Abstract
In this study, the potential of an n-butanol fraction from Ricinus communis to prevent metastasis in MCF-7 breast cancer cells was investigated. The effect of the fraction on BUD-8 and MCF-7 cell viability was assessed using the MTT assay. Apoptotic cell death was analyzed by Hoechst staining assay. The antimetastatic effect of the fraction on MCF-7 cell was evaluated using the wound healing, adhesion and Boyden chamber invasion assays. Gelatin-zymography was used to assess the effect of the fraction on MMP-2 and MMP-9 activity. The expression profile of proteins implicated in metastasis and angiogenesis was determined using the human angiogenesis antibody array kit, following treatment with the fraction. BUD-8 cell viability was significantly reduced at concentrations between 300 and 500 µg/ml of the extract. In contrast, a significant reduction in cell viability was seen in MCF-7 cells treated with 400 to 500 µg/ml of the fraction. At sub-lethal concentrations (100 and 200 µg/ml) of the fraction, no nuclei morphological changes associated with apoptotic cell death were observed in MCF-7 cells. In addition, the fraction showed to have an inhibitory effect on MCF-7 cell migration, adhesion, invasiveness, and MMP-2 activity. Moreover, the fraction was seen to modulate the expression of several proteins, such as MMP-9, uPA, VEGF, and TGF-β1, playing a role in the metastasis process. This study demonstrates that the n-butanol fraction of R. communis can inhibit major steps of the metastatic cascade and modulate metastasis regulatory proteins. Thus, the fraction can be considered a potential source of antimetastatic agents that could be useful in the treatment of malignant cancers.
Introduction
The burden of cancer and cancer-related diseases is on the rise particularly in African countries, due to inefficient prevention strategies, poor prognosis following late diagnosis. 1 Cancer metastasis, which involves the spreading and invasion of distant body tissues by cancer cells, accounts for most cancer-related deaths. 2 During metastasis, several morphological changes result in altered cellular function. These include the loss of cell-cell and cell-extracellular matrix (ECM) adhesion, cleavage, and remodeling of the basement membrane or ECM. 3 When normal endothelial and epithelial cells lose their cell-cell and cell-matrix interactions, they trigger anoikis response. Malignant tumor cells, however, can escape or suppress anoikis which leads to successful migration through the body’s circulatory systems to distant sites. 4 Matrix metalloproteinases are mostly responsible for the remodeling and degeneration of the major components of the ECM. Matrix metalloproteinases are a family of zinc-dependant endopeptidases, and of the five major classes of metalloproteinases, MMP-2 (gelatinase A), and MMP-9 (gelatinase B), which are both type IV collagenases, have gained a huge interest based on their involvement in cancer invasiveness and metastasis. 5
Medicinal plants have been solely relied on as remedies to cure different diseases in ancient times. 6 Recently, the value of medicinal plants as a promising source of active drugs has gained more interest due to availability, cost-efficiency, safety, and better bioactivity with less or no side effects. Ricinus communis, commonly known as castor oil plant, 7 has been reported to possess several phytochemicals that have therapeutic significance. The leaves, seed oil, and roots are reported to have biological activity in the treatment of inflammation, diabetes, infertility, and liver disorders.8,9 Extracts from different parts of this plant have also been evaluated for various biological activities (antioxidant, anti-bacterial, and anti-inflammatory) 8 and its leaf extracts found to be cytotoxic to some human tumor cells. 10 The plant is also considered as one of the many natural crude remedies with the potential to combat different diseases in traditional medicine. 11 The type of chemical constituents present in the plant and its various pharmacological, including anticancer activity, have been recently reviewed.12,13 Furthermore, the fruit extract of R. communis was demonstrated to have in vitro pro-apoptosis and anti-metastasis activity against MCF-7 and MDA-MB-231 breast cancer cell lines as well as reduced tumor volume in vivo. 14 MCF-7 and MDA-MB 231 breast cancer cells are phenotypically/genotypically different thus the difference in their metastatic capabilities. MCF-7 cells which are hormone-dependent are known to be less metastatic as compared to the hormone-independent MDA-MB-231 cells. 15 However, the phenotypic and genotypic differences between these cell lines make it imperative to investigate cancer metastasis using less aggressive and/or highly aggressive cancer cells. To the best of our knowledge, no study exists on the antimetastatic mechanism of action of R. communis on MCF-7 cells. In this study, responding to the global and national challenges affecting the lifespan of cancer patients through the search for novel anti-metastatic cancer agents, the semi-purified extract (n-butanol fraction) of R. communis leaves was examined in vitro for anti-metastatic cancer activity in human breast MCF-7 cancer cells.
Materials and Methods
Plant Collection and Preparation of Semi-Purified Leaf Extracts by Solvent-Solvent Fractionation
The leaves of R. communis, voucher specimen UNIN 11352 deposited to the Larry Leach (UNIN) herbarium, were harvested, air-dried at room temperature and ground into a fine powder. The powdered leaves (20 g) were exhaustively extracted using 200 ml of acetone with continuous shaking at 150 rpm for 24 hours at room temperature using a rotary shaker. The resultant acetone extract was filtered using Whatman No. 2 filter paper and air-dried under a fan. Sub-fractions of the extract were obtained by the solvent-solvent fractionation procedure as described by Suffness and Douros 16 with slight modification. A schematic representation of the procedure is outlined in Figure 1. The n-butanol fraction was selected for further use based on its phytochemical composition. The fraction was reconstituted in dimethyl sulphoxide (DMSO) and stored at −20°C until required for cell-based assays.

Solvent-solvent fractionation scheme of R. communis crude acetone extracts separated into 6 different fractions. 15
Cell Culture and Maintenance
Breast MCF-7 (ATCC® HTB-22™) cancer cells and normal skin BUD-8 (ATCC® CRL-1554™) cells were maintained in Dulbecco’s Modified Eagle Medium (DMEM) (Gibco, New Zealand), supplemented with 10% (v/v) heat-inactivated fetal bovine serum (HyClone, USA), and 1% antibiotic mixture, Penicillin, Streptomycin, and Neomycin, (PSN; Gibco, USA) at 37°C in a humidified atmosphere of 5% CO2.
Determination of n-Butanol Fraction Effect on Cell Viability
The MTT assay as described by Mosmann 17 was used to assess the effect of the n-butanol fraction on MCF-7 and BUD-8 cell viability. After 24 and 48 hours of treatment, 25 µl of MTT (Sigma-Aldrich, USA; 5 mg/ml) solution was added to the cells and further incubated for 2 hours at 37°C. The absorbance of the solubilized formazan, by 100 µl DMSO (Sigma, Germany), was measured at 560 nm using the GloMax®-Multi+Detection system (Promega, USA).
Hoechst 33342 Nucleic Acid Fluorescent Staining
The effect of the n-butanol fraction on MCF-7 nuclear morphology was analyzed by Hoechst 33342 (Sigma-Aldrich, USA) nucleic acid staining following the description by Paterna et al 18 Briefly, cells were grown to 90% confluency and then treated with 100 and 200 µg/ml of the n-butanol fraction. Cells with media only served as negative control and 20 µM curcumin (Sigma-Aldrich, USA) as a positive control for 24 hours. Cells were washed with 1× PBS, fixed with 3.7% (w/v) paraformaldehyde (Sigma-Aldrich) and stained with Hoechst 33342 dye at 20 µg/ml for 15 minutes. Fluorescence intensity and morphology of the stained nuclear DNA were observed and photographed under a fluorescence microscope (Nikon ECLIPSE Ti, Tokyo, Japan) at a maximum excitation/emission spectra of 350/461 nm.
Wound Healing Migration Assay
The wound healing migration assay as described by Chia et al, 19 was used to assess the effect of the n-butanol fraction on the migration of MCF-7 cells. MCF-7 cells were seeded in 24-well plates and allowed to grow overnight to form monolayers. The cell monolayers were scratched with a pipette tip to create a wound and washed 3 times with serum-free medium to remove suspended cells and debris. The wounded cell monolayers were treated with 100 and 200 µg/ml of the fraction and 20 µM of curcumin (Sigma-Aldrich, USA) as a positive control for 6 and 24 hours. Wound closure was monitored by taking photographs of the wounded monolayers at 0 hours and after each incubation time using an inverted light microscope (10× magnification; Nikon ECLIPSE Ti, Tokyo, Japan)
Cell Adhesion Assay
The effect of the n-butanol fraction on the attachment of MCF-7 cells onto the surface of cell culture plates was assessed using the cell attachment assay as described previously by Yahayo et al 20 MCF-7 cells were seeded at a density of 2 × 105 cells/ml and treated with 100 and 200 µg/ml of the n-butanol fraction, and 20 µM of curcumin as a positive control for 2 hours. Following treatment, cells were plated in 24-well cell culture plates and incubated for 6 and 24 hours. Non-adherent cells were removed by gently washing the plate three times with 1× PBS. The attached cells were fixed with 3.7% paraformaldehyde and stained with 0.5% (w/v) crystal violet (Sigma-Aldrich, USA) solution. Photographs of stained cells were captured under an inverted light microscope at 10× magnification. Percentage cell adhesion was determined by solubilizing the crystal violet stained-cells with 10% (v/v) acetic acid and quantified at 560 nm using the plate reader GloMax®-Multi+Detection system (Promega, USA).
Boyden Chamber Invasion Assay
Invasiveness of MCF-7 cells following treatment was evaluated by the Boyden chamber invasion assay as reported by Zhang et al 21 MCF-7 cells at a density of 1 × 106 cells/ml were harvested, centrifuged and re-suspended in cell culture media containing 0.1% FBS, and treated with 100 and 200 µg/ml of the n-butanol fraction and 20 µM curcumin as a positive control. Cells were added into cell culture inserts (Corning, USA) coated with a matrigel matrix (BD Biosciences, USA). The inserts were placed into 24-well cell culture plates containing cell culture media with 10% FBS (serving as a chemo-attractant) and incubated at 37°C for 6 and 24 hours. Following incubation, non-invasive cells were removed from the inserts by wiping using a cotton swab. Cells that migrated to the bottom of the polyethylene terephthalate (PET) membrane of the cell culture inserts were fixed with 3.7% paraformaldehyde and stained with 0.5% crystal violet solution. Photographs of migrated cells were captured under an inverted light microscope (10×).
Gelatin-Zymography Assay
The effect of the n-butanol fraction on the expression of matrix metalloproteinases (MMP-2 and MMP-9) following treatment was assayed using gelatin-zymography as previously described by Kupai et al. 22 Briefly, MCF-7 cells were grown in serum-free media and incubated with 100 and 200 µg/ml of the fraction and 20 µM of curcumin as a positive control for 6 and 24 hours. After incubation, cell culture media from untreated and treated cells was collected and protein concentration quantified using the BCA protein assay kit (Thermo Scientific, USA), following the manufacturer’s protocol and gel photographs captured using ChemiDoc XRS image analyzer (Bio-rad Laboratories, USA).
Analysis of Relative Protein Expression
The effect of the n-butanol fraction on the expression levels of angiogenesis and metastasis-related proteins was assayed using the human angiogenesis antibody array kit (R&D Systems, USA), as described by the manufacturer (R&D Systems, Minneapolis, MN, USA). Briefly, 300 µg of protein from each sample was incubated with the angiogenesis array membranes at 4°C overnight. The membranes were then washed with an array wash buffer for 5 minutes at room temperature. The Streptavidin-HRP solution was added to the membranes which were then incubated for 30 minutes at room temperature on an orbital shaker. Pictures of the arrays were captured using the ChemiDoc XRS and the signal density of each spot representing the protein of interest was determined using imager Quantity One software (Bio-Rad Laboratories, USA). The background signal was subtracted from the total signal value of each spot and each pair of duplicate spots was averaged for each protein in each treatment group.
Statistical Analysis
Statistically significant levels were calculated using the Graphpad Instat 3 software using one-way ANOVA, followed by Dunnett`s comparison test. The data were expressed as mean ± standard deviation (SD) of 3 independent experiments. P-value ≤.05 was considered significant.
Results
Effect of n-Butanol Fractions of R. communis on BUD-8 and MCF-7 Cell Viability
The effect of the n-butanol fraction on cell viability in the MTT assay on BUD-8 and MCF-7 cells is presented in Figure 2A and B respectively. The fraction reduced BUD-8 and MCF-7 percentage cell viability in a concentration- and time-dependent manner. A significant decrease in cell viability was observed in BUD-8 cells at concentrations between 300 and 500 µg/ml at 24 and 48 hours of exposure (Figure 2A). On the other hand, a significant decrease in MCF-7 cell viability was observed at a concentration above 400 µg/ml after 48 hours of exposure (Figure 2B).

Effect of the n-butanol fraction of R. communis extracts on BUD-8 (A) and MCF-7 (B) cell viability. The cells were treated with or without the fraction or Curcumin for 24 and 48 hours. The data shows the mean ± SD of 3 independent experiments. **P ≤ .01, indicate a significant difference to the control.
Effect of n-Butanol Fraction of R. communis on the Nuclei Morphology of MCF-7 Cancer Cells
The effect of the fraction on the nuclei morphology of MCF-7 cells is presented in Figure 3. The results show that n-butanol fraction did not induce nuclei morphological features, such as chromatin condensation, the formation of apoptotic bodies and total fragmentation of the nucleus, associated with apoptosis. Curcumin at 20 µM also did not induce nuclei morphological changes associated with apoptosis in MCF-7 cells.

Hoechst 33342 fluorescent staining of the nuclei in human breast MCF-7 cancer cells treated with or without the n-butanol fraction or Curcumin for 24 hours.
n-Butanol Fraction of R. communis Inhibits the Migration of MCF-7 Cells
The effect of the fraction on MCF-7 cell migration is shown in Figure 4. The n-butanol fraction is shown to exert its inhibitory capacity of MCF-7 cell migration in a time- and concentration-dependent manner. Untreated cells show partial closure of the scratch wound after 6 hours, with complete closure at 24 hours. Unlike untreated cells, treated cells show inhibition of wound closure at both time intervals following exposure to the fraction. The fraction exhibited higher inhibitory potential at 200 µg/ml than curcumin (positive control; Figure 4).

Effect of n-butanol fraction of R. communis extracts on MCF-7 cell migration. Cell monolayers were wounded and treated with or without the fraction or Curcumin for 6 and 24 hours. Photographs of the wounds were captured under an inverted light microscope (10×) after 0, 6, and 24 hours of incubation.
n-Butanol Fraction of R. communis Inhibits the Adhesion of MCF-7 Cells
The ability of MCF-7 cells to adhere, following treatment with n-butanol fraction, was determined using the attachment assay. The n-butanol fraction shows a time- and concentration-dependent effect on the adhesiveness of the cell. Untreated cells were able to attach to the surface of the cell culture plate within 6 hours with the attainment of confluency within 24 hours of incubation (Figure 5) while exposure to 100 and 200 µg/ml of the n-butanol fraction show reduced ability of the cells to attach to the surface of the cell culture plate evident by the less number of attached cells compared to the control after 6 hours of incubation with a more pronounced effect at a concentration of 200 µg/ml at 24 hours.

Effect of n-butanol fraction of R. communis extract on MCF-7 cell adhesion. Data represent the mean ± S.D of 3 independent experiments done in triplicates. **P ≤ .01, indicate a significant difference to the control.
n-Butanol Fraction of R. communis Inhibits MCF-7 Cell Invasiveness
Matrigel matrix coated trans-well inserts were used to assess the effect of the n-butanol fraction on the invasiveness of MCF-7 cells. As represented in Figure 6A, the n-butanol fraction showed a strong inhibitory effect on the invasiveness of cells in a concentration-dependent manner. To assess the effect of the n-butanol fraction on the activity of MMPs, gelatin-zymography was employed. As shown in Figure 6B, untreated cells produced MMP-2 (72 kDa) with detectable gelatinolytic activity while no detectable gelatinolytic activity of MMP-2 was observed in MCF-7 cells treated with concentrations of 100 and 200 µg/ml of the fraction. The constitutively secreted MMP-2 activity was completely inhibited to undetectable levels by the n-butanol fraction. Furthermore, treated and untreated MCF-7 cells showed no production of active MMP-9 through gelatin-zymography seen by the lack of activity corresponding to the molecular weight of MMP-9 (92 kDa).

Effect of n-butanol fraction of R. communis extracts on MCF-7 cell invasiveness (A) and MMP-2 activity (B).
Effect of R. communis n-Butanol Fraction on the Expression of Pro-Angiogenesis/Metastasis Proteins and Anti-Angiogenesis/Metastasis-Related Proteins
The human angiogenesis antibody array kit was used to assess the effects of n-butanol fraction of R. communis on the expression of pro-metastatic proteins, pro-angiogenic proteins and factors that inhibit angiogenesis and metastasis. As shown in Figure 7A, cells treated with the extract exhibit low expression levels of membrane degrading proteins (MMP-8/9) as well as urokinase-type plasminogen activator (uPA) which are responsible for the activation of latent proteases and growth factors implicated in cancer progression. The effect of the fraction also shows a slight inhibition of thrombospondin-1, a multifunctional protein that binds to matrix proteins important in cell adhesion, cell motility, angiogenesis, and inflammation. Both treated and untreated MCF-7 cells are shown to produce elevated levels of thrombospondin-1 (TSP-1) as compared to other proteins, although a decrease in expression levels was seen in extract-treated cells at 100 µg/ml. Figure 7B shows the fraction to down-regulate the expression of cytokines or growth factors (angiogenin, CXCL8, FGF-1, PIGF, TGF-β1, and VEGF) that play a key role in angiogenesis in a concentration-dependent manner. On the contrary, the expression of endostatin (Figure 7B), a strong inhibitor of angiogenesis and tumor growth, is shown to be up-regulated in cells treated with the fraction. The effect of the fraction further shows an increase in the expression of TIMP-1 and TIMP-4, which are inhibitors of matrix remodeling and basement membrane degrading enzymes (MMPs; Figure 7C). Overall, the n-butanol fraction selectively regulated the expression of pro-angiogenesis/metastasis and anti-angiogenesis/metastasis-related proteins.
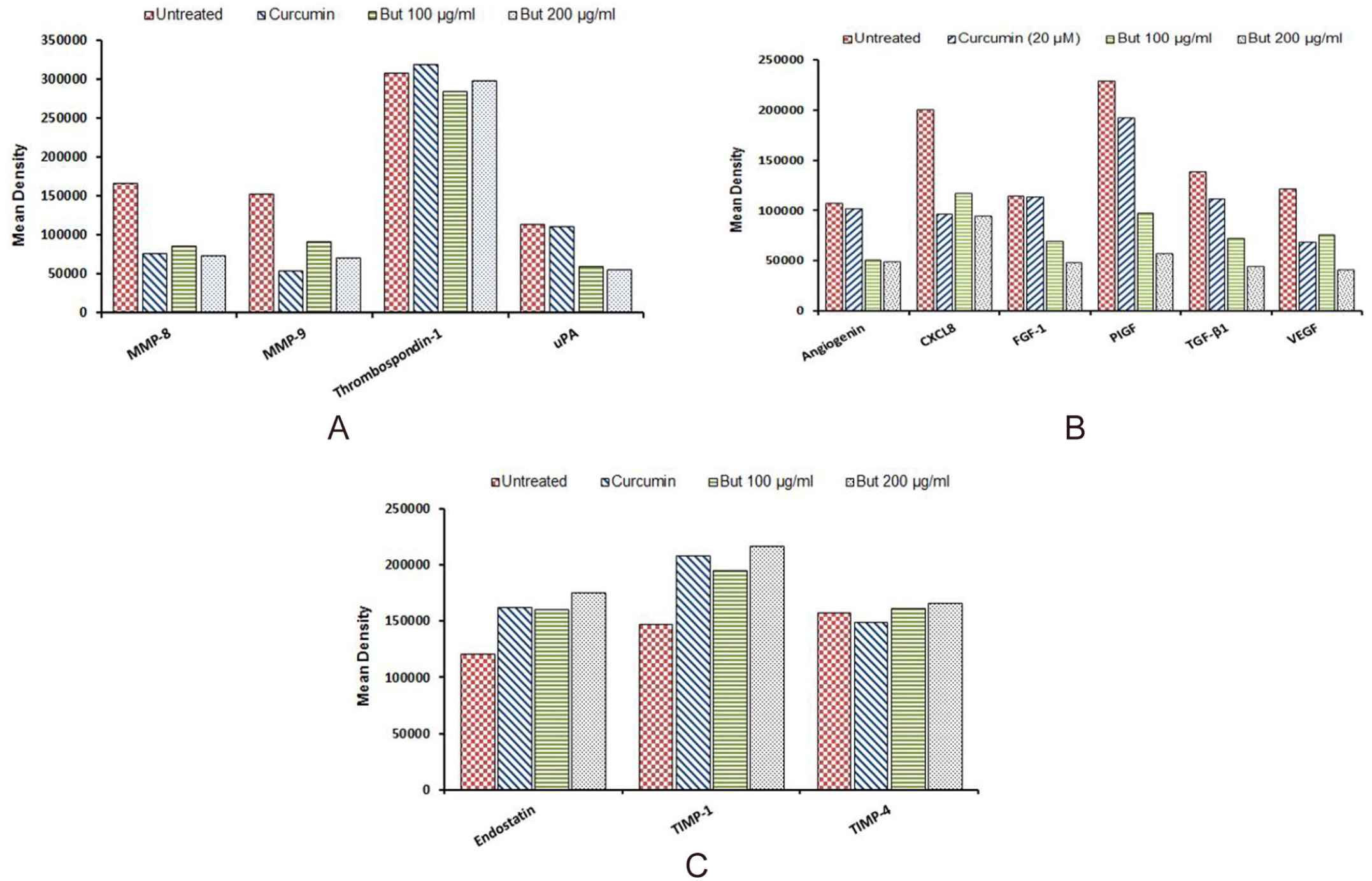
Effect of n-butanol fraction of R. communis extracts on expression levels of pro-metastatic proteins (A), proangiogenic growth factors (B), and as well as inhibitors of angiogenesis and MMPs (C). Data show the mean signal density of each representative pair of duplicate spots on the array.
Discussion and Conclusion
Cancer invasiveness has for a long time been poorly treated by apoptosis-inducing conventional anticancer drugs, which has led to increased mortality in cancer patients. 23 To promote patient survival and prevent cancer recurrence, treatments that reduce the spread and inhibit the invasiveness of tumor cells as well as prevent them from colonizing healthy tissues are required.24,25 Most plant extracts present pharmacological actions attributed to their nature of phytochemical constituents, particularly secondary metabolites such as phytosterols, tannins, coumarins, alkaloids, flavonoids, etc. In addition, phenolic compounds with antioxidant potential have been widely assessed for their ability to counteract the progression of adverse conditions such as inflammation, diabetes, and cancer. 26 In this study, the n-butanol fraction of Ricinus communis leaf extracts were evaluated for their anti-metastatic potential against MCF-7 breast cancer cells.
Prior to anti-metastatic assays, the effect of the extract on non-cancerous cells (Human Caucasian skin fibroblast: BUD-8 cells) was determined. With these cells, the effect of the n-butanol fraction was shown to decrease viability in a concentration and time-dependent manner; although viability was sustained at the highest concentration of 500 µg/ml tested in the study at both exposure time intervals. Of interest, the cells were shown to regain their proliferative ability at prolonged incubation time (48 hours) in the presence of the fraction (Figure 2A). This suggests that a longer exposure time may have allowed the cells to develop the capacity to resist the growth inhibitory effect of the fraction. The effect of the fraction on breast cancer (MCF-7) cells, on the other hand, at the highest concentration tested (500 µg/ml) did not have an adverse effect on cell viability (Figure 2B), even though viability is shown to decrease with increase in concentration. Since toxicity is concentration-dependent, concentrations of 100 and 200 µg/ml did not exhibit a marked effect on BUD-8 cell viability were employed in subsequent assays to avoid false-positive results that might occur due to cytotoxic effects of the fraction at high concentrations. The spread of cancer cells involves complex changes in the structure and function of tumor cells which contributes to architectural modifications of the extracellular matrix (ECM) and exchange in cell-cell and cell-ECM adhesive interactions.27-29 These events allow an individual or collective tumor cells to migrate toward and past the basement membrane through the blood vessels and into distant parts of the body. 30 In this study, the hindrance of MCF-7 cell migration in a concentration- and time-dependent manner was observed (Figure 4). During cell migration, metastatic cells must carefully maintain their attachment to the ECM and within the circulatory system apart from the development of anchorage-independent survival. 31 The inability to maintain adherence could lead to cell-detachment induced apoptosis, known as anoikis. Cell migration can also be suppressed through inhibition of cell adhesion by moderating the expression of critical adhesion proteins such as integrins, cadherins, and selectins. 32
Adhesion of migrating cells to the endothelial cell receptors is another key process during intravasation and spread of cancer cells into the circulatory or lymphatic system. 33 The in vivo presentation of cell-ECM interactions is defined by interactions of cell surface receptors (integrins) with fibronectin, laminin, collagen, glycoproteins and growth factors as constituents of the ECM. In this study, polystyrene (as a sort of matrix of cell attachment) treated cell culture plates were used to determine the effect of the n-butanol fraction on the ability of MCF-7 cells to adhere to a support matrix. Findings revealed the n-butanol fraction to significantly decrease cell-matrix adhesion. However, the proliferative ability of the attached cells was not affected as was observed by an increase in cell number after 24 hours of exposure (Figure 5). Cell adhesion may be regulated by the dynamics of the ECM. The ECM is essentially remodeled by MMP-2/-9 to suit the interactions of exchanged cell adhesion molecules to protein components of the ECM. 34 This suggests that these fractions may be inhibiting the adhesive ability of MCF-7 cells by regulating adhesion proteins that are also substrates of MMPs.
Over-expression of MMPs, specifically MMP-2 and MMP-9, has been shown in several types of human cancers including breast, lung, and ovarian cancers.35-37 The secretion of these proteases is one of the widely explored factors that relate to the invasiveness of tumor cells. Invasive tumor cells are characterized by their ability to induce proteolysis of the surrounding ECM as well as degradation of the basement membrane. This process allows cells to break out of the primary tumor and disseminate to target sites in the body. 21
To assess the effect of the n-butanol fraction on MCF-7 cell invasiveness, matrigel matrix-coated trans-well inserts were used. The results showed that treatment with the n-butanol fraction reduced the amount of MCF-7 cells that transited through the matrigel matrix barrier (as the basement membrane) to the bottom of the trans-well (Figure 6A) which is indicative of the fractions ability to inhibited MCF-7 cell invasiveness. To investigate the mechanism by which the invasiveness of MCF-7 cells was suppressed, the effect of the n-butanol fraction on activities of MMP-2/-9 was assessed through gelatin-zymography. Findings revealed the presence of a band that corresponded to the molecular weight of MMP-2 (72 kDa) only in the control conditioned media, while no band was comparable to the molecular weight of MMP-9 (92 kDa; Figure 6B). From this, it is evident that the gelatinolytic activity of MMP-2 was completely inhibited when cells were exposed to the fraction. However, the lack of MMP-9 activity on the zymogram suggests that MCF-7 cells may be secreting very low basal levels of MMP-9. Cancer cell lines do not secrete the same levels of MMPs and can as well be classified based on the amount of MMPs they express. The expression of MMP-9 is highly controlled by growth factors, cytokines, and signal transduction pathways. A study 38 in which PMA was used to stimulate the expression of MMPs reported the ovarian cancer SK-OV-3 cell line to express between 0% and 2% basal MMP-9 levels. The study further demonstrated that the breast cancer MCF-7 cell line only shows the expression of MMP-9 after PMA stimulation. Thus, no band correlating to both MMP-2 and MMP-9 is observed in media without PMA. It was also demonstrated that MMP-2 is constitutively expressed in many cell lines and its secretion is not affected by inducers, while MMP-9 is typically not expressed at substantial levels, though inducible. 38
The expression of MMPs activity is regulated at many levels, that is, pro-enzyme activation, protease inhibition, secretion, translation, and transcription. 39 One of the critical steps in defining the effects of the n-butanol fraction on the activity of MMPs, particularly MMP-2, is determining the point of regulation at which the fraction acts on the protease(s). Therefore, it may be concluded that constituents of n-butanol fraction hindered the activity of MMP-2 by inhibiting its enzymatic activities or by down-regulation of the protease’s expression. Furthermore, various cytokines, growth factors (TGF-β, VEGF, FGF-1, etc.), and inhibitors have a defined involvement in metastasis and angiogenesis; for example, they have been shown to have regulatory properties on the expression of MMP-2 and MMP-9. 38
The human angiogenesis array kit was used to assess the effects of the n-butanol fraction on the expression levels of pro-metastatic proteins, pro-angiogenic cytokines, and growth factors, and inhibitors of angiogenesis and metastasis. The fraction was shown to decrease the expression levels of promoters of metastatic processes including urokinase plasminogen activator (uPA; Figure 7A). Several studies have reported uPA to initiate the activation of latent MMP-2 and MMP-9, supporting its function as a system that catalyzes the transformation of the basement membrane and extracellular matrix-degrading proteases40,41 and thereby showing its direct involvement in the promotion of cell migration and invasion in many cancers. The expression and activity of MMPs were shown to decrease in MCF-7 cells exposed to the n-butanol fraction. This then suggests that the observed inhibition of cell invasiveness and MMP-2 activity may be due to the down-regulation of uPA. The n-butanol fraction was shown to decrease the expression of all the growth factors investigated in this study (Figure 7B). The fraction’s ability to down-regulate the expression levels of TGF-β1, VEGF, angiogenin, and other pro-angiogenic factors suggest the fraction’s potential as a source of antitumor agents. On the contrary, inhibitors of angiogenesis and metastasis were shown to be up-regulated in cells treated with the fraction (Figure 7C). It may be concluded that the n-butanol fraction selectively suppresses the expression of factors that promote metastasis. Thus, the focus should be given to illustrate the molecular network induced by the fraction between the selected metastatic and angiogenic proteins.
In conclusion, the potential of R. communis n-butanol fraction as an effective source of anti-metastatic agents has been demonstrated in this study. This study shows that the n-butanol fraction of R. communis leaves impedes metastasis by inhibiting the migration, invasion (through the repressed activation of MMP-2, known to participate in the promotion of cancer invasiveness) and adhesion of metastatic breast (MCF-7) cancer cells. Metastasis and angiogenesis linked proteins investigated in this study participate in many signal pathways applicable to cancer. However, further studies should be undertaken to determine the interconnectivity of these pathways and the effect of the extract on more aggressive metastatic breast cell lines such as the MDA-MB-231. Moreover, the ability of R. communis semi-purified extract to down-regulate the expression levels of cancer-associated proteins suggests that it can block or limit the progress of pathways implicated in metastasis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation (NRF) Thutuka Grunt and the University of Limpopo.
