Abstract
Protosappanin B (PSB) is a key active component of Lignum Sappan extract. Although the antiproliferative effects of Lignum Sappan extract have been demonstrated in various cancer cells, relatively little is known about the effects of PSB on tumor progression. The aim of this study was to explore the anti-tumor effects of PSB on human colon cancer cells by regulation of intracellular signaling pathways and Golgi phosphoprotein 3 (GOLPH3) expression in vitro and in vivo. Our results showed that PSB effectively inhibited the viability and migration of SW620 cells and induced apoptosis, but had poor effect on HCT116 cells. Furthermore, PSB significantly reduced the expression of p-AKT, p-p70S6K, β-catenin, and p-ERK1/2 proteins in SW620 cells, and this effect was reversed by the corresponding signaling pathway agonists. Interestingly, PSB could also suppress GOLPH3 expression of SW620 cells in a concentration-dependent manner, but SW620 cells transfected with lentiviral vectors overexpressing GOLPH3 can effectively resist the cytotoxic activity of PSB in vitro. The xenograft experiment of SW620 cells with LV-GOLPH3 confirmed that PSB distinctly inhibited the tumor growth via suppressing GOLPH3 expression. Collectively, these findings clarified a new anti-cancer mechanism of PSB through inhibition of GOLPH3 expression and intracellular signaling pathways in colon cancer cells. PSB may be a potential new drug for colon cancer.
Introduction
Colorectal cancer is one of the most common malignancies in the world and ranks third in terms of incidence and the second leading cause of cancer death among 36 cancer types in 2018 worldwide according to 2018 Global Cancer Statistics report. 1 At present, surgical resection is the only curative modality for colorectal cancer. Unfortunately, most patients with colorectal cancer in developing countries are in the advanced stage. According to National Comprehensive Cancer Network (NCCN) Guidelines, these patients must receive individualized treatments such as adjuvant chemotherapy, targeted therapy, and immunotherapy. However, the high cost, adverse reactions, and drug resistance limit the treatment of patients in these countries. Therefore, it is necessary to research new cheap drugs for adjuvant treatment of colon cancer, which would benefit more patients.
Caesalpinia sappan L. is a member of the leguminous plant family and is widely distributed in tropical and subtropical regions of the world. Lignum Sappan extracts isolated from its dried heartwood by different methods have been investigated for anti-tumor roles in lung cancer, breast cancer, liver cancer, and acute myelocytic leukemia (AML) cells.2-5 However, the monomers in the extracts are very complex. Various active chemical compounds have been isolated from Lignum Sappan extract and were classified into different types according to molecular structure, including hematoxylins, protosappanins, homoisoflavonoids, sappan chalcones, and phenylpropanoids. 6 Some monomeric ingredients were demonstrated to improve the effect of combined therapy, for example, the combination of cisplatin and brazilin or brazilein showed synergistic effect on WiDr cells through induction of apoptosis. 7 Brazilin can enhance cytotoxic activity of doxorubicin, induce apoptosis through suppression of Bcl-2 protein expression and inhibit migration through downregulation of MMP9, MMP2, human epidermal growth factor receptor 2 (HER2), Rac1, and p120 protein expression in MCF-7/HER2 cells, 8 and 3-deoxysappanchalcone can suppress colon cancer cell growth by directly targeting the TOPK-mediated signaling pathway. 9 However, there are few studies on the antitumor effect of Protosappanin B (PSB).
PSB has been effectively isolated from extract of Caesalpinia sappan L. heartwood by different extraction methods,10,11 and showed anti-inflammatory effect in lipopolysaccharide (LPS)-induced macrophages via inhibited the IL-6 and TNF-α secretion. 12 Recently, it was discovered that PSB can inhibit the proliferation of human bladder cancer cell lines and colon cancer cell lines,13,14 but its mechanism is not clear.
Aberrant activation of phosphoinositide 3 kinase (PI3K)/serine/threonine-protein kinase (Akt)/mammalian target of rapamycin (mTOR), Wingless/Integrated (Wnt)/β-catenin, and mitogen-activated protein kinase (MAPK)/ERK signaling pathway is usually found in colorectal cancer cells, which promotes cell proliferation, invasion, and metastasis. These signaling pathways are regulated by various upstream proteins.15-17 Meanwhile, the Golgi phosphoprotein 3 (GOLPH3) is an oncoprotein and upstream component of these signaling pathways. Our previous studies have shown that the overexpression of GOLPH3 in colon cancer cells can activate the PI3K/Akt/mTOR, Wnt/β-catenin, and MAPK/EKR signaling pathways, promote proliferation and induce apoptosis, and is involved in the resistance of colon cancer cells to 5-fluorouracil (5-Fu) and cisplatin chemotherapy.18-20
Therefore, the present study evaluated the inhibitory mechanism of PSB in human colon cancer cells, and observed its implications on intracellular signaling pathways and GOLPH3 expression.
Materials and Methods
Chemicals and Reagents
Protosappanin B was purchased from Shanghai Yuanye Bio-Technology Co. Ltd. (B21622-20 mg, Shanghai, China). Reagents, signaling pathway agonists and antibodies were purchased from the following companies: RPMI-1640 medium, DMEM medium (Shanghai Yuanye Bio-technology, Shanghai, China); Streptavidin Peroxidase immunohistochemistry kit (GS4961, GS4962), cell counting kit-8 (QN1293), Annexin V-FITC/PI double staining apoptosis detection kit (SNM530), and ECL western blotting detection kit (QN1155) (Beijing BioRab Technology Co. Ltd., Beijing, China); BCA protein assay kit (C05-02001), western blocking buffer (5% BSA, C05-06002) (Beijing Biosynthesis Biotechnology Co., Ltd, Beijing, China); IGF-1 (K002504P), AZD2858 (A126819), and TPA (bs-1545R) (Shanghai Hengfei Biotechnology Co. Ltd. Shanghai, China); Anti-GOLPH3 (ab98023), Anti-p-AKT (ab8805), Anti-AKT (ab38449), and anti-PCNA (proliferating cell nuclear antigen) (ab18197) (Abcam, Cambridge, UK); Anti-p-p70S6K (ribosomal protein S6 kinase, 70 kDa) (sc-8416), Anti-p70S6K (sc-8418), Anti-β-catenin (sc-7963), Anti-p-ERK1/2(extracellular signal-regulated kinase 1 and 2) (sc-81492), Anti-ERK1/2 (sc-514302), Anti-GAPDH (sc-365062), and secondary antibodies (sc-516102, sc-2357) (Santa Cruz Biotechnology, Inc., Dallas, TX, USA).
Cell Culture
The human colon cancer SW620 cell line and HCT116 cell line were purchased from the Cell Bank of Type Culture Collection of the Chinese Academy of Sciences (Shanghai, China) and cultured in RPMI-1640 medium supplemented with 10% fetal bovine serum in 5% CO2 at 37°C. The cells were harvested in the logarithmic growth phase for further study.
Cell-Counting Kit 8 (CCK-8) Assay
Cell proliferation was assessed by CCK-8 assay. SW620 and HCT116 cells were seeded in a 96-well plate at a density of 1 × 103 cells/well. First, 6 double cells were set up in each group, and 6 control wells that only contained cells without any other treatment were set up in each group. Second, 10 µl of different concentrations of PSB (6.25, 12.5, 25, 50, 100, and 200 µg/ml) were added to each well for 24, 48, 72, and 96 hours respectively, 10 µl CCK-8 solution was added into each pore, and the cells were continuously cultured for 4 hours in 5% CO2 at 37°C cell incubator. The experiment was repeated 3 times. OD490 was measured with a microplate reader (Huadong Electronics, Nanjing, China). The following formula was used to calculate the cell viability ratio was:
Cell viability ratio (%) = OD490 (drug groups)/OD490 (Control) × 100.
Colony Formation Assay
SW620 and HCT116 cells in the logarithmic growth phase were seeded into 6-well plates (2 × 103 cells/well) containing complete culture medium. After 24 hours, different concentrations of PSB (17.5, 35, and 70 µg/ml) were added and cultured at 37°C for 7 days. Once the clones became visible, their culture was terminated and cells were washed twice with PBS and fixed in 100% methanol at 25°C for 10 minutes. After staining with crystal violet dye for at 37°C 30 minutes, cells were washed twice with PBS and the number of colonies (≥50 cells) were counted with Image J Software. The above steps were repeated 3 times.
Apoptotic (Annexin V-FITC/PI) Assay
SW620 and HCT116 cells in the logarithmic growth phase were seeded into 6-well plates (5 × 105 cells/well) containing complete culture medium. After 24 hours, 35 µg/ml of PSB was added. In another 24 hours, cells were washed with PBS and trypsinized. The culture medium was centrifuged at 1000 × g for 5 minutes. The supernatant was discarded, and the cells were resuspended gently in PBS. A total of 5 to 10 million resuspended cells were centrifuged at 1000 × g for 5 minutes, and the supernatant was discarded. The cells were resuspended in a mixture containing 195 µl Annexin binding buffer, 5 µl Pacific Blue Annexin V and 10 µl propidium iodide working solution for 10 to 20 minutes at room temperature, placed in an ice bath and detected by flow cytometry (FC500; Beckman Coulter, Inc.).
Transwell Migration Assay
SW620 and HCT116 cells were treated with 35 µg/ml PSB for 24 hours. The migration assay was carried out according to the manufacturer’s protocol. Briefly, migration chambers were placed in a 24-well plate, SW620 and HCT116 cells (2 × 105 cells/well in serum-free medium) were plated into the upper chambers, and RPMI 1640 (600 µl) with 10% FBS was added to the lower chambers as a chemoattractant. After incubation at 37°C in 5% CO2 for 24 hours, the non-migrating cells were carefully scraped from the upper chamber with cotton swabs. The migrated cells were subjected to 4% formaldehyde fixation and then 0.5% crystal violet staining. Five fields (×200) were randomly selected under a light microscope to count the number of cells migrated to the lower side. Experiments were performed independently 3 times and the counts were averaged.
Western Blot Analysis
Each cell line in the logarithmic growth phase was washed with PBS. The cells were lysed in RIPA lysis buffer for 20 minutes. The protein concentration was determined using a BCA assay kit. The protein lysates (40 µg per lane) were separated by 10% SDS-PAGE and transferred onto nitrocellulose membranes (Beijing Solarbio Science & Technology Co., Ltd.). The membranes were blocked in phosphate-buffered saline with 0.1% Tween-20 (PBST) plus 3% BSA for 1 hour at room temperature. The blots were incubated with antibodies against GOLPH3 (1:500), p-AKT (1:500), AKT (1:500), p-p70S6K (1:500), p70S6K (1:500); β-catenin (1:500), p-ERK1/2 (1:500), ERK1/2 (1:500), and GAPDH (1:1000) at 4°C overnight. The membranes were washed and incubated with horseradish peroxidase-conjugated secondary antibodies (1:5000) for 1 hour at room temperature. The bands were visualized with a ECL kit. The blots were scanned and analyzed using the Image J software (National Institutes of Health, Bethesda, MD, USA). All experiments were performed independently in triplicate.
Construction of GOLPH3 Overexpression Vector and Transfection
The lentivirus system was used to up-regulate GOLPH3 expression in SW620 cells. The lentiviral vectors expressing GOLPH3 (LV-GOLPH3) and the empty vector (LV-NC) were purchased from Miaoling Bio Co., Ltd. (Wuhan, China). SW620 cells were plated in 96-well plates and transduced with LV-GOLPH3 or LV-NC vectors, according to the manufacturer’s protocol. The GOLPH3 expression in SW620 cells was confirmed by Western blotting.
Nude Mouse Tumorigenicity Assay
A total of 16 Balb/c nude mice (6 weeks old) were purchased from Beijing Vital River Laboratory Animal Technology Company (Beijing, China). All animal experiments were approved by the Animal Experimental Ethics Committee of The Second Affiliated Hospital of Fujian Medical University (ethics approval no. 201983; Quanzhou, Fujian, China). All mice were free to eat and drink, and the temperature was kept at 22 ± 2°C with a humidity of 50% to 60%. The lighting was controlled in the room with alternating cycles of 12 hours-light (8:00-20:00) and 12 hour-darkness (20:00-8:00). All experimental procedures involving animals were performed in accordance with the Guide for the Care and Use of Laboratory Animals and conformed to the institutional ethical guidelines for animal experimentation. Nude mice were handled and cared for at the Experimental Animal Center of Fujian Medical University.
The female nude mice (16-17 g) were randomly divided into 4 groups (n = 4/group). The LV-NC group (control group) consisted of mice that were xenografted with SW620 cells transduced with empty vector. The LV-GOLPH3 group comprised mice that were xenografted with LV-GOLPH3-infected SW620 cells. PSB group involved mice that were xenografted with empty vector-infected SW620 cells and injected intraperitoneally with PSB. PSB +LV-GOLPH3 group consisted of mice that were xenografted with LV-GOLPH3-infected SW620 cells and injected intraperitoneally with PSB. Forty-eight hours after lentivirus infection, the SW620 cells were trypsinized, collected by centrifugation (400 × g for 5 minutes at 4°C) and suspended in PBS. Subsequently, 0.2 ml of DMEM containing 2 × 107 cells was injected subcutaneously into the axillary region of each mouse. The mice were housed in a pathogen-free environment. When an appreciable tumor had formed subcutaneously at 7 days, nude mice in the PSB group and the PSB + LV-GOLPH3 group received an intraperitoneal injection of PSB (20 mg/kg, every 4 days), while nude mice in the control group and LV-GOLPH3 group were treated with normal saline. Tumor volumes were measured every 4 days by caliper and calculated as V = 1/2 × (length × width 2 ). The mice were sacrificed after 27 days, and the tumors were resected. The GOLPH3 expression in the tumor tissue was detected by Western Blot, and the remaining tumor tissue was fixed in paraformaldehyde, embedded in paraffin, and sectioned for H&E staining and immunohistochemistry.
H&E Staining
The dissected colon cancer transplanted tumor was fixed in 10% formalin buffer, dehydrated with low to high concentration of ethanol, and embedded in paraffin to make 4 µm paraffin sections. The sections were dewaxed with xylene, then passed through high-concentration to low-concentration ethanol, and finally washed with distilled water for 5 minutes; the sections were dewaxed to stain. The stained sections were dehydrated and made transparent with absolute ethanol and xylene, and finally sealed with neutral gum.
Immunohistochemistry
Sections of 4 µm-thick tissue slices were dried overnight at 37°C, and then deparaffinized with xylene and rehydrated. The sections were treated with 3% hydrogen peroxide for 20 minutes to quench the endogenous peroxidase and then microwaved for antigenic retrieval using ethylene diamine tetraacetic acid (EDTA) buffer. Nonspecific antibody binding was blocked. Subsequently, the sections were incubated with anti-PCNA antibody (1:500) overnight at 4°C. After rinsing, the sections were incubated with biotin-labeled secondary antibody bound to a streptavidin-horseradish peroxidase complex. The peroxidase reaction was developed in DAB buffer substrate for visualization. The sections were then counterstained, mounted, and observed under microscope. Phosphate buffered saline (PBS) replaced the primary antibody as a negative control. Images were acquired using a digital camera (Olympus, Tokyo, Japan) at 400× magnification, and the results were analyzed by Image J Software. The results were defined as the number of positive cells/total number of cells.
Statistical Analysis
Statistical analysis was performed using the Statistical Package for the Social Sciences, version 20.0 (SPSS, Inc. Chicago, IL, United States). Numerical data are expressed as the mean ± standard deviation. The Student’s t-test was used to compare each experimental group with the control group. The differences between experimental groups were compared by 1-way analysis of variance. The differences between paired experimental groups were compared by the Least Significant Difference t-test. Statistical significance was set at P < .05.
Results
PSB Inhibits the Proliferative Viability of Colon Cancer Cells
SW620 cells and HCT116 cells were exposed to different concentrations (6.25, 12.5, 25, 50, 100, and 200 µg/ml) of PSB for 24 hours, 48 hours, 72 hours, and 96 hours. CCK-8 assay showed that PSB can inhibit the proliferation of SW620 cells in a dose-dependent and time-dependent manner (Figure 1A). However, the inhibitory effect of PSB on HCT116 cells was very poor. PSB could not significantly suppress proliferation of HCT116 cells at concentrations below 100 µg/ml (Figure 1B). The IC50 value of PSB in SW620 cell at 24 hours was 35.25 µg/ml. Under different concentration (17.5, 35, and 70 µg/ml) of PSB treatment for 24 hours, the colony number of SW620 cells significantly reduced with the increasing concentration of PSB, but there was no significant change in the colony number of HCT116 cells (Figure 1C and D). The results suggested that the inhibition effect of PSB on the SW620 cells viability is better than HCT116 cells viability.

Effects of PSB on the viability of SW620 cells and HCT116 cells. (A) Inhibition curve of SW620 cells. (B) Inhibition curve of HCT116 cells. (C) Cloning formation of SW620 cells and HCT116 cells. (D) Comparison of the colony number in SW620 cells and HCT116 cells. CCK-8 assay was used to detect the proliferation of SW620 and HCT116 cells. Colon cancer cell proliferation viability was tested by colony formation assay.
PSB Induces Apoptosis and Suppresses Migration in Colon Cancer Cells
SW620 cells and HCT116 cells were treated with PSB (35 µg/ml) for 24 hours. The apoptosis rate of SW620 cells was significantly increased relative to that in the untreated SW620 cells (Figure 2A and B), and the cell migration number of SW620 cells was significantly reduced (Figure 2C and D). However, these factors did not alter after PSB treatment in HCT116 cells (Figure 2B and D). The results indicated that PSB can induce apoptosis and inhibit migration in SW620 cells, but had no effect on HCT116 cells.

Effects of PSB on apoptosis and migration in colon cancer cells. SW620 cells and HCT116 cells were treated with 35 µg/ml PSB for 24 hours. (A) Apoptosis induced by PSB in colon cancer cells. Apoptosis was measured by the Annexin V-FITC/PI assay. (B) Comparison of apoptosis rate between SW620 and HCT116 cells. ***P < .001 versus the untreated cells. ###P < .001, comparison between SW620 cells and HCT116 cells. (C) Migration of SW620 and HCT116 cells under PSB treatment. Migration ability was detected by Transwell migration assay. (D) Comparison of the migration numbers of SW620 cells and HCT116 cells. ***P < .001 versus the untreated cells. ##P < .01, comparison between SW620 cells and HCT116 cells.
PSB Down-Regulated the Related Protein Expression of the Signaling Pathways in SW620 Cells
SW620 cells were treated with PSB (35 µg/ml) for 24 hours. The relative expression levels of p-AKT, p-p70S6K, β-catenin, and p-ERK1/2 in the SW620 cells were significantly reduced relative to the untreated SW620 cells (P < .05) (Figure 3A and B). However, when PI3K/Akt/mTOR, Wnt/β-catenin and MAPK/ERK signal pathway agonists (IGF-1, AZD2858, and TPA) were respectively added to SW620 cells under the same concentration of PSB treatment, the p-AKT, p-p70S6K, β-catenin, and p-ERK1/2 expressions were significantly elevated in the PSB treated SW620 cells (P < .05) (Figure 3A and C). These results suggested that PSB can simultaneously inhibit the activity of PI3K/Akt/mTOR, Wnt/β-catenin, and MAPK/ERK signaling pathways in SW620 cells, and hinted that PSB may suppress the key point in the upstream of these signaling pathways.
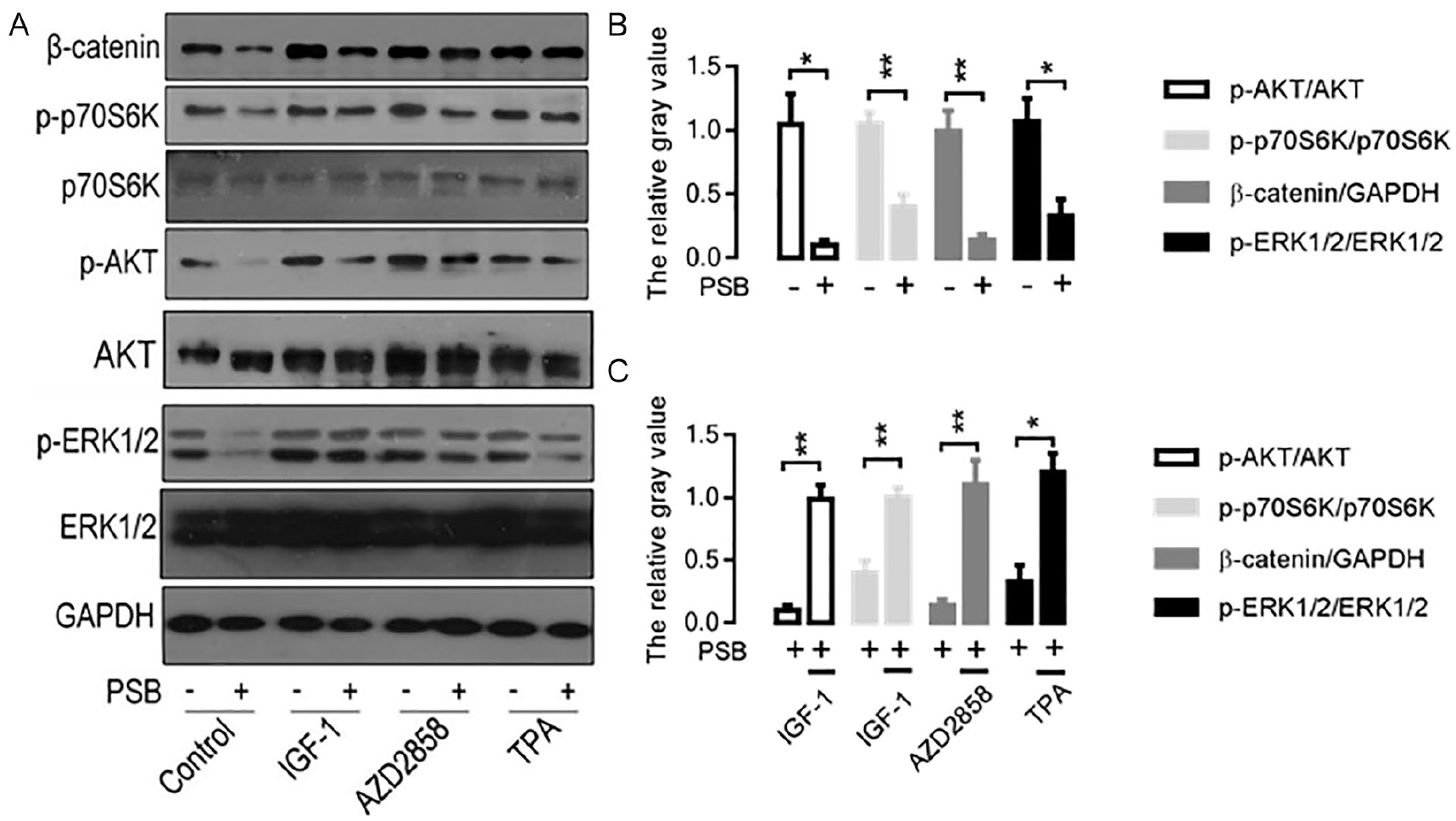
Effect of PSB on the related protein expression of signaling pathways in SW620 cells. (A) Western blot protein band images. The related protein expression of cell signaling pathways are analyzed by Western blot. (B) Comparison of the relative expression level of proteins in signaling pathways under PSB treatment. *P < .05. **P < .01, versus the untreated SW620 cells. (C) Comparison of the relative expression level of protein in signaling pathways under PSB and agonist (IGF-1, AZD2858, or TPA) treatment. *P < .05. **P < .01, versus the PSB treated SW620 cells.
PSB Inhibits the Expression of GOLPH3 Protein in Colon Cancer Cells
SW620 cells and HCT116 cells were treated with PSB in different concentration (17.5, 35, and 70 µg/ml) for 24 hours. As shown in Figure 4, with increasing PSB concentration, the relative expression of GOLPH3 protein in SW620 cells gradually decreased in a dose-dependent manner, while there was no significant difference in the GOLPH3 expression of HCT116 cells at different PSB concentration. When the PSB concentration reached 35 µg/ml, the GOLPH3 expression in SW620 cells was significantly downregulated relative to that in HCT116 cells (P < .05).

Effect of PSB on the expression of GOLPH3 protein in colon cancer cells. (A) Western blot protein band images. The expression of GOLPH3 protein was assessed by Western blot. (B) Comparison of the GOLPH3 expression in SW620 and HCT116 cells. Colon cancer cells were treated with PSB (concentration: 0, 17.5, 35, and 70 µg/ml) for 24 hours.
Effect of PSB on Proliferation in SW620 Cells Overexpressing GOLPH3
SW620 cells were transduced with lentiviral vectors overexpressing GOLPH3 (LV-GOLPH3) or empty vectors (LV-NC) for further experiments. Under PSB treatment (35 µg/ml) for 24 hours, the survival rate of SW620 cells with LV-NC was obviously decreased and the LV-GOLPH3-infected SW620 cell viability improved significantly (Figure 5). The result shows that the GOLPH3 overexpression in SW620 cells can antagonize the cytotoxic effect of PSB.

Effect of PSB on proliferation in SW620 cells overexpressing GOLPH3. CCK-8 assay was used to assess the survival rate of SW620 cells. PSB: 35 µg/ml.
PSB Inhibits the Growth of SW620 Cell Transplantation Tumor in Nude Mice by Suppressing the GOLPH3 Expression
The xenograft experiment was terminated at 27 days following the start of the experiment. The volume of subcutaneous xenografts in LV-GOLPH3 group was larger than that in control group, but the difference was not statistically significant. The tumor volume in PSB group was significantly lower than that in control group, and the volume in PSB + LV-GOLPH3 group was significantly larger than in PSB group (Figure 6A and B) (Table 1).

Effect of PSB on the growth of SW620 cell transplanted tumors in nude mice. (A) The specimens of subcutaneous xenograft in each group. (B) Volume growth curves of transplantation tumor in each group. (C) Expression of GOLPH3 protein in each group. The GOLPH3 expression of transplanted tumor tissues in each group was assessed by western blot. **P < .01, versus control group. ##P < .01, versus PSB group. (D) H&E staining and immunohistochemical staining of xenograft tissue. The magnification of images was 200×. (E) The positive expression rates of PCNA protein.
Comparison of Transplantation Tumor Volume in Each Group (X ± S).

P < .05, versus control group.
P < .05, versus PSB group.
Compared with control group, the GOLPH3 expression of the transplanted tumor was significantly increased in LV-GOLPH3 group and decreased in PSB group. Moreover, the GOLPH3 expression in the PSB + LV-GOLPH3 group was significantly upregulated relative to that in the PSB group (Figure 6C). This showed that PSB can inhibit the growth of SW620 cell transplanted tumors by down-regulating the GOLPH3 expression in vivo. In addition, the histopathological changes of transplanted tumors were further observed by H&E staining and the immunohistochemical method (IHC). H&E staining exhibited that the necrotic area was found in the transplanted tumor tissue. Degeneration and apoptosis of cancer cells were obviously observed in PSB group under the light microscope. In contrast, these features were inapparent in the PSB + LV-GOLPH3 group, which exhibited significant angiogenesis and karyokinesis in xenograft tissue. Furthermore, IHC staining indicated that PCNA-positive cells of xenograft tissue in the PSB group were lower than in the PSB + LV-GOLPH3 group (Figure 6D and E).
Discussion
PSB is one of the main components of Caesalpinia Sappan in Chinese traditional medicine and also an important reference for the quality of Caesalpinia Sappan L. in the Chinese Pharmacopoeia. At present, its molecular formula has been determined as C16H16O6, and it was found to effectively protect PC12 cells and neurons from ODG-induced neuronal damages by increasing the mitochondrial membrane potential and blocking mitochondria-dependent caspase apoptosis pathway. 21 However, there are few reports on the anticancer effect of PSB. It has been observed that PSB can significantly inhibit the growth of bladder cancer cell lines except SV-HUC-1 cells. 13 Our results showed that the inhibitory effect of PSB on the proliferation and migration in SW620 cells was better than that in HCT116 cells. This indicated that the sensitivity of PSB varies greatly in different cell lines. Interestingly, it has been demonstrated the GOLPH3 expression in SV-HUC-1 and HCT116 cell lines were significantly lower than bladder cancer cell and other colon cancer cell lines in Liu’s study and our previous experiments.18,22 Therefore, we speculated that the antitumor effect of PSB may be related to the GOLPH3 expression in colon cancer cells.
Our results found that the GOLPH3 expression was significantly downregulated in SW620 cells after PSB treatment in vitro and in vivo; moreover, exogenous GOLPH3 overexpression can attenuate the antiproliferative effect of PSB in SW620 cells. These data confirmed our hypothesis that PSB exerts the antitumor effect on colon cancer cells by inhibiting GOLPH3 expression.
The GOLPH3 gene is known as oncogene and identified as a frequent amplification in several human solid tumors including colon cancer. 23 GOLPH3 protein localizes to the trans-Golgi via the direct interaction with PI4P and drives cancer progression.24,25 Experimental data have validated that GOLPH3 overexpression correlates with cancer-associated phenotypes, and promotes proliferation, cell migration and angiogenesis by activating the downstream cell signaling pathways, including PI3K/Akt/mTOR, JAK2/STAT3, MAPK/ERK, Wnt/β-catenin, NF-κB, and AKT/FOXO1.26-28
Proteomic analysis indicated that PSB significantly changed the 1188 proteins expression in the bladder cancer T24 cells, which involved in TNF, p53, FOXO, and phosphatidylinositol signaling pathways. 13 Our results also revealed that PSB can suppress the PI3K/Akt/mTOR, Wnt/β-catenin, and MAPK/ERK signaling pathways in SW620 cells. Collectively, these data demonstrated that PSB could inhibit the activity of multiple cell signaling pathways which are regulated by the upstream GOLPH3 protein in many tumors. Therefore, our results further explain the antitumor mechanism of PSB.
In response to DNA damage, GOLPH3 expression in cancer cells is upregulated and can enhance cellular survival via regulation of the Golgi ribbon, 29 and this can result in colon cancer cell resistance to platinum drugs, which remain as first-line agent for colon cancer chemotherapy. PSB may improve the chemosensitivity of colon cancer to DNA-damaging agents via inhibiting GOLPH3 expression. Of course, further insights into the molecular mechanism of PSB may offer therapeutic strategies for tumors.
Conclusion
PSB can inhibit the viability and migration of colon cancer SW620 cells, and induced apoptosis. We demonstrated a new anti-cancer mechanism of PSB, which suppressed the expression of GOLPH3, resulting in inhibition of PI3K/Akt/mTOR, Wnt/β-catenin, and MAPK/ERK signaling pathways in SW620 cells. These results suggest that PSB may be a potential new drug for colon cancer.
Footnotes
Authors’ Note
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Science and Technology Project of Quanzhou (No. 2017Z015), Traditional Chinese Medicine Research Project of Fujian Province (No. 2017FJZYLC403), and Educational Research Project for Young and Middle-aged Teachers of Fujian Educational Committee (No. JAT190216).
