Abstract
Pathological angiogenesis and apoptosis evasion are common hallmarks of cancer. The present work was an endeavor to evaluate the influence of bee venom (BV) or its major constituent melittin (MEL) as antiapoptotic and angiogenic regulator modifier on the tumor growth and the cell sensitivity to ionizing radiation targeting the improvement of cancer therapeutic protocols. BV (0.56 mg/kg/day) and MEL (500 µg/kg body weight/day) were injected intraperitoneally to mice bearing 1 cm3 solid tumor of Ehrlich ascites carcinoma (EAC) for 21 consecutive days. Mice were whole-body exposed to 1 Gray (Gy) of γ-radiation (2 fractionated doses). Treatment with BV or MEL markedly suppresses the proliferation of tumor in EAC mice. The concentrations of m-RNA for angiogenic factors (TNF-α, VEGF) as well as MMPs 2 and 9 activities and NO concentration were significantly decreased, combined with improvements in apoptotic regulators (caspase-3 activity) and normal cells redox tone (catalase and free radicals content) compared with EAC mice. Moreover, the histopathological investigation confirms the improvement exerted by BV or MEL in the EAC mice group or EAC + R group. Exposure to γ-radiation sustained the modulatory effect of BV on tumor when compared with EAC + BV mice. Convincingly, the role of BV or MEL as a natural antiangiogenic in the biological sequelae after radiation exposure is verified. Hence, BV and its major constituent MEL might represent a potential therapeutic strategy for increasing the radiation response of solid tumors.
Introduction
Cancer is an irregular growth of cells arising from the accumulation of genetic mutations inherited and/or acquired causing oncogene activation or tumor suppressor gene inactivation. This contributes to dysregulation of the normal cellular system for cell division, cell differentiation, deregulated cell proliferation balance, and cell death, and ultimately to a population of cells that can invade tissues and metastasize to distant locations, causing substantial morbidity. 1 The basal membrane becomes the first barrier for an invading epithelial tumor. Degradation of the basement membrane and the extracellular matrix (ECM) in tissue around the tumor is necessary for the invasion and metastasis cycle. 2 Matrix metalloproteinases (MMPs) are powerful proteolytic enzymes that play key roles in this process, and tissue architecture alterations are a hallmark of malignant disease. 3 Approximately 25% of malignancies worldwide are believed to be caused by chronic inflammation, for example, colorectal, bronchial lung cancer, gastric, cervical cancer, hepatocellular carcinoma, gall bladder, pancreatic cancer, Burkitt’s lymphoma, melanoma, mesothelioma, and adenocarcinoma, 4 where altered rates of proinflammatory and proangiogenic factors are found in various cancers. Activation of major proinflammatory cascade inducers, including transcription factors signal transducer and activator of transcription 3 (STAT3) and nuclear factor–κβ (NF-κβ), leads to an increase in cell proliferation, apoptotic evasion, invasion. and metastasis, as well as angiogenesis, all of which are well-established cancer hallmarks. 5 Apoptosis is a cellular cycle, organized and coordinated, occurring under physiological and pathological conditions. It is the main event that regulates the occurrence and/or spread of cancer. 6
The interaction of anticancer agents with the infected cells is considered critical for choosing and optimizing a drug in order to achieve the most successful treatment for cancer. It is well known that the role of an anticancer agent is to cause the target cancer cells to become apoptotic. Cellular morphological changes, such as shrinkage, membrane blebbing, DNA cleavage, caspase activation, and mitochondrial dysfunction, are characteristic of apoptosis. 7 Recently, several antitumor compounds have been extracted from natural products with new structural features and mechanisms of action. Natural products serve as a good and affordable source for new drug companies. Apitherapy can be used as a preventive cancer agent or as a supplement to traditional cancer therapies. 8
Apitherapy (Apis is a Latin word meaning bee) is the practice of using bee products like honeybee venom and propolis for disease prevention or treatment. It can be also described as the science (and art) of using honeybee products, maintaining health, and assist the individual in regaining health. 9 Bee venom therapy is the part of apitherapy that uses bee venom (BV) in the treatment of health problems. 10 However, BV is a complex mixture of a variety of proteins and peptides, some of which have strong immunogenic 11 and neurotoxic properties. 12 It was stated that BV therapy is used for treatment of many different autoimmune disorders including rheumatoid arthritis, 13 lupus, scleroderma, and multiple sclerosis. 10 BV is a mixture of varying concentrations of proteins, polypeptides, and low-molecular-weight aromatic and aliphatic constituents. It also contains several important enzymes, that is, phospholipase A, hyaluronidase, acid phosphatase, and D-glucosidase, which are highly antigenic. Honeybee and wasp venom contain many substances such as bradykinin and mast cell and degranulating peptide; some of them show potential cytotoxic effects for tumor cells. These toxin components have wider therapeutic applications. 14
Melittin (MEL) is the main component of BV, accounting for about 50% of its dry matter. MEL is a peptide of 26 amino acids. It is the main active ingredient in honeybee venom. MEL has numerous biological, pharmacological, and toxicological activities, including strong cell lipid membrane surface activity, hemolyzing activity, antibacterial and antifungal activity, and possible antitumor activity.15,16
Radiotherapy is an important modality in the treatment of cancer, either employed alone in inoperable tumors or following surgery in many neoplasms. 17 Despite significant advances in medical physics, resistance of tumors to therapy in addition to systemic and normal tissue toxicity has limited the success of anticancer treatment. Several investigations during the past decade have shown that low-dose radiation is even more effective in cancer therapy than the conventional daily doses of 1 to 2 Gray (Gy). 18
This study aims to evaluate the antitumor modality of BV or MEL as proapoptotic and antiangiogenic molecules in Ehrlich ascites carcinoma (EAC) solid tumor irradiated mice. Thus, the action of BV or MEL was examined in vitro in EAC cells as well as in vivo in irradiated mice bearing Ehrlich cells (a model of solid carcinoma tumor).
Materials and Methods
Chemicals
Bee venom and other chemicals and reagents used in this study were obtained from Sigma-Aldrich Chemical Co. MEL was purchased from Shaanxi GTL Biotech Co, Ltd. BV or MEL was dissolved in sterile physiological saline (1:10 v/v).
Dose: mice received a daily BV intraperitoneal (ip) injection of 0.56 mg/kg body weight for 21 consecutive days. 19 MEL was injected ip at a dose of 500 µg/kg body weight for 21 consecutive days. 20
Radiation Facility
Whole-body γ-irradiation of mice was performed with a Canadian Gammacell 40 (137Cs) at the National Center for Radiation Research and Technology (NCRRT), Cairo, Egypt, at a dose rate of 0.46 Gy/min
Assay for In Vitro Cytotoxicity
In vitro short-term cytotoxicity of BV or MEL was assayed using the EAC cell line. Briefly, 2.5 × 106 EAC cells suspended in a mixture of 100 µL of phosphate-buffered saline (PBS 0.2 M, pH 7.4), and Roswell Park Memorial Institute (RPMI) 1640 media were mixed with 100 µL of various concentrations (0, 30, 60, 120, 240, 480,960 µg) of BV or MEL in a 96-well microtiter plate and incubated for 24 hours at 37 °C; suspension in PBS without treatment served as control. Stock solution of MTT in PBS (5 mg/mL) was prepared, and 10 µL of this solution was added to each well of microtiter plate, 24 hours after treatment with BV or MEL. The plate was incubated for an additional 4 hours to develop color. After incubation, the supernatant was carefully removed with the help of a micropipette. A total of 200 µL of dimethyl sulfoxide/well of microtiter plate was added to dissolve MTT and absorbance was measured at 570 nm. 21 The absorbance of formazan formed in the untreated cells was considered as 100% viable. Percentage of cell survival was calculated as follows:
The percentage of cytotoxicity was determined by calculating % inhibition of BV or MEL.
Tumor Transplantation
In the present study, the cell line of EAC was used as a model of solid carcinoma by inoculation in the right thigh of albino mice. The parent line was supplied by the Egyptian National Cancer Institute, Cairo University, as a donation. EAC, a spontaneous murine mammary adenocarcinoma modified to grow in female Swiss albino mice, is a well-established model in tumor biology. The cell line of EAC was preserved by ip injection of 2.5 million cells per animal. To count the EAC before ip, the bright line hemocytometer was used. Physiological sterile saline solution was used for the injection and dilution. In order to grow Ehrlich solid tumor (EST) in the thigh, 0.2 mL EAC cells (2.5 × 106 cells/mouse) were subcutaneously inoculated at the right thigh of the lower female mouse limb. 5
Animal Grouping
In this study, we used 105 mice weighing about 25 g each. All the experiments were conducted under national research center guidelines for the use and care for laboratory animals and were approved by an independent ethics committee of the NCRRT.
The animals were categorized into 7 groups of 15 mice each as follows:
Group 1. Control (C): Mice neither treated nor irradiated.
Group 2. (EAC): Mice bearing solid Ehrlich tumor.
Group 3. (EAC + R): Mice bearing solid Ehrlich tumor were subjected to 1 Gy whole-body γ-irradiation (2 fractionated doses) first at 11 days post EAC inoculation second at the day 14 post EAC formations.
Group 4. (EAC + BV): Mice bearing solid Ehrlich tumor were injected ip with BV for 21 consecutive days at 11 days after EAC inoculation.
Group 5. (EAC + BV + R): Mice bearing solid Ehrlich tumor were injected with BV as in group 4 along with a first whole body γ-irradiation at a dose level of 0.5 Gy 30 minutes after the first injection of BV and the second dose of irradiation as in group 3.
Group 6. (EAC + MEL): Mice bearing solid Ehrlich tumor were injected ip with MEL for 21 consecutive days at 11 days after EAC inoculation.
Group 7. (EAC + MEL + R): Mice bearing solid Ehrlich tumor were injected with MEL as in group 6 along with a first whole body γ-irradiation at a dose level of 0.5 Gy 30 minutes after the first injection of MEL and the second dose of irradiation as in group 3.
Tumor Volume Monitoring
Tumor volume was measured at different time intervals during the experimental period using a Vernier caliper on the 7th, 15th, and 21st days from the time the tumor reached 1 cm3 during the experimental period. The volume of solid tumor was calculated using the following formula: [A × B 2 × 0:52], where A and B are the longest and the shortest diameter of tumor, respectively. 22
Mice were anesthetized at the end of the experiment using diethyl ether. The skeletal muscle (normal control), tumor tissues, and liver were collected for biochemical investigations.
Quantitative Real-Time PCR
RNA Isolation and Reverse Transcription
RNA was extracted from the tumor tissue homogenate using the RNeasy plus mini kit (Qiagen), according to the manufacturer’s instructions. Genomic DNA was eliminated by a DNase-on-column treatment supplied with the kit. The RNA concentration was determined spectrophotometrically at 260 nm using the Nano Drop ND-1000 spectrophotometer (Thermo Fisher Scientific), and RNA purity was checked by means of the absorbance ratio at 260/280 nm. RNA integrity was assessed by electrophoresis on 2% agarose gels. RNA (1 µg) were used in the subsequent cDNA synthesis reaction, which was performed using the Reverse Transcription System (Promega). Total RNA was incubated at 70 °C for 10 minutes to prevent secondary structures. The RNA was supplemented with MgCl2 (25 mM), RTase buffer (10×), dNTP mixture (10 mM), oligod (t) primers, RNase inhibitor (20 U) and AMV reverse transcriptase (20 U/µL). This mixture was incubated at 42 °C for 1 hour.
Quantitative real-time polymerase chain reaction (qRT-PCR) was performed in an optical 96-well plate with an ABI PRISM 7500 fast sequence detection system (Applied Biosystems) and universal cycling conditions of 40 cycles of 15 seconds at 95 °C and 60 seconds at 60 °C after an initial denaturation step at 95 °C for 10 minutes. Each 10 µL reaction contained 5 µL SYBR Green Master Mix (Applied Biosystems), 0.3 µL gene-specific forward and reverse primers (10 µM), 2.5 µL cDNA, and 1.9 µL nuclease-free water. The sequences of PCR primer pairs used for each gene are shown in Table 1. Data were analyzed with the ABI Prism sequence detection system software and quantified using the v1·7 Sequence Detection Software from PE Biosystems. Relative expression of studied genes was calculated using the comparative threshold cycle method. All values were normalized to the endogenous control GAPDH. 23
Primers Used for QRT-PCR.

Abbreviation: QRT-PCR, quantitative real-time polymerase chain reaction.
Gelatin Zymography for MMPs 2 and 9 Detection
The presence and activity of specific MMP species (MMPs 2 and 9) were initially detected in the serum using substrate (gelatin) gel electrophoresis. 24 A buffer of 4% SDS, 0.15 mol/L Tris (pH 6.8), 20% glycerol, and 0.5% (w/v) bromophenol blue was added to the serum sample. Serum samples mixed with buffer were directly added to 10% SDS-acrylamide gel containing 0.1% (w/v) gelatin (Sigma) and separated by running on a mini gel apparatus at 15 mA/gel, and then gels were gently rocked in a 2.5% Triton X-100 solution for 30 minutes at room temperature (RT). Gels were then incubated overnight at 37 °C in substrate buffer containing 50 mmol/L Tris-HCl (pH 8), 5 mmol/L CaCl2, and 0.02% NaN3. Gels were subsequently stained for 30 minutes in 0.5% Coomassie Blue R-250 dissolved in a 1:3:6 solution of acetic acid, isopropyl alcohol, and water. The gel was scored for the presence/absence MMP activity by a blinded evaluator and photographed. MMPs 2 and 9 could be detected on the SDS gel as transparent bands.
ELISA Detection
Enzyme-linked immune sorbent assay (ELISA) for levels of caspase-3 was determined by using ELISA Kit (R& D Systems) according to the manufacturer’s instructions on the supernatants of sample tissue homogenates. In brief, microplates were coated with 100 µL/well of capture antibody, and then they were incubated overnight at 4 °C. After washes, the plates were blocked with assay diluent at RT for 1 hour. One hundred microliters of a serum sample was added to each well of the plate, followed by incubation for 2 hours at RT. Working detector was added into each well, and the plate was incubated for an additional 1 hour at RT before the addition of substrate solution. The reaction was stopped by adding stop solution. The absorbance was read using ELISA reader. The concentrations were calculated from standard curve according to the instructions in the protocol.
Measurement of Radical-Scavenging Ability in Hepatic Tissue Using Electron Spin Resonance (ESR) Spectroscopy
The liver tissues were quickly removed from mice and were gently lyophilized and were evaporated to dryness under vacuum. All samples were dissolved in a small volume of toluene, and were transferred to a round ESR cell. The cells were capped with a rubber septum and were thoroughly deoxygenated by nitrogen bubbling before ESR spectroscopy was performed. The ESR spectra were recorded at RT using a Bruker EPR ER-200D spectrometer, and spectral accumulation was done by using a Bruker ER-140 (ASPECT 2000) data systems. The microwave power was 2 mW, the modulation amplitude was 1 G, and 1 E4 receiver gain. The response time constant was 10 ms, with a field-sweeping rate of 100 G/42 s. The height of powder sample inside the quartz tube was about 10 mm. ESR spectral analyses were performed through the use of a computer simulation program. 25
Biochemical Assay
Catalase (CAT) activity was assayed in liver homogenate according to the method described by Sinha. 26 The dichromate/acetic acid reagent can be thought of as a stop bath for CAT activity. By addition of acetic acid to the reaction medium, CAT activity is destroyed and any hydrogen peroxide that has not been split by the CAT will react with the dichromate to give a blue precipitate of perchromic acid. This unstable precipitate is then decomposed by heating to give the green solution; this green color is measured photometrically at 570 nm. Nitric oxide (NO) level in the tumor tissue was determined as nitrite by Griess reaction. 27 Tumor homogenate was incubated with an equal amount of Griess reagent (1 part of 0.1% N(1-naphthyl)-diamine dihydrochloride in distilled water and 1 part 1% sulfanilamide in 5% concentrated H3PO4) for 10 minutes at RT, and the absorbance of color developed was measured at 540 nm.
Histopathological Study
Tissue samples were taken from the thigh muscle of mice in different groups and fixed in 10% formol saline for 24 hours. Washing was done in tap water, then serial dilutions of alcohol (methyl, ethyl, and absolute ethyl) were used for dehydration. Specimens were cleared in xylene and embedded in paraffin at 56 °C in hot air oven for 24 hours. Paraffin bees wax tissue blocks were prepared for sectioning at 4-µm thickness by sledge microtome. The obtained tissue sections were collected on glass slides, deparaffinized, and stained by hematoxylin and eosin stain for routine examination through the light microscope. 28
Statistics
Statistical analysis was performed by 1-way analysis of variance (ANOVA) followed by Duncan’s Multiple Range test using the Statistical Package for Social Science (SPSS) version 20.0 for Windows with P ≤ .05 as a significance level.
Result
Impact of BV or MEL on the Tumor Proliferation
A significant EAC viability alteration was observed in the EAC cells incubated with different concentrations of BV or MEL by MTT viability test.
Viable EAC cells incubated with RPMI complete media are considered a positive control (100% viability). BV or MEL inhibit EAC cell growth in a dose- and time-dependent manner. The inhibitory concentration (IC50) values of BV or MEL are reached at approximately 120 µg/mL and 55 µg/mL respectively, after 24 hours incubation (Figure 1).

Antiproliferative effect of (a) bee venom and (b) melittin on Ehrlich ascites carcinoma cells.
Impact of BV, MEL, and/or γ-Irradiation Exposure on Tumor Volume in Different Groups of Mice
Administration of BV or MEL according to the present study resulted in a noticeable retarding in tumor volume of EAC + BV mice and EAC + MEL mice when compared with EAC mice. However, the exposure to whole-body γ-irradiation displayed maximum repression especially on the 21st day (Figure 2).

Impact of BV, MEL, and/or γ-irradiation exposure on tumor volume in different mice groups.
Impact of BV, MEL Administration, and/or γ-Irradiation Exposure to Mice on Angiogenic Parameters
Treatment of mice bearing solid tumors with BV or MEL (EAC + BV and EAC + MEL groups) induced significant decreases (P < .05) in the expression levels of tumor necrosis factor–α (TNF-α), vascular endothelial growth factor A (VEGF-A), and serum MMPs 2 and 9 activities. Also, the NO contents of tumor tissue were significantly ameliorated. More pronounced decreases in TNF-α, VEGF-A expression, serum MMPs 2 and 9 activities, and NO concentrations were observed in EAC + BV + R, EAC + MEL + R group than that observed in EAC + BV, EAC + MEL, or EAC + R mice groups (Table 2).
Impact of BV, MEL, and/or γ-Irradiation Exposure on the Expression of (TNF-α, VEGF), the Activity of MMPs 2 and 9, and Nitric Oxide Concentration in Different Mice Groups*.

Abbreviations: BV, bee venom; MEL, melittin; TNF, tumor necrosis factor; VEGF, vascular endothelial growth factor; MMP, matrix metalloproteinase; C, control; EAC, Ehrlich ascites carcinoma; R, radiation.
Each value represents the mean ± standard error of mean (n = 6).
Significantly different from control.
Significantly different from EAC.
Impact of BV, MEL Administration, and/or γ-Irradiation Exposure to Mice on Tumor Cells Apoptotic Regulator Molecule
The data represented in Figure 3 point to significant increases (P < .05) in the activity of caspase-3 in the tumor cells of EAC + BV and EAC + MEL mice when compared with their corresponding values in EAC mice. The activity of caspase-3 was significantly induced after treatment of mice bearing solid tumor were exposed to whole-body γ-irradiation (EAC + R) group with BV or MEL.

The effect of exposure to γ-radiation and treatment with BV or MEL on caspase-3 in different animal groups.
Impact of BV or MEL Administration and/or γ-Irradiation on Catalase Concentration and the Peak Intensity for Liver Tissue ESR-Free Radical in Different Mice Groups
The results obtained (Table 3) displayed a significant recovery in normal tissue redox tone. The concentration of CAT (μmol consumed H2O2/min/g tissue) was significantly increased (P < .05) in the liver cells of mice bearing solid tumor (EAC + BV, EAC + MEL and EAC + BV + R, EAC + MEL + R) when compared with control mice (EAC). On the other hand, the level of liver-free radicals was markedly ameliorated (Table 2). The treatment of mice bearing solid tumor exposed to whole-body γ-irradiation (EAC + R) with BV or MEL was more effective in restoring and correcting the biochemical parameter level where the liver-free radical level was significantly decreased and liver CAT was significantly increased.
Impact of BV, MEL, and/or γ-Irradiation Exposure on Liver Catalase and the Peak Intensity for Liver Tissue ESR-Free Radical in Different Groups*.
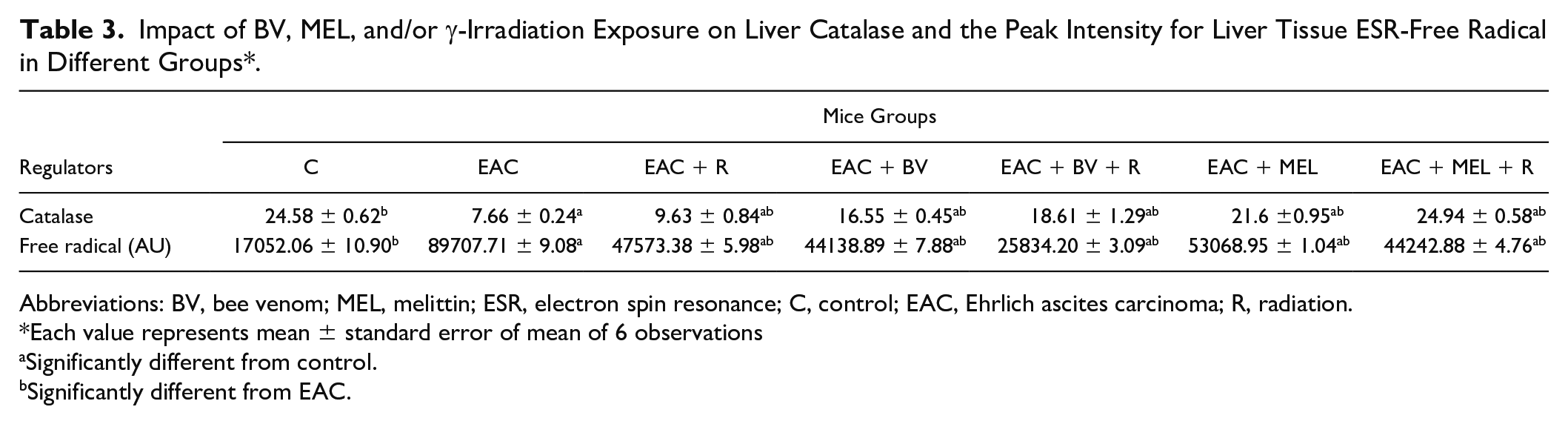
Abbreviations: BV, bee venom; MEL, melittin; ESR, electron spin resonance; C, control; EAC, Ehrlich ascites carcinoma; R, radiation.
Each value represents mean ± standard error of mean of 6 observations
Significantly different from control.
Significantly different from EAC.
Impact of BV or MEL Treatment on Histopathology of EAC-Bearing Mice Irradiated or Not Irradiated
Photomicrographic examinations of control mice thigh muscles tissue demonstrate normal histological structure of striated bundles (Figure 4A). However, photomicrographs of EAC mice displayed compactness of the tumor cells scattered within the muscular tissues and aggregated in a focal manner, infiltrating and penetrating the muscle bundles. Groups of large round and polygonal cells with pleomorphic shapes, hyperchromatic nuclei and binucleation, and several degrees of cellular and nuclear pleomorphism were observed (Figure 4B). Furthermore, in mice exposed to whole-body γ-radiation, a focal area of aggregated intact Ehrlich tumor cells appeared rather than musculature necrosis in other sides (Figure 4D). On the other hand, the histopathological investigation performed on tumor tissue sections of EAC + BV, EAC + MEL, EAC + BV + R, and EAC + MEL + R mice showed a great destruction of tumor tissue represented by the appearance of dead and necrotic cells after BV and MEL administration (Figure 4C and F). Combined treatment of irradiated female mice bearing solid tumor by BV or MEL represented more necrotic cells and remnant tumor cells and aggregation of the Ehrlich tumor cells in between the degenerated and necrosed skeletal muscle (Figure 4E and G).

Photomicrographs sections of right thigh muscles. (A) Normal control; C displayed normal histological structure of striated bundles (→). (B) Mice bearing solid tumor; Ehrlich ascites carcinoma (EAC) Ehrlich tumor cells exhibiting pleomorphism, hyperchromatism (*), infiltrating and penetrating the muscle bundles and aggregated in focal manner in between (→). (C) Bee venom (BV) showed malignant cells (red arrow) surrounded by 10% of tumor necrosis (blue arrow). (D) Irradiated mice bearing solid tumor; EAC + R, showed malignant cells (blue arrow) surrounded by 20% of tumor necrosis (red arrow). (E) Irradiated mice bearing solid tumor treated with BV; EAC + BV + R showed individual malignant cells (red arrow) surrounded by 60% of tumor necrosis (blue arrow). (F) Melittin (MEL) group displayed malignant cells (red arrow) surrounded by 30% of tumor necrosis (blue arrow). (G) Irradiated mice bearing solid tumor treated with MEL: EAC + MEL + R showed individual malignant cells (red arrow) surrounded by more than 80% of tumor necrosis (blue arrow).
Discussion
The metastatic spread of tumor cells is responsible for most cancer deaths, and all cancers will metastasize, with few exceptions. Clinical studies have long indicated that tumor-induced neovascularization (angiogenesis) is a key component of metastatic spread via the development of a route for tumor cell dissemination. Targeting tumor vasculature has a clear biological basis in radiation therapy, enhancing the radioresponsiveness of tumors with radiosensitizers, is a promising approach to radiation therapy effectiveness. 5 In an attempt to improve cancer therapeutic protocols, this study was undertaken to evaluate the antitumor effect of BV or MEL together with γ-irradiation against solid EAC tumors in female mice. Different molecular targets were analyzed in order to explore the underlying mechanisms by which BV or MEL suppressed the growth of solid tumor and/or enhanced the radiosensitivity of cancer cells to γ-irradiation.
In this study, a significant upregulation was recorded in TNF-α messenger RNA (mRNA) expression in solid EAC tumors, compared with non–EAC-bearing mice. The increase in TNF-α gene expression could be interpreted in the view of its role in neovascularization process. TNF-α is a key player in cancer-related inflammation in clinically important models of human and mouse epithelial carcinoma tumor through several mechanisms, including tumor inflammation, tumor angiogenesis, and inducing epithelial mesenchymal transformation (EMT). 29 Interaction of cancer cells with endothelial cells is a pivotal step in both tumor angiogenesis and metastatic dissemination. Mortensen et al 30 have observed spontaneous fusion between cancer cells and endothelial cells in vitro and in vivo that provide a novel type of tumor-endothelial cell interaction and may strongly modulate the biological behavior of tumors. 30 TNF-α is a major inflammatory mediator that induces several changes in the expression of endothelial cell genes, including the activation of adhesion molecules, integrins, and MMPs, and thus acts as an autocrine growth factor for angiogenesis of tumors. 31
Moreover, the obtained data displayed a significant elevation in the expressions of the angiogenic markers VEGF-A, MMPs 2 and 9 in EAC mice (Table 2). VEGF-A is a potent, endothelial specific permeability mediator and mitogen. It was found that tumor-induced angiogenesis is initiated by angiogenic cytokines such as basic fibroblast growth factor (bFGF) and VEGF that are expressed in the tumor itself. 32 In addition, increases of MMPs 2 and 9 are indices for the initiation of new vascularization. The significant increase in MMPs 2 and 9 activities in EAC-bearing mice could be attributed to TNF-α over-expression. TNF-α stimulates MMPs 2 and 9 activities in human corneal epithelial cells via focal adhesion kinase (FAK)/extracellular regulated protein kinase (ERK) signaling activation. 33 TNF-α has already been shown to be involved in cell-cell contact, and in a coculture of breast cancer cells and tumor-associated macrophages, macrophage-derived TNF-α increased the MMPs 2 and 9 levels in tumor cells. 34
MMPs are a family of enzymes involved in many physiological processes involving matrix remodeling, and tend to be important for angiogenesis, invasion of tumor cells, and metastasis. MMPs are held mainly responsible for degradation of the ECM. These enzymes are zinc-dependent and secreted in inactive proenzymatic forms where activation is necessary. In several invasive tumor cells, MMPs 2 and 9 (gelatinases A and B) have been found to be overexpressed. Several experiments confirmed the essential role these enzymes play in angiogenesis. 35 Several studies have shown that elevated levels of MMP-9 are expressed by cancer cells due to metastasis at distant organs. Hence, MMP-9 is not only critical in recognizing signs of invasion and diagnosis, but also a promising therapeutic target to prevent invasion and metastasis of cancer. 36
Furthermore, the significant increase (P < .05) in NO concentration of EAC-bearing mice might be due to TNF-α overexpression. It has been demonstrated that TNF-α is a mediator of NO synthesis. 37 A family of NO synthase (NOS) isoenzymes produces NO out of L-arginine. Such enzymes consist of 3 distinct isoforms encoded by 3 separate genes, including neuronal (nNOS codified by NOS-1), inducible (iNOS/NOS-2), and endothelial (eNOS/NOS-3) forms. Although, at baseline, the main source of plasma NO is related to eNOS, during several clinical conditions, such as inflammation, iNOS is activated. 38 The inducible form of NOS is expressed mainly through the TNF-α-activated pathway. 5 The earliest stages of angiogenesis are defined by vasodilatation-mediated NO and an increased vascular permeability of preexisting capillaries or postcapillary venules in response to VEGF, which are overexpressed by TNF-α. 35
The disturbance in the angiogenic and apoptotic regulators leads to tumor proliferation and growth, which was clearly demonstrated by the EAC tumor volume increase. Neovascularization enhances the ability of the tumor to grow and increases invasiveness and metastatic ability of it. 39 Apoptosis is a programmed cell death that maintains the stability of the internal environment by removing genetic mutations and unstable cells. However, this process is inhibited in cancer that leads to the accumulation of various genetically unstable cells. Our results demonstrated a significant decline in the level of apoptotic molecule (caspase-3) in the solid EAC tumors, compared with non–EAC-bearing mice. Caspase-3–mediated apoptosis is a major focus in the field of cancer growth inhibition, as activation of the proteolytic caspase cascade is a critical component in the execution of apoptotic cell death. 40 The increase in TNF-α expression is accompanied with decrease in caspase-3 activities. These findings are in line with the study by Zhang et al 41 who found that transmembrane TNF-α (tmTNF-α) expression knockdown enhances the therapeutic efficacy of doxorubicin (DOX) in a xenograft mouse model where the combination of tmTNF-α inhibition and DOX treatment resulted in much more pronounced suppression of tumor growth than treatment with either DOX or silence of tmTNF-α.
Furthermore, free radical level was elevated in liver tissue of EAC mice in parallel with a decrease in CAT concentration (Table 3). The presence of tumor in the human body or in experimental animals could affect many functions of vital organs in the body, even when the site of tumor does not interfere directly with this function. Gönenç et al 42 reported that enlargement in tumor size might causes disturbances of antioxidant and oxidative stress elevation in vital organs of the tumor host. The redox tone disturbance in normal cells could be attributed to influx of certain cytokines released from proximal tumor cells. Moreover, Khanna et al 43 identified that carcinoma cells implantation into mice exerted significant increase in lipid peroxidation represented by free radical content in vital organs that reflected cell growth rate of tumor. CAT plays an important role as an endogenous antioxidant system, which is found in particularly high concentration in liver, and is known to have key functions in cells protection by scavenging reactive oxygen species and modulating cellular redox status. The decrease in CAT activity was observed in the liver of tumor-bearing mice. 44
TNF-α is reported to be produced as a part of the host pro-inflammatory response to tumor cells during the initial stages of hepatic metastasis. Although TNF-α is cytotoxic, it also facilitates the formation of hepatic metastasis by increasing the expression of adhesion molecules, by promoting tumor cell migration, survival and invasion, and by inducing an angiogenic response. 45 It is well known that TNF-α activates NF-κB in many types of cells through binding to TNF receptors. It has also been demonstrated that hydrogen peroxide is involved in the TNF-α–induced NF-κB activation. 45 Administration of CAT derivatives was found to be effective in inhibiting the NF-κB activation in both liver cells and tumor cells. Targeting delivery of CAT leads to inhibiting the increase in the expression of antiapoptotic factors in tumor cells and increases the expression of adhesion molecules, pro-inflammatory cytokines, and MMP in host cells by suppression of NF-κB activation. 45
The current data revealed that the upregulation of TNF-α genes and induction of MMPs’ activity and NO concentration along with apoptosis suppression in solid EAC-bearing mice collectively enhance tumor cell proliferation and neovascularization that ultimately resulted in tumor growth acceleration, invasiveness, and metastatic ability of tumor cells.
Exposure of EAC-bearing mice to γ-irradiation produced a significant delay in tumor volume, which was achieved through the downregulation of angiogenic regulators expression, suppression of MMP activities, and induction of apoptosis. Radiation-induced DNA damage activates a variety of signaling pathways; the intrinsic apoptotic pathway has been considered the primary mechanism mediating ionizing radiation–induced apoptosis, making tumor cells unable to divide and grow.5,46,47 Many cancer cells, including prostate, lung, immortalized keratinocytes, and colon cancer cells, are committed to apoptotic cell death when exposed to radiation.48,49 A noticeable alteration in muscle bundles and the cells’ architectures in addition to the occurrence of tumor cell infiltration and penetration is seen in micrographs of EAC and EAC + R when matched with the micrograph of normal control thigh muscle bundles (Figure 4). Moustafa et al 50 attributed these changes to the alteration in biochemistry of tumor where the changes in signaling of STAT-3 and malondialdehyde (MDA) accumulation in the cells of muscle bundles destroyed the cellular integrity and paved the way to tumor proliferation. The MDA accumulation at the end of the lipid peroxidation process had destroyed the intact cellular membrane.
Present cancer treatment approaches consist mostly of surgery, radiation-based therapy, chemotherapy, gene therapy, and/or hormonal therapy. Another technique that has become increasingly important is the use of biotoxins or api-toxins, such as animal venoms, as cancer therapies.51-53 These biotoxins are produced by living organisms as a defense mechanism against predators and are known to have both toxicological and pharmacological effects. 54 Recently, data suggest that toxin from BV has some potential as antitumor agent. 6 On the other hand, apitherapy, the medical uses of honeybee products ranging from royal jelly to BV, has been introduced as a natural therapeutic in cancer chemotherapy. 55 BV is a biotoxin or api-toxin synthesized and secreted by a gland that is present in the abdominal cavity of the bee and is composed of a complex mixture of several biologically active peptides including MEL, bioactive amines, enzymes, and non-peptide components that have a variety of pharmaceutical properties. 56
The result of this study revealed that treatment of EAC-bearing mice with BV or MEL significantly reduced the growth of solid tumors, and a synergistic effect was demonstrated following combination of both treatments with γ-irradiation. As a key player in tumor proliferation, angiogenesis, and inflammatory cascade induction, the down regulation of TNF-α expression following treatment of irradiated and non-irradiated EAC-bearing mice with BV or MEL reduced tumor growth via multiple mechanisms, including suppression of tumor cell proliferation (reduction in tumor volume), angiogenesis (downregulation of tumor TNF-α expression and NO concentration), invasion (decrease in VEGF expression, reduction of serum MMPs 2 and 9 activities), and enhancing apoptosis (induction of tumor caspase-3). BV prevents tumor angiogenesis and metastasis by inhibiting tyrosine phosphorylation of VEGFR-2 in LLC tumor–bearing mice. 32 The valuable effect of BV may be mediated through its inhibitory effect on serum IL-6 concentration, ovarian cyclooxygenase 2 (COX-2), and VEGF expression. 57 BV or MEL effects on TNF-α prevent NO via JNK pathway–dependent inactivation of NF-κB, and induction of apoptosis via suppressions of NF-κB.58-60 BV has demonstrated anticancer activities, it inhibited cancer cell growth through induction of apoptosis in lung cancer cells, 61 and hepatocellular carcinoma cells. 62 MEL suppresses tumor progression through tumor-associated macrophage regulation in a Lewis lung carcinoma mouse model. 63
Honeybee venom significantly inhibits lipid peroxidation and displays a considerable hydroxyl radical scavenging activity. 64 The apoptotic effect of BV was demonstrated on human MDA-MB-231 breast cancer cells using Raman spectroscopy and principal component analysis. 7 BV has been observed to inhibit prostate cancer under in vitro and in vivo conditions, and these effects are mediated through activation of caspase via inactivation of the NF-κB pathway. BV or MEL inhibited LNCaP cells, DU145 cells, and PC-3 cells growth (prostate cancer cell) through induction of apoptotic cell death. They inhibited growth of the late- and early-stage prostate cancer cells. This cancer cell growth inhibitory effect was correlated with the downregulation of various proliferative and antiapoptotic gene products, including Bcl-2, cIAP-2, XIAP, iNOS, COX-2, and cPLA2, which are regulated by NF-κB.65-68
The results of our study was paralleled with the findings of Badria et al 8 who found that BV affects cell proliferation, MMP-2 activity, and interferon-β production in a time- and dose-dependent manner. MEL is the main compound found in BV, exerts multiple effects on cellular functions of cancerous cells such as apoptosis, proliferation, angiogenesis metastasis, and cell cycle, and the anticancer processes involve diverse signal molecules and regulatory pathways by regulating the expression of caspases, Ca2+ concentration, death receptors (DRs), ECM degradation enzymes, angiogenesis factors, and a variety of signal pathways. 69 BV was observed to inhibit PMA-induced MMP-9 expression and activity by inhibition of NF-κB via p38 MAPK and JNK signaling pathways in MCF-7 cells. In addition, MMPs 2 and 9 were inhibited by MEL, apamin and phospholipase A2 (PLA2). 70
Where the earliest stages of angiogenesis are defined by vasodilatation mediated by NO and an increased vascular permeability of preexisting capillaries or postcapillary venules in response to overexpressed VEGF, the inducible form of NOS is expressed mainly through TNF-α–activated pathway, So TNF-α is the maestro for controlling angiogenesis process.35,71 BV or MEL has demonstrated potential efficacy in inhibiting angiogenesis and invasion markers in a variety of human cancers tested under preclinical model systems by inhibiting the expression of TNF-α, MEL inhibits TNF-α secretion and expression of IL-1β and IL-6 in the TNF-α–treated hepatic stellate cells (HSCs). 68 It attenuates inflammation and fibrosis through NF-κB signaling pathway inhibition, and was observed to suppress HIF-1α/VEGF expression by inhibition of ERK and mTOR/p70S6K pathway in human cervical carcinoma CaSki cells. 72 It was stated by Yang et al. that MEL enhances radiosensitivity of hypoxic head and neck squamous cell carcinoma by suppressing HIF-1α, Additionally MEL dose-dependently inhibited proliferation and tube formation in human umbilical vein endothelial cells (VEGF-A-HUVECs), without affecting cell viability in native HUVECs; it also decreased the expression of VEGF receptor-2, COX-2, and prostaglandin E2 in VEGF-A–transfected HUVECs. These effects were accompanied by a reduction of the phosphorylation of extracellular signal-regulated kinase 1/2 and c-jun N-terminal kinase, whereas it increased the phosphorylation of p38 MAPK.73,74
In the present study, treatment of EAC and EAC + R groups with BV or MEL exerted a marked effect in the retardation of tumor growth as compared with the tumor-bearing mice group. These observations could be attributed to the antiangiogenic, antiapoptotic, and antioxidant capacity of BV or MEL.
Furthermore, the results of histological examination of the tumor micrograph of EAC + BV, EAC + MEL, EAC + BV + R, and EAC + MEL + R mice groups as compared with EAC mice reveals noticeable amelioration in cell architecture of the muscle bundle in addition to certain improvements in the state of tumor cells infiltration, penetration, area of necrosis, and enhancing tumor cells apoptosis (Figure 4). The results give the impression that BV or MEL plays an affordable role in the inhibition of the TNF-α activation event. Tumoral cells contain anionic phospholipids, mainly phosphatidylserine on the external leaflet of the plasma membrane. 75 To these leaflets MEL binds preferentially as it possesses cationic charges more than normal cells. It displays antiproliferative,76,77 and antiangiogenic activity against cancer cells in vivo.72-74
Conclusion
In conclusion, our results provide evidence that BV and its main constituents, especially MEL, may be developed as potential therapeutic agents for cancer metastasis via enhancing the sensitivity of tumor cells to ionizing radiation in addition to protection of normal cells against radiation and chemotherapeutic toxicity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Statement
The animal procedure was approved by the Ethics Committee of the National Center for Radiation Research and Technology.

