Abstract
San Huang Decoction (SHD), a Chinese herb formula, has been popularly prescribed in the clinical treatment of patients suffering from breast cancer. The aim of this study was to explore the anti-angiogenic effects of SHD in breast cancer and explain the underlying mechanism. Transwell and Matrigel assays showed that SHD reduced human umbilical vein endothelial cell migration and tubule formation and ELISA and qRT-PCR assays demonstrated its mediation of vascular endothelial growth factor (VEGF) expression. siRNA silencing of aurora kinase A (AURKA) produced results similar to those obtained by inhibition of AURKA with SHD. In addition, a chorioallantoic membrane assay was carried out to directly examine the effect of SHD on breast cancer anti-angiogenesis and immunofluorescence and immunohistochemical staining analysis showed that SHD reduced the expression of CD31, AURKA, and VEGF in a xenograft model. Furthermore, SHD regulated extracellular signal-regulated kinase expression in breast cancer cells, which was examined by western blotting. In conclusion, our findings indicated that SHD treatment mimicked the decrease in tumor neovascularization in breast cancer cells after the siRNA-mediated knockdown of AURKA. Thus, SHD may inhibit tumor angiogenesis in breast cancer by targeting AURKA and downregulating the ERK signaling pathway.
Keywords
Introduction
Breast cancer is the most common cancer leading to death in the female population globally. Although current therapeutic strategies enable spontaneous progress in treating this tumor for some people, tumor heterogeneity means others are classified into a high-risk group with more aggressive carcinoma subtypes according to age at diagnosis, histological grade, status of estrogen receptor (ER), progesterone receptor (PgR), and human epidermal growth factor receptor-2 (HER2), and the value of ki67.1-3 In light of this, our research continues to examine the internal mechanisms at play in breast cancer and search for effective regimens against specific targets.
Aurora kinase A (AURKA), a crucial member of the serine/threonine kinase family, plays an important part in regulating cellular mitosis and cytokinesis. 4 The amplification of AURKA is strongly accompanied by chromosomal instability, aneuploidy, cell cycle delay, and centrosomal abnormalities. 5 It has been widely reported that AURKA is frequently amplified in tumor cells, such as those in bladder cancer,6,7 prostate cancer,8,9 colon cancer, 10 pancreatic cancer, 11 and breast cancer. 12 In breast carcinoma, dysregulation of this oncogene is closely related to high histological grade, negative or extremely low expression of ER and/or PgR, and high ki67 value, resulting in progression to advanced disease and distant metastases.13-18 In addition, oncogene amplification is associated with the increased transcription of vascular endothelial growth factor (VEGF), contributing to disordered tumor angiogenesis—a key factor of carcinoma development. 19
San Huang Decoction (SHD), a Chinese herbal formula designed by a notable traditional Chinese Medicine (TCM) physician of Jiangsu Province, Prof. Zhi-yin Xu, has been popularly prescribed in the clinical treatment of patients suffering from breast cancer. Previously, we have demonstrated that SHD is able not only to inhibit the proliferation and induce the apoptosis of breast cancer cells but also to target AURKA to downregulate its expression. Moreover, we have primarily examined its effect on mediating the expression of VEGF in cells and breast cancer patients orally receiving SHD treatment.20-23 However, the effects of SHD on downregulating angiogenesis in breast cancer and the underlying mechanism have not been thoroughly explored. In this study, according to the molecular classification of breast cancer, which includes luminal A/B, Her-2 positive, and triple-negative breast cancer (TNBC), we investigated the effect of SHD on anti-angiogenesis and AURKA expression in ER and PgR positive breast cancer MCF-7 and TNBC MDA-MB-231 cells, both in vitro and in vivo, and elucidated its mechanism of action.
Materials and Methods
Preparation of the SHD Extract
The herbs of the SHD extract (Astragalus membranaceus, 30 g; rhubarb, 10 g; and turmeric, 10 g) were acquired from Jiangsu Province Hospital of TCM (Nanjing, China). The total weight of the crude herbs was 50 g. To begin with, the herbs were blended in 1000 mL of double-distilled water (1:10, w/v) for 1 hour and then heated to 100°C for 2 hours. After boiling continuously for 2 hours with another 1000 mL of distilled water, the remainder was concentrated to 1 g/mL and filtered through a 0.2 μm filter. Finally, the extract was stored at −20°C until administration.
Cell Culture
Human breast cancer MCF-7 and MDA-MB-231 cells and HUVECs (KeyGEN BioTECH, Nanjing, China) were maintained in complete Dulbecco’s modified Eagle medium (DMEM) with 10% (v/v) heat-inactivated fetal bovine serum and antibiotics. All cells were maintained at 37°C in a humidified atmosphere of 95% air and 5% CO2.
Cell Migration Assay
Migration assays were performed using a Matrigel invasion chamber. The Matrigel-coated plates were rehydrated in warm serum-free DMEM for 2 hour at 37°C. HUVECs were starved overnight and seeded at a density of 10 000 cells per well in 24-well inserts with 8 mm pore polycarbonate membranes in the presence or absence of SHD. The supernatant of breast cancer cells with or without AURKA knockdown, serving as a conditioned medium for the HUVECs, was added in the plating chamber. Cells were incubated in 5% CO2 at 37°C for 24 hours. Then, cells were removed from the upper side of the membrane by scraping with cotton swabs and those on the lower surface were stained with crystal violet cell staining solution and mounted on slides. The number of cells was quantified by counting 5 random fields and averaging the results of each group.
Endothelial Cell Tube Formation Assay
After being grown to approximately 80% confluence, HUVECs were collected and seeded at a density of 75 000 cells per well in 24-well plates coated with 250 µL of Matrigel (BD Biosciences, Bedford, MA, USA). Each sample was periodically observed with a microscope in the process of forming capillary-like tubule structures. After 8 hours of incubation, samples were photographed, and the average number of tubules was assessed from observation of 5 independent microscopic fields.
ELISA
The supernatant of cultured cells was collected and stored at −70°C until detection. Before the assay, samples were thawed and centrifuged, and the secretion of VEGF was determined using a human VEGF ELISA kit according to the manufacturer’s instructions (R&D Systems, Inc. Minneapolis, MN, USA).
AURKA Silencing by siRNA
siRNA targeting AURKA was used for the knockdown of AURKA in MCF-7 and MDA-MB-231 cells. For siRNA transfections, 2 × 105 cells, suspended in 2 mL of medium, were seeded into each well of a 6-well plate. After incubation for 24 hours, cells were transfected with 500 nM siRNA (Cell Signaling Technology, Beverly, CA, USA) and 5 µL of Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA) diluted in Opti-MEM I Reduced Serum Medium (Invitrogen, Carlsbad, CA, USA). After 48 hours of incubation, proteins were collected and gene silencing efficiency was measured using western blotting.
Quantitative Real-Time Reverse Transcription-PCR (qRT-PCR)
Total RNA was isolated using TRIzol reagent (Life Technologies, Grand Island, NY, USA). Reverse transcription into cDNA was conducted using the TaKaRa RT reagent kit (Dalian, China). The sequences of primers used in this study are listed in Table 1. VEGF and GAPDH (internal control) mRNA levels were quantified by the ABI 7500 fast qRT-PCR System using SYBR Green master mix (Life Technologies, Grand Island, NY, USA), resulting in cycle threshold (Ct) values for VEGF and GAPDH. Each group was tested in triplicate. ΔCt = Cttarget genes − CtGAPDH. ΔΔCt = ΔCttreated samples − ΔCtcontrol samples. The relative mRNA expression level is represented by the value of 2−ΔΔCt.
Sequences of Primers Applied in the qRT-PCR Assay.
Western Blotting
Cells were treated with selected concentrations of SHD and cell lysates were prepared. Proteins were resolved by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis at 20 μg/lane and electrophoretically transferred to polyvinylidene difluoride membranes. The membranes were blocked with 5% albumin bovine serum (BSA) in Tris-buffered saline Tween (TBST; 20 mM Tris-HCl, pH 7.6, 150 mM NaCl, and 0.1% Tween-20) for 1 hour at room temperature and incubated in the primary antibodies overnight at 4°C. The primary antibodies (Cell Signaling Technology, Beverly, CA, USA) used were p-AURKA (1:1000), AURKA (1:1000), p-ERK (1:1000), ERK (1:1000), and β-actin (1:2000). The next day, the membranes were washed with TBST for 5 minutes 3 times and incubated with horseradish peroxidase (HRP)-linked anti-rabbit secondary antibody at a dilution of 1:1000. Then, after washing with TBST for 5 minutes 4 times, proteins were detected with an enhanced chemiluminescence detection kit (Millipore, Billerica, MA, USA). β-Actin was used as a loading control for densitometry normalization.
Chick Embryo Chorioallantoic Membrane Assay
The chorioallantoic membrane (CAM) assay was performed according to the method designed by Li-Jing et al 24 On day 9, small windows (10 × 10 mm2) were opened in fertilized chicken eggs (Kai Ji Biotechnology Co., Ltd., Nanjing, China) using a bistoury. The eggshells were then opened with sterile forceps. The cell suspension, with or without SHD (1 × 106 cells/100 μL phosphate-buffered saline [PBS, pH 7.4]), was applied directly to the surface of the CAM through the openings. The eggshell openings were covered and the eggs were placed in an incubator at 37.8°C and 60% humidity and rotated for 48 hours. The eggshells were then cut with scissors with great care to form a square and the contents were thrown away. The vasculature of the CAM at the eggshell openings was observed and recorded as digital images captured with a camera, and the number of vessels in the photographs were then counted. Each experiment was carried out twice, with 10 embryos in each group. The results are presented as the mean number of vessels ± SD.
Mouse Xenograft Model Establishment and Specimen Collection
The cell suspension solution was collected and adjusted to a density of 1 × 105 cells/μL PBS and 100 µL was mixed with 200 μL of Matrigel, which was subcutaneously injected into the right posterior dorsal of BALB/c mice (n = 10 per group). All mice, with or without treatment with SHD, were euthanized after 2 weeks, and the Matrigel tissue was excised and fixed in 10% formalin solution for 24 hours, then embedded in paraffin. The paraffin Matrigel tissue was subsequently cut into 4 μm sections. Then, the tissue sections were dewaxed with xylene and ethanol consecutively. Matrigel sections from mice were prepared and used for immunofluorescence or immunohistochemical staining. Each experiment was carried out twice. The study was approved by the IRB Committee of Simcere (SL-007-01).
Immunofluorescence Staining Analysis
After dewaxing and antigen recovery, the tissue sections were blocked with 5% BSA for 30 minutes at room temperature and stained with rabbit-anti-CD31 antibody (Abcam, Cambridge, UK) at a dilution of 1:20 overnight at 4°C. CD31, a cell marker that is constitutively expressed in the cells lining the endothelium, plays a role in tumor angiogenesis and growth. The next day, the sections were incubated with the goat anti-rabbit IgG diluted secondary antibody (Jackson ImmunoResearch, West Grove, PA, USA) for 1 hour and DAPI for 5 minutes at room temperature in the dark. After washing, photographs were immediately captured with the Nikon TE2000-U fluorescence microscope system (Nikon Instruments Inc., Melville, NY, USA). The results are shown as the mean number of CD31 positive cells ± SD.
Immunohistochemical Staining Analysis
After dewaxing and antigen recovery, the tissue sections were incubated with 3% H2O2 for 10 minutes to quench endogenous peroxidase and then blocked with 5% BSA for 30 minutes at room temperature. Then, sections were stained with rabbit-anti-aurora antibody at a dilution of 1:100 and mouse-anti-VEGF antibody at a dilution of 1:50 overnight at 4°C. The sections were then incubated with HRP-linked goat anti-rabbit IgG antibody (Sigma) at room temperature for 1 hour. Tissue sections were immersed in 0.05% 3-3′-diaminobenzidine solution (Sigma) for 3 minutes. After washing with TBS 4 times for 5 minutes, all sections were counterstained with hematoxylin. The semiquantitation of AURKA and VEGF was performed according to the method published by Li-Jing et al 24 Results are the mean expression scores ± SD.
Statistical Analysis
All data are expressed as means ± standard error of the mean (SEM) and evaluated by a one-way ANOVA test with SPSS 16.0. Differences between the control and treated groups were considered significant when P < .05 or .01. The intensities of western blot bands were analyzed by Image J software.
Results
SHD Downregulated AURKA to Reduce Tumor Angiogenesis by Mediating VEGF
AURKA plays a critical role in promoting tumor angiogenesis by upregulating the transcription of VEGF and increasing its secretion in cells, resulting in their aggressive behavior, 25 and our previous data have shown that SHD targets AURKA to inhibit the growth and increase the apoptosis of breast tumor cells. 23 In this study, we continued to determine whether the downregulation of AURKA by SHD had an effect on anti-neovascularity. The dose of SHD in this study was selected based on previous studies as well as the IC50 values of SHD in these respective cell lines.20-23 The expression of aurora A in MCF-7 and MDA-MB231 cells was knocked down by silencing AURKA, and the results revealed that the protein level (Figure 1) of AURKA notably decreased in MCF-7 and MDA-MB-231 cells. The supernatant of the cancer cells was collected and used in assays measuring the migration and tubule formation of HUVECs. The results revealed that SHD significantly inhibited the migration of HUVECs as effectively as it silenced AURKA (Figure 2A). Furthermore, the tubule formation of HUVECs was significantly decreased by treatment with SHD in the supernatant from MCF-7 and MDA-MB-231 cells. SHD treatment achieved a similar effect to that observed when HUVECs were grown in the supernatant from AURKA-knockdown tumor cells (Figure 2B). We then continued to use SHD and AURKA siRNA to treat MCF-7 and MDA-MB-231 cells. To investigate whether SHD regulates tumor neovascularity by altering the secretion of VEGF, ELISA was employed to evaluate the level of VEGF in each group. The VEGF secretion levels in cells from SHD-treated groups were remarkably lower than those in the control group (Figure 2C), paralleling the results of the si-AURKA group. This was consistent with HUVEC migration and tubule formation assay results. Finally, a qRT-PCR assay was performed to determine the effect of SHD on VEGF mRNA transcription. Consistent with VEGF mRNA expression in AURKA-knockdown cells, the mRNA level of VEGF was notably reduced by SHD in MCF-7 and MDA-MB-231 cells compared to the control (Figure 2D). Taken together, these results indicated that SHD inhibited AURKA to decrease the neovascularity of breast cancer via the regulation of VEGF transcription.

Silencing AURKA downregulated the expression of AURKA in breast cancer cells. Western blot analysis showing the knockdown of AURKA in MCF-7 and MDA-MB-231 cells by siRNA.

SHD-induced AURKA suppression inhibited angiogenesis and decreased VEGF mRNA level and its secretion. (A) SHD inhibited the migration of HUVECs cultured in cell supernatant from MCF-7 and MDA-MB-231 cells, which was consistent with the migration of HUVECs incubated in AURKA-silenced tumor cell supernatant. (B) The tubule formation of HUVECs cultured in MCF-7 and MDA-MB-231 cell culture supernatant was downregulated by SHD, similar to HUVECs grown in AURKA-silenced breast carcinoma cell culture supernatant. (C) ELISA results showed the level of VEGF in SHD-treated MCF-7 and MDA-MB-231 cell supernatants and AURKA-knockdown tumor cell supernatant. (D) SHD significantly downregulated mRNA levels of VEGF in MCF-7 and MDA-MB-231 cells as assessed by real-time qRT-PCR.
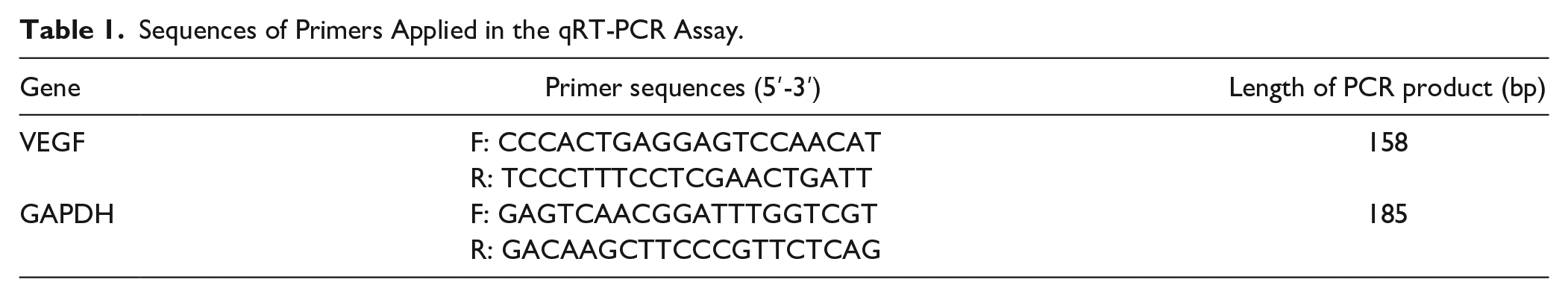
SHD Downregulated AURKA to Inhibit Tumor Angiogenesis in Chick Embryo CAM
The expression of AURKA in MBA-MD-231 cells was much higher than that in MCF-7 cells, and the effect of SHD on reducing the expression of AURKA in MBA-MD-231 cells was stronger than that in MCF-7 cells, as evidenced by qRT-PCR and western blotting. 23 Triple-negative breast cancer, the phenotype of MDA-MB-231 cells, is more aggressive and has a higher recurrence rate than any other type. Therefore, we used MDA-MB-231 cells treated with 4 mg/mL SHD for the remainder of the experiments in vivo; the dose of SHD was selected based on the IC50 values in the respective cell lines. 23 We first investigated whether SHD, by downregulating AURKA, could inhibit tumor neovascularity in the chick embryo CAM assay. Compared with the control, the MDA-MB-231 cell suspension significantly induced angiogenesis and increased the number of vessels (P < .01) (Figure 3A and B). When treated with SHD, the number of vessels was significantly reduced (P < .01), which was similar to the effect of knocking down AURKA. We further treated AURKA-silenced tumor cells with SHD and observed that the combined use of SHD and AURKA knockdown only mildly improved the effect of SHD on blocking cancer neovascularity. Consequently, we speculated that SHD suppressed breast cancer neovascularity by inhibiting the expression of AURKA.
SHD-induced AURKA suppression inhibited angiogenesis in CAM. Fertilized chicken eggs were treated according to the study protocol. (A) Representative vasculature photographs of the CAM assay. (B) The number of vessels was noticeably decreased by treatment with SHD, similar to si-AURKA. However, combined administration of SHD and si-AURKA only slightly enhanced the inhibition of angiogenesis in the CAM.
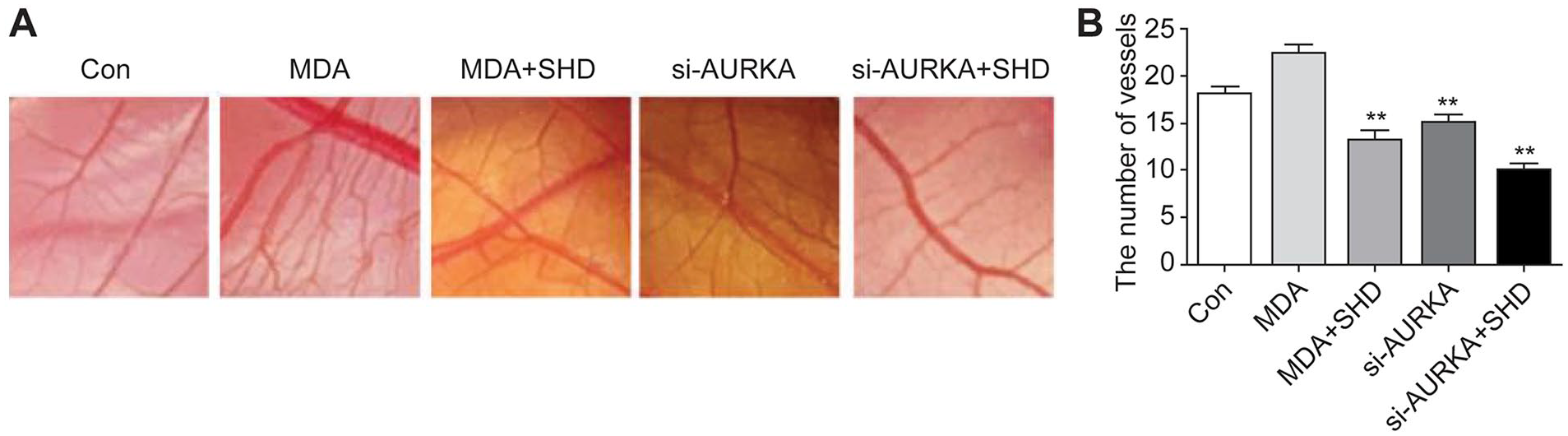
SHD Downregulated AURKA to Inhibit Tumor Angiogenesis in a Mouse Xenograft Model
We further examined whether the administration of SHD could suppress tumor neovascularity by downregulating AURKA in a mouse xenograft model. Macroscopically, MDA-MB-231 cells significantly increased the number of blood vessels in Matrigel plugs, whereas the number of blood vessels in the SHD-treated group significantly decreased, which was similar to the results in the AURKA knockdown group. Furthermore, the number of blood vessels in the group with combined SHD treatment and AURKA knockdown was only slightly less than that in the other groups (Figure 4A). The results of CD31 immunofluorescence staining showed that the number of CD31 positive cells notably increased when mice were implanted with MDA-MB-231-cell-loaded Matrigel plugs (P < 0.01). However, treatment with SHD decreased the number of CD31 positive cells as effectively as AURKA silencing. Additionally, we observed fewer CD31 positive cells than in any other group, but to a limited extent when compared with the SHD-treated group (Figure 4B and C). Immunohistochemical staining revealed that the level of AURKA in MDA-MB-231 cells in implanted Matrigel was moderately higher than that in the control group, whereas the level of AURKA significantly decreased with SHD treatment (Figure 4D and E). The effect of SHD on decreasing AURKA expression was comparable to si-AURKA treatment in MDA-MB-231 cells. Furthermore, the addition of SHD slightly improved the effect of AURKA knockdown on suppressing the expression of AURKA. Notably, we also observed that the VEGF level was reduced by SHD treatment as effectively as si-AURKA treatment. Additionally, the VEGF level was reduced by the combined treatment of SHD and si-AURKA, but not significantly so. Thus, on the basis of mouse xenograft analysis, it seems that SHD suppresses breast cancer angiogenesis by mediating the expression of AURKA.
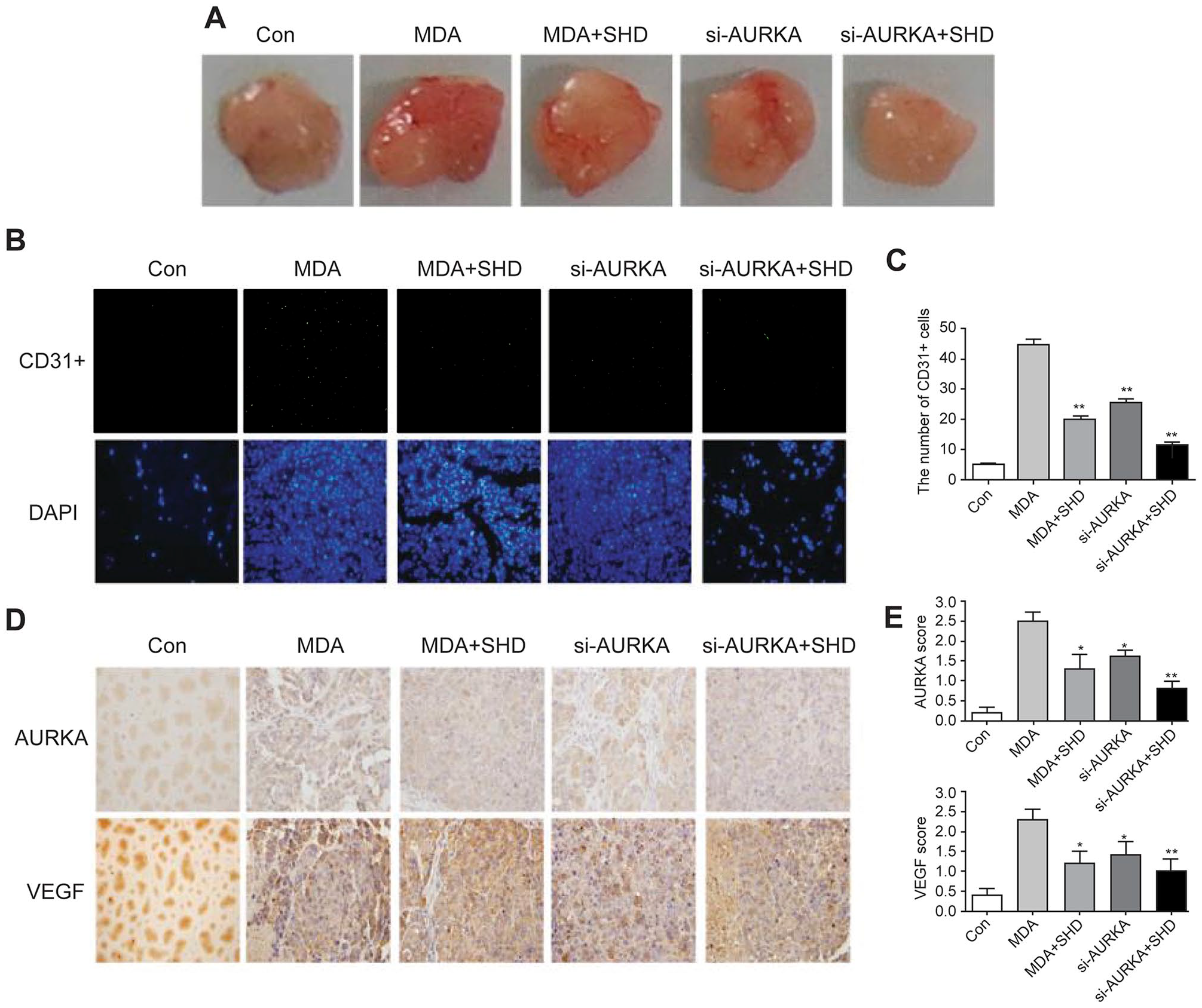
SHD-induced AURKA suppression inhibited angiogenesis in a xenograft model. (A) Matrigel was mixed with breast cancer cells and injected subcutaneously into the back of nude mice. After 2 weeks, the Matrigel plug was removed. (B) and (C) The expression of CD31 in Matrigel sections was detected by immunofluorescence staining, and the number of CD31 positive cells was calculated. (D) and (E) The expression of AURKA and VEGF was examined by immunohistochemical staining, and the scores of AURKA and VEGF were calculated.
SHD Targeting of AURKA Led to Decreased Expression of ERK
As AURKA overexpression increases tumorigenesis by moderating the ERK signaling pathway, 26 the activation of which frequently leads to angiogenesis by regulating VEGF, 27 we sought to explore the effect of SHD-suppressed AURKA on the status of ERK protein and mRNA expression in MCF-7 and MDA-MB-231 cells. Western blot analysis revealed that the ERK protein level was significantly downregulated by SHD and the knockdown of AURKA (Figure 5A and B). Consequently, we speculated that SHD can reduce AURKA levels, leading to the downregulation of the downstream ERK signaling pathway, resulting in the inhibition of tumor angiogenesis.
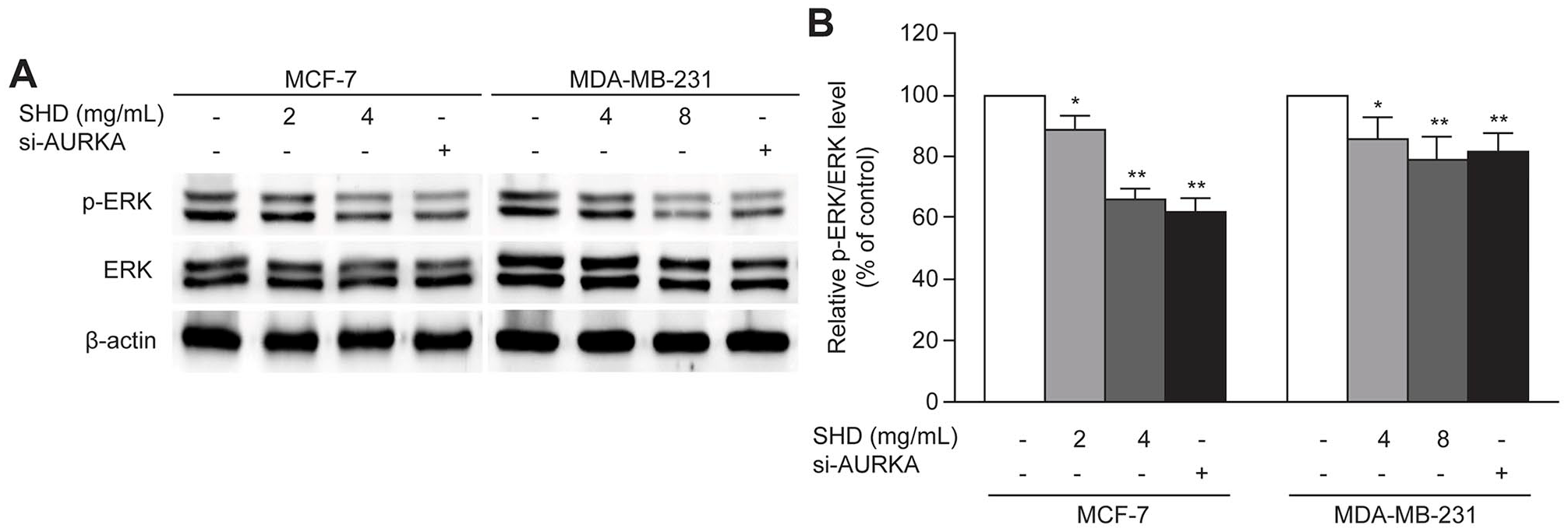
SHD targeting AURKA reduced the protein level of ERK. (A) P-ERK and ERK protein level was decreased by SHD and AURKA siRNA in MCF-7 and MDA-MB-231 cells, which was determined by western blotting with β-actin as the loading control. The value of P-ERK/ERK was calculated by semiquantitative densitometry after normalization to the corresponding β-actin. The results were from at least 3 independent experiments.
Discussion
Breast cancer can be classified into distinct subtypes according to the status of ER, PgR, and HER2, a notion that has been used to guide target-based clinical practice. For instance, tamoxifen, anastrozole, exemestane, and fulvestrant are used as endocrine therapy for patients with hormone receptor-positive breast cancer, and trastuzumab, lapatinib, pertuzumab, and TDM1 are the main anti-HER2 drugs in the treatment of HER2 enriched early and advanced carcinoma.28-30 The therapeutic strategies for breast cancer have been revolutionized in the past few decades by the use of molecular-based approaches, contributing to the promotion of overall survival, progression-free survival, and quality of life in patients with breast cancer. Unfortunately, TNBC, accounting for approximately 20% of all breast carcinoma, is still a challenge that cannot be cured with the available molecular-based therapies, such as endocrine and anti-HER2 therapies, because of the lack of ER, PgR, and HER2 expression in these patients. Moreover, as oncogenes related to TNBC have not been well elucidated, targeted treatment of TNBC is difficult to achieve. As a result, surgery, chemotherapy, and radiation therapy are the usual treatments for this kind of carcinoma. Therefore, owing to the aggressiveness of and the limited treatment options for TNBC, it is prudent to search for more effective regimens against the tumor.
It has been demonstrated that AURKA is frequently amplified in breast cancer and plays an important part in carcinoma metastasis.12,14,31,32 Currently, an AURKA-targeting treatment has been used against cancer in preclinical research and clinical trials. As an orally administered AURKA-targeted inhibitor, MLN8237 has been revealed to induce cell cycle arrest, apoptosis, and autophagy via the regulation of p38 MAPK and Akt/mTOR signaling pathways in human breast cancer cells. 33
Carcinoma neovascularization is crucial to sustain and develop a tumor. The overexpression of VEGF has been reported to be deeply involved in malignancy, aggressive behavior in breast cancer, and unfavorable prognosis.34,35 Targeting AURKA is known to downregulate angiogenesis by mediating VEGF transcription.19,25 Furthermore, thienopyridine urea, a selective inhibitor, has been orally administered to reduce the VEGF level in patients in Phase I clinical trials. 36 Our results revealed that SHD, similarly to the knockdown of AURKA, resulted in the failure of HUVEC migration and tubule formation and caused a decrease in the transcription and secretion of VEGF in tumor cells, providing an explanation for the anti-angiogenesis effect of SHD observed in the HUVEC Transwell and Matrigel assays.
There was a similar effect of SHD on inhibiting tumor angiogenesis in a CAM assay and mouse xenograft model. In the CAM assay, SHD decreased the number of vessels in the CAM, which was similar to the effect of ARUKA knockdown. However, the co-administration of SHD and si-AURKA was unable to remarkably inhibit the growth of vessels. Immunofluorescence staining analysis revealed that SHD, similarly to si-AURKA treatment, could suppress the number of CD31 positive cells in a mouse xenograft model. Furthermore, the effect of SHD on suppressing tumor angiogenesis was supported by our immunohistochemical staining analysis of cancer cell-loaded Matrigel plugs, in which we found that SHD reduced the expression of AURKA and VEGF, and its effect was mimicked with AURKA knockdown. Nevertheless, the combined treatment with SHD and AURKA silencing failed not only to further suppress CD31 positive cells but also to further inhibit the expression of AURKA and VEGF. As a result, we speculated that SHD suppressed breast tumor angiogenesis by targeting AURKA.
Crosstalk between oncogenes and substrates in cellular signaling pathways encompasses the principle of tumorigenesis and development. AURKA interacts with various downstream oncogenic pathways that play a critical role in tumor angiogenesis and progression. As described by Karar and Maity, 37 the constitutive activation of the PI3K/AKT/mTOR signaling pathway causes the abnormal formation of blood vessels that further contribute to the disorder of tumor vessels, playing a critical role in mediating neovascularization in cancers. Furthermore, the activation of ERK 1/2 reportedly promotes angiogenesis and the upregulation of VEGF.38,39 In this study, SHD treatment and the silencing of AURKA resulted in reduced ERK expression in MCF-7 and MDA-MB-231 cells compared to that in the control group. Our findings support the opinion that SHD may target AURKA, thereby mediating the downstream ERK signaling pathway, subsequently regulating tumor behavior such as angiogenesis. Further research is required to elucidate exactly how SHD interacts with AURKA and ERK in the mediation of tumor angiogenesis and VEGF expression.
Conclusion
In conclusion, our study demonstrated that SHD mimicked the anti-angiogenesis effect of AURKA silencing. SHD-induced AURKA suppression may regulate the downstream ERK pathway, thereby inhibiting tumor angiogenesis in breast cancer. Considering the close relationship between tumor neovascularization and the poor prognosis of this breast cancer, the results from these experiments provide patients and physicians, especially those facing TNBC, a promising outlook for a positive clinical outcome.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Project (Grant No. 81804103) supported by the National Natural Science Foundation of China, Jiangsu Hospital of TCM (Grant No. Y17022), and Innovative Development Foundation of Department in Jiangsu Hospital of TCM (Grant No. Y2017CX20).
