training can improve scapular kinematics.22,23,25 The gains in the MVIC strength of the UT and MT are essential to elevate and upwardly rotate the scapula. 4 Increased scapular muscle strength might provide benefits in scapular kinematics during arm elevation, and a corresponding increase in the AROM of shoulder abduction in the motor-control group was also found in the present study. Our results recommend conscious control of scapular orientation during scapular muscle training for patients with neurogenic-caused scapular dyskinesis.
Although reinnervation of the trapezius muscle was demonstrated in the ninth month after neck dissection, the lowest EMG activity was found in the third month after operation 14 ; axonotmesis of the accessory nerve was suspected. 13 Even though subacromial pain was found to influence EMG activity under MVIC, 52 evidence showed high test-retest reliability of EMG activity under MVIC in patients with shoulder pain. 40 Several studies have reported that the muscle activity of the trapezius muscle can increase through EMG biofeedback and verbal or tactile feedback from therapists.22,23 The present study also found increased EMG activity under MVIC conditions in each muscle of the motor-control group after a 3-month intervention, but the LT showed no improvement in the regular-exercise group. The present study showed that 3-month intervention with scapular-focused exercises might improve axonal regeneration. 53 Notably, combining these exercises with conscious control of scapular orientation provided proprioception training for scapula orientation and restored scapular muscle balance in patients with oral cancer and scapular dyskinesis.
The present study showed that the muscle activation of the SA increased when performing shoulder horizontal adduction and flexion in the regular-exercise group but not the motor-control group. The SA stabilizes the medial border of the scapula on the chest wall when elevating the arm overhead and protracting the scapula 1 and acts with the trapezius muscle as a force couple to stabilize the scapula during arm elevation. 4 Increased SA activity may be a compensatory effect due to insufficient strength of the trapezius muscle, 13 which might indicate that more SA motor units were recruited to compensate for reduced trapezius muscle activation (eg, LT) in the regular-exercise group. The present study provides evidence that strengthening exercises combined with conscious control of scapular orientation for scapular muscles are effective in improving neuromuscular efficiency in oral cancer patients with scapular dyskinesis.
Health-related QoL is associated with shoulder AROM and strength in head and neck cancer survivors.10-12 Reduced shoulder mobility might result in shoulder dysfunction and physical impairment.11,21 Our results revealed that early exercise intervention for 3 months had a positive impact on shoulder mobility (ie, AROM and strength), particularly in the motor-control group, and greatly improved health-related QoL accordingly.
This study has several limitations. First, the scapular kinematics (eg, scapular upward rotation) were not measured. These can provide information on scapular movement and the biomechanical effects of the motor-control intervention. Second, observational scapular dyskinesis might represent normal movement variability in healthy adults. 54 Alteration tests (eg, EMG examination) to objectively assess spinal accessory nerve dysfunction may prevent potential bias. The third limitation is the sample size. Fortunately, the GEE model can explore the overall average effects in the case of missing data, 45 and we recruited more participants (n = 36) than were necessary to achieve the required sample size (n = 28). Further study is needed to evaluate the biomechanical effects of motor-control interventions coupled with scapular-focused exercises.
Conclusions
In summary, scapular-focused exercises combined with conscious control of scapular orientation more effectively improved shoulder AROM, scapular muscle strength, muscle activity, and QoL than scapular-focused exercises alone. Additionally, conscious control of scapular orientation led to a superior scapular muscle activation pattern with better neuromuscular efficiency in the muscle activity of the SA in patients with oral cancer and scapular dyskinesis.
Supplemental Material
sj-docx-1-ict-10.1177_15347354211040827 – Supplemental material for Effects of Conscious Control of Scapular Orientation in Oral Cancer Survivors With Scapular Dyskinesis: A Randomized Controlled Trial
Supplemental material, sj-docx-1-ict-10.1177_15347354211040827 for Effects of Conscious Control of Scapular Orientation in Oral Cancer Survivors With Scapular Dyskinesis: A Randomized Controlled Trial by Yueh-Hsia Chen, Cheng-Ya Huang, Wei-An Liang, Chi-Rung Lin and Yuan-Hung Chao in Integrative Cancer Therapies
Supplemental Material
sj-docx-2-ict-10.1177_15347354211040827 – Supplemental material for Effects of Conscious Control of Scapular Orientation in Oral Cancer Survivors With Scapular Dyskinesis: A Randomized Controlled Trial
Supplemental material, sj-docx-2-ict-10.1177_15347354211040827 for Effects of Conscious Control of Scapular Orientation in Oral Cancer Survivors With Scapular Dyskinesis: A Randomized Controlled Trial by Yueh-Hsia Chen, Cheng-Ya Huang, Wei-An Liang, Chi-Rung Lin and Yuan-Hung Chao in Integrative Cancer Therapies
Footnotes
Acknowledgements
The authors sincerely thank and acknowledge the support of the Maintenance Project of the Center for Big Data Analytics and Statistics (Grant CLRPG3D0045) at Chang Gung Memorial Hospital for statistical consultation and data analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Chang Gung Memorial Hospital, Linkou (grant numbers CMRPG3H0891, 2018), Taiwan. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
