Abstract
Background. Kefir is a unique cultured product that contains beneficial probiotics. Kefir culture from other parts of the world exhibits numerous beneficial qualities such as anti-inflammatory, immunomodulation, and anticancer effects. Nevertheless, kefir cultures from different parts of the world exert different effects because of variation in culture conditions and media. Breast cancer is the leading cancer in women, and metastasis is the major cause of death associated with breast cancer. The antimetastatic and antiangiogenic effects of kefir water made from kefir grains cultured in Malaysia were studied in 4T1 breast cancer cells. Methods. 4T1 cancer cells were treated with kefir water in vitro to assess its antimigration and anti-invasion effects. BALB/c mice were injected with 4T1 cancer cells and treated orally with kefir water for 28 days. Results. Kefir water was cytotoxic toward 4T1 cells at IC50 (half-maximal inhibitory concentration) of 12.5 and 8.33 mg/mL for 48 and 72 hours, respectively. A significant reduction in tumor size and weight (0.9132 ± 0.219 g) and a substantial increase in helper T cells (5-fold) and cytotoxic T cells (7-fold) were observed in the kefir water–treated group. Proinflammatory and proangiogenic markers were significantly reduced in the kefir water–treated group. Conclusions. Kefir water inhibited tumor proliferation in vitro and in vivo mainly through cancer cell apoptosis, immunomodulation by stimulating T helper cells and cytotoxic T cells, and anti-inflammatory, antimetastatic, and antiangiogenesis effects. This study brought out the potential of the probiotic beverage kefir water in cancer treatment.
Introduction
Cancer is infamous as the leading cause of death in the world. In the United States, breast cancer is the most common type of cancer for women. 1 In addition, the majority of breast cancer deaths are mainly because of cancer metastasis to other parts of the body. 2 For years, research into a cure for cancer has been ongoing, and inhibition of the metastatic cascade is one of the goals. The association of functional foods, such as probiotics, with cancer therapy has become an emerging field of study. Although the use of probiotics as a therapeutic agent is most common in colon-related diseases3,4 because the bacteria can interact directly with the colon after consumption, probiotics also demonstrate antitumor properties in breast cancer cells. 5
Kefir was introduced more than a thousand years ago in the Caucasus Mountains in Central Asia. It is a traditional beverage made from the fermentation of kefir grains with milk or water. 6 Kefir grains contain various kinds of microbial flora, including the most commonly studied species with probiotic potential such as Lactobacillus acidophilus, Lactobacillus casei, and Lactococcus lactis subsp Lactis.7,8 Because these species have numerous different strains, not every single strain has the potential to be probiotic. This important characteristic makes kefir suitable as a probiotic beverage. Probiotics refer to live microorganisms, which when administered in adequate amounts, confer a health benefit on the host. 9 Kefir has been consumed extensively for years because it is believed to promote good health. This claim was verified by recent studies, which showed that kefir can aid in alleviating lactose intolerance, 10 inflammation,11,12 and high total cholesterol13,14 as well as exhibiting immunomodulation properties. 15 It has also been reported that kefir has the ability to induce cytotoxicity and reduce cancer growth in vivo.5,16
Furthermore, studies by de Moreno de LeBlanc et al5,17 on the administration of commercial milk kefir from Canada in a breast cancer murine model determined that it can slow down cancer by elevating local immune response and selected cytokines. Nevertheless, an in-depth study is needed on the antitumor and antimetastatic effects of kefir in a murine breast cancer model. Thus, this study focused on the antimetastatic effects of kefir water made from kefir grains grown in Malaysia on 4T1 cells in in vitro and in vivo murine models.
Material and Methods
Sample Preparation
Kefir grain was obtained from a local store in Kuala Lumpur (http://mykefirworld.com/); 200 mg of kefir grain was added into a glass vessel containing brown sugar (200 g) dissolved in 1.5 L of mineral water. The fermentation was performed for 24 hours at room temperature. After that, the grain was filtered out from the fermented medium, and the kefir water was freeze dried. Dried kefir water was diluted in phosphate-buffered saline (PBS; 500 mg/mL) and stored at 4°C until used.
Animal and Experimental Groups
Female BALB/c mice at the age of 5 to 6 weeks (20-22 g) were obtained from the Animal House of Univesiti Putra Malaysia, Serdang (UPM, Malaysia). The mice were separated into 3 groups (n = 7 for each group): (1) normal group; (2) untreated tumor group, in which mice were fed orally with distilled water; and (3) kefir water–treated group, in which mice with tumor were fed orally with kefir water (150 mg/kg body weight per day). Kefir dosage was determined based on previous studies.16,18,19 All mice in the tumor group were inoculated with 1 × 105 4T1 cells subcutaneously, and treatment was administered orally with samples on the same day of tumor inoculation for 28 days. The mice were maintained under standard animal house conditions, with a regular 12-hour dark-light cycle at 25°C. Water and standard diet pellet were provided ad libitum. All the experiments using animals were performed under the ethical guidelines set by Universiti Putra Malaysia for the care of lab animals (UPM/IACUC/AUP-R042/2015).
TUNEL Assay
The terminal deoxynucleotidyl transferased UTP nick end-labeling (TUNEL) DNA fragmentation assay was carried out according to the user’s guidelines in DeadEnd colorimetric TUNEL assay protocol (Promega, USA). Briefly, tumor tissue sections from untreated and kefir water–treated groups on microscope slides were deparaffinized in xylene for 5 minutes. Then, the slides were immersed in sequential concentrations of ethanol (100%, 95%, 85%, 70%, and 50%) and in 0.85% NaCl for 5 minutes. Next, the slides were immersed in PBS (pH 7.0) before they were fixed in 4% paraformaldehyde for 15 minutes. The slides were washed with PBS, and the tissues were permeabilized with the provided proteinase K. The fixing steps were repeated before the tissues were equilibrated using an equilibrium buffer. Then, the tissue sections were labeled with terminal deoxynucleotidyl transferase and blocked in hydrogen peroxide. The slides were incubated in streptavidin-horseradish peroxidase (HRP) and developed using 3, 3’-Diaminobenzidine (DAB) substrate. Finally, the slides were mounted and viewed under bright field microscopy (Motic, Hong Kong), and the apoptotic cells were counted from 5 different fields in every tumor slide (n = 7 per group).
Hematoxylin and Eosin (H&E) Staining
The tumor tissues from untreated and kefir water–treated groups were fixed in 10% buffered formalin, embedded in paraffin, and sectioned. The sections were stained with hematoxylin and eosin and viewed under bright field microscopy (Motic, Hong Kong) for mitotic cell identifications. The mitotic cells were counted from 5 different fields in every tumor slide (n = 7 per group).
Nitric Oxide Assay
NO in tumor samples from untreated and kefir water–treated groups was measured according to the user’s guidelines in the Griess reagent kit (Sigma, USA). Tumors were meshed using a plunger and a 70-µm cell strainer in PBS. The supernatants were collected and mixed with the provided Griess Reagent and deionized water. After the sample mixtures were incubated for 30 minutes, the absorbance was measured using a microplate reader (Bio-Tek Instruments, Winooska, VT) at 548 nm.
Malondialdehyde (MDA) Assay
The level of lipid peroxidation in tumor samples from untreated and kefir water–treated groups was measured using the MDA assay by thiobarbituric acid reaction, according to Ohkawa et al. 20 Tumors were meshed using a 70-µm cell strainer in PBS. The supernatants were collected and mixed with butylated hydroxytoluene (Sigma, USA) and 2,4,6-trichloroanisole before they were vortexed and kept on ice for 2 hours. Afterward, the mixtures were centrifuged, and the supernatants were mixed with thiobarbituric acid and boiled for 15 minutes. The absorbance of the mixtures was measured at 532 nm using a spectrophotometer (Beckman Coulter, USA). Tetramethoxypropan (Sigma, USA) was used as standard.
Serum Cytokine Detection of Interleukin 2 (IL-2), IL-1β, IL-10, and Interferon γ (IFN-γ)
Blood serum was collected from normal, untreated, and kefir water–treated groups and subjected to cytokine detection using a Duoset ELISA assay kit (R&D Systems, USA; catalog no: DY401, DY485, DY417, DY402) per the user’s guidelines. The assay plate was coated with capture antibody overnight. The plate was washed with PBS 0.5% Tween 20 and incubated with block buffer followed by serum samples for 2 hours. Next, the plate was washed again and incubated with detection antibody for 2 hours. Streptavidin-HRP was added to the plate for 20 minutes after the plate was washed. Then, the plate was incubated with substrate solution for 20 minutes and the reaction was ended with the addition of stop solution. The absorbance was measured using a microplate reader (Bio-Tek Instruments, Winooska, VT) at 450 nm.
Immunophenotyping
Immunophenotyping of spleen CD3, CD4, and CD8 were done based on Yeap et al. 21 Briefly, the harvested spleens from normal, untreated, and kefir water–treated groups were meshed, and red blood cells were lysed using lysis buffer (8 g NH4Cl, 1 g Na2EDTA, 0.1 g KH2PO4 at pH 7.4). The cells were washed with PBS and stained with the CD3, CD4, and CD8 antibodies (Abcam, USA; Catalog No: ab11089, ab25475, ab22378) for 2 hours. The cells were then washed, fixed in 4% paraformaldehyde for a week, and run through a FACS-Calibur flow cytometry system (BD, USA).
Lung Clonogenic Assay
Clonogenic assay was performed to determine the number of metastatic 4T1 cells in the lung. 22 The harvested lungs from untreated and kefir water–treated groups were washed with PBS, minced into small pieces, and subjected to digestion in 2 mg/mL collagenase type IV for 1 hour in a 37°C water bath. The digested lungs were meshed and filtered through 70-µm strainers using syringe plungers. Then, the digested lungs were centrifuged at 2000 rpm for 5 minutes and washed twice with PBS. The cell pellets were resuspended in Roswell Park Memorial Institute medium (RPMI) containing 60-µm 6-thioguanine for clonogenic growth. The suspensions were serially diluted in 6-well plates. After 10 days, the clonogenic cells were fixed in methanol and stained with 0.5% crystal violet. Clonogenic cells were counted for each group.
Bone Marrow Smearing
Femurs from untreated and kefir water–treated mice were harvested; the bone marrows were located and washed out with PBS. The bone marrow solution was dropped onto clean glass slides and smeared across the slide. After the slides dried, they were fixed in methanol for 30 minutes and stained with Giemsa for 2 hours. The slides were viewed under an inverted microscope (Nikon, Japan).
Quantitative Real-Time Polymerase Chain Reaction (qPCR)
Tumors from untreated and kefir water–treated groups were snap frozen with liquid nitrogen and ground using a mortar and pestle. Total RNA was extracted from the ground samples using the Qiagen RNEasy Mini Kit (Qiagen, Germany). The RNA (1 µg) obtained was converted into cDNA using Maxima First Strand cDNA synthesis kit (Thermo Scientific, USA) and loaded into a thermal cycler (Labnet, USA). The cDNA was subjected to real-time polymerase chain reaction using SYBR Select Master Mix (Life Technologies, USA) in Eco Illumina (Illumina, USA) to quantify gene expression of the selected genes described in Table 1. The primers were obtained from Primer-Blast NCBI (http://www.ncbi.nlm.nih.gov/tools/primer-blast/) and Primer Bank (http://pga.mgh.harvard.edu/primerbank/). GAPDH and ACTB were used as housekeeping genes. The expressions were analyzed using Eco Study (Illumina, USA).
The Gene Name, Accession Number, and Sequence of Primers Used in Real-Time qPCR.

Proteome Profiler Angiogenesis Antibody Array
The detection of multiple cytokines in protein samples was done using the Mouse Angiogenesis Antibody Array kit (Ray Biotech, USA). The proteins from tumors harvested from untreated and kefir water–treated groups were extracted from ground samples using QIAShredder (Qiagen, Germany) and RIPA-Protease Inhibitor buffer (50 mM Tris-HCl pH 8.0, 150 mM NaCl, 1.0% TritonX-100, 0.5% sodium deoxycholate, 0.1% SDS, Protease inhibitor [Roche, USA]). Then, the protein samples were subjected to the protein array per the user’s guidelines. In brief, the provided glass chip was blocked using blocking buffer for 30 minutes, and samples were incubated in the chip overnight at 4°C. After 24 hours, the chip was washed 3 times with selected wash buffer. The chip was then incubated with biotin-conjugated anticytokine reagent for 2 hours and with streptavidin-fluor for 2 more hours. The chip was washed twice with selected wash buffer and distilled water. The chip was allowed to dry and scanned using the Axon Gene Pix 4000B (Molecular Devices, Sunnyvale, CA), and the values were extracted using the Axon Gene Pix Pro 6.1 (Molecular Devices, Sunnyvale) software. The data were then analyzed based on calculations provided in the user’s guidelines.
Statistical Analysis
Unless stated otherwise, the results were expressed as the means and SDs of all mice for each group (n = 7 for each group). Statistical significance (P < 0.05) was determined using Student’s t-tests and performed using GraphPad Prism (version 6.0) against the untreated group.
Results
Kefir Water Induced Cell Cytotoxicity and Positively Suppressed Cell Migration and Invasion In Vitro
MTT assay was performed on 4T1 cells to evaluate the cytotoxicity of the kefir water at the highest dosage of 50 mg/mL. The half-maximal inhibitory concentration (IC50) values were determined and selected for further bioassays. The IC50 values after 48 hours and 72 hours were 12.5 and 8.33 mg/mL, respectively. The migration and invasion ability of 4T1 cells were challenged with the exposure to 3 concentrations of kefir water at 3.125, 6.25, and 12.5 mg/mL through a Matrigel membrane. The number of migrating cells was significantly decreased in the treatment groups as compared with the untreated group (supplementary data, available at http://ict.sagepub.com/supplemental). The percentage of migrating cells was reduced by 88.03% ± 1.44% when treated with 12.5 mg/mL kefir water. The same pattern can be observed in the invasion assay, where the invaded cells were reduced in number when exposed to higher concentration of kefir water, with 72.83% ± 4.01% of cell invasion being suppressed at 12.5 mg/mL. Hence, it can be concluded that the rate of 4T1 cell migration and invasion was dose dependent, whereby as the concentration of kefir water increased, the rate decreased. In the wound healing assay, a significant reduction in wound healing in comparison to untreated cells was observed at 3 concentrations of kefir water (3.12, 6.25, and 12.5 mg/mL) at 12 and 24 hours (supplementary data, available at http://ict.sagepub.com/supplemental). The wound closures between the 3 concentrations were distinguishable only at 24 hours, where the percentages were 67.9%, 62.5%, and 39.6%, at the respective ascending concentrations.
Kefir Water Obstructed Ex Vivo Angiogenesis in Mouse Aortic Ring
Sections of mouse aortic rings were embedded in Matrigel and exposed to 12.5 mg/mL of kefir water for 10 days to allow vessels to grow. The formation of microvessels sprouting from the aortas was observed and counted in every well. As compared with the untreated aortas, the aortas in the kefir water–treated group produced minimal microvessel growth (supplementary data, available at http://ict.sagepub.com/supplemental).
Kefir Water Reduced 4T1 Tumor Growth in Mice
Figure 1A shows the image of tumor harvested from cancer mice after 28 days of treatment. Tumor size in kefir water–treated mice was smaller than that in untreated mice. The tumor weight and volume were also reduced significantly by 29.53% and 32.92%, respectively, in kefir water–treated mice as compared with the untreated mice (Figure 1).

Tumor size, weight (g) and volume (cm3), obtained from 4T1 cell-challenged mice. (A) Images of tumor harvested from untreated group and kefir water–treated group after 28 days. (B) Tumor weight of untreated group and kefir water–treated group after 28 days. (C) Tumor volume of untreated group and kefir water–treated group after 28 days. Each value represents the mean ± SD (n = 7 mice per group). Significant difference (*P < .05) from the untreated group was calculated using the t-test.
Kefir Water Inhibited Cell Mitosis and Enhanced Tumor Apoptosis in Mice
Tumor sections were paraffin embedded on glass slides and subjected to H&E staining and TUNEL assay. The histology analysis of untreated tumor sections displayed a greater number of differentiated cancer cells (Figure 2A). Black circles in Figure 2A indicate mitotic cells that underwent cell division. Apoptosis of cancer cells in tumor was observed in the TUNEL assay. A higher number of apoptotic cancer cells were observed in the kefir water–treated tumor section as compared with the untreated section (Figure 2B).
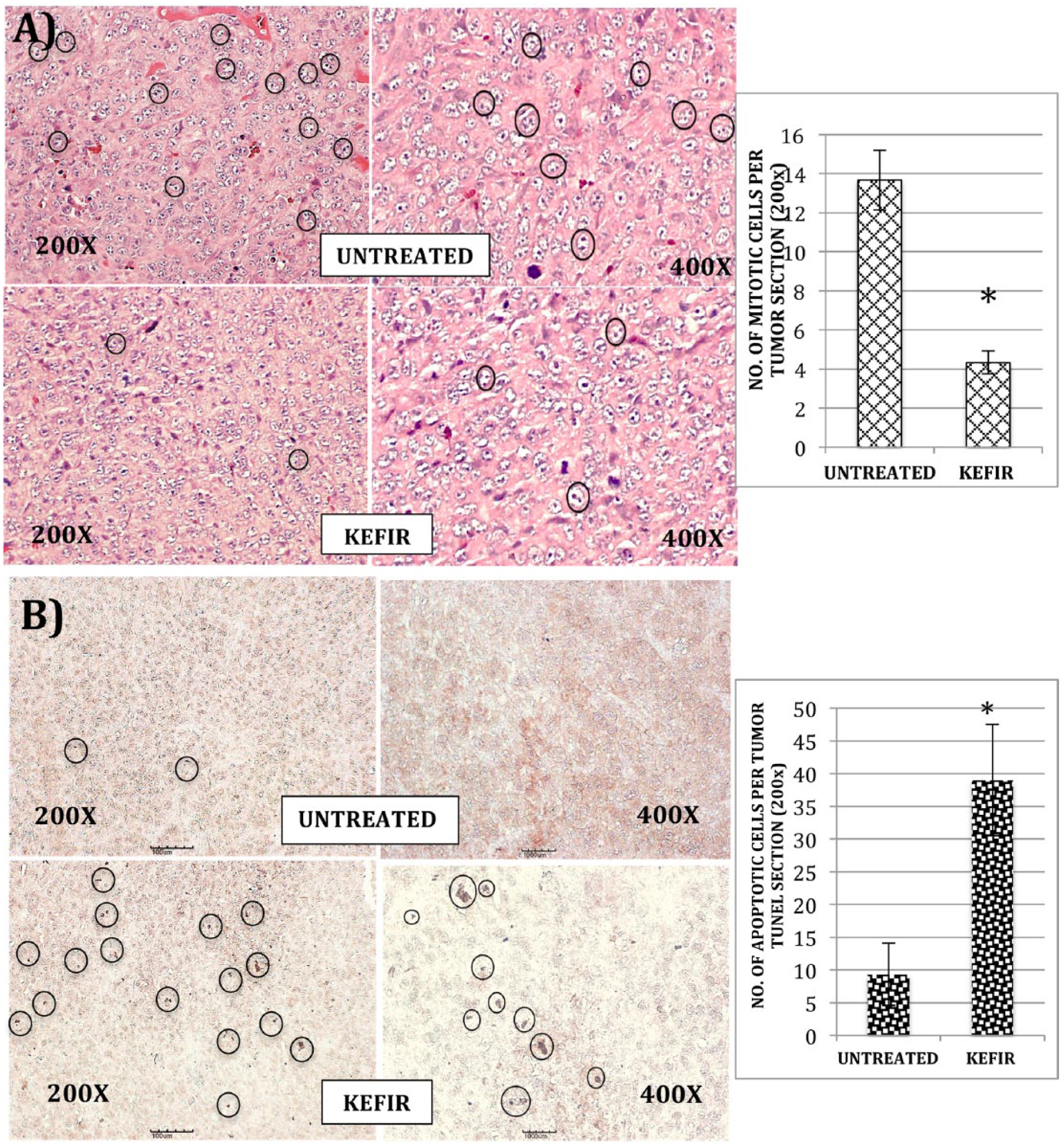
Hematoxylin and eosin stain image of tumor from untreated group and kefir water–treated group. Black circles show mitotic cells. (A) The histogram shows the number of mitotic cells per tumor section in the untreated group and kefir water–treated group (n = 3 slides per mice). Significant difference (*P < .05) from the untreated group was calculated using the t-test. (B) Terminal deoxynucleotidyl transferased UTP nick-end labeling (TUNEL) assay image of the tumor from untreated and kefir water–treated groups. Black circles show DNA fragmentation and apoptotic cells (dark brown spots). The histogram shows the number of apoptotic cells per tumor section in the untreated group and kefir water–treated group (n = 3 slides per mice). Significant difference (*P < .05) from the untreated group was calculated using the t-test.
Kefir Water Decreased Metastasis to Lung and Bone Marrow
The antimetastatic effects of kefir water on tumor were further analyzed in a clonogenic assay and bone marrow–smearing assay. In the clonogenic assay, 6-thioguanine is a selective medium to grow 4T1 cells. The metastatic potential of 4T1 cells to the lung was observed after 10 days. The number of 4T1 cells metastasized in the lung of kefir water–treated mice was significantly lower than in untreated mice, as shown in Figure 3A. In the bone marrow–smearing assay, metastatic cells were identified by the presence of large and abnormal cells. Based on Figure 3B, metastatic 4T1 cells were only observed in the untreated group.

(A) The images of clonogenic colonies in lung harvested from 4T1 cell–challenged mice at a dilution factor of 104 and the number of 4T1 colonies formed after 10 days of incubation (n = 6 mice per group). Significant difference (*P < .05) from the untreated group was determined using the t-test. (B) Bone marrow smear for untreated and kefir water–treated group at 200× and 400× magnification.
Kefir Water–Controlled Cytokine Expressions in Serum
The expression levels of cytokines involved in angiogenesis and inflammation were measured using blood serum. The levels of tumor pro-growth-related cytokines (IL-10 and IL-1β) were significantly decreased in the kefir water–treated group (Figure 4). On the other hand, the levels of immunomodulation related cytokines (IFN-γ and IL-2) were significantly increased in the kefir water–treated group (Figure 4).

The results from cytokine (IL-10, IFN-γ, IL-1β, IL-2) enzyme-linked immunosorbent assay (ELISA) assay on blood serum from the untreated cancer group and kefir water–treated cancer group. Data are presented as means ± SD (n = 6 mice per group). Significant difference (*P < .05) from the untreated group was determined using the t-test.
Kefir Water Enhanced Immune Response in Spleen
The population of selected immune markers (T-helper CD4+/CD3+) and cytotoxic T cells (CD8+/CD3+) was calculated based on immunophenotyping by flow cytometry. As shown in Figure 5, the population of T-helper CD4 cells in the kefir water–treated group as compared with the untreated group was significantly higher, by 5-fold. The same pattern was observed in the population of cytotoxic CD8 T cells, where it was increased by 7-fold. In addition, the population of the immune cells between the normal group and the kefir water–treated group showed no statistical difference.

Flow cytometry dot plots and histograms of immune markers (CD3, CD4, and CD8) on splenocytes harvested from the normal group, untreated cancer group, and kefir water–treated cancer group. Data are presented as means ± SD. Significant difference (*P < .05) from the untreated group was determined using the t-test.
Kefir Water Decreased Lipid Peroxidation and NO Levels in Tumor
The levels of lipid peroxidation of MDA and NO in tumor supernatant were determined, and reductions were seen in both assays (Figure 6). Kefir water had decreased the levels of both inflammation-related chemicals in the tumors by about 3-fold.

(A) NO levels in tumors from the untreated and kefir water–treated groups. (B) Malonaldehyde (MDA) levels in tumors from the untreated group and kefir water–treated group. Data are presented as means ± SD. Significant difference (*P < .05) from the untreated group was determined using the t-test.
Kefir Water Reduced the Expression of Inflammation and Metastatic-Related Genes and Proteins In Vivo
The mRNA expressions of ICAM, iNOS, MMP-9, IL-1β, NF-κB, G-CSF, GM-CSF, IL-4, and TNF-α genes were downregulated in the kefir water–treated group as compared with the untreated group (Figure 7A). A significant downregulation of iNOS and IL-1β by 3-fold were observed in the kefir water–treated group. In the angiogenesis proteome profiler assay, the levels of selected proangiogenesis proteins were downregulated and that of antiangiogenesis proteins were upregulated in the kefir water–treated group (Figure 7B).
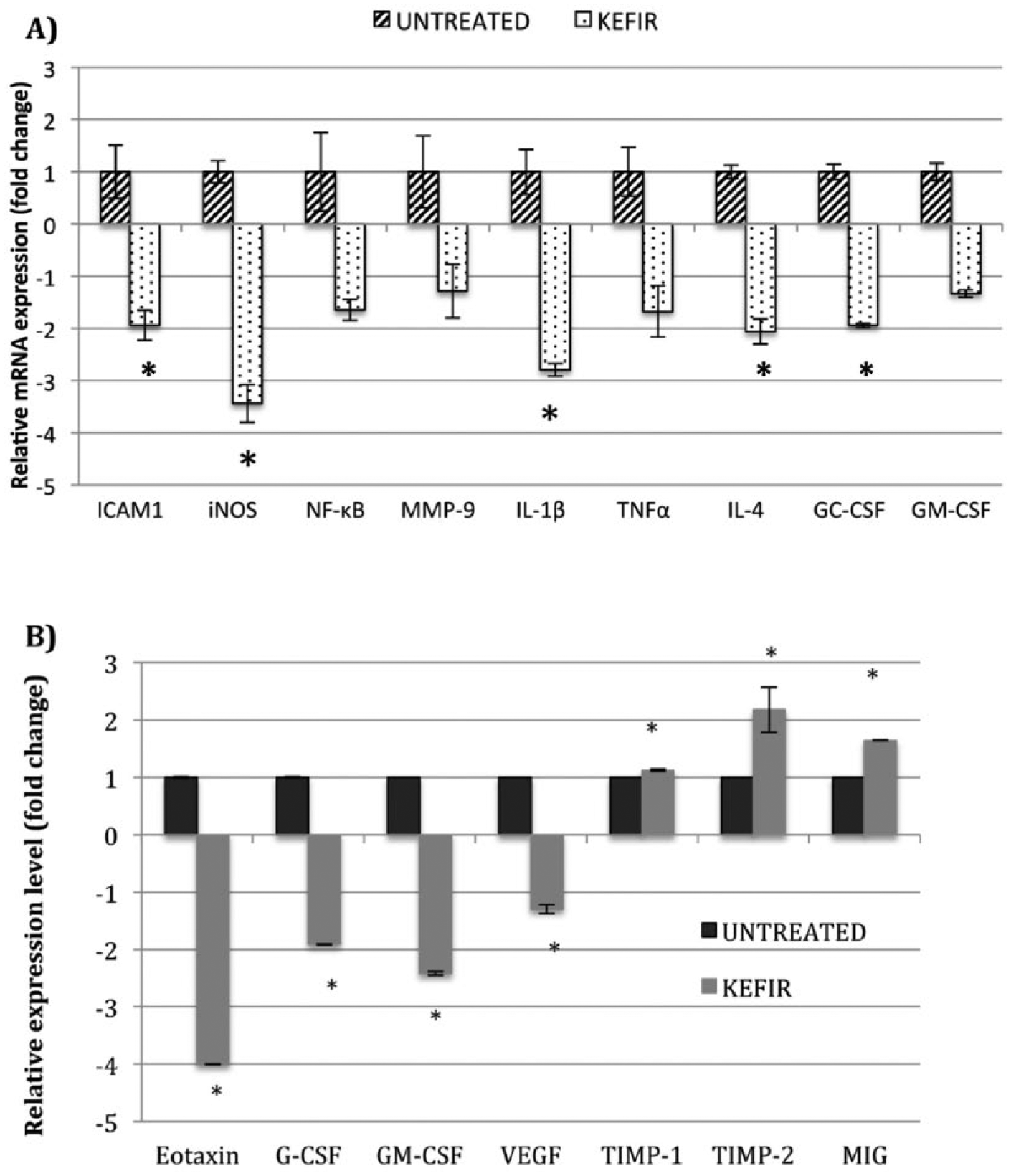
(A) Relative mRNA expression levels of ICAM, iNOS, MMP-9, IL-1β, NF-κB, G-CSF, GM-CSF, IL-4, and TNF-α. Each value represents the mean ± SD. Significant difference (*P < .05) from the untreated group was determined using the t-test. The experiment was done in triplicate. (B) The angiogenesis-related proteome array test was performed to evaluate the level of change of selected proteins. Data are presented as means ± SD. Significant difference (*P < .05) from the untreated group was determined using the t-test.
Discussion
Extensive research has been done to tackle breast cancer progression, but complete understanding is still a long way off. Diet is one of the factors that leads to cancer development, and the association between diet and cancer is currently being investigated. Scientists have found that the occurrence of tumors in animals can be manipulated by their diet intake. 23 A study on milk kefir provides an insight into its antiproliferative effects on breast cancer cells in vitro. 24 Although kefir has been commercialized in some places in the world, the mechanism of action behind the anticancer claim of kefir, especially kefir water, is still underresearched. Therefore, this study was carried out to further investigate the anti-metastasis and antiangiogenesis potential of kefir from Malaysia in triple negative 4T1 murine breast cancer cells. Triple negative breast cancer (TNBC) is known to have poor prognosis and highly metastasis compared to other breast cancer types. Women with TNBC developed a significantly higher visceral metastasis as the first site of distant recurrence than women with other breast cancer types. 25 Other than that, the development of brain metastases is higher in the triple-negative phenotype compared with other types of breast cancer. 26
Kefir water showed cytotoxicity toward 4T1 cells in the MTT assay, and the IC50 obtained was used in subsequent in vitro migration assays. The significant reduction in cell migration and invasion in kefir water–treated cells in vitro demonstrated the potency of kefir water as an antimetastasis agent. Angiogenesis is the formation of new blood vessels from existing vessels, and it has an important role in wound healing and development of tumors. 27 This feature is stimulated by proangiogenic factors, which are essential for tumor metastasis. 28 The ability of kefir water to inhibit angiogenesis was shown in the ex vivo mouse aortic ring assay, where lower microvessel growth was significantly observed in treated aortas (supplementary data, available at http://ict.sagepub.com/supplemental).
The effectiveness of kefir water in the inhibition of breast cancer metastasis was further tested in an in vivo model using BALB/c mice. After 28 days of treatment, the size and weight of tumors in the kefir water–treated mice were reduced significantly. Normal cell growth and proliferation are regulated within the cell cycle. Cancer cells divide rapidly because they deregulate the cell cycle signal and, thus, proliferate at their own pace. 28 Dividing cells in tumor sections were analyzed using H&E staining, and as can be seen in Figure 2A, the number of dividing cells was reduced in the kefir water–treated tumor section. TUNEL assay was performed on tumor tissue sections to detect DNA damage. An increase in DNA fragmentation was observed in kefir water–treated tumor sections in the TUNEL assay. DNA fragmentation is one of the indicators for cell apoptosis. 29 Therefore, the increase in DNA fragmentation indicated that the tumor cells were undergoing apoptosis.
The correlation between inflammation and cancer progression has been discussed for centuries. The chronic inflammatory conditions that correlate with infection and irritation may prompt genomic lesions and tumor initiation. 30 Reports have found that breast cancer patients exhibit elevated lipid peroxidation marker, MDA, 31 and inflammatory mediator, NO. 32 In this study, kefir water successfully reduced the levels of MDA and NO in kefir water–treated mice. The decrease in NO was associated with significant downregulation of iNOS in the kefir water–treated group (Figure 7A). The overexpression of iNOS is highly correlated with tumor progression and angiogenesis ability. 33 The activation of NF-κβ, which follows infectious agents or cytokines such as TNF-α, regulates the inflammatory response of iNOS and also ICAM-1, an adhesion molecule closely related to metastasis.34,35 Therefore, the reduction of these proinflammatory genes will help in providing a microenvironment that disfavors tumor growth.
The suppression of IL-2 and IFN-γ, which were generated by T-helper 1, are common in the case of chronic infectious diseases. 35 The effects of kefir water in the regulation of IL-2 and IFN-γ cytokines were also analyzed, and the kefir water–treated group showed increments in both cytokines. IL-2 is a product of activated CD4+ T cells and CD8+ T cells. 36 Furthermore, IFN-γ is known to increase the production of major histocompatibility complex and obstructs tumor angiogenesis. 35 This antitumor immunity stimulation was validated by the in immunophenotyping assay, where notably increased numbers of immune markers (CD4+/CD3+, CD8+/CD3+) were observed in the kefir water–treated group (Figure 5). In fact, kefir has also been reported to exert immunomodulating effects, when IgA+ and IgG+ were increased in the healthy treatment group. 37 Both CD4 helper T cells and CD8 cytotoxic T cells are highly important in immune response and producing antitumor response. 38 The inability of the immune system to eradicate tumors was mainly a result of the immunosuppressive environment produced by the developing tumor. The tumor suppressed both T cell populations in the untreated group as compared with the normal group and kefir water–treated group (Figure 5). However, this observation was dissimilar to that in a previous kefir study. 17 IL-4 and IL-10 are some of the Th2 cytokines that are formed in a COX-2 expressing environment, and these cytokines are able to obstruct the production of Th-1 cytokines by CD4+ T helper lymphocytes. 35 A study by Sharma et al 39 on the Lewis lung tumors in mice suggested that the overexpression of IL-10 decreased the cytolytic activity of CD8+. A similar outcome was observed in a study by Jarnicki et al 40 in which a corresponding increase in IL-10 diminished cytotoxic T cell responses and IFN-γ secreting T cells during tumor growth. An increase of IL-10 expression and reduction in the CD8+ population were observed in the untreated group when compared with the kefir water–treated group.
The role of kefir water as an antiangiogenic agent in breast cancer was further investigated through the expression of several angiogenesis-related cytokines, genes, and proteins. For tumor growth to obtain the capability to activate angiogenesis, it requires an angiogenic switch. 41 The angiogenic factors of IL-1β that encourage tumor invasiveness were observed in various experimental models.42,43 In the cytokine and gene expression assay, kefir water significantly reduced the level of IL-1β in the tumor environment. Furthermore, kefir water also inhibited the level of IL-10 cytokine, which is associated with poor prognosis. 44
Growth factors cause tumors to develop and migrate. Some important hematopoietic growth factors studied in this experiment included vascular endothelial growth factor (VEGF), granulocyte-colony stimulating factor (G-CSF), and granulocyte macrophage-colony stimulating factor (GM-CSF). The expression of VEGF is an essential mediator for angiogenesis, and it is abundant in many types of cancer malignancies, especially in breast cancer.45,46 In addition, the activation of VEGF promotes the growth of endothelial cells and migration. 47 G-CSF was identified as a macrophage regulator to promote proliferation, differentiation, and survival. 48 However, G-CSF along with GM-CSF can also act as proto-oncogenes, in which they are highly expressed in various tumors49-51 and also frequently found in invasive breast cancer cells. 52 They were also found in abundance in the cytokine profile of metastatic 4T1 tumors. 53 The levels of VEGF, G-CSF, and GM-CSF were significantly downregulated in the kefir water–treated group proteome profiler as compared with the untreated group. In addition, kefir water reduced the expression level of eotaxin, which is a member of the CC chemokine family. Chemokines are involved in the angiogenesis process by stimulating proangiogenic immune cells or by activating G protein–coupled chemokine receptors, which affect the tissue microenvironments. 54
The disruption and deregulation of the extracellular matrix (ECM) often leads to cancer progression. In chronic diseases, the disruption and remodeling of ECM by matrix metalloproteinases (MMPs) will affect several attributes of the cancer cells, such as the invasiveness, and metastatic and angiogenesis potentials. 55 MMP-9 is one of the important players in the MMP family, promoting vascular events in tumors as well as stimulating VEGF. 56 Tissue inhibitor of metalloproteinases (TIMPs) is an endogenous inhibitor that regulates the activities of MMP-9 in cancer cells. 55 Keeping the level of MMP-9 and TIMPs in check is important in reducing angiogenesis in cancer cells. MMP-9 expression is commonly abundant in metastatic breast cancer cells. 57 On the other hand, the increase in TIMPs such as TIMP-1 and TIMP-2 in the tumor microenvironment can kill cancer cells and reduce cancer proliferation. 58 The balance between the downregulation of MMP-9 and upregulation of TIMP-1 and TIMP-2 were demonstrated in the kefir water–treated group, and this equilibrium was important in reducing cancer. This antiangiogenic potential of kefir water was validated with lung clonogenic and bone marrow smearing assays, where significantly fewer 4T1 cells were observed in the lung of the kefir water–treated mice, and no abnormal cells were found in the bone marrow of the kefir water–treated mice. The immunomodulation potential of kefir water, which aids in inhibiting tumor cell growth has also been observed in other studies using murine models of breast cancers and sarcomas, whereby tumor growth was inhibited by enhancing the local immune system response and by increasing the total IgA level.5,59
The constituents of kefir that could have antitumor properties are mostly the polysaccharide and exopolysaccharides isolated from kefir grains as well as components released during fermentation, as suggested by previous studies.11,16,60 Interestingly, we have detected gallic acid as the major phenolic acid in the kefir water (result not shown). The antitumor effects and cytotoxicity of gallic acid extracts on many cell lines have been extensively observed over the years. 61 Thus, further studies on kefir water should include the effects of antitumor and immunomodulation activity of the gallic acid detected in kefir water.
Conclusion
In this study, kefir water was able to reduce 4T1 cell proliferation, migration, and invasion in vitro. An ex vivo angiogenesis assay was also performed, and kefir water showed positive results in inhibiting angiogenesis. In addition, kefir water exhibited antimetastatic and antiangiogenesis effects in 4T1 cell–challenged mice in vivo through reducing 4T1 cell mitosis, promoting apoptosis, modulating immune systems, inhibiting inflammation in the tumor microenvironment, and regulating angiogenesis-related proteins and genes. Thus, these findings are crucial in promoting kefir water as part of the diet intake for cancer patients.
Footnotes
Acknowledgements
The authors wish to thank Chai-Youn Wong from My Kefir World, Kuala Lumpur, Malaysia, for kindly providing the kefir grains for analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
